Abstract
Lameness in sows is reported as the most frequent cause of early culling from commercial farms and results in reduced productivity, economic losses, and a negative impact on animal welfare. Osteochondrosis was reported as the leading cause of lameness in North American sows and, although more recent European studies report infectious arthritis as the leading cause, lameness in US production facilities using group housing for gestating sows has not yet been evaluated. This study’s aim was to characterize lesions associated with lameness in the appendicular musculoskeletal system of 26 sows euthanized for lameness using pathologic, radiologic, and microbiologic analyses. Of 178 total lesions, infectious lesions were most common (54%), predominated in distal limb segments (ie, at or distal to carpi and tarsi) and more often correlated with the clinically lame limb, whereas osteochondrosis and degenerative osteoarthritis predominated in proximal limb segments (ie, at or proximal to cubital and stifle joints) and rarely correlated with the clinically lame limb. The location and characteristics of infectious lesions, including mixed bacterial growth isolated from 22/22 orthopedic sites representing 19 sows with Trueperella pyogenes isolated in 16/22 (73%) of samples, suggest an etiologic component involving trauma. Radiography had a 70.6% sensitivity and 93.9% specificity for detecting infectious lesions affecting tarsocrural, antebrachiocarpal, and digital (ie, claw) regions combined. The frequency, type, and location of infectious lesions identified in this cohort of sows euthanized for lameness differ from previous reports, indicating the need for further investigation of the etiopathogenesis, earlier detection methods, and prevention.
Keywords
Sow lameness is a welfare concern and an economic concern for swine farmers. As farmers transition away from single-housing sows in gestation stalls to group-housing sows in pens that allow more mobility and social interaction, but may increase their risk of injury, there is an increased importance for early detection, assessment, and treatment of lameness. The prevalence of lameness in sows housed in group pens varies by study and is frequently above 20%.4,24,31 Advanced lameness leads to involuntary removal of the sow through culling or euthanasia, thereby decreasing lifespan and lifetime productivity, and incurring cost to farmers. 2 One survey of sow removal records identified lameness as the cause of 10% of sow removals from herds with 55% of those sows culled immediately and an additional 40% of these eventually euthanized on the farm. 3
To undertake effective prevention and treatment strategies, it is necessary to understand the etiologies and pathogenesis of lameness. Studies characterizing appendicular orthopedic lesions associated with lameness in sows from the United States and Canada are limited. One Canadian study of 50 sows culled for lameness identified osteochondrosis as the most prevalent postmortem lesion, identified within 50% of lame sows; followed by infectious arthritis, identified within 22% of sows; and “foot lameness” defined by the presence of hoof cracks, sole ulcers, or injured paradigits (dewclaws) with associated external soft tissue swelling, identified within 20% of sows. 8 However, soft tissues, bones, and synovial structures (joints, tendon sheaths, or bursae) within the digital segments associated with the hoof were not included in the examination. Moreover, lesions identified as infectious arthritis were limited to gross evaluation of intra-articular structures without microbial cultures or further evaluation of adjacent bones (ie, epiphyses or metaphyseal physes flanking joint inflammation), and histologic evaluations were limited to the medial condyles of the femur and distal humeri to confirm and characterize lesions associated with osteochondrosis. Although a more recent study of 107 autopsied sows representing one US herd reported infectious polyarthritis as the primary cause for euthanasia in 18/49 culled sows, the anatomic site(s) of the arthritis were not specified, nor were histologic or microbiologic evaluations of musculoskeletal lesions pursued. 26 Another US study performed in a large slaughterhouse cohort of 3158 sows identified 10% of sows having shoulder and claw abscesses associated with cutaneous ulcers and fistulae compared with 17% having visceral inflammatory lesions, but again, characterization of the musculoskeletal lesions was limited to external evaluation without further exploration of underlying structures. 20 One European study showed osteochondrosis often associated with degenerative osteoarthritis as the predominant lesions with osteochondrosis affecting 48% of 117 euthanized sows, as well as frequent claw lesions associated with clinically discernible leg weakness. In this study, joint evaluations comprised unilateral gross dissections with sagittal sectioning limited to the proximal limb segments (ie, glenohumeral, cubital, coxofemoral, and femorotibial), and claw evaluations were limited to external features. Although more recent European studies have report infectious arthritis as a frequent, 1 if not predominant cause of lameness, affecting 17% to 36% of sows examined,9,19 dissections were also limited to gross evaluation of proximal limb joints with histology limited to osteochondrosis lesions. 9 Thus, systematic and complete characterization of appendicular orthopedic lesions in breeding sows is warranted. Moreover, as only one of the European studies surveyed a more modern management operation where sows were housed in groups ranging from 9 to 15, or up to 50 sows, depending on the farm and the stage of gestation, 9 more studies are needed, especially as sow farms are transitioning to groups of sows in pens as the predominant method of housing gestating animals.
The primary objective of this descriptive study was to systematically identify and rigorously characterize lesions within the appendicular musculoskeletal system of group-housed breeding sows that were euthanized for lameness by correlating gross, histologic, microbiologic, and radiologic analyses. Given the previous research in sows culled for lameness in North America, we hypothesized that noninfectious lesions such as osteochondrosis and degenerative osteoarthritis would predominate in our regional cohort of breeding sows group-housed in pens and euthanized for lameness. A secondary objective was that information pertaining to the distribution, classification, and severity of orthopedic lesions identified in this study would provide additional insight into the etiopathogenesis of lameness affecting group-housed sows to inform prognosis or treatment of live animals.
Materials and Methods
A convenience sample of sow farms within a 200-mile radius of the University of Pennsylvania, School of Veterinary Medicine, were enrolled in the study if pregnant sows were housed using pen gestation (ie, group housing) and agreed to donate the appendicular limbs of sows euthanized for lameness based on industry standardized protocols in the Pork Quality Assurance Plus program (doi.org/10.31274/safepork-180809-18), during the study period between May 2018 and October 2019. Limitations pertaining to transport of entire carcasses limited pathologic evaluation to the locomotor system (all 4 limbs). All sows had a history of non-weight-bearing lameness in at least one limb. The age/parity of the animal was provided, and both the systemic health of the animal and location of lameness (ie, affected limb or limbs) were confirmed by the herd veterinarian or barn staff by antemortem physical examination (Supplemental Table S1). Sows were euthanized at the farm and all 4 limbs were harvested by either disarticulation of the coxofemoral joint (ie, hind limbs) or excision of the scapulae from the muscular attachment to the body wall (ie, front limbs), leaving the glenohumeral joints intact. Limbs were transported in a chilled container to the University of Pennsylvania, New Bolton Center, where they underwent radiographic and pathologic evaluation of soft tissues and bones, and bacteriology within 24 to 36 hours of their delivery to the postmortem facility.
Radiography
Prior to gross dissection, digital radiographs including 2 orthogonal views (1 dorsopalmar or dorsoplantar exposure and 1 lateral exposure) of the distal front and hind limbs of 26 sows were obtained using a digital radiography system (Vet Rocket X1; Vet Rocket, Santa Clara, California) with a wireless digital detector (CXDI-701C; Canon, Tokyo). A distal limb was defined as the manus or pes (including and distal to the antebrachial carpal joint in front limbs and the tarsocrural joint in the hind limbs, respectively). The hind limbs of 2 sows were not radiographed due to inadvertent dissection and disarticulation prior to radiography. An additional 3 sows had the limbs disarticulated at the radiocarpal joint prior to radiography and therefore radiocarpal joints were not scored on those sows and for 2 of those 3 sows; the tarsocrural joint was disarticulated before radiography and not scored. Anatomic locations segregated by synovial joints included within distal limb segments (carpal, tarsal, metacarpophalangeal, metatarsophalangeal, proximal, and distal interphalangeal joints, including any persistent physes) were evaluated and scored for the presence (score = 1) or absence (score = 0) of each category of lesion: infectious (INF), characterized by ill-defined subchondral or physeal osteolysis and joint space widening, ill-defined periarticular osseous proliferation or periosteal reaction, severe regional soft tissue swelling or intra-articular effusion (INF, score = 1); osteochondrosis, characterized by subchondral sclerosis and flattening or free mineral body with accompanying intra-articular effusion (OC, score = 1); and degenerative osteoarthritis, characterized by smoothly marginated periarticular osseous proliferation, subchondral sclerosis with or without accompanying joint space narrowing (OA, score = 1) (Supplemental Table S2). Radiographs were evaluated individually and randomly without knowledge of findings at other sites and scored by a board-certified radiologist (KBW) blinded to the identity or antemortem clinical history of the sow.
