Abstract
Pithomycotoxicosis (facial eczema) is a seasonal hepatogenous photosensitization of sheep caused by the ingestion of sporidesmin contained in the spores of the fungus Pithomyces chartarum. We describe 4 cases of obstructive rhinopathy associated with chronic pithomycotoxicosis naturally occurring in the north of Spain. Sheep were 5 to 7 years old and Latxa breed. A detailed clinical study was conducted together with computerized tomography examination and completed by necropsy and histopathology. All sheep developed a permanent narrowing of the nasal lumen close to the nostrils causing inspiratory dyspnea and snoring. Computerized tomography demonstrated a significant increase of soft tissue in the rostral nasal cavity. Elevated gamma-glutamyl transferase, alanine aminotransferase, and lipase were noted on serum biochemistry. At necropsy, liver atrophy and fibrosis associated with chronic pithomycotoxicosis was identified in 3 of the sheep. All sheep had whitish elevations and rough surfaces on the alar folds and areas adjacent to the nasal surfaces. Histopathologic assessments, which included histochemical and immunohistochemical techniques, of the nasal lesions identified moderate to severe arteriosclerosis in 21.5% to 61.9% of the small arteries evaluated with surrounding fibrosis and edema. No changes associated with hypersensitivity reactions were found. These lesions were similar to the ones described in blood vessels of the liver in chronic pithomycotoxicosis and in our cases. The results of this study suggest a direct action of the sporidesmin on the rostral nasal cavity. Further studies are needed to analyze the impact of the sporidesmin on the sheep nasal mucosa.
Pithomycotoxicosis (facial eczema) is a seasonal hepatogenous photosensitization of sheep and other ruminants grazing in pastures, in which there has been accelerated sporulation of the fungus P. chartarum. This species is a polyphagous saprophyte that is usually found in the decomposing stem and foliar debris of a wide range of grasses and lives endophytically in them. 10 Pithomycotoxicosis frequently occurs in New Zealand but is recorded globally and negatively impacting the sheep industry.9,28 Spores of some strains of this fungus contain sporidesmin (epidithiodioxopiperazine), a secondary metabolite made only by the fungus and a potent hepatotoxin. By ingestion, this toxin can cause liver inflammation and bile duct obstruction that lead to retention of photosensitizing pigment (phylloerythrin) in the circulation. When animals are exposed to sunlight, photodermatitis can develop on unpigmented or hairless areas of the skin. This disease is named acute facial eczema.9,13,25 The exposure to sporidesmin also can drive to the so-called “chronic facial eczema” in sheep, which is observed with no sign of clinical photosensitivity. Chronic facial eczema is characterized by progressive liver atrophy, fibrosis, biliary hyperplasia, and eventually cirrhosis. 22 In addition to lesions in hepatocytes and the biliary collecting system, hepatic arteries and veins are also damaged. Eccentric intimal thickening in hepatic arteries and portal veins are described in natural and experimental cases.11,12,18,20
As a consequence of hepatic damage, the affected animals have elevated gamma-glutamyl transferase (GGT), aspartate aminotransferase (AAT), and serum bilirubin.9,25 Gamma-glutamyl transferase has been shown to correlate with the severity of the liver damage caused by the fungal toxin, and may be used to reveal animals with liver damage, but no clinical signs to predict which animals will subsequently develop severe, long-term liver lesions.22,25
A chronic obstructive nasal syndrome has been described in sheep flocks where P. chartarum is endemic in Spain and France, and its relationship with sporidesmin intoxication is under debate.2,14,23 There are no references for this disease in any other countries where pithomycotoxicosis is common in sheep. It occurs either in association with the ingestion of pastures containing spores of P. chartarum or is reported in cases of acute facial eczema.2,14,23 The condition was named either ovine chronic nasal stertor or obstructive nasal syndrome.2,23 The affected animals show complete or partial bilateral obstruction located close to the nares, and up to 80% of the sheep in the flock can be permanently affected.2,23 The condition has been described primarily in milk breeds such as Latxa or Manech tête rousse and also in other breeds.2,14,23 The disease seems to be more prevalent in autumn, but it is present throughout the year.2,23 Histopathological descriptions of the nasal mucosa indicated perivasculitis, inflammatory infiltration, edema, and in severe cases, fibrosis. Thus, these lesions were associated with local hypersensitivity or allergic reactions to an unidentified allergen in pastures or common nasal parasites (Oestrus ovis), but not with sporidesmin intoxication.2,23
We report in this article, a clinical and pathologic investigation in a flock with positive identification of P. chartarum in their grazing area that is located in a region where previous cases of facial eczema and the referred nasal disease were described in Spain.2,14 In this flock, a high proportion of sheep showed clinical signs of obstructive rhinopathy, similar to those detailed in preceding publications.2,14 However, we describe very different histopathological findings, with arteriosclerotic changes in small arteries, interstitial fibrosis, and edema in a particular area of the nasal mucosa in 4 sheep from this flock. Inflammatory changes related to allergies or hypersensitivity reactions were not significant in the studied cases. For the first time, we describe vascular lesions in small arteries of the nasal mucosa that are similar to those found in the hepatic blood vessels in cases of chronic pithomycotoxicosis.11,12,18,20 These similarities suggest a putative direct action of sporidesmin on the nasal mucosa causing degeneration and fibrosis, but not an allergic or hypersensitivity reaction.
Materials and Methods
Clinical Investigation
The study was carried out in a pure breed Latxa sheep flock located in the north of Spain (Basque Country) with a history of facial eczema and records of P. chartarum spores in their grazing areas that were detailed in previous studies developed by local sanitary authorities. The veterinary practitioners’ early clinical examination of the flock indicated that around 90% of the adult sheep showed a variable degree of nasal obstruction together with sero-mucous nasal secretions and snoring. In addition, cases of photosensitization and oestrosis were recorded in previous clinical visits.
