Abstract
Influenza A viruses (IAV) cause mammalian infections following several transmission routes. Considering the anatomic proximity and connection between the nasopharynx and periocular tissues, there is a need to understand the dynamics of virus spread between these sites following both respiratory and nonrespiratory viral transmission. We examined virus distribution and associated inflammation within nasal and periocular tissues during the acute phase of H1N1 IAV infection in ferrets following intranasal or ocular inoculation. Ocular and intranasal inoculations with IAV caused comparable viral antigen distribution and inflammation in the nasal passages, though infection kinetics and magnitude differed by inoculation route. Ocular inoculation was associated with inflammation in the conjunctiva and lacrimal glands. Although intranasal inoculation was also associated with periocular inflammation, the onset was delayed relative to ocular inoculation. This work underscores the importance of investigating extrapulmonary tissues following mammalian infection with respiratory pathogens, even after intranasal inoculation.
Studies of influenza A virus (IAV) pathogenicity, transmissibility, and tropism in ferrets have revealed critical roles for upper respiratory tract tissues (URT, including the nasal respiratory epithelium and the soft palate) in virus reassortment, transmission, and elicitation of host responses following IAV infection.10,12,16 Although these studies have been generally limited to ferrets inoculated intranasally, 2 IAV deposition and replication in ferret respiratory tract tissues can be modulated by virus subtype,17,18 host features, 7 and volume. 14 Inoculation methods that restrict virus to different anatomic sites8,11,15,20 can also influence virus replication and dissemination. Importantly, ocular (conjunctival) exposure to IAV represents another route of infection that can lead to replication in the respiratory tract and tissue-specific host responses.1,3–6 As humans are exposed to IAV at a range of doses and by various routes,9,19 there is a need to evaluate multiple exposure modalities to understand how nontraditional inoculation routes can modulate virus pathogenicity and induction of host responses.
IAV possess a predominant tropism for the respiratory tract, though conjunctivitis has been associated with H1N1 virus infection in humans (reviewed in Belser et al 5 ). Furthermore, recovery of infectious virus in periocular tissues and swabs from ferrets intranasally inoculated with IAV supports the capacity for fluid exchange between periocular and URT tissues. 4 Although viral antigen in respiratory and periocular tissues following conjunctival (ocular) inoculation with seasonal IAV has been reported, 4 the exact mechanism and timing of how virus spreads between periocular and URT tissues remains poorly understood. Here, we inoculated ferrets with IAV by either intranasal or ocular routes and performed histopathologic assessments of the URT (nasal passages, sinuses, and soft and hard palates) and periocular tissues (conjunctiva, lacrimal glands, and nasolacrimal ducts) through the acute phase of infection, to more clearly identify the dynamic processes involved in virus spread throughout these tissues.
Previous studies have identified that ocular exposure to IAV can result in robust infection of the URT of ferrets, albeit with delayed kinetics relative to intranasal inoculation.4,6 Consistent with these findings, all ferrets inoculated by either the intranasal or ocular route with the H1N1 IAV A/Nebraska/14/2019 were productively infected (Table 1, n = 3 per group, per day unless otherwise specified, methods provided in Supplemental Methods). Virus titers were assessed with a plaque assay (Supplemental Methods). Virus titers in nasal wash (NW) specimens peaked at 1-day postinoculation (d.p.i.) in both groups. Peak mean weight loss and rise in body temperature were more pronounced in ferrets inoculated by the intranasal route compared with the ocular route. Nasal discharge or conjunctivitis were not observed in any ferrets. Conjunctival wash (CW) specimens were used to assess ocular viral loads and were collected by rinsing and swabbing the right eye. 4 Ferrets inoculated by the ocular route had infectious virus present in all CW specimens collected, with virus detected more frequently and to higher titer than in intranasally inoculated ferrets (Table 1). Conjunctival wash specimens detect virus on the ocular surface, but cannot distinguish if this virus is replicating within periocular tissues or if its presence is due to fluidic exchange from the nasal passages. Therefore, we histologically and immunohistochemically assessed the pathologic changes and presence of viral antigen in oculonasal tissues to evalute how inoculation route modulates the distribution of viral antigen and magnitude of inflammatory processes.