Pathology and Microbiology
Gross and histopathological evaluations were performed by a board-certified pathologist (JBE). Gross evaluation included soft tissue dissection and disarticulation of all synovial structures (ie, diarthrodial joints, tendon sheaths, and bursae) within all 4 limbs, including the femoral portion of the coxofemoral joint, stifle, and tarsocrural joints in the proximal portions of the hind limbs, and glenohumeral, cubital, and carpal joints in the proximal portions of the front limbs, as well as the manus and pes as described for radiology. To facilitate disarticulation and evaluation of synovial structures distal to the antebrachiocarpal and tarsocrural joints (ie, distal limbs), sagittal sections that extended from the hoof of each digit through the proximal row of carpal/tarsal bones were performed with a bandsaw. Macroscopically, normal and abnormal structures were photodocumented. All gross lesions involving soft tissues (ie, skin/subcutis, muscle, tendon, synovial structures, and coronary band/lamellae/sole of the digits) and hard tissues (ie, bone with or without overlying articular cartilage), as well as an anatomically matched subset of macroscopically normal structures, were further evaluated histologically, incorporating targeted bandsaw subsections performed in a plane orthogonal to the articular surface along sagittal/parasagittal and/or coronal planes as previously described.11,30 Representative samples of soft and hard tissue lesions were fixed in 10% formalin prior to trimming and routine processing, including bone decalcification in an aerated and refreshed 15% formic acid solution for days to weeks until the bones were soft enough for further processing. Lesions were classified by regional location (eg, front versus hind limb, proximal versus distal limb), and specific anatomical location (eg, tarsocrural joint, distal tibial epiphysis, digital tendon sheath, and subcutis), and further classified by the 1 of 4 processes based on macroscopic criteria described below that were confirmed with histology: infectious (INF), osteochondrosis or osteochondrosis dissecans (OC), degenerative osteoarthritis (OA), and other (eg, traumatic fracture). Criteria for defining INF lesions were expanded from those described in the work by Dewey et al, to incorporate lesions present within bones as well as tendon sheaths, bursae, and joints, and included presence of fibrinopurulent exudate (ie, cloudy to opaque, yellow to green-gray effusion containing strands or mats of fibrin) expanding and coating synovial structures or effacing the cortical or trabecular architecture of bones or physes. INF synovial structures often had concurrent hypertrophied, hyperemic synovial membranes with expansion of the joint capsule by fibrosis. Criteria for defining INF lesions in the subcutis or muscle included presence of fibrinopurulent exudate with or without fibrous encapsulation or extension as sheets through edematous myofascial planes. Criteria for defining OC lesions were the same as previously described.8,11,30 Criteria for defining OA lesions were expanded from Dewey et al, to include clear to orange-tinged synovial effusion of reduced viscosity with synovial hypertrophy and hyperemia; focal to diffuse cartilage thinning with yellowing or softening of the matrix with or without discrete erosions to ulcers, osteophytes, or capsular fibrosis. Although many joints with OC lesions had concurrent lesions of OA, only joints without gross or histologic evidence of OC were classified as OA.
For each anatomic location and using a semi-quantitative grading scale from 0 to 3, representative lesions of each type (INF, OC, OA) were further classified by severity based on the following gross criteria confirmed histologically (Supplemental Table S1). INF: 0—lesions not present; 1—lesions limited to nonsynovial soft tissues (ie, cellulitis, myofasciitis, subcutaneous abscesses); 2—lesions involved synovial structures (ie, septic arthritis, bursitis, or tenosynovitis); 3—lesions involved bone (ie, septic physitis or osteomyelitis). OC: 0—lesions not present; 1—osteochondrosis manifesta comprising mild focal cartilage surface defects (ie, depressions, infolding, plaques, or surface distortion); 2—osteochondrosis manifesta comprising multifocal or regionally extensive cartilage surface defects; 3—osteochondrosis dissecans (ie, presence of cartilage fissures or flaps extending to subchondral bone). OA: 0—lesions not present; 1—focal shallow cartilage erosions or fissures with synovial edema and hyperemia, and mild clear-yellow effusion with slightly reduced viscosity; 2—multifocal to regionally extensive articular cartilage erosions with mild synovial hypertrophy/capsular fibrosis, mild osteophytosis, and moderate yellow to orange effusion with reduced viscosity; 3—severe degenerative OA characterized by focal, multifocal to regionally extensive cartilage ulcers with synovial villous hypertrophy/capsular fibrosis, pronounced osteophytosis, and moderate to severe yellow to orange effusion with reduced viscosity.
Aerobic and anaerobic cultures obtained in conjunction with postmortem dissections were collected and transported using the BBL CultureSwab Plus cotton-tipped single-swab system (Copan Italia S.p.A., Brescia). Cultures representing a subset of lesions classified as INF (ie, sites containing fibrinopurulent material) were obtained using standard postmortem microbiologic sampling techniques that targeted INF sites uncontaminated by dissection instruments, or INF sites where superficial or previously contaminated surfaces were heat-seared immediately prior to obtaining the culture through an incision made with a sterile scalpel blade that extended into the underlying uncontaminated deeper tissue. Microbiology samples were submitted to a diagnostic lab accredited by the American Association of Veterinary Laboratory Diagnosticians (AAVLD). For isolation of aerobic bacteria, cultures were plated on tryptic soy agar (TSA) with 5% sheep’s blood, Columbia CNA agar, and MacConkey (MAC) agar. Plates were incubated at 35°C ± 1°C for 48 hours. TSA plates were incubated in 7% carbon dioxide–enriched environment and the CNA and MAC were incubated in standard oxygen environment. Anaerobic cultures were inoculated onto Brucella agar with 5% sheep’s blood, anaerobic CDC PEA (phenylethyl alcohol) agar, LKV (Laked Brucella Blood Agar with Kanamycin and Vancomycin) agar and Bacteroides Bile Esculin Agar. Anaerobic plates were placed into an anaerobic chamber with BD GasPak EZ anaerobic container system pouch (Becton, Dickinson and Company, Sparks, Maryland) and incubated at 35°C ± 1°C for 7 days. Isolated colonies were identified using the Bruker Maldi-TOF system (Bruker Co., Billerica, Massachusetts). Microbiologic results were correlated with histopathology including Gram stains (Supplemental Table S3).
Statistics
Statistical analyses were performed with Stata IC v.15 software (StataCorp LLC, College Station, Texas). The anatomic location was treated as the statistical unit. Data were examined for normality using the Shapiro-Wilk test. All data were nonparametric. The number of affected joints was calculated by summing the presence or absence of a lesion in each of the examined anatomical locations (Supplemental Tables S2 and S4). Two by two tables with front versus hind and each lesion type, as well as proximal versus distal limb locations and each lesion type (ie, INF, OC, and OA), were made and a chi-square test of association was applied to look for the relationships between location and lesion type. Significance for odds ratios (ORs) for lameness and lesion types was calculated using Fisher exact test. Given the rarity of OA (n = 2) and OC lesions (n = 4) identified in the distal limb, the test characteristics for radiography were limited to detection of septic lesions (n = 23). The lesions for all the joints in the distal limb were combined and the diagnosis of a lesion on histopathological examination was treated as the gold standard for comparison with detection by radiography.