Four, 5- to 7-year-old sheep showing nasal obstruction were transported to the Veterinary Faculty of Zaragoza. A comprehensive clinical examination of all animals was conducted, emphasizing respiratory examination. Blood samples with anticoagulant (ethylenediaminetetraacetic acid [EDTA]) and without anticoagulant were collected from the jugular vein through a vacutainer system from all the animals. Blood analysis was carried out using an automatic hematological counter IDEXX Procyte Dx (IDEXX laboratories, Westbrook, ME, USA). Biochemical analysis was also performed with sera from affected animals using an IDEXX Catalysis One device (IDEXX Laboratories, Westbrook, ME, USA). Parameters studied were as follows: glucose (mg/dL), creatinine (mg/dL), urea (mg/dL), phosphorus (mg/dL), calcium (mg/dL), total protein (g/dL), albumin (g/dL), globulin (g/dL), alanine aminotransferase (ALT) enzyme (U/L), alkaline phosphatase enzyme (U/L), GGT (U/L), total bilirubin (mg/dL), and cholesterol (mg/dL).
In addition, a computerized tomography scan of the head of the affected animals was performed with a Bivro CTS of 2 sides (General Electric Healthcare, Spain). The selected parameters for this study were helicoidal mode and 2 series in each study with bone and soft tissue filters. Amperage limits were 80 mA and 120 kV of power. Two-mm cuts were obtained, and studies with and without contrast were included using intravenous injection of Ominpaque® (iohexol) 300 mg/mL, 1.5 mL/kg body weight.
Isolation and Characterization of P. Chartarum From the Grass
Samples consisting of approximately 500 to 700 g of fresh grass were collected from 4 different locations in the sheep grazing areas. Selected plant materials were investigated to detect P. chartarum in them. First, plant fragments were surface sterilized (ethanol 75% 0.5 min, sodium hypochlorite 3% 1 min, and 4 to 5 washes in autoclaved bi-distilled water), placed in Petri plates containing potato dextrose agar (Panreac, Spain), and medium supplemented with streptomycin sulfate (0.5 g/L; Merck, Germany) to avoid bacterial contaminations. They were incubated 3 to 5 days at 26°C in the dark to obtain endophytic isolates of P. chartarum. The second procedure was to determine the presence of the level of conidia in the grass. With this objective, sterile double-distilled water was passed through funnels containing approximately 50 gm of plant material selected from the prospected samples. The eluent was collected and subsequently analyzed, quantifying the presence of P. chartarum conidia under the optical microscope with the aid of a hemocytometer.
Postmortem Studies
All sheep were euthanized following authorized protocols by the Ethics Committee for Animal Experiments from the University of Zaragoza. A complete postmortem investigation was conducted on 4 animals with particular attention to the liver and nasal mucosa. Several sections representing all liver lobes and gallbladder were obtained from each sheep. In the case of cirrhotic livers, 5 to 6 selected sections representing the complete organ were collected. To investigate the nasal cavities, we obtained 8 to 10 sections from an area between the nostrils and the beginning of the ventral turbinate. It included anatomical structures such as nostrils, nasal vestibulum, alar fold, ventral fold, and rostral portion of dorsal and ventral turbinates from both nasal cavities. Sections followed dorsal to ventral orientation, trying to replicate the same cut planes of the computerized tomography scan. Nasal septum samples from the same area were also included. The collection of samples was completed with representative sections from all organs, including skin from the head (nasal, dorso-lateral nasal, frontal, masseteric, orbital, and auricular regions) and other body areas (thoracic vertebral and lumbar regions).
All samples were immersed in 10% buffered formalin for a minimum of 48 hours and processed for automatic paraffin embedding following routine protocols. Four mm sections were obtained and stained with Carazzi’s hematoxylin and eosin combination (H&E) as routinely used in our laboratory. Additional techniques were used to investigate vascular changes in more detail. To accomplish this objective, a group of classical histochemical methods were carried out in sections of the nasal cavity and liver from all sheep. Elastic fibers were investigated using acid orcein solution. 19 The orcein stain was combined with picrocarmin solution used in Cajal’s trichrome (saturated picric acid mixed with 1% aqueous indigo carmin) to simultaneously investigate muscle cells, collagen, and elastic fibers. 24 The total number of arteries with lesions was assessed in relationship with the total number of arteries found in the sections. We also counted the number of arteries according to the arteriosclerotic changes and the number of arteries with these degenerative lesions orientated to the mucosa epithelium. Based on the characteristics of lesions observed in arteries, they were grouped as type A or type B. This classification was obtained after histopathological observation of large numbers of arteries and the characteristics of each type are defined in the results section. Then, arterial counts were split into the 2 groups, and the results were summarized in Table 1. We have tried to use this classification as an indicator of severity of arteriosclerotic changes and comparison with liver lesions in this study (Table 1). The presence of calcium was investigated following Kossa’s method. 17 Toluidine blue method for metachromasia was used to identify mast cells. 19
Assessment of arteriosclerosis in the nasal mucosa of 4 sheep in relation to liver damage severity and blood biochemistry of GGT, ALT, and Lipase in U/L.
