Abstract
Hepatic lipidosis is a common disease of captive bearded dragons (Pogona vitticeps). Diagnosis, prevention, and treatment of this condition are challenging, as there is minimal information in the literature. Our study determined the prevalence and epidemiological risk factors associated with the grade and severity of hepatic lipid changes in bearded dragons submitted for necropsy in 2 North American institutions. A total of 571 postmortem cases were retrieved, and from each pathology report the demographic data (age, sex) and the list of final diagnoses were extracted. For each case diagnosed with hepatic lipidosis, the archived sections of liver were reviewed and the severity of lipid change was stratified using a standardized histologic grading system. Descriptive statistics were used to estimate the prevalence of each grade and severity class. Associations between grade and severity, as well as demographic data and concurrent diseases, were explored using ordinal logistic regression analysis. On multiple logistic models, the occurrence of infectious disease and neoplasia was associated with decreased grade and severity of hepatic lipid changes, while the female sex and adult age were associated with an increased grade and severity. None of the other variables were significantly associated with hepatic lipid changes. These results suggest that reproductively active females and adult bearded dragons are predisposed to increasing hepatic lipid changes, while those with an underlying disease process have reduced hepatic lipid accumulation and changes, possibly due to increased fat catabolism. Data in this study can serve to benchmark the prevalence of hepatic lipidosis in bearded dragons and allow further investigations.
Hepatic lipidosis is a frequently reported debilitating condition that affects several reptilian species including the bearded dragon (Pogona vitticeps), a very popular pet worldwide.6,20 The basis of this condition involves progressive accumulation of triacylglycerols within hepatocytes resulting in impaired hepatic metabolism and function, and dyslipidemic changes.4,7–9,21 Classic clinical signs in pet reptiles are nonspecific and include progressive and chronic anorexia, lethargy, weight loss, and ultimately liver failure and death.7,12,14,23 Reptile lipid metabolism mainly occurs in the liver, with limited lipogenesis in the coelomic fat pads, while mammals mainly utilize the adipose tissue. 17 This difference may be relevant to the etiology behind the increased susceptibility to hepatic triacylglycerol accumulation. Currently, the prevalence and risk factors of this disease are unknown. High-fat and carbohydrate diets, deficiency in lipotrophic dietary factors, excess vitellogenesis in females, decreased activity, low environmental temperatures, lack of hibernation of temperate species, hypothyroidism, hyperparathyroidism, systemic illness, and chronic stress are considered potential contributing factors to hepatic lipidosis in captive reptiles.4,5,7,8,14,16,18,22,23 Some reptile veterinarians are of the opinion that nonbreeding females chronically store lipids in their livers in anticipation of reproductive activity, thus exceeding their metabolic ability to prevent hepatocyte damage. 23 These reproductive abnormalities may be secondary to abnormal photoperiods and temperatures, as well as long-term isolation of induced ovulators. 14
Most reptiles are intermittent feeders or undergo brumation, and are accustomed to various periods of anorexia, making anorexia unlikely to be a predisposing factor as in cats and guinea pigs.3,17 Reptiles have mechanisms in place for these periods of fasting as they are able to reduce their metabolic rate with prolonged fasting, as demonstrated in some snake and turtle species.2,3,17 However, the pathogenesis of hepatic lipidosis and the reasons for its frequent diagnosis in captive bearded dragons are poorly understood, though the lack of hibernation for use of stored lipid may play a role.