Results
Lameness Correlation of Infectious Versus Noninfectious Lesions
A total of 104 limbs from 26 sows (average parity = 1.46; median parity = 1 ± 1.0; parity range = 0–6) representing 5 farms (Supplemental Table S1) were fully dissected; comprising 416 anatomic locations. Sows were all mixed breed with commercially available genetics. All farms, including 1 internal facility and 5 external facilities, used an electronic sow feeding system and housed sows on fully slatted concrete flooring. Farm inventory ranged from 230 sows to 5000 sows (median = 1625) with pen inventory ranging from 70 to 290 gestating sows. Prior to being euthanized, 2 sows (8%) were reported as nonambulatory and recumbent, 1 sow (4%) was reported as nonambulatory in both hind limbs, and the remaining 23 sows (88%) were reported as having unilateral non-weight-bearing lameness. Of the 26 sows, 24 (92%) had INF orthopedic lesions determined as the primary cause of the clinical lameness with only 2/26 sows (8%) having their primary cause of lameness attributable to non-INF orthopedic lesions (Supplemental Table S1). One sow recorded as lame in all 4 limbs had a combination of severe (grade 3) INF lesions involving 3 limbs with a mild (grade 1) OC lesion involving a single limb. Although pain disproportionate to the mild OC lesion or a nonorthopedic cause for the lameness could not be ruled out, it is likely the INF lesions were the primary cause of lameness, potentially producing gait abnormalities manifesting in all 4 limbs. In the 2 sows with non-INF lesions determined to be the primary cause of lameness, 1 had bilateral coxofemoral luxation associated with grade 3 OC lesions (ie, bilateral femoral head epiphysiolysis) and the other had a complete, displaced comminuted humeral fracture. Although non-INF lesions were identified in 17/26 sows (65%) with osteochondrosis OC identified in 13/26 sow (50%) and OA identified in 16/26 sows (62%), all other OC and OA lesions were designated as nonprimary causes of clinical lameness due to the preponderance and severity of concurrent INF lesions identified in one or multiple limbs of this cohort, aside from the bilateral proximal femoral epiphysiolysis and humeral fracture.
As there were no indications of prior or concurrent visceral or systemic disease within the production records of these sows, only 4/26 (15%) sows received complete postmortem examinations, all of which were submitted from our internal swine facility. One sow euthanized for severe right forelimb lameness had bacterial pulmonary valvular endocarditis, multifocal disseminated suppurative pneumonia with intralesional gram-positive cocci, and chronic fibrinosuppurative arthritis of the right radiocarpal joint with gram-positive cocci associated with Splendore-Hoeppli reaction. Although bacteria (Escherichia coli and Streptococcus pyogenes) were only isolated from aerobic cultures of the valvular lesion, the presence of bacteria within pulmonary and chronic synovial lesions having morphology compatible with Streptococcus spp, suggests an embolic etiopathogenesis. The other 3 sows lacked gross or histologic visceral lesions.
Prevalence and Anatomic Distribution of Infectious Lesions
Of 416 anatomic locations assessed, INF was the most common lesion type, affecting 43/416 (10.43%) locations and 37/104 (36%) limbs, followed by OA and OC lesions (Table 1). Most INF lesions were grade 3 (23 lesions identified within 104 limbs), involving bone as well as synovial and periarticular soft tissue structures. In comparison, 7 grade 3 OC lesions and 3 grade 3 OA lesions were identified within 106 limbs. The odds of an INF lesion corresponding to the limb identified as lame on antemortem examination was significantly higher than for the non-lame limbs (OR = 2.7; confidence interval [CI] = 1.05–6.96; P < .05). The odds of identifying an OC (OR=1.43; CI = 0.45–4.27; P > .05) or OA (OR=1.0; CI = 0.30–3.01; P > .05) lesion in the lame limb was not significantly higher than for the non-lame limbs.
Total and percent (parentheses) of infectious, osteochondrosis, and degenerative osteoarthritis lesions grossly categorized by semi-quantitative lesions severity scores (0–3) within each limb of 26 lame sows.
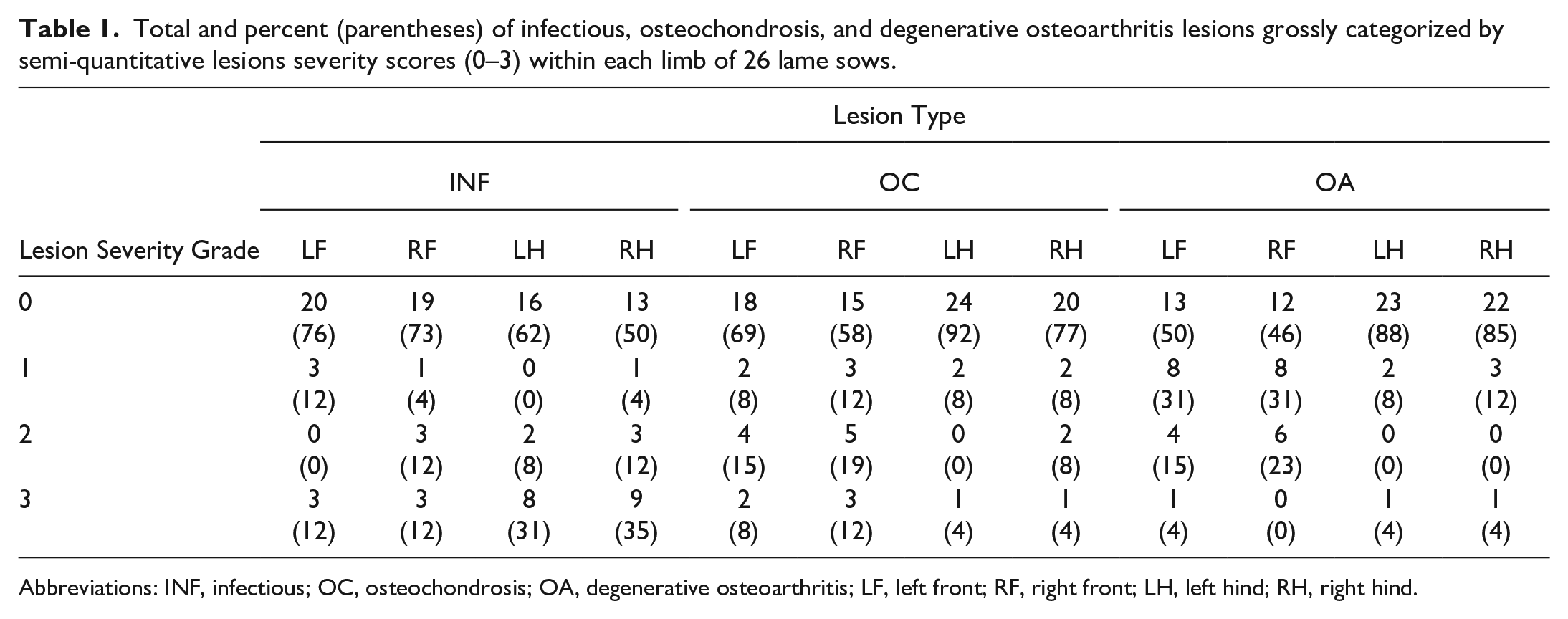
Abbreviations: INF, infectious; OC, osteochondrosis; OA, degenerative osteoarthritis; LF, left front; RF, right front; LH, left hind; RH, right hind.