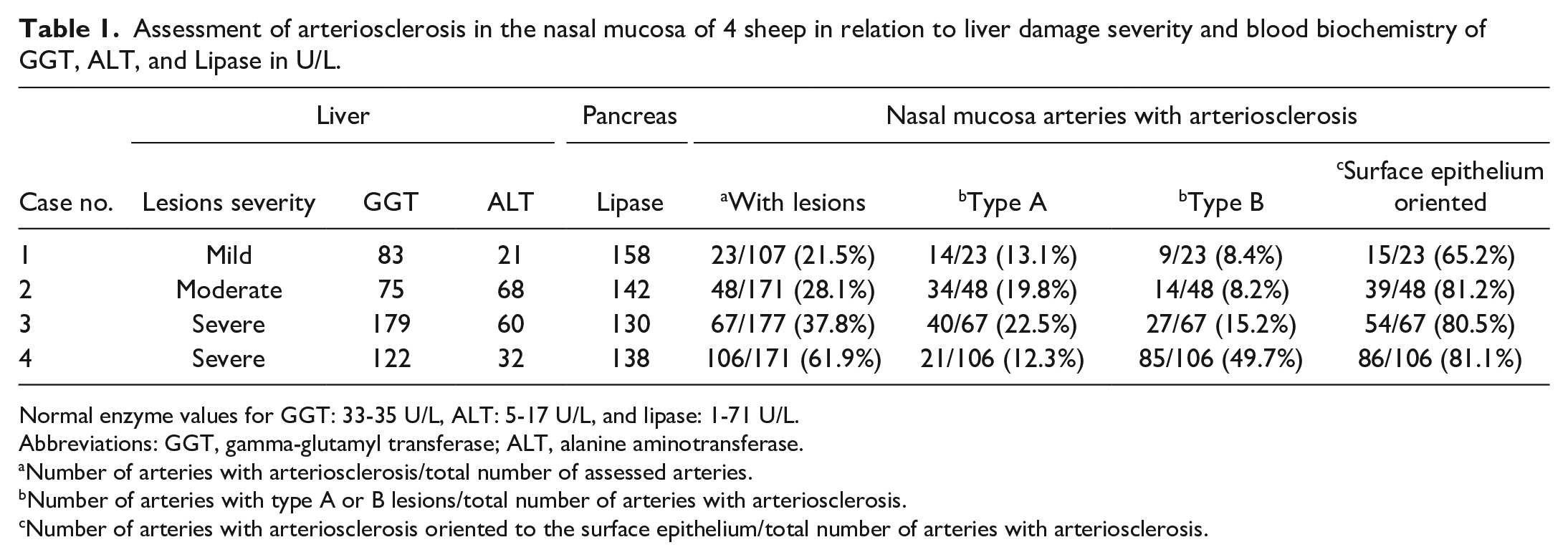
Normal enzyme values for GGT: 33-35 U/L, ALT: 5-17 U/L, and lipase: 1-71 U/L.
Abbreviations: GGT, gamma-glutamyl transferase; ALT, alanine aminotransferase.
Number of arteries with arteriosclerosis/total number of assessed arteries.
Number of arteries with type A or B lesions/total number of arteries with arteriosclerosis.
Number of arteries with arteriosclerosis oriented to the surface epithelium/total number of arteries with arteriosclerosis.
The histopathological analysis was completed with dual-labeling immunohistochemistry (IHC) technique using smooth muscle actin (SMA) and collagen III markers for sheep tissues. Briefly, hydrated tissue sections were immersed in Tris-buffered saline (TBS; 0.05 M Tris HCl, 0.15 M NaCl, pH 7.4-7.6) for 10 minutes, repeated 3 times. Peroxidase activity was quenched using Bloxall™ endogenous peroxidase and alkaline phosphatase blocking solution (Vector Laboratories Inc, Burlingame, California) for 10 minutes. This step was followed by another 3 immersions in TBS for 10 minutes each. Sections were treated with 2.5% horse serum (Vector Laboratories Inc, No 30022) for 30 minutes to remove nonspecific tissue antigens. After discharging excess solution, the slides were incubated at 4°C overnight with rabbit anti-SMA antibody (Thermo Scientific, Spain, RB-9010) diluted 1/800 in TBS. Diluted normal rabbit serum and TBS replaced primary antibodies as routine controls for nonspecific reactions or residual endogenous peroxidase activity. Postincubation, 3 10-min TBS washes at room temperature (RT) were followed by covering the slides with ImmPRESS universal polymer peroxidase (Vector Laboratories Inc, No MP-7500) for 30 minutes. After a further 3 TBS washes (as described above), the sections were covered with the solution of ImmPACT DAB peroxidase substrate (Vector Laboratories Inc, No SK-4105) and incubated for 3 to 4 minutes at RT. The sections were then washed with distilled water for 10 minutes, followed by 3 changes of TBS. Sections were again treated with 2.5% horse serum (Vector Laboratories Inc, No 30022) in TBS for 30 minutes. After removing the excess solution, the slides were incubated at 4 C overnight with the rabbit anti-collagen III antibody (Abcam, Spain, ab7778) diluted 1/1500 in TBS. 8 It was followed by 3 TBS washes of 10 minutes, each at RT, and the slides covered with ImmPRESS universal polymer peroxidase (Vector Laboratories Inc, No MP-7500) for 30 minutes. After another 3 TBS washes, the sections were covered with the ImmPACT DAB VIP substrate solution (Vector Laboratories Inc, product No SK-4605) and incubated for 3 to 4 minutes at RT. Nuclear fast red (Vector Laboratories Inc, No H-3403) was used as a counterstain with 2 minutes of staining on these sections. Finally, the sections were washed with tap water, dehydrated, and mounted.
The rabbit polyclonal anti-human CD3 (DAKO code A0452) was used to identify T-lymphocytes in sheep. 8 In this case, only the first part of the previously described IHC procedure was applied. Normal sheep lymph node tissue sections were used for positive controls.
Nasal cavity samples were similarly processed from 4 Rasa Aragonesa sheep of the same age group (4–7 years old) as the 4 Latxa sheep investigated in this study. These sheep belonged to flocks located close to the Veterinary Faculty of Zaragoza. They were used as age-matched controls for normal nasal vascular microanatomy and had a low probability of contact with P. chartarum conidia. A dry climate is characteristic of this region, and pithomycotoxicosis has never been diagnosed in this area.
Results
Clinical Findings
The computerized tomography scans revealed increased soft tissue in the rostral nasal cavity with variable degrees of obstruction of the 3 meatuses in all sheep. Both nasal cavities were blocked with different degrees of severity. Obstruction was greater in the right nasal meatus in 3 sheep and in the left of 1 sheep (Fig. 1a–c). Intravenous injection of contrast did not result in the differentiation enhancement.