Several cases of hepatic lipidosis suspected to be secondary to poor diet and husbandry have been reported. 26 Based on ecological studies, an adult bearded dragon should consume a diet composed of 50% to 90% vegetation and 10% to 50% adult insects.13,15 These insects should be fed a high calcium diet (gut-loading) and dusted with pure calcium prior to being consumed by the bearded dragon.4,18 However, many bearded dragons that present to veterinarians for evaluation consume high-fat larval insects (18%–29% crude fat)10,11 throughout their lives. This chronic increase in dietary fat results in increased absorption of fatty acids, which are transported to the liver for lipogenesis to produce triacylglycerols.17,23 At the time of diagnosis, husbandry and diet change alone seem relatively ineffective at reversing hepatic changes and clinical signs at a reasonable pace in most cases. 7
Despite hepatic lipid accumulation and changes being commonly observed in bearded dragons, there is a considerable gap of knowledge in the epidemiology and predisposing factors for these changes. While several risk factors have been proposed in the literature, these hypotheses have not been tested.4,5,7,8,14,16,18,21,23 Therefore, determination of risk factors would help identifying husbandry practices that could mitigate the progression to hepatic lipidosis and severity of disease at the time of veterinary intervention. A diagnosis of hepatic lipidosis is not possible based on the current knowledge of the disease, and so the association between demographic variables and a recently developed grading scheme 1 that considered the microscopic changes of hepatic lipid accumulation in this species was evaluated. The objective of this study was to estimate the prevalence of each hepatic lipid change grade and severity class based on the established grading scheme, as well as the basic epidemiological risk factors associated with hepatic lipid change in pet bearded dragons submitted for necropsy at the Animal Health Laboratory (AHL) and the Zoo/Exotic Pathology Service (ZEPS). We hypothesized that there would be a greater than 20% prevalence of cases classified with moderate to severe lipid changes, and that these changes would be more common in females, older animals, and those with concurrent reproductive diseases.
Materials and Methods
Inclusion Criteria
From the cohort of cases used for the development of the grading scheme, pathology reports of bearded dragons (P. vitticeps) submitted to the AHL and the ZEPS from 1998 to 2018 were retrieved. Biopsy cases were excluded as these cases did not have known comorbidities to determine risk factors and the hepatic biopsy samples may not have been representative of the whole liver. For further inclusion in the study, archived liver histology slides for any case diagnosed with lipidosis had to be available for review, to have a homogeneous assessment of the lesions over time and between institutions. All bearded dragon necropsy cases that were not diagnosed with hepatic lipidosis on initial histologic evaluation of the liver were included without further evaluation of the histology slides due to the large number of cases. However, a subset of these nonlipidotic cases (n = 55) from the AHL were evaluated histologically. Cases that were not diagnosed with hepatic lipidosis in the original histopathology report, and for which slides were not retrieved, were given a grade of 0. All of the other cases underwent histology review, and the changes associated with hepatic lipid change were scored based on a previously described microscopic grading scheme (see below).
Slide Evaluation
The liver sections from 252 cases stained with hematoxylin and eosin were reevaluated and assessed for 2 semiquantitative microscopic parameters and 1 quantitative microscopic parameter (Supplemental Table S1), using a histopathology grading system previously developed from the same cohort of cases. 1 A total of 197 cases were originally diagnosed with hepatic lipidosis, and 55 cases did not have a diagnosis of hepatic lipidosis from the AHL. Upon review, cases were stratified according to the final histological grade (0–12) and severity score (mild, moderate, severe; Supplemental Table S1).
Database Formatting
For each case in the cohort, the identification number, institution of submission, demographic data (age and sex), and the final diagnoses provided in the pathology report were imported into a spreadsheet. Each disease process listed as a final diagnosis was categorized by anatomic location, by giving a binary code for the affected (1) or not affected (0) organ systems (hepatic, reproductive, cardiovascular, renal, neurological, dermatological, respiratory, gastrointestinal, and endocrine). In addition, the presence (1) or absence (0) of hepatic lipidosis, atherosclerosis, infectious diseases, and neoplasia, independent of the body system affected, was also recorded. These disease processes were included separately as they are known to affect the metabolism of bearded dragons. Further differentiation of the infectious etiology or type of neoplasm was not recorded.
Statistical Analysis
Missing data handling
Data for age, sex, or both were missing in 31.9%, 23.1%, and 14.5% of cases, respectively (unknown or unreported on submission sheets or necropsy reports), and were assumed to be missing at random. To limit selection bias and loss of information associated with listwise deletion (default for statistical software), the missing values were dealt with via a multiple imputation procedure. Five data sets with imputations for missing values were generated by predictive mean matching using chained equations with the R-package “mice.” 4 Imputation diagnostics were performed by comparing the distribution of the imputed data with the complete data. The prevalence of final histological grades and severity tiers, using reevaluated cases, was calculated from necropsy cases with their respective binomial confidence intervals ([CIs] asymptotic method).