The distribution of INF, OC, and OA lesions varied by anatomic location (Table 2). INF lesions were more common in distal limbs (81%) than proximal limbs (19%) (P < .01) with similar distribution for front (44%) and hind (56%) limbs (P = .42), but a high prevalence of INF lesions localized to the hind digits (79% affected) compared with the front digits (13% affected). Most sows (14/26; 54%) had INF lesions involving more than one limb, including 6/26 (23%) and 3/26 (12%) having INF lesions in 3 or all 4 limbs, respectively (Supplemental Table S1).
Numbers and percentages of infectious, osteochondrosis and degenerative osteoarthritis lesions grossly identified and confirmed histologically per anatomic region in 26 lame sows.
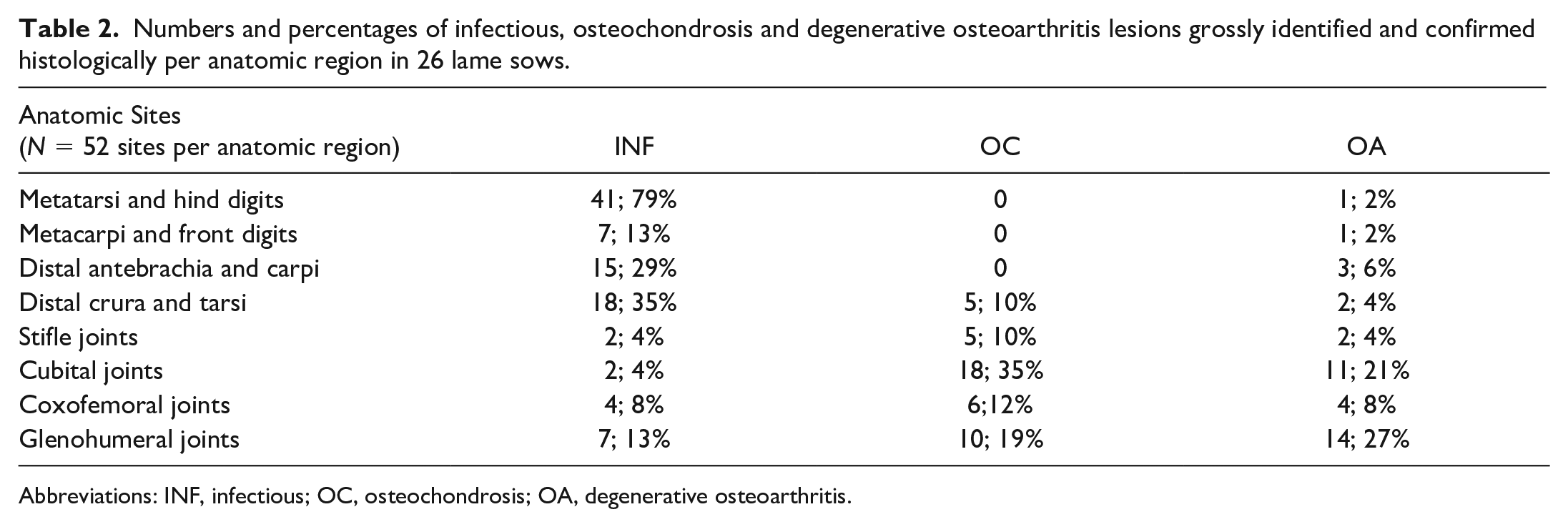
Abbreviations: INF, infectious; OC, osteochondrosis; OA, degenerative osteoarthritis.
Radiologic, Pathologic, and Microbiologic Features of Infectious Lesions
Radiographically, INF lesions were characterized by soft tissue swelling and widened joint space with irregular, ill-defined subchondral osteolysis. INF lesions of the digits were most common (Supplemental Table S2), often involving the distal interphalangeal joints (Fig. 1), and occasionally metacarpo- and metatarsophalangeal joints. Grossly, lesions involving the digits frequently had a fistulous tract extending from the cutaneous surface into the affected joint with concurrent, regionally extensive necrosuppurative cellulitis, and occasionally tenosynovitis. In sows with involvement of the distal interphalangeal joint, the fistulous tract was located at or immediately proximal to the coronary band (Fig. 2). These lesions were also associated with soft tissue swelling and ulceration of the sole and hoof cracks. There was good correlation between radiographic and postmortem INF lesions. While INF lesions identified within distal limbs segments on postmortem examination had an 18% prevalence, radiography had a 73.5% sensitivity (95% CI = 55.6%–88.1%) and 91.6% specificity (95% CI = 86.1%–95.5%) for detecting INF lesions within the combined regions of the distal limb segments (ie, manus and pes), with positive and negative predictive values at 72.7% (95% CI = 54.5%–86.7%) and 93.2% (95% CI = 87.9%–96.7%), respectively. INF lesions were grossly characterized by necrotizing and fibrinosuppurative arthritis, often with concurrent osteomyelitis (Fig. 2). Histologically, articular cartilage and subchondral bone were effaced by swaths of neutrophils, fibrin, and cell debris (Fig. 3) with marked active osteoclastic osteolysis, necrotic bone islands, and intralesional bacteria (Fig. 4).
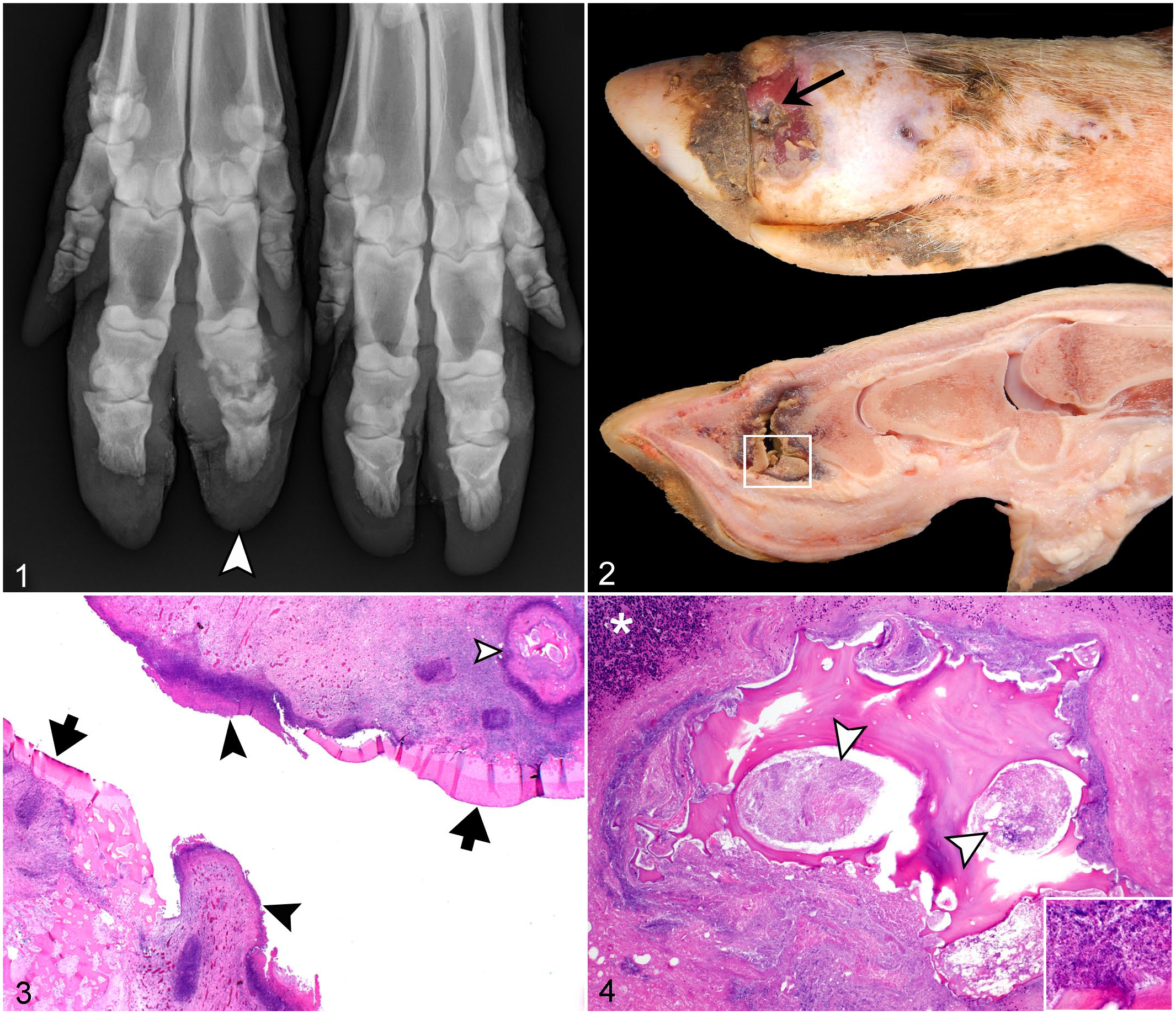
Grade 3 infectious (INF) distal interphalangeal arthritis with flanking osteomyelitis, right manus, sow. HE, hematoxylin and eosin.