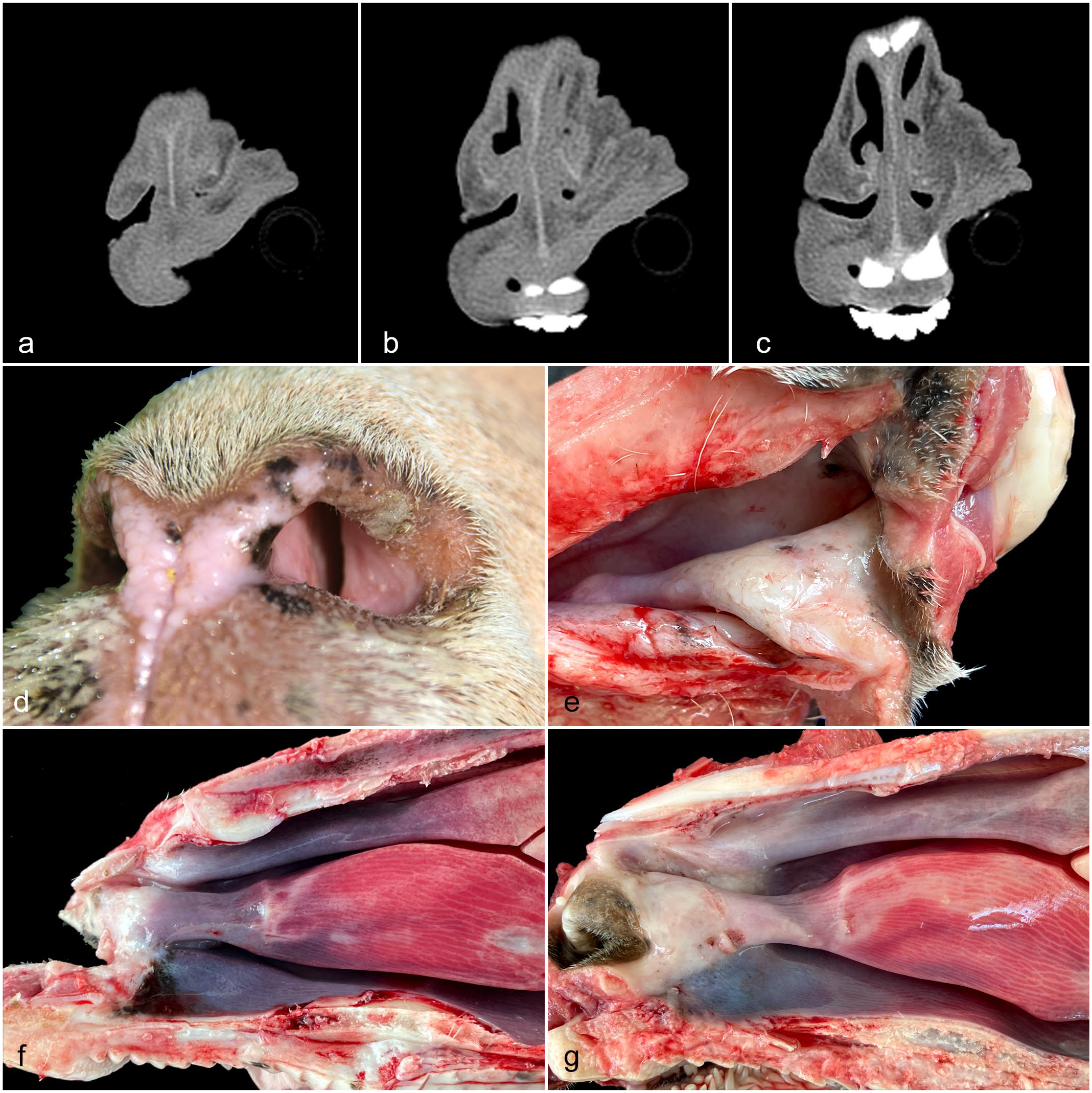
Computerized tomography results, clinical picture, and gross pathology of sheep with obstructive rhinopathy. (a, b, c) Sheep no. 4. Three cuts of the nasal cavity adjacent to the alar fold. Severe blockage of the nasal cavities. Computerized tomography scanner. (d) Sheep no. 4. Left nostril, showing whitish tissue narrowing the nasal lumen together with sero-mucous secretions. (e, f, g) Gross pathology of the nasal cavity. (e) Sheep No 4. Dorsolateral view of the right nasal cavity. Alar fold and adjacent areas of the nasal mucosa are thickened, pearly white color, and have rough surfaces. Spots of black color, interpreted as melanotic stains, are present on the dorsal conchae. (f, g) Sagittal section of the skull, rostral part of the right nasal chamber. (f) Sheep no. 2. Gross changes described in Fig. 1e are observed in small area on the alar fold. Spots of black color are present on the mucosa of the ventral fold. (g) Sheep no. 3. In this sheep, gross changes described in Fig. 1e extend from the alar fold to the dorsal, ventral nasal conchae, and ventral fold.
The 4 sheep presented a variable degree of nasal obstruction that led to inspiratory dyspnea and snoring on clinical examination. When observing the nostrils close to the vestibulum, it was possible to detect in all of them that the lumen was visibly restricted by whitish tissue narrowing the nasal meatus, and sero-mucous secretions were painting nasal orifices (Fig. 1d). The sheep were monitored for several weeks to observe their evolution after being removed from the contaminated pastures. The occlusion was preserved with different degrees of oscillation but with no apparent improvement throughout the observation period. No clinical evidence of photosensitization was observed in any of the studied sheep.
Hematology showed a slight degree of anemia in 2 analyzed animals with hematocrit values of 23.9% and 25%, respectively (normal values 27%-42%). However, leukocytes were within normal parameters. Blood biochemistry analysis detected elevations of GGT, ALT, and lipase in all sheep (Table 1). Gamma-glutamyl transferase showed higher values in animals with severe gross pathology lesions, but ALT did not show the same relationship. Lipase was also elevated, suggesting pancreatic damage. Serum bilirubin levels were slightly elevated (0.5-0.7 mg/dL), but close to the normal values (0.1-0.4 mg/dL).
Presence of P. Chartarum in the Grass
After incubation in a synthetic nutrient medium of the plant fragments, fungal colonies compatible with the morphology of P. chartarum were detected in the culture. In addition, conidia of P. chartarum were recovered and identified in the eluents of the 4 washed subsamples, at an average rate of 102 conidia/g of plant matter.