Virus pathotyping, incidence of viral replication, average compiled histologic score for inflammation, and average immunohistochemical score from ferrets inoculated with IAV by the intranasal or ocular route.
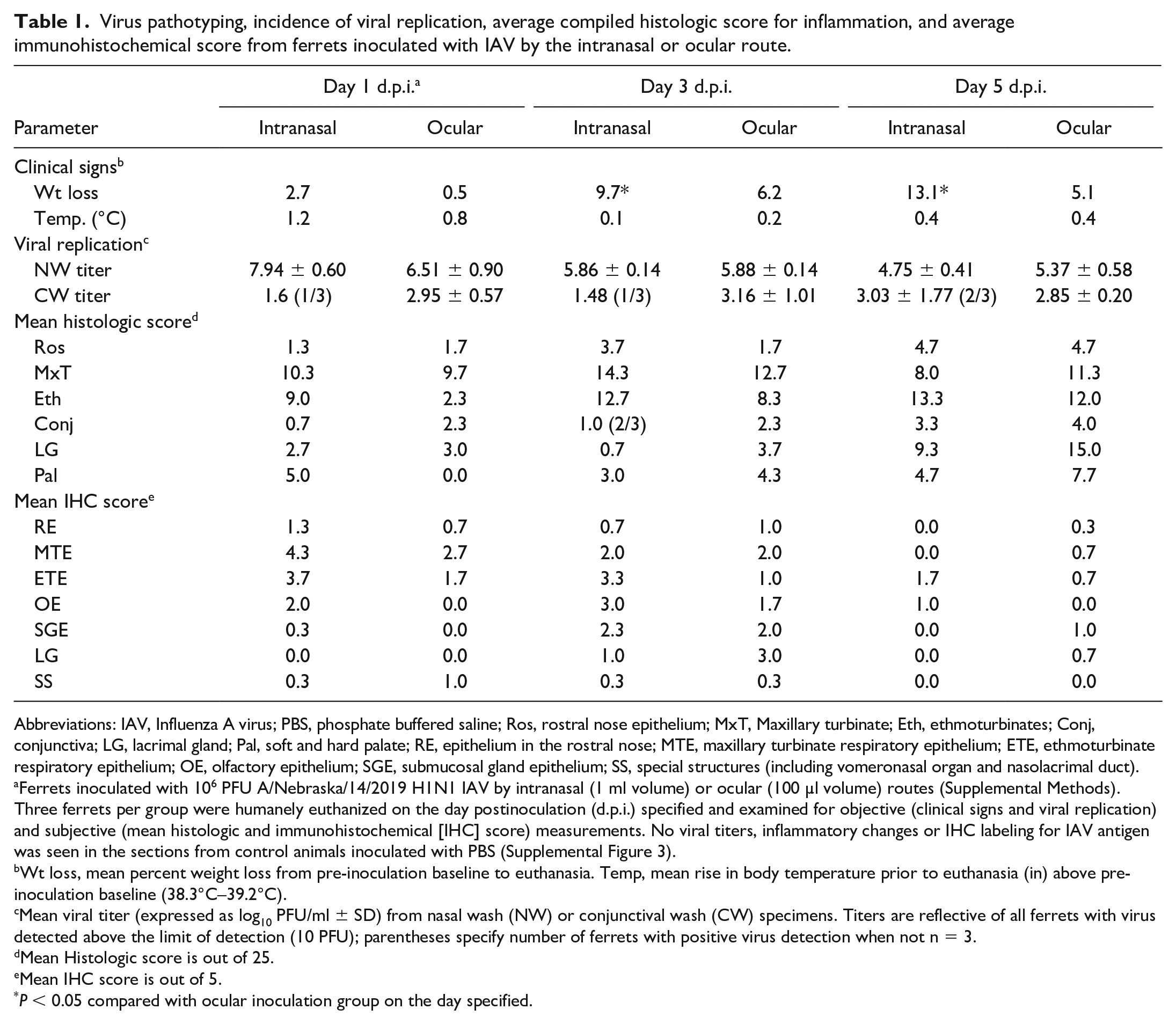
Abbreviations: IAV, Influenza A virus; PBS, phosphate buffered saline; Ros, rostral nose epithelium; MxT, Maxillary turbinate; Eth, ethmoturbinates; Conj, conjunctiva; LG, lacrimal gland; Pal, soft and hard palate; RE, epithelium in the rostral nose; MTE, maxillary turbinate respiratory epithelium; ETE, ethmoturbinate respiratory epithelium; OE, olfactory epithelium; SGE, submucosal gland epithelium; SS, special structures (including vomeronasal organ and nasolacrimal duct).