A high prevalence of INF lesions located within antebrachiocarpal and tarsocrural regions had bone involvement including extensive osteolysis within adjacent epiphyses and physes (n = 16 sows) (Fig. 5). These lesions were associated with extensive soft tissue swelling (Figs. 5, 6) that corresponded to severe necrosuppurative cellulitis, myofasciitis, tenosynovitis, and arthrosynovitis (Fig. 7), often including cavities of malodorous, friable, gray-green to yellow tan exudate that distorted articular and periarticular tissues. Similar to INF lesions involving the claws, epiphyseal subchondral bone was often distorted by extensive bone lysis (Fig. 8) in association with necrosis and suppurative inflammation that effaced the articular cartilage and subchondral bone (Fig. 9) and sometimes extended into metaphyseal regions. A subset of sows (n = 5) had physitis or epiphysitis of the distal tibia/fibula, radius/ulna, or distal metatarsus without concurrent involvement of the antebrachiocarpal or tarsocrural joints. Three of these sows had concurrent INF lesions involving the ipsilateral claw. One sow had a concurrent INF lesion involving the claw of a different limb, and 2 sows had bilaterally symmetrical INF lesions involving their distal ulnas (Figs. 10–14). Although these 2 sows had minimal swelling at the level of the carpus, radiographically, ill-defined osteolysis caused expansion of the distal ulnar physes, corresponded to gross and histologic lesions of severe necrosuppurative bacterial physitis. In this cohort, no septic lesions were identified within the middle carpal, carpometacarpal, intertarsal, or tarsometatarsal joints.
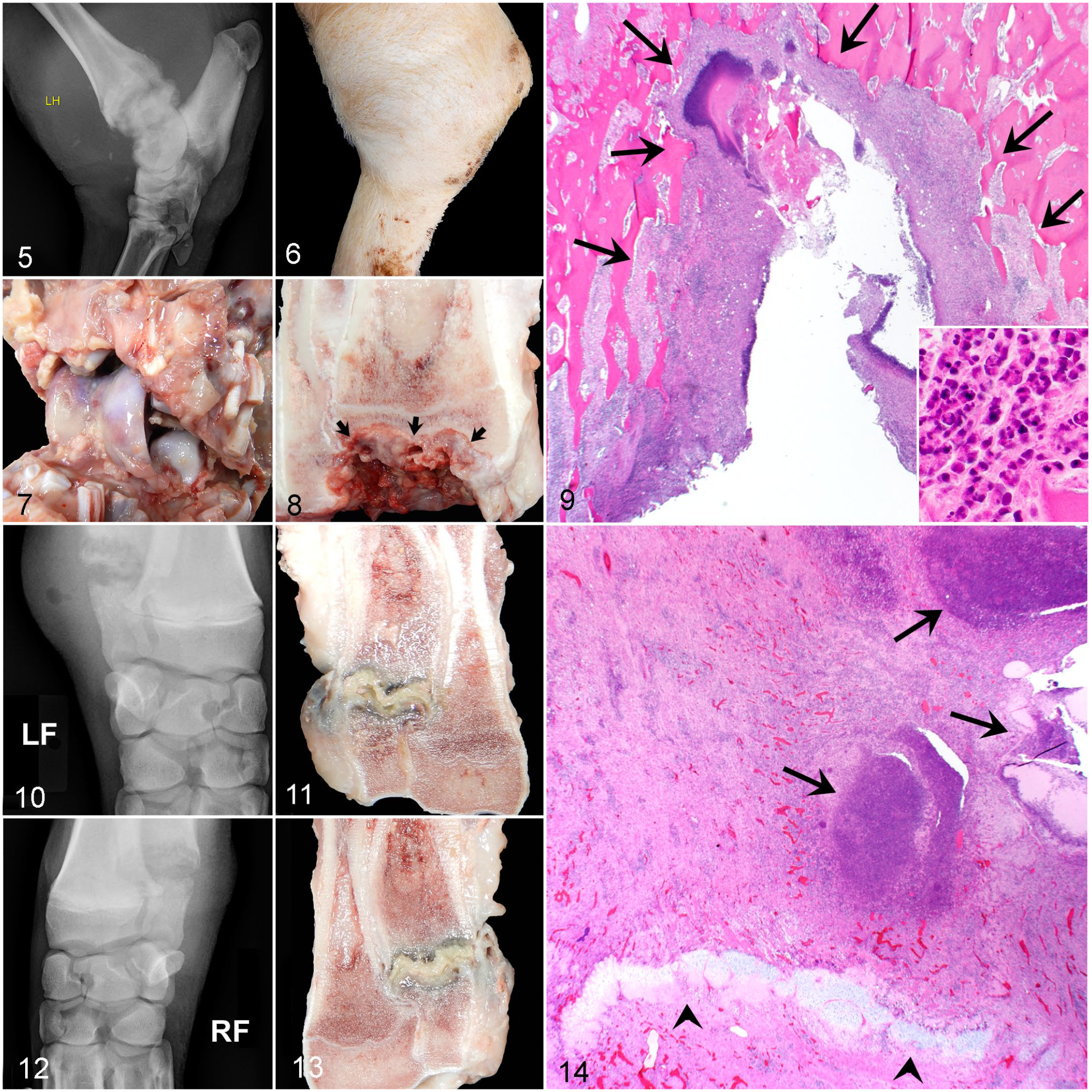
Grade 3 infectious (INF) fibrinosuppurative arthritis and osteomyelitis, pes and manus, sow. HE, hematoxylin and eosin.
Bacterial cultures of INF lesions (Supplemental Tables S3) included 22 aerobic and 18 anaerobic cultures from 22 sites from the appendicular skeleton of 18 sows. Mixed bacterial growth was isolated from aerobic cultures of 18/22 (82%) orthopedic sites with only 2 samples producing single isolates. Trueperella pyogenes was the predominant isolate in 16/22 (73%) of samples. While the second most common aerobic isolate was E. coli, identified in 12/22 (56%) of samples, it was recovered as a component of mixed growth and classified as “light growth” in 10/12 (83%) of these isolates. Streptococcus suis was not isolated in any of the INF orthopedic sites sampled in this study. Of the 18 anaerobic cultures representing 16 sows, 5/18 (28%) yielded mixed growth, 5/18 (28%) yielded single isolates, and 8/18 (44%) produced no growth. Fusobacterium necrophorum was the most common isolate, comprising 6/10 (60%) samples yielding pure or mixed growth. Gram stains performed on histologic sections of 22 INF lesions identified bacteria that correlated with expected morphology of bacteria isolated from cultures in 16 (73%) samples; however, bacteria could not be identified by Gram or hematoxylin and eosin (HE) stains in 4 samples, whereas in 2 samples, bacteria identified on Gram stains did not match the expected morphology based on cultures. Of note, in the 6 samples without bacteria identified, 4 of these sows had a history of antimicrobial therapy, which may have affected bacterial populations within these lesions.
