Abstract
Rhodococcus equi was isolated from lung, liver, spleen, and stomach content of two aborted equine fetuses of 7 and 8 months gestation from two different farms. Lesions included diffuse pyogranulomatous pneumonia with numerous Gram-positive coccobacilli within the cytoplasm of macrophages, multinucleated Langhans giant cells and neutrophils, and enhanced extramedullary hematopoiesis with megakaryocytosis within the liver and spleen. Detection of R. equi was made by bacteriology and immunohistochemistry for R. equi and VapA, the virulence factor of R. equi. R. equi and VapA were identified within the lungs of both fetuses, and its distribution correlated with lesions. Fetal lesions were similar to those observed in foals. We speculate that the fetuses contracted infection from the placenta by normal breathing movements or by swallowing of the amniotic fluid contaminated with R. equi.
Rhodococcus equi is a ubiquitous soil bacterium able to infect humans, horses, pigs, goats, cattle, sheep, cats, and llamas. 4 R. equi virulent strains carrying one of several plasmids that encodes the VapA (15–17 kd) antigen consistently cause disease when used for experimental infection of horses. 16 Horses with R. equi lesions are infected almost exclusively with virulent strains. 15 However, in humans, goats, and occasionally, foals, R. equi strains lacking this virulence factor can also produce disease. 4, 14, 15 R. equi causes pyogranulomatous pneumonia, lymphadenitis, polyarthritis, and ulcerative enteritis in 2–6 month old foals. 5 Sporadically, it produces numerous other lesions in horses, such as peritonitis, pleuritis and pericarditis, renal infarction, septicemia, retrobulbar abscess formation, suppurative osteomyelitis, nephritis and hepatitis with abscesses, ulcerative lymphangitis, and dermatitis with abscesses. 2, 18 Rarely, R. equi can be isolated from the genital tract of mares 1, 2, 3 and has been cultured from aborted equine fetuses. 3, 11, 18 There are two reports describing the lesions associated with infection in aborted equine fetus and placenta. 6, 9 We describe the gross, histologic, immunohistochemical, and bacteriologic findings of two cases of equine abortion caused by R. equi.
A 6-year-old, Hungarian half-breed mare aborted a 7-month-old, male fetus (fetus No. 1) in October 1990. A 6-year-old, Hungarian half-breed mare from a different farm aborted her 8-month-old, male fetus (fetus No. 2) in November 1995. Neither premonitory clinical signs nor postabortion complications were observed in either horse. Both fetuses were submitted for necropsy, without fetal membranes, to the Central Veterinary Institute, Budapest, Hungary.
Samples collected for bacterial cultures were swabs from kidney, liver, spleen, lungs, and stomach content from fetus No. 1; a nasal and a cervical swab from its dam (7th day after abortion); and spleen, liver, and lungs from fetus No. 2. These samples were inoculated on agar containing 10% sterile sheep blood and also on common agar. Petri dishes were incubated under aerobic conditions at 37°C for 48 hours. Smears from fetus No. 1 were made from the stomach content and the cut surface of the lungs and stained according to Stamp 12 for bacterium and fungi. Virus isolation was attempted from the viscera of both fetuses using the RK-13 cell line. Tissues from both fetuses were fixed in 10% buffered formalin, processed routinely, and paraffin embedded. Four-micron sections were deparaffinized, and serial sections were stained with hematoxylin and eosin, PAS, Brown-Brenn, and Ziehl-Nielsen. Following deparaffinization, sections for immunohistochemistry were treated with 3% H2O2 solution for inactivation of endogenous peroxidases, blocked with 2% skim milk powder solution at room temperature, and digested for 20 minutes with 0.1% protease XIV solution (Sigma Aldrich Co., St Louis, MO) at 37°C. Subsequently serial sections were incubated overnight with R. equi specific polyclonal affinity purified rabbit immunoglobulin G (IgG) 13 (dilution 1 : 12,000; produced at the Central Veterinary Institute, Budapest, Hungary) and murine monoclonal antibody against VapA (15–17 kd) antigens 7 (dilution 1 : 20,000; produced at the School of Veterinary Medicine and Animal Sciences, Kitasato University, Towada, Japan). Further serial sections were treated for 10 minutes with 0.1% protease XIV solution at 37°C, and subsequently, they were incubated overnight with one of the following antibody preparations: Mycobacterium spp.–specific polyclonal rabbit immunoglobulin (DAKO Co., Glostrup, Denmark, dilution 1 : 1,000); murine monoclonal antibody against the glycoprotein of equine herpes virus 1 (EHV-1; dilution 1 : 1,500; courtesy of Dr. G. Allen, Gluck Equine Research Center, University of Kentucky, Lexington, KY); and murine monoclonal antibody against a 30-kd protein of equine arteritis virus (EAV; dilution 1 : 10; courtesy of Dr. A. Glaser, New York State Diagnostic Laboratory, Cornell University, Ithaca, NY). Irrelevant mouse and rabbit serum were substituted for the primary antibodies. Lung from a foal containing R. equi and associated lesions was used as a positive control. The antigen–antibody binding was detected by the use of horseradish-peroxidase–labeled streptavidin–biotin kit (Universal LSAB2 Kit-HRP, DAKO Co.). The sections were treated at room temperature for 10 minutes with 3-amino-9-ethylcarbazole (Sigma Aldrich Co.) solution, also containing H2O2, counterstained with Mayer's hematoxylin and covered with glycerol gelatin.