Ferrets inoculated with 106 PFU A/Nebraska/14/2019 H1N1 IAV by intranasal (1 ml volume) or ocular (100 µl volume) routes (Supplemental Methods). Three ferrets per group were humanely euthanized on the day postinoculation (d.p.i.) specified and examined for objective (clinical signs and viral replication) and subjective (mean histologic and immunohistochemical [IHC] score) measurements. No viral titers, inflammatory changes or IHC labeling for IAV antigen was seen in the sections from control animals inoculated with PBS (Supplemental Figure 3).
Wt loss, mean percent weight loss from pre-inoculation baseline to euthanasia. Temp, mean rise in body temperature prior to euthanasia (in) above pre-inoculation baseline (38.3°C–39.2°C).
Mean viral titer (expressed as log10 PFU/ml ± SD) from nasal wash (NW) or conjunctival wash (CW) specimens. Titers are reflective of all ferrets with virus detected above the limit of detection (10 PFU); parentheses specify number of ferrets with positive virus detection when not n = 3.
Mean Histologic score is out of 25.
Mean IHC score is out of 5.
P < 0.05 compared with ocular inoculation group on the day specified.
Multiple coronal sections through the nose of PBS-inoculated control ferrets and ferrets intranasally or ocularly-inoculated with IAV virus were examined by routine H&E staining and an immunohistochemical (IHC) assay targeting influenza A virus nucleoprotein (A3) (Table 1, Supplemental Methods, Supplemental Figure 1). For both inoculation routes, the patterns of IAV antigen distribution and inflammation correlated with the viral titers observed in the NW and CW specimens. Immunolabeling for IAV antigen in the rostral nose was minimal to mild and was most prominent in the ventral epithelium in the nasal group on 1 d.p.i., and for the ocular group at 3 d.p.i. Acute inflammation and necrosis were most prominent at 3 d.p.i. for both groups. IAV immunolabeling was seen in glandular epithelium starting at 3 d.p.i. in both groups with neutrophilic inflammation and necrosis of submucosal glands delayed in the ocular group until 5 d.p.i. Mild reparative changes, primarily epithelial hyperplasia, were noted at 5 d.p.i. in both groups. Moderate neutrophilic inflammation in the vomeronasal organ was seen in 1/3 animals at 1 and 3 d.p.i. in the ocular group, with focal viral antigen immunolabeling. IAV immunolabeling was in the vomeronasal organ of 1/3 animals at 1 d.p.i. in the nasal group with minimal associated inflammation.
In the middle nose, IHC labeling for IAV was seen in the respiratory epithelium of the maxilloturbinates in both nasal and ocular groups by 1 d.p.i. (Supplementary Figure 2a, 2c), with decreased abundance by 5 d.p.i. In both groups, the mucosa of the maxilloturbinates was multifocally expanded by edema and mixed inflammation composed of neutrophils with occasional macrophages on 1 d.p.i. There were associated areas of epithelial loss, and sloughed epithelial cells mixed with cell debris, mucin, neutrophils, and occasional macrophages in the nasal vestibule (Supplementary Figure 2b, 2d). Mucosal inflammation was most severe in the nasal group, especially on 3 d.p.i. By 5 d.p.i., inflammatory scores were reduced in both groups. Immunolabeling in the submucosal glands was associated with inflammation and necrosis and was most pronounced on 3 d.p.i. in the nasal group.