Necropsy Findings
Postmortem examination of the 4 sheep showed significant changes in nasal cavities, liver, and pancreas. Both nasal cavities from all sheep were investigated by opening the skull either at the sagittal plane following the projection of the nasal septum or by observing the rostral nose by dorso-lateral incision. Nasal mucosa showed significant alterations in the rostral nasal cavities in the 4 animals. The rostral segment of the dorsal nasal turbinate, alar fold, basal fold, and adjacent areas of ventral nasal turbinate showed changes with variable caudal extension. The nasal mucosa in these areas was pearl white, slightly elevated, and with patchy roughness on the surface. These lesions were more marked at the alar folds (Fig. 1e–g) (Supplemental Fig. S1). Changes described in the mucosa extended to the adjacent nasal septum. Gross lesions were present in both nasal cavities, although there was variation in the severity of lesions between them. In 2 sheep, dots or poorly demarcated black color areas, interpreted as melanotic stains, were observed on alar folds, ventral folds, and dorsal turbinate (Fig. 1e, f).
Gross lesions, which varied between individuals, were observed in the liver of 3 sheep. One sheep had atrophy and fibrosis of the left lobe (estimated 40% of reduction). In 2 sheep, the liver was severely atrophied (estimated 60%-70% of reduction), kidney-shaped, with a rough surface and hard consistency. According to liver gross pathology severity assessment published for long-term facial eczema, 2 were severe, 1 was moderate, and 1 was grossly normal. 22 Gallbladders in these 2 livers were reduced to a heavily fibrosed cordon that were displaced dorsally (Supplemental Fig. S2). The presence of a few Dicrocoelium dendriticum adults was recorded inside 2 gallbladders.
Histopathology, Histochemistry, and IHC
Liver and gallbladder
The histopathological examination of the livers confirmed the level of severity established on gross examination. Moreover, even the liver with no detectable gross lesions (classified as normal at gross examination) showed mild inflammation and fibrosis of portal areas under histopathological examination (Table 1). In larger bile ducts, parasitic structures (compatible with Dicrocelium dendriticum) were identified in the lumen, and the epithelium was hyperplastic and surrounded by inflammation (lymphocytes and eosinophils), together with fibrosis. No vascular lesions were observed in this case. The other 3 livers showed moderate or severe fibrosis (bridging fibrosis) of portal areas, moderate or severe biliary ducts hyperplasia, inflammation (lymphocytes and macrophages), and scattered foci of hepatocellular necrosis (Table 1). Vascular lesions were primarily present in blood vessels running together with interlobular bile ducts, and in more significant number and severity in the 2 livers with severe lesions (Fig. 2a). Intimal proliferation together with fibrosis and an increase of elastic fibers in arteries and veins were observed mainly in interlobular areas. Intimal proliferations were eccentrically located and orientated in parallel to the position of bile ducts (Fig. 2a, b). Affected vessels had increased elastic fibers or fragmented fibers combined with collagen and mild mononuclear infiltration (Fig. 2b; Supplemental Fig. S2). The gallbladder lesions were observed in the 2 sheep. The main changes were patchy epithelial necrosis or complete denudation, multifocal proliferation of mucous glands, and fibrosis of the muscular and adventitial tunic. No lesions like the ones recorded in the liver were observed in blood vessels of the gallbladder.
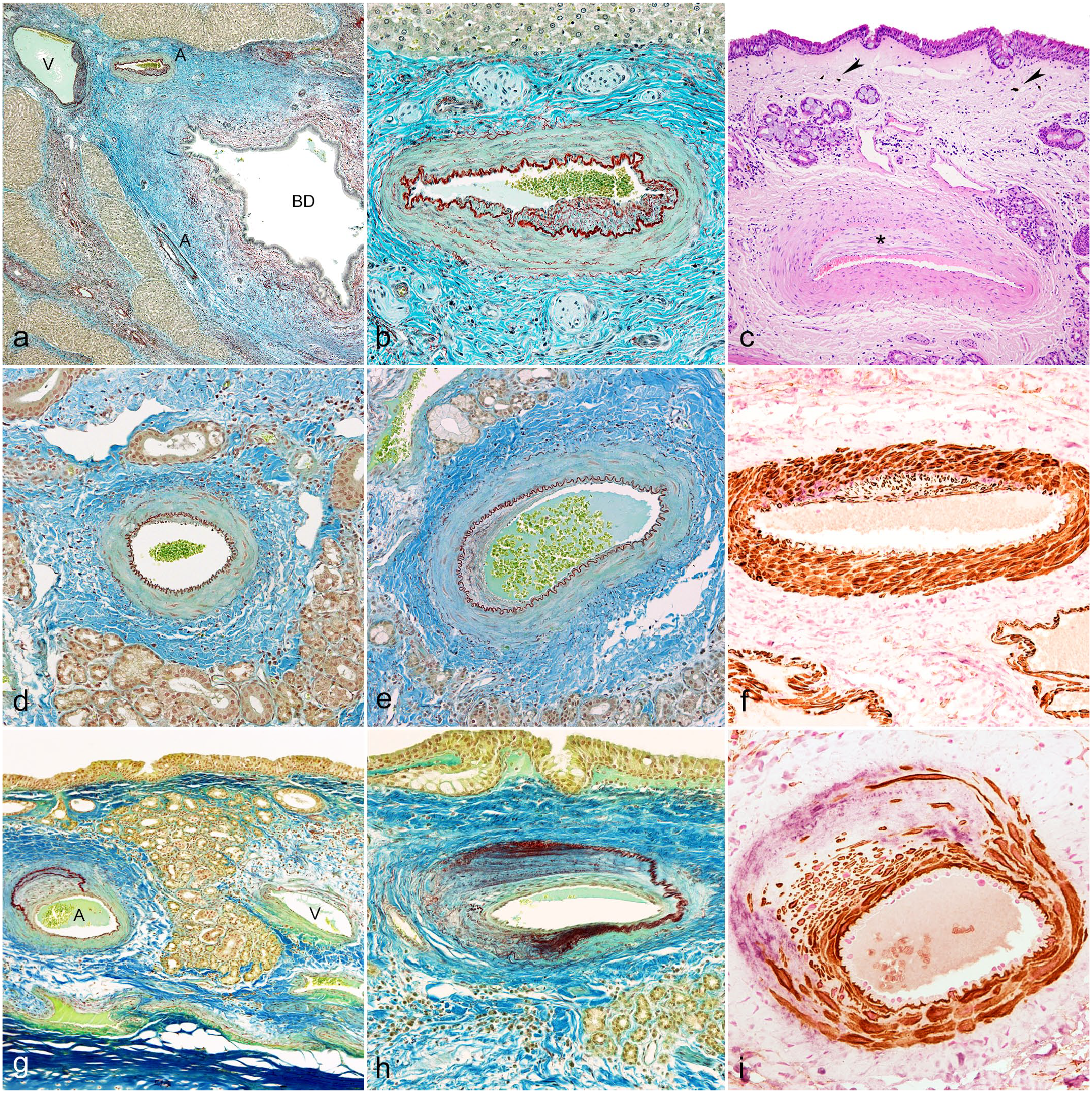
Histopathology and immunohistochemistry of the liver and the nasal mucosa. (a, b) Histopathology of the liver. Orcein-Cajal’s picrocarmin combination (a) Sheep no 3. Interlobular septum showing 1 vein (V), 2 arteries (A), and 1 biliary duct (BD). Arteries and vein show sclerosis and intense fibrosis with proliferation of collagen and elastic fibers of interlobular septa. Intima vascular degenerative lesions are orientated to damaged BD. There are few mononuclear cells, mostly surrounding dilated biliary duct. (b) Sheep no. 3. One artery showing eccentric intimal proliferation and increase of elastic and collagen fibers in all arterial layers. (c) Histopathology of the nasal mucosa. Sheep no. 4. Small artery located close to the surface of the nasal mucosa. Nasal epithelium remains unaltered. Intima of the artery shows eccentric proliferation (asterisk). Edema and