Fetus No. 1 had icterus and mild hepatomegaly. Lungs were diffusely consolidated, and on the cut surface, there was grayish-whitish marbling with purulent exudates. Bronchial lymph nodes were enlarged and edematous. Fetus No. 2 presented with similar changes, and the lungs contained numerous grayish, 4–10-mm foci. Histopathologic examination of fetus No. 1 revealed pyogranulomatous pneumonia, characterized by numerous intra-alveolar neutrophils and macrophages and multinucleated Langhans giant cells containing intracytoplasmic coccobacilli. The alveolar epithelium was multifocally necrotic. The perivascular and peribronchiolar, as well as the interlobular and subpleural connective tissues, were edematous, slightly fibrotic, and infiltrated with numerous macrophages containing coccobacilli (Fig. 1). In fetus No. 2, similar histopathologic changes were observed, but perivascular and peribronchiolar fibrosis was more pronounced, and there were a few fibrin thrombi in the blood vessels. Lymphoid organs were characterized by mild lymphocyte depletion and moderate-to-prominent extramedullary hematopoiesis with megakaryocytosis, particularly in the liver and spleen. There was also mild fibrous thickening of the hepatic capsule. The coccobacilli were Gram positive mainly intracytoplasmic (Fig. 2) and stained with HE and PAS but not with acid-fast stain.
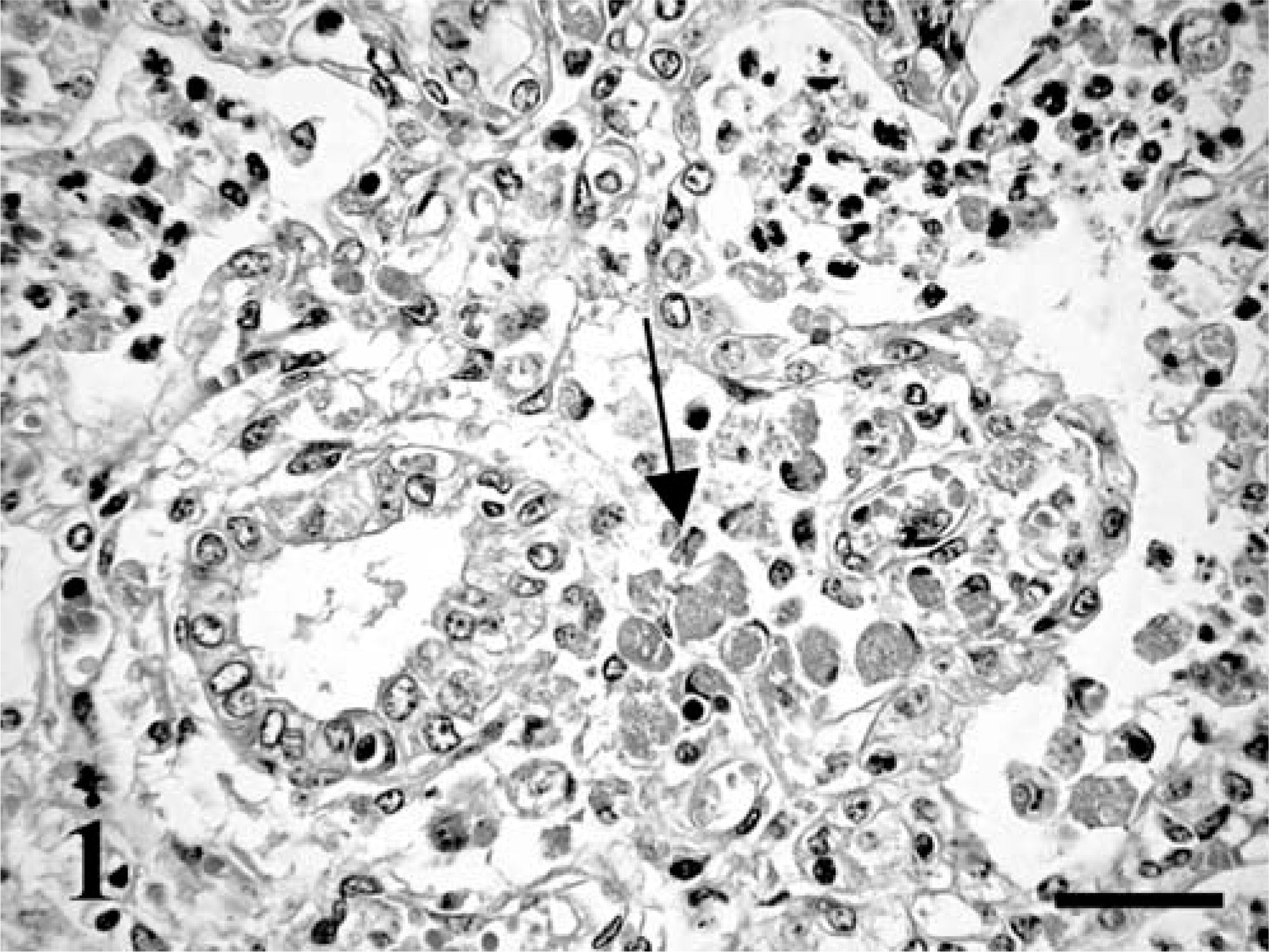
Lung; equine fetus No. 1. Numerous intra-alveolar neutrophils and several macrophages (arrow) in the connective tissues around a bronchiole. HE. Bar = 60 µm.
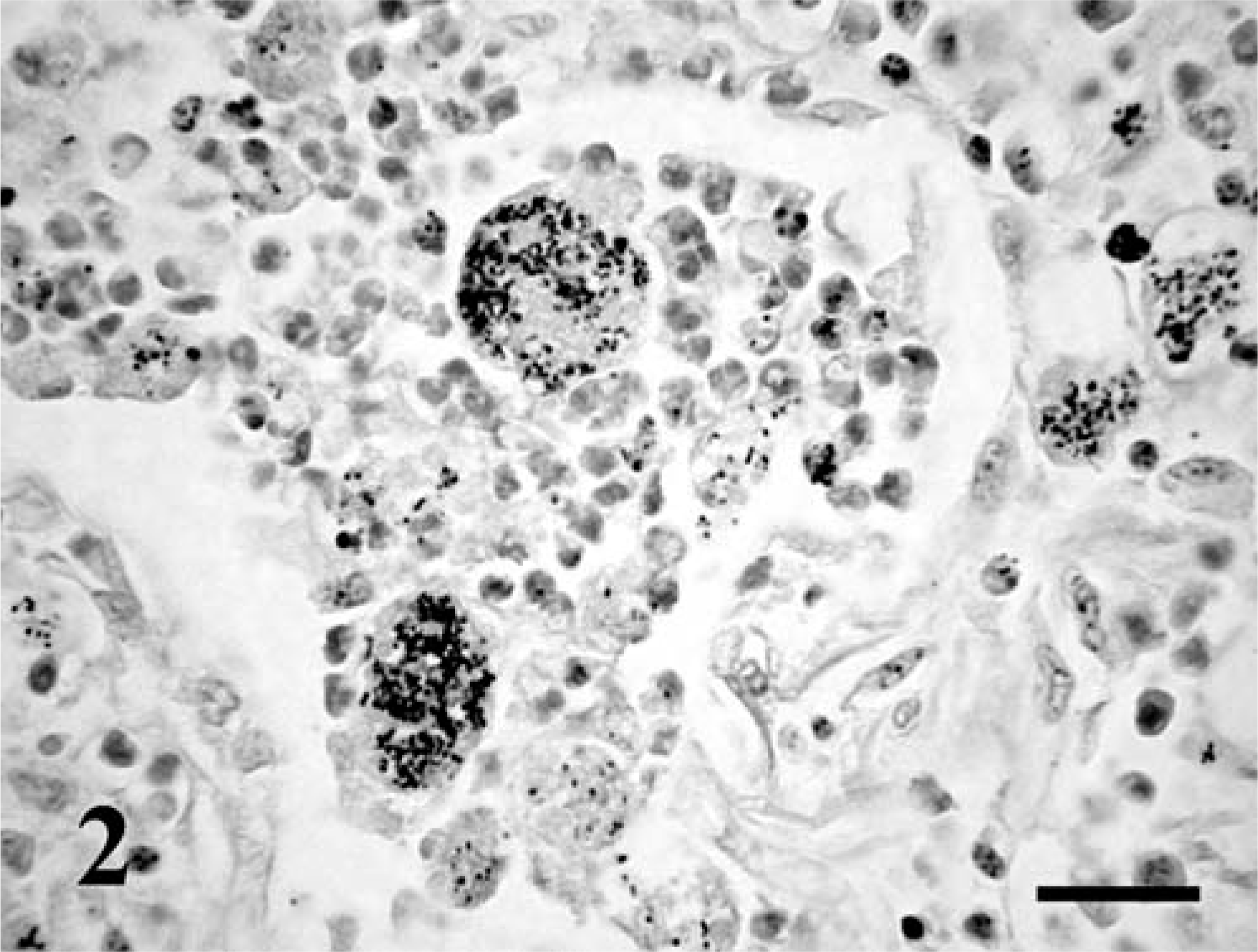
Lung; equine fetus No. 2. Numerous Gram-positive coccobacilli in the cytoplasm of macrophages. Brown-Brenn. Bar = 30 µm.