In the caudal nose, immunolabeling for IAV was primarily localized to the respiratory epithelium of the ethmoturbinates and was most prominent at 3 d.p.i. for both groups, with the most extensive labeling in the nasal group (Fig. 1a, b). IAV immunolabeling increased in the olfactory epithelium from 1 to 3 d.p.i. in the nasal group (Fig. 1c), and increased in the ocular group from 3 to 5 d.p.i. Ethmoturbinates showed inflammatory changes similar to those observed in the maxilloturbinates, with inflammatory scores staying relatively consistent between 1 and 3 d.p.i. (Fig. 1b) for the nasal group and decreasing slightly by 5 d.p.i. Inflammatory scores increased between 3 and 5 d.p.i. for the ocular group. Inflammation and necrosis were seen in submucosal glands starting at 3 d.p.i. and were more notable by 5 d.p.i. for both groups. IHC labeling for IAV was present at 1 and 3 d.p.i. in the maxillary sinuses of all nasal-inoculated animals with sinus available in evaluated sections (Fig. 1d). Only 1 animal from the ocular group had maxillary sinus IAV immunolabeling. Inflammation in the maxillary sinuses echoed the changes seen in the ethmoturbinates and were most pronounced at 3 d.p.i. in the nasal group and at 5 d.p.i. in the ocular group.
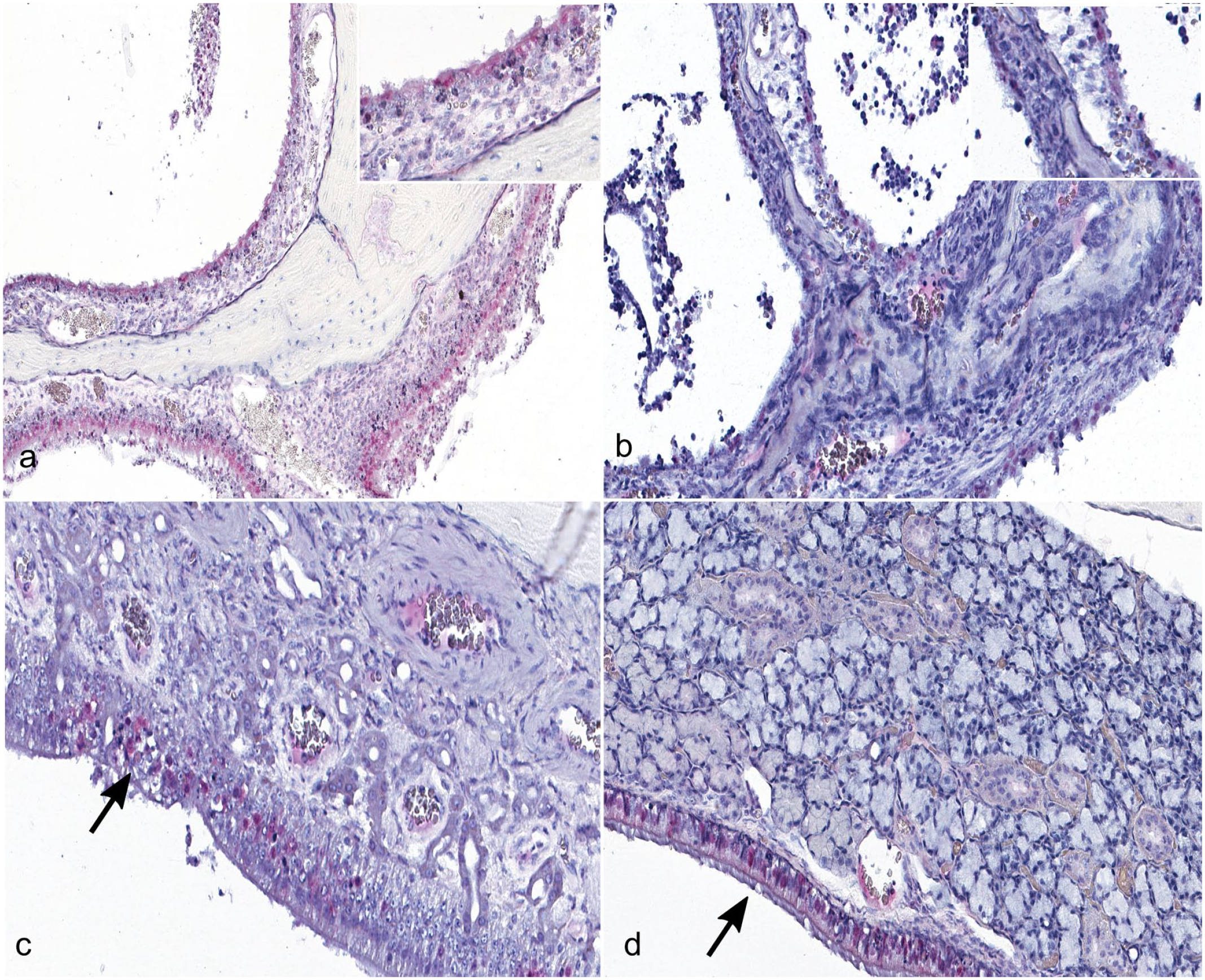
Immunolabeling for Influenza A virus (IAV, in red, primarily nuclear) is most extensive in structures of the caudal nose and sinuses of nasal-inoculated ferrets at day 1 postinoculation. (a) Immunolabeling is diffuse in the respiratory epithelium of the ethmoid turbinates on day 1 postnasal inoculation. The inset shows a higher magnification of immunohistochemical (IHC) labeling in the respiratory epithelium. (b) Immunolabeling for IAV is less extensive in the ethmoid turbinate respiratory epithelium on day 1 and day 3 (shown in figure) postocular inoculation than in the nasal inoculation group. The inset shows a higher magnification of the patchy IHC labeling in the ethmoid turbinate respiratory epithelium of this day 3 postocular inoculation ferret. (c) Immunolabeling for IAV is most extensive within the olfactory epithelium in day 3 