Univariate ordinal logistic models were first performed using the final histological grade and severity classification of hepatic lipid change as outcome variables (in different models), and the following predictors: age, age as a nominal variable (juvenile [less than 18 months] vs adult), 18 sex, and concurrent disease categories, as described above. As simply adding variables significant on the univariate models to a multiple logistic model has been shown to be erroneous, 25 all initial variables were added to a multiple ordinal logistic model instead. Subsequent nonsignificant variables were eliminated using a backward elimination strategy. All ordinal logistic regression models were fitted with the 5 data sets (resulting from multiple imputations for missing data) simultaneously with model parameters being automatically combined using Rubin’s rule by the “mice” R-package. 19 Associations between hepatic lipid change histological grades, severity classification, and explanatory vafriables were reported as proportional odds ratio (OR) with 95% CIs by exponentiation of the parameter estimates of the ordinal logistic models. For all tests, an α of 0.05 was used for statistical significance, and statistical analysis was performed using R (R Version 4.0.3, 2020, R foundation for statistical computing, Vienna) and related programming packages for multiple imputations (“mice”), 5 ordinal logistic models (“ordinal,” Christensen RHB, Version 2019-12-10), and plots (“ggplot2”). 27
Results
Inclusion Criteria and Slide Evaluation
A total of 809 pathology reports were initially collected from the AHL and the ZEPS. Of these, 571 cases met the inclusion criteria and were used to determine the risk factors and prevalence associated with the grade and severity of hepatic lipid change. One hundred and ninety-seven cases were diagnosed with hepatic lipidosis in the original report and were reevaluated histologically according to the grading scheme. 1 Three hundred and seventy-four cases were not diagnosed with hepatic lipidosis in their original reports: 319 of these were scored 0 for final grade and severity tier, while 55 cases were reevaluated and graded. 1
Descriptive Statistics
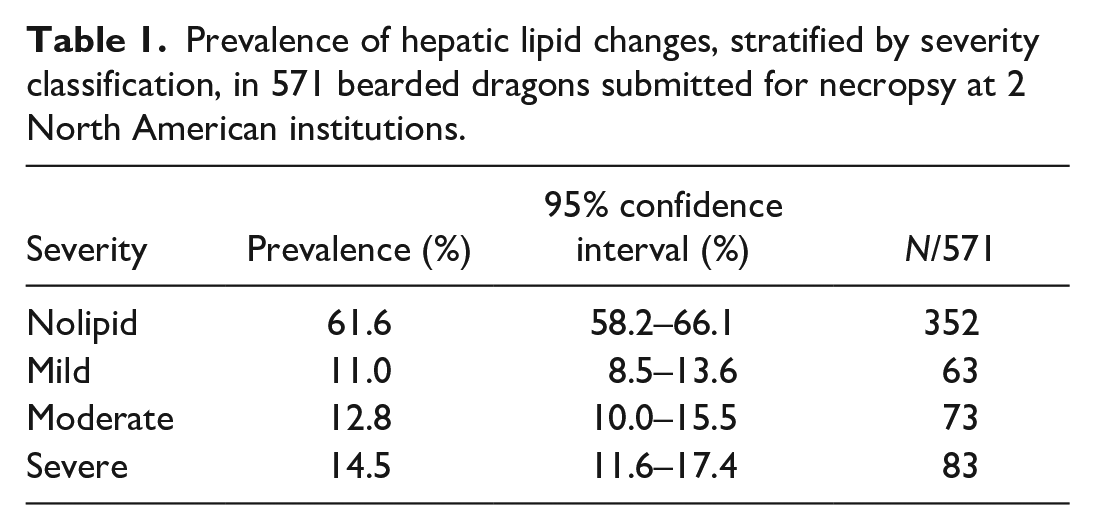
The median (interquartile range) reported age was 3 (4) years. There were 231 females, 208 males, and 132 bearded dragons of unknown sex. The proportion of cases in each severity category of the disease is shown in Table 1, and stratification by grade of hepatic lipid change is presented in Table 2. Severe (grades 8–12) hepatic lipid change was the most common category (prevalence 14.5% and CI 11.6%–17.4%) found in this cohort.
Prevalence of hepatic lipid changes, stratified by severity classification, in 571 bearded dragons submitted for necropsy at 2 North American institutions.
Prevalence of hepatic lipid changes, stratified by histologic grade, in 571 bearded dragons submitted for necropsy at 2 North American institutions.