fibrosis together with small groups of mononuclear cells, few neutrophils, and melanotic cells are present (arrowheads). Hematoxylin-eosin. (d, e) Histopathology of the arteries of the nasal mucosa lesion type A. Sheep no. 3. Orcein-Cajal’s picrocarmin combination. (d) Small artery with minimal intima proliferation and slight collagen proliferation. (e) Small artery with a more extensive proliferation of elastic and collagen fibers in the intima, media, and adventitia. (f) Immunohistochemistry of the arteries of nasal mucosa lesion type A. Sheep no 3. Small artery with intimal proliferation of smooth muscle cells and scarce intermingled collagen III. Smooth muscle cells are shown with brown labeling and collagen III with light purple. Double immunohistochemistry smooth muscle actin-collagen III. (g, h) Histopathology of the arteries of the nasal mucosa lesion type B. Sheep no. 4. Orcein-Cajal’s picrocarmin combination. (g) Nasal mucosa showing one artery with intense intimal proliferation together with herniation and duplication of internal elastic membrane (A). Vein showing minimal changes in the wall (V). (h) Small artery with severe intimal proliferation of fibromuscular cells and increased collagen and elastic fibers. The internal elastic membrane is enlarged, reduplicated, and fragmented, and narrows the vascular lumen. Fibrosis surrounds the vessel. (i) Immunohistochemistry of the nasal mucosa arteries, lesion type B. Sheep no. 4. Small artery with intense intimal proliferation of smooth muscle cells and collagen III. Smooth muscle cells are shown with brown labeling and collagen III with light purple. Double immunohistochemistry smooth muscle actin and collagen III.
Nasal mucosa
As we did not have detailed microscopic descriptions of the normal nasal mucosa of these areas, we studied nasal samples of 4 clinically normal Rasa Aragonesa sheep. Microscopic observation of the nasal mucosa of these sheep identified several small arteries (according to the criteria of the Nomina Histologica Veterinaria) that run in parallel near the epithelial surface. These arteries were between 80 and 300 µm in diameter, closer to the alar fold surface, and run deeper in caudal portions. The lining epithelium of this part of the mucosa started with as non-keratinized stratified squamous and transitioned to respiratory epithelium close to the beginning of the ventral nasal turbinate. The mucosa was rich in sero-mucous glands, and gland ducts embedded in slight, fibrillar connective tissue that contained variable numbers of small veins. Mononuclear cells and a few polymorphonuclear cells were in small number, mostly in the sub-epithelial zones or crossing the epithelium. Continuous cartilage was the main support of the mucosa and several sections of the lacrimal duct were observed because the nasolacrimal duct opens on the alar fold. We did not record any significant degenerative change in these arteries in any animal.
The nasal epithelium was unaltered in most of the sections. In 2 sheep, melanotic cells were scattered individually throughout the nasal mucosa or in small groups (Fig. 2c). Melanotic cells in the nasal mucosa may be considered unusual but normal if compared with humans. 32 Based on this, we considered that the presence of melanotic cells in one of our cases as unrelated to the disease we are describing, although, in our experience, melanotic cells in nasal mucosa are rare in the Rasa Aragonesa breed. No remarkable changes were observed in the sections of the crossing lacrimal duct.
In the 4 Latxa sheep with obstructive rhinopathy, lesions were primarily observed in small arteries running in parallel and close to the surface of the epithelium. Hematoxylin and eosin stain revealed intimal proliferation, mostly eccentric, in many of arteries accompanied by a variable degree of edema and fibrosis of the surrounding mucosa (Fig. 2c; Supplemental Fig. S3). Although isolated or small groups of mononuclear cells and few neutrophils were present, we did not find any clear evidence of inflammation, increased number of mast cells, or calcification of these vessels or in other areas of the nasal mucosa.
Orcein-picrocarmin histochemical method and IHC demonstrated degenerative changes in a significant number of the arteries, mainly the ones located on the alar fold. The percentage of arteries with arteriosclerosis ranged from 21.5% to 61.9%, and sheep with more severe liver lesions developed a higher percentage of arteriosclerosis (Table 1). We describe here 2 types of degenerative changes according to severity.