postnasal inoculation ferrets. Immunolabeling corresponds with disruption of the epithelium (indicated by an arrow) (d) Immunolabeling is present in the lining epithelium, but not in the glandular epithelium, of the maxillary sinus 1 day postnasal inoculation. Immunolabeling of the lining epithelium is indicated by an arrow.
IAV antigen has been detected in ocular tissues following conjunctival inoculation, 4 but periocular structures, including the conjunctiva, lacrimal glands, and nasolacrimal duct, are not routinely examined following virus inoculation by any route. In this study, these structures were evaluated in sections adjacent to the eye; immunolabeling was not observed in these regions in a subset of animals with corresponding inflammation likely due to the lengthy decalcification process needed to section the calvarium. In animals where labeling was observed, periocular structures were more likely to show IAV immunolabeling in the ocular group. IAV immunolabeling was present rarely within the conjunctival epithelium and lacrimal gland of 1/2 and 1/3 nasally inoculated animals, respectively, at 3 d.p.i., and in the lacrimal glands in 2/3 and 1/3 animals in the ocular groups at 3 and 5 d.p.i., respectively (Fig. 2a) with associated inflammation (Fig. 2b). Mononuclear inflammation was most prominent in the conjunctival submucosa; expansion of the conjunctiva-associated lymphoid tissue was more pronounced in the ocular group at all time points compared with controls and with the contralateral (noninoculated) eye (Fig. 2c and Supplemental Figure 3d). Inflammation was also observed around adjacent lacrimal ducts. Conjunctival inflammatory scores were greater at later time points in the nasal group. Moderate, mixed inflammation, characterized by neutrophils with occasional macrophages, lymphocyte aggregates, and necrosis was seen in the lacrimal glands. Inflammation was increasingly prominent at 3 and 5 d.p.i. in the ocular group (Fig. 2b, c), and was less severe in the nasal group. It is interesting to speculate that virus replication within the lacrimal gland could contribute to virus detected in CW specimens.