Prevalence, Anatomic Distribution, Pathologic Features, and Radiologic Detection of Osteochondrosis and Osteoarthritis Lesions
For non-INF lesions (Figs. 15–22), OC lesions were identified in 29/416 (6.97%) locations and 22/104 (21%) limbs. OA lesions were identified in 26/416 (6.25%) locations and 21/104 (20%) limbs. Thirteen sows (50%) had OC lesions identified within at least one of their appendicular joints, but only 3/26 sows (12%) had grossly evident lesions characterized as severe OC, whereas 10/26 sows (38%) had mild to moderate OC lesions. OC occurred more frequently in the proximal limbs (86%) than distal limbs (14%) (P < .001). Although OC occurred more commonly in the front limbs (62%) compared with hind limbs (38%), this difference was not statistically significant (P = 0.18). OC was most frequently identified in the cubital joints, comprising 41% of all OC lesions identified, and affecting 35% of all cubital joints evaluated. OC lesions were identified in 19% of glenohumeral, 12% of coxofemoral, and 10% of all stifle and tarsocrural joints evaluated. OC lesions were bilateral in 6/13 (46%) with 12/13 (92%) of sows with OC lesions having concurrent OA lesions within the affected joints (Figs. 15, 16). Gross lesions ranged from mild focal distortion of the articular cartilage surface (ie, irregular roughening, thickening, undulation) to more severe, regionally extensive lesions, including retention of cartilage beyond the articular-epiphyseal complex and cartilage infolding (ie, osteochondrosis manifesta) with the formation of fissures that dissected to the articular surface (ie, osteochondrosis dissecans) (Fig. 17). Histologically, most severe cartilage lesions were associated with subchondral lysis, necrosis, or collapse (Fig. 18). Only 1 sow had lameness attributed to OC due to physeal dysplasia of the proximal femurs resulting in bilateral epiphysiolysis.
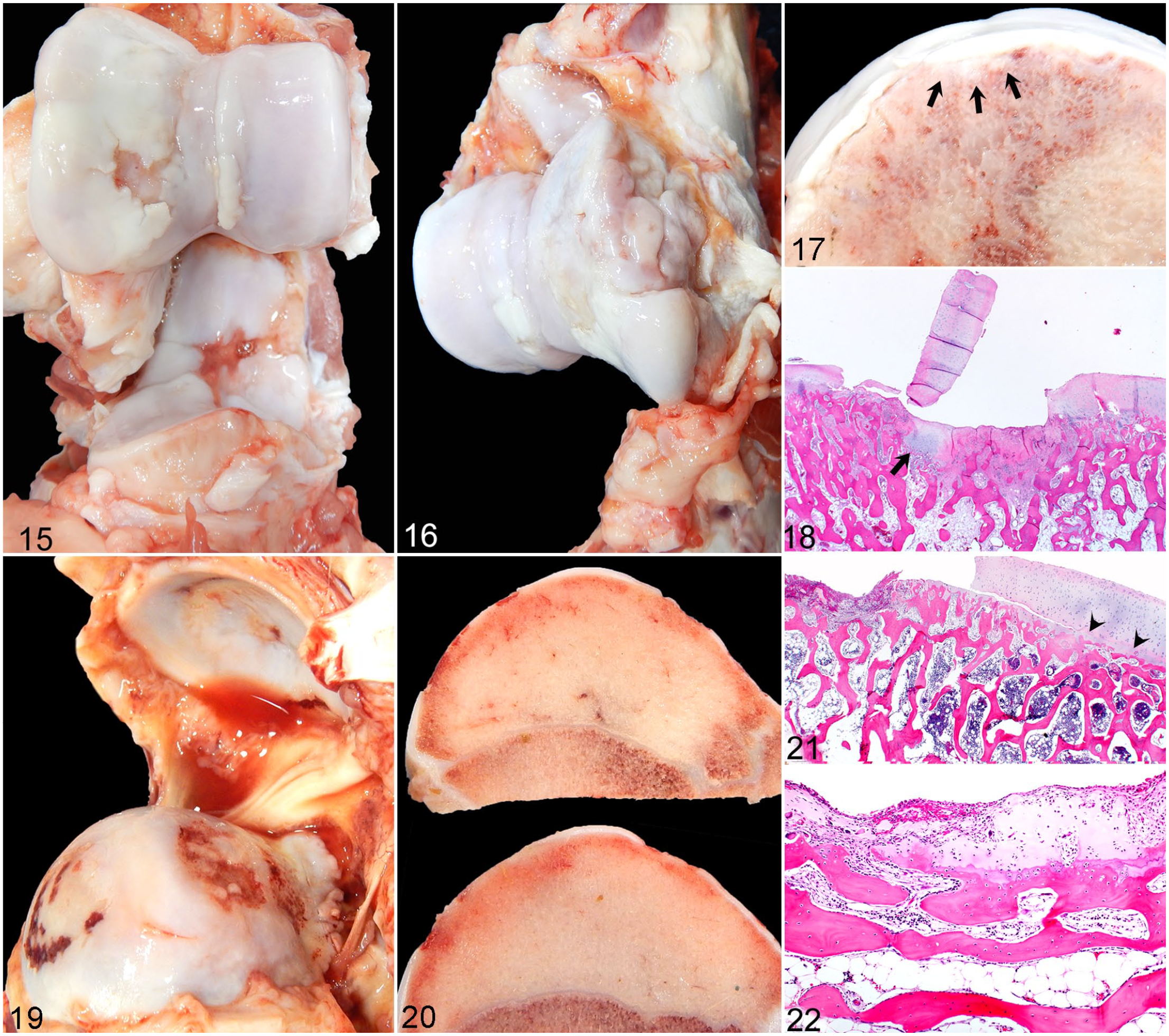
Grade 3 OC and OA lesions, cubital and glenohumeral joints, sows. Both sows manifested hind limb lameness due to septic processes. HE, hematoxylin and eosin.
OA lesions were identified in 16/26 sows (62%). The distribution of OA was similar to OC with the majority of OA lesions identified in the front limbs (77%) compared with hind limbs (23%) (P < .01), and a predilection for proximal limb joints (88%) compared with distal limb joints (12%) (P < .001). OA was most frequently identified in the glenohumeral joints, comprising 37% of all OA lesions identified and affecting 30% of all glenohumeral joints examined. OA was identified concurrently with OC lesions in 12 sows and interpreted as a secondary process. Only 4 sows, ranging in age from 10 months to 5 years of age, had OA lesions of varying severity that ostensibly occurred independently of OC lesions (ie, OC score for all 4 limbs = 0, confirmed histologically). Three out of 4 of these sows had glenohumeral joint involvement with 2 sows having unilateral involvement (severity range = 2–3) and 1 sow with bilateral involvement (severity = 1). One sow had bilateral cubital joint OA (severity = 1). OA severity also varied for sows having OC lesions identified within other joints, with the majority of sows having mild to moderate OA (N = 7 and 8, respectively) and a single sow having severe OA. None of the OA lesions were identified within limb(s) designated as clinically lame as all sows with OA lesions had a septic process affecting other sites associated with the clinically lame limb(s). Mild to moderate OA lesions were characterized by roughened and/or thinned articular cartilage with matrix degeneration and fibrillation. Effusions were clear with mild reduced viscosity and mild-moderate synovial hypertrophy, edema, and hyperemia. Severe OA lesions were characterized by pink to orange (ie, blood-tinged), watery effusion with synovial hypertrophy, edema, and hyperemia; regionally extensive to diffuse cartilage thinning with ulcers exposing hyperemic to eburnated subchondral bone; and osteophytosis (Figs. 19, 20). Histology of severe OA lesions showed eroded to ulcerated cartilage subtended by intact epiphyseal-articular complexes, and sclerotic subchondral bone plates containing multifocal ulcers filled with proliferating fibrovascular tissue (Fig. 21). Remaining islands of degenerate, eroded cartilage were often covered and infiltrated by a pleocellular fibrovascular membrane, ie, synovial pannus (Fig. 22) that extended across the articular surface. Delayed ossification fronts (eg, retained cartilage cores or thickened epiphyseal cartilage compatible with OC manifesta) or necrotic cartilage canals surrounded by necrotic chondrocytes and chondrones (eg, features compatible with OC latens) 30 were not identified in lesions classified as OA, although it is possible that severe advanced lesions associated with grade 3 OA may have obscured prior OC lesions that progressed to early OA in this relatively young cohort of sows.