Mucoid, pink bacterial colonies grew and were identified from the inoculated plates as R. equi. R. equi was not identified in the vaginal and nasal swabs of the mare. Numerous intracellular, red-staining cocci were identified in smears prepared from the fetal stomach content and from the cut surface of the lungs stained by Stamp's method. No viruses were identified.
In the immunohistochemical examination the R. equi–specific IgG and the monoclonal antibody produced against the VapA (15–17 kd) antigens gave a strong reaction and detected the pathogen (Fig. 3). Bacteria were mainly contained within the cytoplasm of pulmonary macrophages, multinucleated Langhans giant cells, and less often, in neutrophils within alveoli and interstitium. Positive-isolated macrophages were occasionally identified within the spleen and in the liver of fetus No. 1. R. equi could also be detected by M. bovis–specific immunoglobulin, which have been demonstrated also by others. 8 However, the staining was less intense and was accompanied by a slight background staining using the M. bovis–specific reagent. All the other immunohistochemical stains gave negative results.
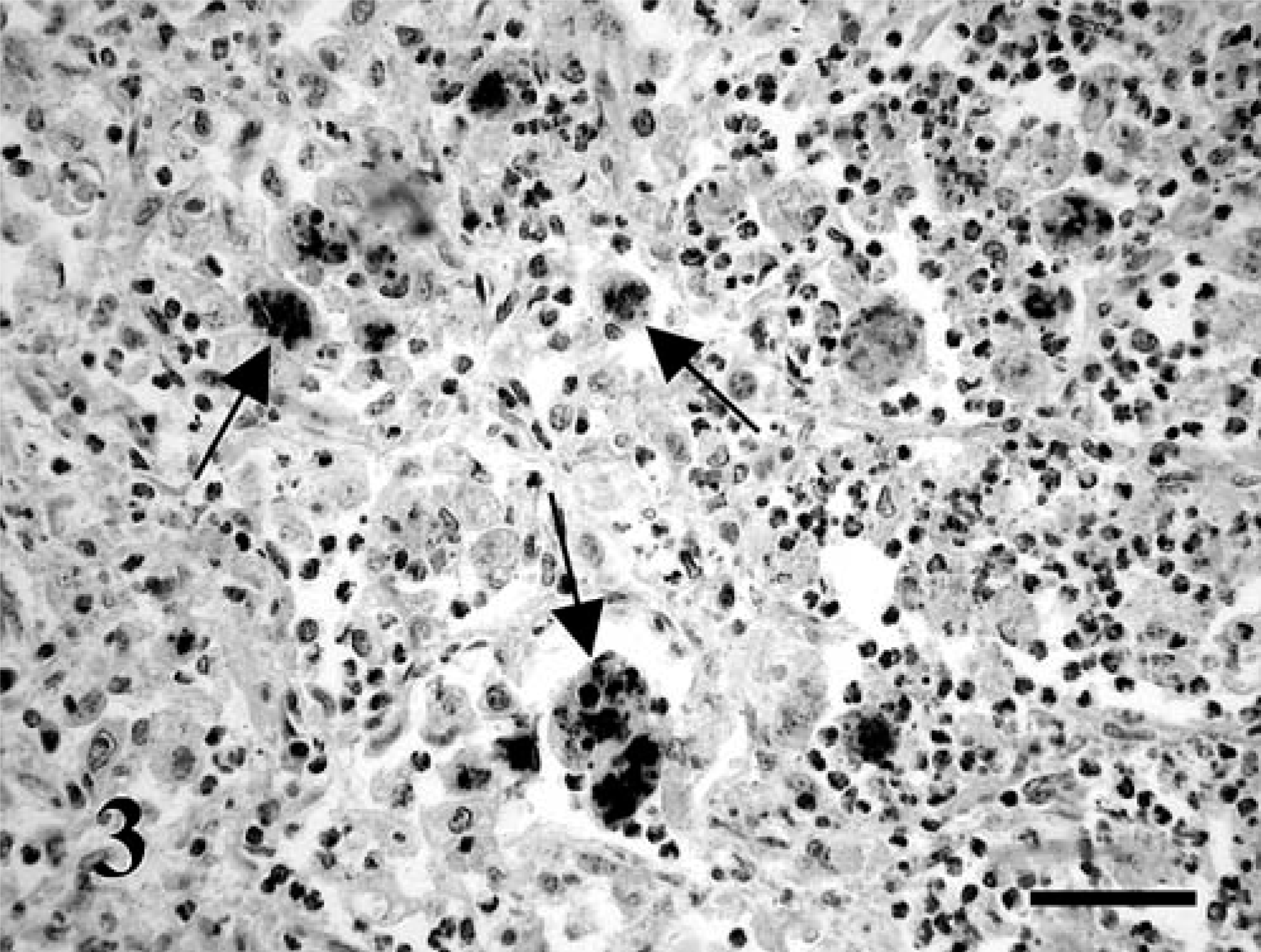
Lung; equine fetus No. 2. R. equi located in the cytoplasm of macrophages (arrows). VapA (15–17 kd)–specific murine monoclonal antibody, labeled streptavidin–biotin method, counterstaining with Mayer's hematoxylin. Bar = 60 µm.
Gross lesions were suggestive of fetal pneumonia, and histologic findings were consistent with R. equi infection of the lungs, confirmed by bacterial cultures and immunohistochemistry. There was no evidence of other etiologic agents. Because the R. equi specific polyclonal IgG, presumably, detects both the virulent and the avirulent strains, 7 in this study, an attempt was made to detect the VapA (15–17 kd) antigens immunohistochemically in samples that had been embedded in paraffin 5 and 10 years previously and demonstrated that abortion had been caused by virulent R. equi strains in both cases.
In foals, R. equi infection usually has a chronic, insidious course, accompanied by extensive necrosis and abscess formation in the lungs. 5 The equine fetus can mount an inflammatory reaction in response to bacterial infections after the 6th month of gestation. 10 Because the immune system of the fetus continues to be immature even beyond that time, the nature of inflammatory changes may differ from those seen in an immunologically mature horse. This may be the explanation of the absence of extensive necrosis and abscess formation in the present report and earlier reports. 