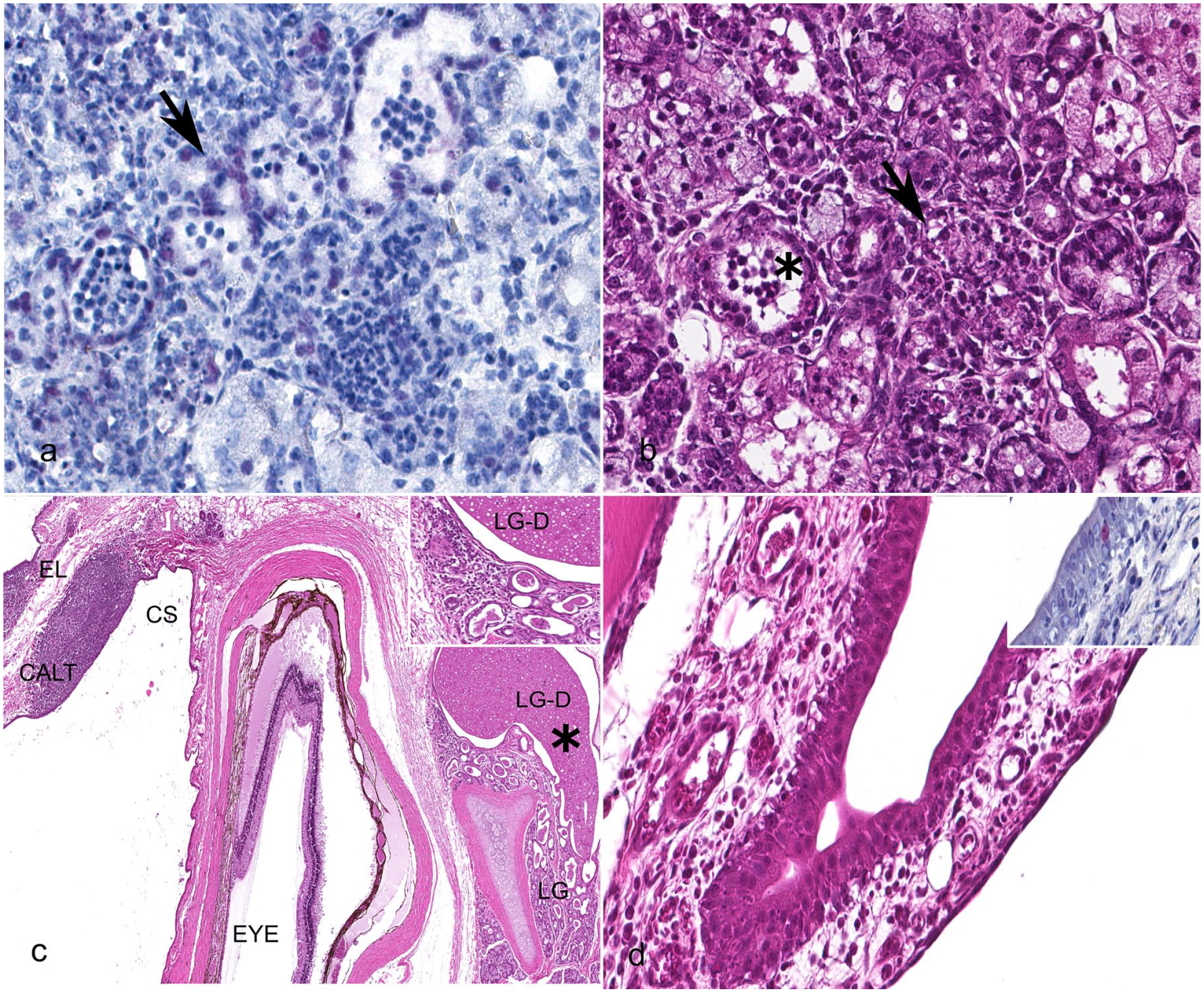
Inflammation and immunolabeling for influenza A virus (IAV, in red) are increased in periocular structures in ferrets inoculated ocularly with IAV compared with those inoculated intranasally. (a) Immunolabeling targeting IAV antigen shows multifocal, faint nuclear labeling in the lacrimal gland acini of a day 1 ocular-inoculated ferret (indicated by arrow). (b) Mixed intraluminal inflammation (indicated by asterisk) and occasional acinar necrosis (indicated by arrow) were seen in the lacrimal gland of the same ocular-inoculated ferret 1 day after inoculation (hematoxylin and eosin, HE). (c) The eyelid, conjunctival sac, eye, and lacrimal gland of a ferret 1 day after ocular inoculation with IAV shows expanded conjunctiva-associated lymphoid tissue (CALT) and dilated ducts and inflammation in the adjacent lacrimal gland. The inset shows a higher magnification of secretory material and cell debris in the dilated lacrimal ducts, and the mixed inflammatory cell infiltrates in the adjacent stroma. An asterisk highlights debris in the lacrimal gland duct on low magnification. (EL, eyelid, CS, conjunctival sac, LG-D, lacrimal gland duct, LG, lacrimal gland; HE). (d) Mixed inflammatory infiltrates surrounding the nasolacrimal duct in a ferret inoculated intranasally, 3 days postinoculation (HE). In the inset, rare nuclear immunolabeling for IAV in the nasolacrimal duct epithelium of this ferret 3 days after ocular inoculation.
IHC labeling was seen in few epithelial cells in the nasolacrimal ducts at 3 d.p.i. of all ocular-inoculated animals (Fig. 2d) and in 1 nasal-inoculated animal. Mild, mixed inflammation surrounded the nasolacrimal ducts of all ocular-inoculated animals at 1 d.p.i, and almost all animals at later time points (Supplemental Figure 3d). This suggests that the duct provides a pathway for spread of ocular infection into the nasal cavity. These findings demonstrate that microscopic inflammation can be detected in periocular structures (notably conjunctiva and lacrimal gland) following ocular inoculation in the absence of macroscopic disease. Moreover, IAV spread to the nasolacrimal duct and adjacent ocular tissues is possible following intranasal inoculation.