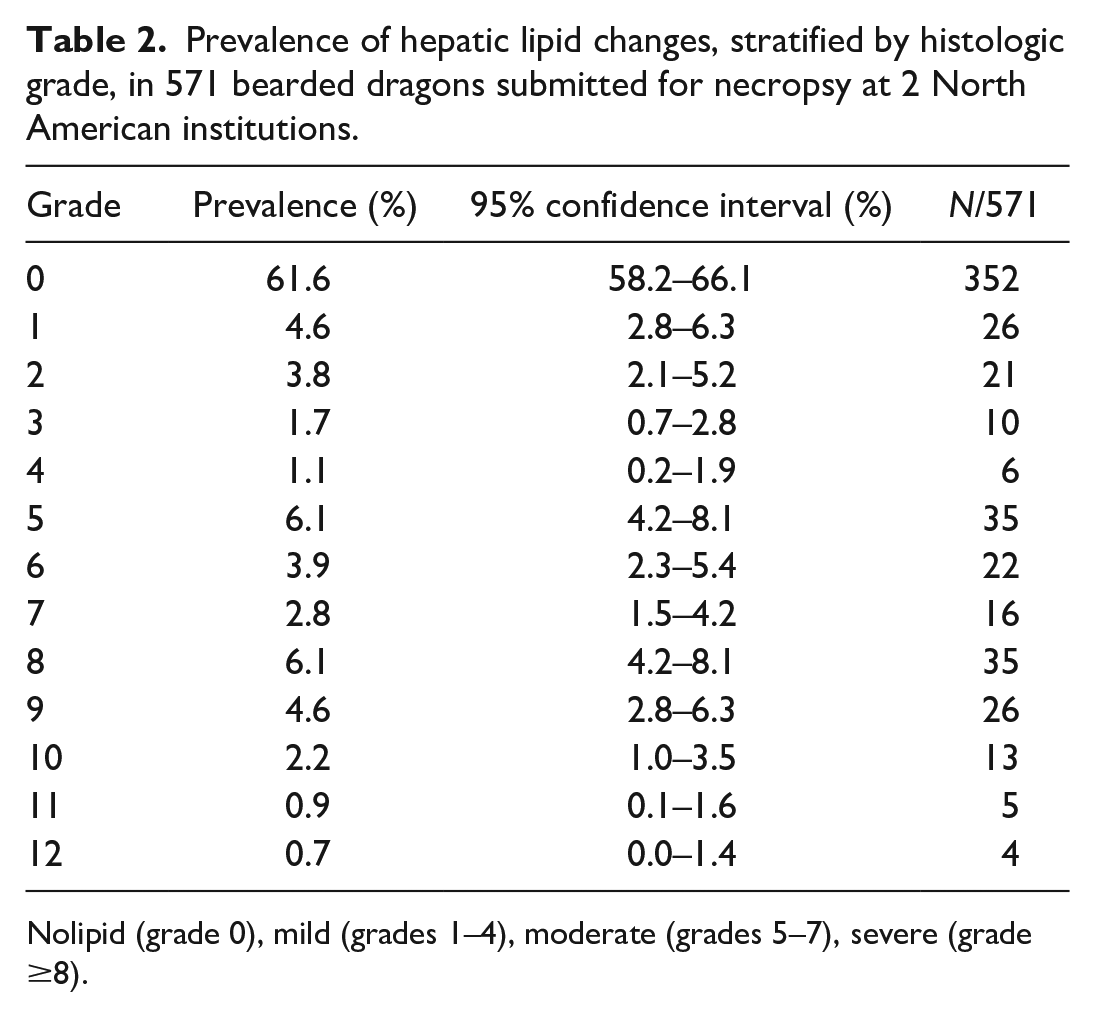
Nolipid (grade 0), mild (grades 1–4), moderate (grades 5–7), severe (grade ≥8).