6, 9 In one previously described case, numerous small inflammatory-necrotic foci were observed in the lungs, as in fetus No. 2. The authors characterized that finding as an acute change. 6 In contrast to that observation, our two fetuses seemed to be rather chronically infected with R. equi, showing mild or moderate fibrosis in the lungs.
We have to rely on hypothesis as to the origin and route of infection of the two examined fetuses. Bacteria most commonly enter the equine uterus through the cervix. 17 It is known that R. equi seldom resides in the genital tracts of mares. 1, 2, 3 In fetus No. 1, the pathogen could not be cultured from the mare's vagina on the 7th day after abortion, indicating that the bacterium did not reside in the vagina as a commensal organism. This, however, does not preclude the possibility of an earlier transient infection of the genital tract. Another, much less-common pattern of fetal infection is when bacteria infect the fetus via the blood stream from a focus of infection present in an organ of the mare. 17 By immunohistochemical examination, R. equi was detected in the largest quantity in the lungs. This suggests that the pathogen entered the amniotic fluid from the contaminated fetal membrane, and then the fetus contracted infection by normal breathing movements or by swallowing the amniotic fluid. In fetus No. 1, this assumption is supported by the detection of R. equi bacteria in cultures and in smears prepared from the fetal stomach content. Others have reported a similar route of fetal infection in an aborted equine fetus. 9
During the 11 years that elapsed between 1990 and 2000, a total of 343 aborted equine fetuses were subjected to diagnostic examination at the Central Veterinary Institute, and R. equi was cultured from only the two fetuses presented in this article (0.6%). In agreement with the literature, this indicates that R. equi rarely plays a role in equine abortion. On the basis of the present findings, it has been established that, like the disease of foals, abortion in mares can be caused by virulent R. equi strains carrying VapA (15–17 kd) antigens.
Footnotes
Acknowledgements
We are grateful to Dr. Pamela A. Wilkins, University of Pennsylvania, New Bolton Center, for previous suggestions, and Edit Karner and Szilvia Lakosi Central Veterinary Institute, Budapest for technical assistance.