Inflammation and viral antigen distribution in the nose of ferrets inoculated with IAV intranasally was similar to previously described work in this model,14,16 though inflammation was seen here at earlier time points in the nasal turbinates. The pattern of inflammation and antigen distribution over time were slightly different for the ocular inoculation group, with more inflammation in periocular structures at early time points, and a slight delay in inflammation in nasal structures of the caudal nose and submucosal glands compared with intranasally inoculated animals. 13
These findings support that IAV distribution and associated inflammation is similar in ferrets inoculated by the intranasal and intraocular routes. The variation in tissues affected in these 2 routes likely reflects local viral loads, though additional methods, such as ISH, are needed to confirm this hypothesis. We observed strong congruence between relative viral loads and mean inflammation scores in URT (NW specimens and turbinates, respectively) and ocular (CW specimens and conjunctiva and lacrimal gland, respectively) sites of IAV replication (Table 1).17,18 Further investigation into whether IAV which exhibit an ocular tropism in mammals are associated with enhanced inflammatory processes in periocular structures is warranted. 5
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221109103 – Supplemental material for Influenza A virus infection and pathology in nasal and periocular tissues after ocular inoculation in ferrets
Supplemental material, sj-pdf-1-vet-10.1177_03009858221109103 for Influenza A virus infection and pathology in nasal and periocular tissues after ocular inoculation in ferrets by Joy M. Gary, Jana M. Ritter, Xiangjie Sun, Taronna R. Maines and Jessica A. Belser in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Brigid Bollweg, Amanda Lewis, and Pamela Fair of the Infectious Disease Pathology Branch Core Pathology Laboratory for their invaluable assistance in preparing and staining tissue sections and performing immunostaining. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention/the Agency for Toxic Substances and Disease Registry.
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