None of the OC or OA lesions macroscopically identified within distal limb segments were detected by radiography. This included OC lesions within 4 tarsocrural joints of 4 sows, and OA lesions identified in 1 antebrachiocarpal joint and 2 tarsocrural joints.
Discussion
The results of this study identify INF lesions as the most frequent cause of lameness in this cohort of group-housed sows and that INF lesions, as opposed to OC or OA lesions, correlated with lameness resulting in euthanasia. Most INF lesions were identified within distal portions of limbs (ie, at or below the carpi and tarsi) with a higher prevalence of INF lesions within hind claws than front claws. Most INF lesions were characterized as severe (ie, involving bones as well as synovial structures) and at the stage that the sow was euthanized, considered end-stage, and not amenable to therapeutic intervention. Although histology was useful to confirm lesions grossly classified as INF, OC, and OA, and enhanced information pertaining to the extent of orthopedic lesions, it was not necessary for accurate identification and classification of lesions as long as detailed orthopedic dissections that incorporated longitudinally oriented bandsaw subsections of proximal as well as distal limb segments (ie, digits and claw) were included. Histology helped identify and morphologically characterize microorganisms with histologic inflammatory foci and provided good correlation with the identity of microbes isolated from microbiologic cultures. One inherent limitation in these postmortem evaluations pertains to advanced OA lesions where prior OC or resolved INF lesions may have initiated OA development resulting in OA identified at the time of euthanasia. Although advanced OA could arise from either of the above scenarios, there are also other initiating causes of OA including mechanical instability that could arise from periarticular soft tissue or subtle articular incongruencies that extend beyond the scope of the study. This study also showed that radiography is a specific technique to identify INF lesions in the distal limbs of lame sows. Although radiology demonstrated good (93.9%) specificity in accurately identifying sows without INF lesions, it had limited (70.6%) sensitivity and thus may have limited use as a screening tool.
The predominance of INF lesions in this cohort of sows is different from a previous report from North America that identified OC lesions as the most frequent cause of lameness in sows. 8 Although INF orthopedic disease has been reported as the most frequent cause of lameness in a more recent Swedish study of group-housed sows, 9 other previous studies also report a predominance of OC lesions.13,15 Our sow cohort also showed a predominance of INF orthopedic lesions within distal limb locations (ie, manus and pes), which is in contrast to prior reports showing a predominance of INF lesions within the proximal portions of limbs (ie, cubital, femoral, glenohumeral, and coxofemoral joints).1,9,19 Although one study performed in preweaning piglets identified a higher prevalence and severity of distal limb lesions in indoor-housed versus outdoor-housed piglets including a subset of lesions confirmed by gross and histopathology, the investigation excluded the proximal limb joints (ie, proximal to the carpus and tarsus).8,18 Thus, differences in anatomic location and distribution of lesion types presented in this report could reflect our employment of a comprehensive orthopedic protocol that included complete dissection of all appendicular soft tissue and synovial structures and incorporation of a series of bandsaw sections to discern bone involvement, or could reflect differences in the etiopathogenesis of lameness between the cohort of sows included in this study versus other swine cohorts.
Studies using radiographs to investigate lameness in other species have helped to establish radiography as a screening tool, and to assist in characterizing lesions associated with lameness. A study from 1975 used radiographs to correctly diagnose septic arthritis in 85.3% of bovine lameness cases. 21 A later study that examined radiographs of digital infections in cattle concluded that joint space widening is among the earliest and most reliable indicators of joint disease in cattle, noting it was most frequently seen in the distal interphalangeal joint. 12 While radiography was able to detect OC in the proximal limb of pigs, 16 the benefit of radiography to detect OC may prove inconsequential if OC is not the most common cause of lameness or if anatomic predilection sites for OC lesions reside in locations that are challenging to image in unsedated or unanesthetized sows. Although radiography may not detect all sources of lameness and implementation of radiography in ambulatory sows may present logistical challenges, given the predominance of INF lesions identified within the distal appendages and the discerning radiographic features of INF lesions, radiography could provide useful information pertaining to lameness etiology, help guide therapy at earlier stages, or improve prognostication, all of which could improve the welfare of lame sows. An inherent limitation in this study is that the lameness in this sow cohort had progressed to a non-weight-bearing stage that corresponded to lesions in severe and chronic stages; therefore, further study is needed to determine the sensitivity and specificity of radiographs to lesions associated with lameness in earlier stages of disease.
Bacteria associated with INF arthritis in swine include Mycoplasma hyorhinis, Mycoplasma hyosynoviae, Streptococcus dysgalactiae subsp. equisimilis, Streptococcus suis, T. pyogenes, Brucella suis, and Glaesserella parasuis. 25 The mixed isolates, most commonly T. pyogenes, and F. necrophorum, recovered from cultures of septic joints in this study suggest the possibility that these INF lesions may be secondary to traumatic wounds that introduced opportunistic pathogens through direct extension of bacteria and/or hematogenous dissemination of bacteria to local, regional, or distant sites. Another possibility is that, especially in INF processes of chronic duration, the high occurrence of mixed growth and frequent recovery of T. pyogenes indicates the primary pathogen may be more difficult to identify via traditional culture methods or is no longer contributing to infection at the time of euthanasia. Although M. hyosynoviae or M. hyorhinis was not identified in this cohort, the use of standard bacterial cultures to identify these organisms from joint samples can be challenging, 22 and polymerase chain reaction testing was not implemented. Moreover, the purulent and necrotizing features of the lesions with concurrent involvement of bone and physes, the staining properties of bacteria identified on gram stains, and the asymmetrical distribution of lesions are features not consistent with previous descriptions of mycoplasma arthritis, which report serofibrinous effusions with mononuclear infiltrates within hypertrophied synovium and fibrous adhesions.23,27 Although mycoplasma pathogens could be contributors to earlier stages of lameness and orthopedic lesions, the results of this study indicate they were likely not involved or the main contributors to lesions at this stage of their progression. Mixed bacterial growth could also result from fistulae communicating between the skin surface and underlying infected structures, which were often present in lesions within the digits. Other considerations include the potential for postmortem overgrowth, inadvertent contamination associated with the dissection process that may have artificially impacted culture results, or impact of antemortem antimicrobial therapy on bacterial populations within infectious lesions. Although specimen processing including formalin fixation and acid-decalcification can impact histochemical staining, given the imperfect correlation between bacteria identified with Gram stain and bacterial culture results, the potential scenarios listed above should be considered for when interpreting microbiologic results of this study. While defining the etiologic cause of infectious lesions was not the focus of this study, future studies that include site-matched control joints from the same animal, correlation of bacteria isolated from traditional cultures with those identified via molecular sequencing techniques, or analyzing infectious lesions at earlier stages of disease are warranted.