Logistic Regression and Risk Factor Analysis
On the univariate models, females had 43% higher odds of having an increased severity classification than males (proportional OR: 1.43/severity class, 95% CI: 1.01–2.04, P = .048; Fig. 1), and 43% higher odds of having an increased lesion grade (proportional OR: 1.43/grade, 95% CI: 1.01–2.04, P = .049; Fig. 2). Adults also had a 2.5-fold increase in odds of having higher severity classification than juveniles (proportional OR: 2.52/severity class, 95% CI: 1.70–3.73, P < .001; Fig. 1) and having a higher lesion grade than juveniles (proportional OR: 2.56/grade, 95% CI: 1.73–3.78, P < .001; Fig. 2). Univariate models also showed that concurrent gastrointestinal (proportional OR: 0.51/severity class, 95% CI: 0.36–0.71, P < .001) or infectious (proportional OR: 0.35/severity class, 95% CI: 0.25–0.49, P < .001) diseases decreased the odds of having a higher severity class by 51% and 66%, respectively. Likewise, the same variables decreased the odds of having a higher lesion grade (gastrointestinal: proportional OR: 0.49/grade, 95% CI: 0.35–0.69, P < .001, infectious: proportional OR: 0.34/grade, 95% CI: 0.24–0.48, P < .001). All other variables were not significantly associated with hepatic lipid change grade or severity on necropsy using the univariate models (P > .05).
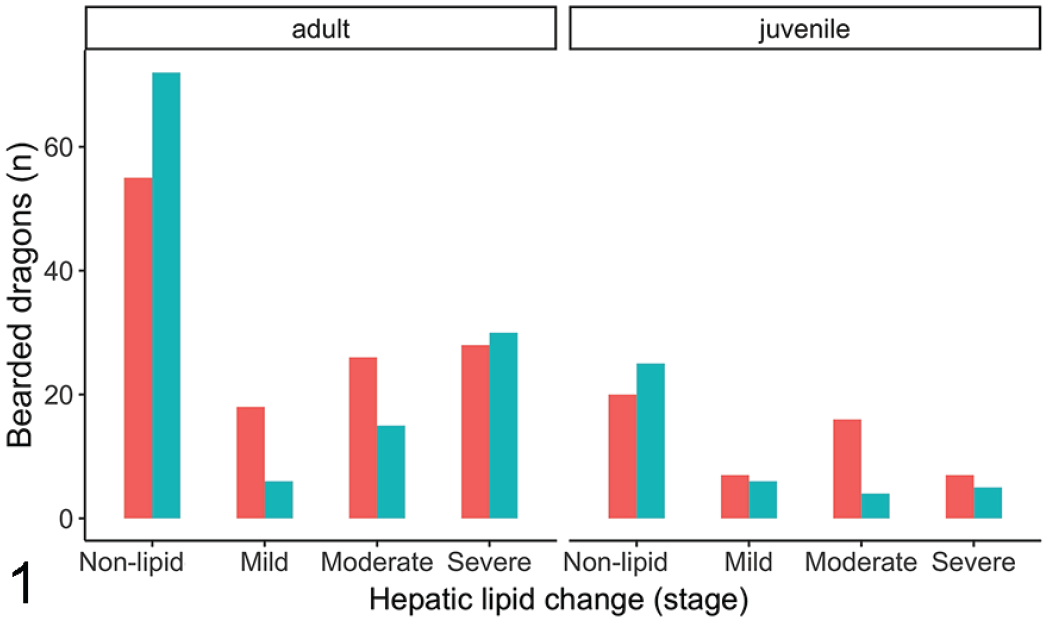
Bar graph of the numbers of bearded dragons with different severities of hepatic lipid change across sexes and age. Unknown sex was excluded. Sex: red—females, teal—males.