Type A: These arteries showed an increase of the intimal thickness with a variable percentage of the intima affected. Most lesions were eccentric and the internal elastic lamina was preserved. Endothelial cells showed focal hyperplasia and hypertrophy in these thickened areas and underneath fibromuscular components had variable degrees of expansion. Orcein-positive material interpreted as fragmented elastic fibers was found mixed with fibromuscular expansion in some of these lesions. The tunica media showed a slight increase of the blue stained fibers interpreted as collagen. Collagen fibers and orcein-positive fibrillar fragments were increased in the adventitia and surrounding connective tissue (Fig. 2d, e; Supplemental Fig. S4). Dual IHC detected a proliferation of SMA-positive cells in subintimal spaces together with collagen III depositions and in the muscular layer in type A lesion (Fig. 2f). CD3-positive cells were found close to the epithelium and small capillaries but seldom in arteries (data not shown). The percentage of type A lesions ranged from 13.1% to 22.5% of the arteriosclerotic arteries assessed (Table 1).
Type B: Arteries showing a severe alteration of the intima, internal elastic lamina, tunica media, and adventitial layer were included in this group. The internal elastic lamina had multiple changes including displacement, interruption, enlargement, or reduplication with substantial alteration of the arteries microanatomy. These changes were combined with more intense intima thickenings with a proliferation of fibromuscular cells and fibrillar connective tissue inducing considerable displacement of the intima layers. In most cases, these lesions were eccentric, but in others, they involved the entire intima of the vessel (Fig. 2g, h; Supplemental Fig. S4). The tunica media was partially or entirely replaced by fibrotic connective tissue, and the lumen was significantly narrowed (Fig. 2h; Supplemental Fig. S4). The adventitia showed similar changes like those described in type A lesion but with more intensity. Immunohistochemistry demonstrated the hyperplasia of SMA-positive cells and the muscular layer was effaced by the proliferation of fibrillar collagen III, narrowing the lumen of the arteries (Fig. 2i). CD3-positive cells were mostly found close to the epithelium and small capillaries but very rarely close to or in the arteries (data not shown). The percentage of type B lesions ranged from 8.4% to 49.7% of the arteries with arteriosclerosis (Table 1).
Type A and B degenerative changes were present in variable proportion in the 4 ewes. However, a high proportion of these degenerated arteries was orientated to the surface of the nasal epithelium (65.2%-81.2%; Table 1).
Lesions in small veins were challenging to assess with H&E. However, orcein-picrocarmin technique showed disorganization of the walls with a proliferation of elastic and collagen fibers replacing the muscular layer and peri-venous edema in some areas where type B lesions in arteries were found (Fig. 2g).
Other tissues
No lesions associated with facial eczema in the skin samples from the head were found in any sheep. A small increase in the mesangial cells and matrix together, and a few hyaline drops in Bowman’s spaces and proximal convoluted tubules together with mild degenerative changes were recorded in the kidneys. In the pancreas, locally extensive or multifocal necrotic changes of acinar cells were found. The locally extensive necrotic foci were mostly recorded close to damaged pancreatic interlobular ducts. Sloughing and loss of the duct epithelium at some points together with peripheral fibrosis were observed. Few macrophages and lymphocytes infiltrated these areas. In 2 cases, main pancreatic ducts were showing similar changes. In addition to locally extensive necrotic foci, a small number of multifocal necrotic foci of acinar cells were found scattered in the pancreatic lobules. They showed a center of homogenous hyaline material resembling the remains of intercalated pancreatic ducts. No significant changes were recorded in other organs.
Discussion
As described in the introduction, naturally occurring obstructive rhinopathy and pithomycotoxicosis coexist in sheep in the north of Spain (Basque Country).2,14 We have studied 4 ewes from a flock located in this region with a long history of pithomycotoxicosis that had reports from the sanitary authorities with evidence of P. chartarum spores in the grazing areas. In this study, we tested grazing areas for confirmation, and high levels of P. chartarum conidia were detected in the grass samples. Our clinical observations replicated those published in previous reports conducted in the same geographic area.2,14,23 Clinical examination, computerized tomography scan, and necropsy studies confirmed that the obstruction is present in the rostral nasal cavity close to the nasal vestibule, involving mainly the alar fold and adjacent anatomical structures. The 4 investigated animals showed high levels of GGT in the serum, and liver and gallbladder lesions consistent with those described in natural or experimental pithomycotoxicosis.9,12,22 Interestingly, lipase levels were high in these animals, and we have described necrosis of pancreatic acini related with ductal changes. However, the association between high lipase levels and histopathological changes is controversial. More natural cases would need to be studied to determine the significance of the pancreatic lesions and elevation of lipase levels in pithomycotoxicosis.
Therefore, obstructive rhinopathy found in the studied ewes confirmed that this disease seems to be strongly associated with pithomycotoxicosis. Notwithstanding, there are disagreements between our histopathology results and those of previous publications. In early descriptions, long-term inflammatory reaction, perivasculitis, and edema were the primarly recorded lesions, and the rhinopathy was associated with allergic and type III or IV hypersensitivity reactions.2,14,23 However, we have not found evidence of abnormal numbers of mucosa infiltrating T-cells, mast cells, eosinophils, vasculitis, or other histological changes generally associated with these types of reactions. 31 Mucosa nodules described in diseases due to type IV hypersensitivity like bovine nasal granuloma were not identified either. 5 In our study, the main change observed in the 4 animals was degenerative lesions in the particular net of small arteries of the alar folds and adjacent areas. Arterial lesions fulfill the criteria of arteriosclerosis, as is detailed in the veterinary literature. 26 We also have not found any calcification or presence of fat accumulations that are characteristic of other types of degenerative arterial lesions. 26 Therefore, allergic or hypersensitivity reactions do not seem to be associated with this rhinopathy because the pathological base we describe consists of degenerative lesions of small arteries together with fibrosis and edema. In addition, the epidemiological observations do not suggest this is due to allergy because this disease does not have a clear seasonal presentation coincident with the highest level of spores in the pastures. 2 Moreover, the 4 sheep investigated were kept away from the contaminated areas, and no improvement in clinical signs was observed.