There are several possible reasons contributing to the predominance of INF lesions identified in this cohort of pigs and their anatomic predilection that are different from prior studies of lameness in pigs, including the sample populations, or management practices (eg, housing). The sows in this study were group-housed which allows for ambulation as well as interaction among pen mates, in contrast to gestation stalls that limit ambulation and potential for aggressive interactions between sows.6,14 As mentioned above, the fact that many lesions within the digits of these sows had mixed infections with transcutaneous fistula communicating with infected synovial structures and bones suggests possible inoculation of opportunistic organisms through a wound. Moreover, many lesions within distal radial/ulnar or distal tibial/fibular physes had concurrent INF cellulitis, arthritis, or osteomyelitis at a different site within either the same limb or a different limb suggesting either local extension of a septic process, bacteremia with embolic localization to structures within a similar region of the primary infected site, or embolic dissemination to distant sites. Although bacteremia seeding appendicular structures could arise from systemic diseases such as pneumonia, enterocolitis, vegetative endocarditis, or polyserositis, due to logistical considerations involving transportation and financial costs associated with obtaining samples from producers enrolled in this study, we were unable to obtain entire bodies to perform complete (ie, visceral and orthopedic) postmortem examinations which could help identify possible underlying or contributing disease from other organ systems. Although there were no indications of prior or concurrent systemic disease within the production records of these sows, the identification of 1 sow having INF arthritis in addition to subclinical bacterial endocarditis and embolic pneumonia identified as part of a complete postmortem examination, and 3 sows with INF physitis, 2 of which had ≥2 physes affected suggests an embolic process and that systemic infections may underlie infectious arthritis or osteomyelitis. It also suggests that identification of systemic illness may be challenging in swine production facilities. Therefore, additional studies are underway that include full postmortem examinations to investigate concurrent visceral or systemic disease associated with infectious orthopedic lesions.
Another explanation for the predominance of infectious lesions identified in this cohort of sows could reflect the advanced clinical stage of the lesions where lameness was the reason for euthanasia, rather than reduced productivity or clinical manifestations of disease in other organ systems. Sows culled for market slaughter are required to ambulate onto a trailer for transport. A European study investigating the clinical deterioration of cull-sows transported to slaughter showed 90% of sows arriving to slaughter had normal ambulation with 9.6% and 0.8% of sows exhibiting mild to moderate lameness, respectively. 28 Therefore, one would expect the lesions associated with lameness in these cohorts to be much less advanced than the sows used in this study that at the time of euthanasia exhibited severe gait abnormalities or were unable to ambulate (ie, downer sows).
One additional factor contributing to lameness could be related to the type of housing that may have predisposed these sows to acquiring traumatically induced lesions. One design characteristic of modern sow farms, including those in this study, is slatted floors, which, when compared with solid flooring, have been associated with lameness in pregnant sows.5,17 Moreover, these sows were housed in pens, which affords them a greater degree of mobility, and has been shown to increase their vulnerability to claw lesions. 3 These lesions may occur as a result of aggressive interactions between animals or as a result of increased mobility as sows with access to a larger pen area have been shown to have an increased prevalence of lameness. 29 The high frequency of septic lesions in this study and their predominance in the distal limb segments, including the digits and claw, may indicate that the combination of group-housing sows and the slatted floor design increased the risk for distal limb trauma, resulting in direct inoculation of microorganisms into synovial structures or bones within the manus or pes, or local extension or embolization of microorganisms from an infection in close proximity (ie, sole ulcer, cellulitis, or myofasciitis). To date, most studies either provide no information regarding housing or flooring, or limited information without specifics, making comparisons pertaining to different types of housing or flooring difficult.
Findings that coincide with prior studies include gross and histologic features of non-INF OC and OA lesions, and their predilection sites, which predominated in the proximal portions of the front limbs, mainly the distal humerus of the cubital joints, followed by proximal humerus and distal scapula of the glenohumeral joints.7,10,30 Although postmortem radiography performed on limbs from boars showed good correlation with OC lesions identified postmortem, 16 OC lesions were most prevalent in proximal limb joints. Therefore, due to the radiographic protocol used in this study, which was limited to the manus and pes, the non-INF (ie, OC and OA) lesions concentrated within anatomically proximal joints were not radiographically identified. However, the finding that only 3 sows in our cohort had lameness that could be attributable to non-INF orthopedic disease (ie, traumatic fracture, epiphysiolysis, and unknown cause) suggests that clinical lameness in group-housed production sows is more often correlated with INF orthopedic lesions than non-INF lesions.
As farms move away from gestation stalls and toward group housing of sows, research must adapt to examine lameness in this modern context of housing and management. Yet, research on group-housed sow populations is lacking. This study informs producers and veterinarians that in this cohort of group-housed sows, INF lesions, not OC or OA, were the most common cause of lameness resulting in euthanasia. These lesions most commonly involved synovial structures and bones at or below the carpi and tarsi, especially the digits, suggesting trauma could be a contributing etiologic factor. Future investigation of pathogens involved in the etiopathogenesis of INF orthopedic lesions could provide information that could apply to prevention strategies, and early and effective methods of therapeutic intervention. Furthermore, complete visceral and orthopedic postmortem examinations could help differentiate between systemic versus local or trauma-associated INF lesions. Results from this study also indicate that radiography is specific but not sensitive at identifying INF lesions within the distal limbs. Although radiography may have limited application as a screening tool, it could improve sow welfare by informing prognosis, guiding therapeutic intervention or, in the case of end-stage lesions, informing the decision to euthanize. Because of the shift in sow housing away from gestation stalls and toward group housing, the findings of this study are applicable to many farms and make lameness prevention an essential consideration during remodeling of sow operations and post-transitional management.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221114470 – Supplemental material for Gross, histopathologic, microbiologic, and radiologic characterization of lesions associated with clinical lameness in a cohort of group-housed sows euthanized for lameness
Supplemental material, sj-pdf-1-vet-10.1177_03009858221114470 for Gross, histopathologic, microbiologic, and radiologic characterization of lesions associated with clinical lameness in a cohort of group-housed sows euthanized for lameness by Julie B. Engiles, Nathan Fanzone, Kathryn B. Wulster, Justin Schumacher and Meghann K. Pierdon in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858221114470 – Supplemental material for Gross, histopathologic, microbiologic, and radiologic characterization of lesions associated with clinical lameness in a cohort of group-housed sows euthanized for lameness
Supplemental material, sj-xlsx-1-vet-10.1177_03009858221114470 for Gross, histopathologic, microbiologic, and radiologic characterization of lesions associated with clinical lameness in a cohort of group-housed sows euthanized for lameness by Julie B. Engiles, Nathan Fanzone, Kathryn B. Wulster, Justin Schumacher and Meghann K. Pierdon in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858221114470 – Supplemental material for Gross, histopathologic, microbiologic, and radiologic characterization of lesions associated with clinical lameness in a cohort of group-housed sows euthanized for lameness
Supplemental material, sj-xlsx-2-vet-10.1177_03009858221114470 for Gross, histopathologic, microbiologic, and radiologic characterization of lesions associated with clinical lameness in a cohort of group-housed sows euthanized for lameness by Julie B. Engiles, Nathan Fanzone, Kathryn B. Wulster, Justin Schumacher and Meghann K. Pierdon in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank Mr Ralph Conti, Mr Robert (Max) Holzman, and Ms Karie Durynski for their technical expertise and assistance in processing samples used in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pennsylvania Department of Agriculture, Animal Health and Diagnostic Commission grant, PDA#44187368 (2019–2020).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