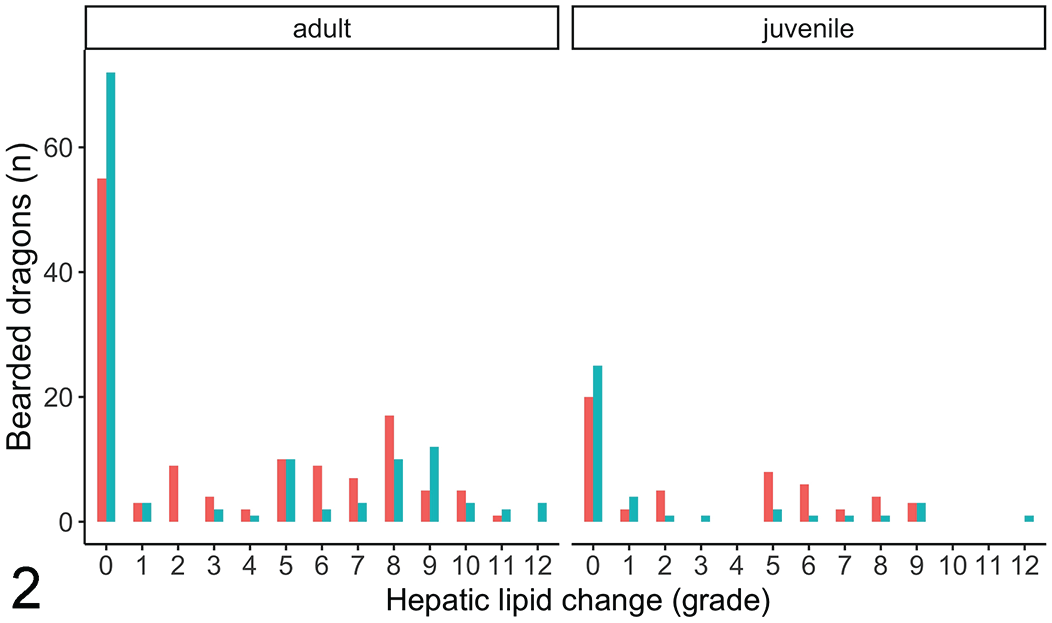
Bar graph of the numbers of bearded dragons with different histologic grades of hepatic lipid change across sexes and age. Unknown sex was excluded. Sex: red—females, teal—males.
On multiple logistic models, the variables sex (proportional OR: 1.44/severity class, 95% CI: 1.01–2.08, P = .047) and age (juvenile vs adult; proportional OR: 2.11/severity class, 95% CI: 1.36–3.28, P = .001) showed a positive association with hepatic lipid change severity, though mild for the female sex. On the other hand, infectious disease (proportional OR: 0.36/severity class, 95% CI: 0.25–0.53, P < .001) and neoplasia (proportional OR: 0.42/severity class, 95% CI: 0.25–0.70, P < .001) showed a significant negative association with hepatic lipid change severity. A similar model showed that sex (proportional OR: 1.45/grade, 95% CI: 1.02–2.06, P = .042) and age (juvenile vs adult; proportional OR: 2.12/grade, 95% CI: 1.38–3.26, P = .001) had a positive association with hepatic lipid change grade, while infectious (proportional OR: 0.36/grade, 95% CI: 0.25–0.52, P < .001) and neoplastic (proportional OR: 0.43/grade, 95% CI: 0.26–0.72, P = .004) diseases had a significant negative association with the grade of hepatic lipid change.
Discussion
Results of the present study indicate a high prevalence (14.5%) of severe hepatic lipid change in pet bearded dragons of North America. The estimated prevalence for mild and moderate lesion severity was 11% and 12.8%, respectively. Assessment of risk factors indicated that the variables female sex and adult age were associated with higher hepatic lipid change grade and severity, while infectious disease and neoplasia were associated with a lower hepatic lipid change grade and severity. All other variables were not significantly associated with hepatic lipid change grade or severity. These results suggest that females are mildly predisposed, likely due to the physiology of follicular development and vitellogenesis, and that adult bearded dragons are more predisposed to this condition compared with juveniles, likely due to the latency period needed for hepatic lipid change. In addition, underlying disease processes that increase fat catabolism may reduce the prevalence of increased hepatic lipid change.
In initial modeling, gastrointestinal lesions and infectious diseases were found to be strong negative predictors of hepatic lipid change. However, when assessed together with other variables, only infectious diseases and neoplasia were found to be strong negative predictors of hepatic lipid change in bearded dragons, possibly because of increased catabolism of fat during these disease processes or prolonged anorexia leading to the use of hepatic lipid stores. Confounding variables, which cannot be accounted for due to the retrospective nature of the study, may influence the associations between infectious disease and neoplasia with the prevalence and severity of hepatic lipid changes. Overall, these results suggest that when bearded dragons have an advanced underlying disease process such as a systemic infection or neoplasia, they may become cachectic and have an increased metabolism or catabolism of fat reducing the prevalence of hepatic lipid accumulation and changes. Alternatively, these individuals with an infectious disease or neoplasia could be chronically anorexic leading to chronic weight loss and remobilization of hepatic triacylglycerols. Since reptiles store their excess energy as lipid and metabolize it to meet their energy demands, 17 it is physiologically possible for a reduction of hepatic lipid during increased energy requirements such as during an infectious process or neoplasia. This ability to metabolize fat when needed raises the question of whether bearded dragons maintained in captivity as pets are generally overfed and have reduced activity? 4 It is plausible that hepatic lipidosis is mainly caused by a chronic increase in dietary fat intake in captive dragons subsequently leading to triacylglycerol accumulation in the liver, compounded by impaired lipid metabolism (eg, triacylglycerol overproduction from both de novo fatty acid synthesis and plasma nonesterified fatty acids, decreased β-oxidation, and insulin resistance). However, the pathogenesis of hepatic lipidosis has not clearly been identified and so anorexia leading to peripheral triacylglycerol hydrolysis and fatty acid mobilization may play a role, albeit likely minor, as in mammals.