Interestingly, pathological descriptions of the liver in natural cases of ovine facial eczema or in experimental intoxications of sheep with sporidesmin include severe damage of bile ducts and associated blood vessels, which are considered of great importance in pithomycotoxicosis cases.7,11,12,18,20 In natural cases of pithomycotoxicosis in sheep, a striking fibro-occlusive lesion consisting of eccentric intimal thickening of the hepatic arteries and portal veins has been recorded.11,12,18 Likewise, in experimental cases, similar lesions were observed in sheep receiving sporidesmin doses from 0.05 to 1 mg/kg body weight at 26 to 28 days.7,20 These vascular lesions were more marked in vessels adjacent to larger bile ducts.11,20 Transforming growth factor-β (TFG-beta) immunostaining was observed in hepatic arterial vessels, and this cytokine plays a role in vascular wall response to fibrosis.6,27 Similar, but less-severe, lesions have been described in experimental intoxication in cows and goats.6,29
Arteriosclerosis in ruminants has been described in relation to hypertension in coronary arteries in long-term bovine laminitis, spontaneously occurring in intramural coronary arteries in calves and beef cattle, in the myocardium of calves with hypoxia-induced pulmonary hypertension, and in large arteries of old ruminants.1,3,21,26 Although hypertension, age, and inflammation can be related to arteriosclerosis in ruminants, the similarities between the hepatic vascular lesions in pithomycotoxicosis and lesions found in the nasal cavity are the basis to suggest common pathogenic mechanisms that not associated with these other factors.
The active biologic center of sporidesmin is the sulfur-bridged dioxopiperazine ring present in other families of fungal metabolites called epipolythiodioxopiperazines (ETPs). 13 Sporidesmin seems to be directly toxic, and its metabolites are either less toxic or inactive. 15 The molecular mechanisms described are oxidative stress generation, direct interaction with proteins, and binding to zinc atoms of some metalloproteins.13,15 Necrotic cell injury, perturbation of some metabolic pathways and mitochondria, and changes in cell adhesion leading to altered permeability of epithelial and endothelial layers seems to be induced by this fungal toxin.4,15 Sporidesmin is very active locally and concentrates the damage in the bile ducts and gallbladder. The toxin appears to cause degenerative lesions and fibrosis in blood vessels running close to the damaged bile duct, and these vascular lesions are found orientated to the bile duct. 15 In the same way, we can speculate that arteriosclerosis and fibrosis observed in the rostral part of the nasal cavity may be related to the direct action of the sporidesmin when these animals are grazing on contaminated pastures. As we have described in healthy sheep, the nasal mucosa of the nasal cavity of this area showed several small arteries running in parallel and located very close to the epithelium. The nasal epithelium is basically respiratory or nonkeratinized squamous, offering less resistance to the absorption of substances. We have also investigated the nostrils and caudal part of the nasal turbinates and arteriosclerosis, edema, and fibrosis were seldom or not observed at all. Perhaps the microanatomy of this part of the nasal mucosa, joined to close contact with the sporidesmin could play a role in the development of this rhinopathy that occurs particularly in this area. In fact, our assessment of the orientation of arteriosclerotic lesions in the nasal mucosa resulted in many arteries orientated to the surface epithelium, similar to the observation that vascular lesions of the liver were orientated to damaged bile ducts described by us and other authors (Table 1). 15
A critical point in the discussion about the obstructive rhinopathy reported here is that it has not been described in other countries where facial eczema is common or in other areas of the north of Spain with similar climatic conditions.9,12,25 Therefore, other factors such as climate, topography, special management practices, or breed susceptibility may be acting in the pathogenesis of the disease. In this regard, the sensitivity to sporidesmin varies according to species, with goats and cattle being more resistant than sheep.9,29 Likewise, experimental evidence indicated differences concerning the sheep breed.28,30 Latxa dairy sheep breed and Manech à-tête rousse were mostly affected in the early descriptions and in our cases.2,14,23 Therefore, breed susceptibility to sporidesmin seems to be an important factor in developing this rhinopathy.
Sheep flocks from this particular geographic area are managed by grazing on the same pastures for a long productive period. Basque Country has a wet and mild climate and P. chartarum is very common in sheep grazing areas.2,14,16 The geography is very hilly and cases are mainly registered in flocks located below 400 m. 2 A combination of factors such as a high concentration of spores, particularly in autumn, short grass, high concentration of sporidesmin sensitive animals in the pasture, and particular management practices may be essential for the development of this syndrome in this geographic area. We can speculate that in these situations, the nares are very close to the contaminated grass, and when eating and breathing, some of these spores can spend enough time to liberate the toxin and cause damage in the more rostral portion of the nasal cavity, with a preference for the alar folds and adjacent areas.
The information contained in this paper would increase the range of organs or tissues that can be damaged by sporidesmin. Thus, obstructive rhinopathy should be added to the list of clinical disorders related to this toxin, such as liver cirrhosis or facial eczema. This fact supports using pithomycotoxicosis to name the disease instead of facial eczema, as it is commonly called.
Future work will have to develop more epidemiological, experimental, and pathological research to fully understand the relationship of this obstructive rhinopathy with pithomycotoxicosis in sheep.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221109095 – Supplemental material for Chronic pithomycotoxicosis associated with obstructive rhinopathy in sheep
Supplemental material, sj-pdf-1-vet-10.1177_03009858221109095 for Chronic pithomycotoxicosis associated with obstructive rhinopathy in sheep by Marcelo De las Heras, Delia Lacasta, Raúl A. Reséndiz, Ane Rivas, Ane Garzianda, Ricardo de Miguel, Héctor Ruiz, Enrique Castells, Vicente González and Luis M. Ferrer in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to acknowledge the farmers from the Farm Premietxeberri for allowing us to research this case and Rosario Puyó and Santiago Becerra for their inestimable technical assistance.
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research group: Welfare and pathology of small ruminants. Universidad de Zaragoza. D Lacasta, LM Ferrer, and V González belong to: Instituto Agroalimentario de Aragón IA II.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