Sex was found to be mildly significant with females more likely to have hepatic lipid change than males, and more significant increase in lesion grade and severity classification. This sex affect is also evident when inspecting graphs of data (Figs. 1, 2). Female reptiles and other sauropsids undergo vitellogenesis during the reproductive season for egg production. 17 This is an estrogen-controlled process whereby vitellogenin, a protein carrying a small amount of phospholipid, and very-low-density lipoproteins, carrying a large amount of phospholipids, are synthetized by the liver for transport to the ovary for follicle formation. 17 Sources of lipid for this synthesis can originate from ingested nutrients, lipids stored in the liver, or from the fat bodies. 17 This active process likely occurs in healthy females, increasing the lipid content of their livers during vitellogenesis. It is possible that those presenting for necropsy had succumbed to hepatic lipidosis or other diseases and the females were not reproductively active in this state, reducing the statistical significance of this sex. However, it is also plausible that females are more predisposed, but the effect is not as significant, as other factors play a larger role in the pathogenesis.
The median age of bearded dragons presented for necropsy was 3 years. Adults, greater than 18 months of age, were found to be predisposed to increased hepatic lipid change (Figs. 1, 2) likely due to prolonged exposure to nutritional imbalances and inadequate husbandry, which are considered key factors in development of hepatic lipidosis. 26 In addition, there is likely a metabolism shift between juveniles and adults, as juveniles require more calories as they grow. Due to the retrospective nature of this study, risk factors such as diet, anorexia, and husbandry were not evaluated as access to the medical files was not associated with the pathology reports. A multicenter retrospective or prospective study involving cases with husbandry information and histopathology reports would be beneficial to understand these risk factors.
Sampling bias could be present in this study as only cases where the liver was submitted were included. It is possible that some primary veterinarians completed the necropsy in-house and did not include liver samples for histology if they believed that the liver looked normal or if they did not think the lipidosis was significant to the case. This could artifactually increase or decrease the prevalence of increased hepatic lipid change, respectively. In addition, all previously diagnosed hepatic lipidosis cases were evaluated prospectively to remove variability such as diagnostic drift, but similar evaluation was not completed for the large number of cases that were not diagnosed with hepatic lipidosis. It is possible that some of these cases had some degree of hepatic lipid change, thus reducing the power of this study. Another limitation of this study is that a grading system, which was not clinically validated, was implemented. The cut-off for hepatic lipid change in the diagnosis of hepatic lipidosis is still unknown and so determining the overall prevalence of this disease was not possible.
Conclusions
In conclusion, this retrospective study shows a high prevalence of severe hepatic lipid change in pet bearded dragons presented for necropsy at the AHL and the ZEPS. Variables that were significantly associated with hepatic lipid change grade or severity classification on necropsy were infectious disease and neoplasia. These variables were found to be strong negative predictors of hepatic lipid change and authors suspect that when bearded dragons have an underlying disease process they have an increased metabolism or catabolism of fat thereby reducing their prevalence of hepatic lipid change. Prospective data would be required to further evaluate this theory. Females also appear to be predisposed; however, this effect is not significant when accounting for comorbid diseases and would have to be confirmed with prospective studies.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221105058 – Supplemental material for Prevalence and risk factors of hepatic lipid changes in bearded dragons (Pogona vitticeps)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221105058 for Prevalence and risk factors of hepatic lipid changes in bearded dragons (Pogona vitticeps) by Trinita Barboza, Leonardo Susta, Drury Reavill and Hugues Beaufrère in Veterinary Pathology
Footnotes
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
