Abstract
Objectives:
Nonalcoholic fatty liver disease is one of the emerging liver diseases affecting 20%–30% of the population creating a burden on public health worldwide and has been associated with the causation of multiple diseases. Besides exercise, several drugs are being used in patients based on clinical evidence especially vitamin E, a potent antioxidant to reduce the oxidative stress responsible for the development and progression of nonalcoholic fatty liver disease. This study aims at evaluating the effect of exercise and pharmacotherapy on nonalcoholic fatty liver disease.
Design:
A prospective follow-up study with purposive sampling was done at a liver clinic for 3 months. Baseline characteristics such as anthropometric measurements and biochemical parameters were recorded and compared after 3 months to determine the effect of therapy. Descriptive analysis using a parametric test was used to assess the change in biochemical parameters and a non-parametric test was applied to find out the association between non-pharmacological and pharmacological approaches.
Results:
Out of 177 patients, 67.2 % were male and 32.8% were female with the mean age ±SD of 46.8 ± 12.06 years. Mean ± SD weight and body mass index of the patients were changed from 74.88 ± 11.61 kg to 72.37 ± 11.61 kg and from 28.41 ± 4.02 kg/m2 to 27.31 ± 4.58 kg/m2 respectively which was found to be statistically significant. There was a significant change in all the biochemical parameters with the p-value < 0.05 through both non-pharmacological and pharmacological approaches.
Conclusion:
Nonalcoholic fatty liver disease management through exercise and pharmacotherapy shows significant improvement in biochemical parameters indicating that alone or in combination with both approaches play an effective role in treating nonalcoholic fatty liver disease.
Introduction
Nonalcoholic fatty liver disease (NAFLD) is defined as the excess accumulation of fat within the hepatocytes. It has affected a wide proportion of the world’s population affecting 30% of Americans and the Western world.1,2 In India, the overall prevalence ranges from 9% to 53%.3–5 Also, in Nepal, the prevalence was found to be 17%. 6
NAFLD, a leading prevalent disease comprises a wide spectrum of liver diseases ranging from simple steatosis to a more severe form known as non-alcoholic steatohepatitis (NASH) which may further progress to cirrhosis and hepatocellular carcinoma. 7 It affects not only the liver but also the multi-organ system best characterised by several cardiovascular diseases along with or without other metabolic syndromes. 8 Similarly, NAFLD is also closely linked with extra-hepatic cancers, especially, bladder cancer which is a most severe cancer in the elderly. 9
A study by Videla et al. 10 showed that insulin resistance (IR) and oxidative stress play a critical role in the development of NAFLD and seem to be multifactorial. Obesity, Type II diabetes mellitus (T2DM), and dyslipidemia along with cardiovascular diseases are the major risk factors that are frequently associated with NAFLD. 11 The prevalence of these risk factors varies between 30% and 100% in the case of obese patients, 10% to 75% in the case of type 2 diabetes and 20% to 92% in the case of dyslipidemia. 3
Abnormal biochemical and imaging studies were observed in NAFLD patients, such as mild to moderately elevated serum levels of alanine transaminase (ALT) or aspartate aminotransferase (AST) or both. Other abnormalities seen are hyperbilirubinemia, and hypoalbuminemia especially when it progresses to cirrhosis. 12 The main management of NAFLD consists of treating the liver disease as well as the associated metabolic comorbidities including obesity, dyslipidemia, IR and T2DM. Lifestyle modification through diet, exercise, and weight loss has been suggested to treat patients with NAFLD. 13 There are multiple Asian data supporting the hypothesis that NAFLD and progression to NASH can be resolved through lifestyle changes.14–16 A difference in prescribing patterns of drugs is observed between patients with and without coexisting conditions such as T2DM, obesity and dyslipidemia. Many clinical trials have been performed using vitamin E with and without other interventions, but no firm conclusion has yet been reached to date.17,18 There are no food and drug administration-approved pharmacotherapies for NAFLD. However, there are many drugs on the pipeline that are reckoned as good candidates to cure NAFLD and NASH.19–21 Due to the small number of patients involved, larger trials are needed to estimate better treatment response through therapy.
To date, there have also been no studies in Nepal on the difference in treatment patterns of different drug therapies to have a significant effect on the improvement of liver function. Thus, this study aims to evaluate the effect of therapy through both pharmacological and non-pharmacological approaches.
Materials and methods
Study setting and population
A cross-sectional study was conducted in a hepatology clinic in Kathmandu from March to May 2022. All the patients visiting the clinic for the treatment of NAFLD constituted the study population. All the NAFLD patients visiting the clinic for follow-up checkup with hepatologist was the sampling unit for this study. The STROBE guidelines were followed while preparing the manuscript.
Eligibility criteria
Patients who were aged above 18 years visiting with a report of fatty liver by ultrasonography were included in the study. Those who were having other diseases such as hepatitis and autoimmune disease; those having other diseases causing fatty liver and participant’s unwillingness to participate were excluded. Also, those with a history of taking more than 40 g of alcohol in a week or taking any potential hepatotoxic drugs were excluded.
Sample size
The sample size was 177. It was calculated by using Cochran’s formula with the assumption: the population size per year (N = 982), a 5% margin of error (e), a 95% confidence interval (CI). The prevalence rate of NAFLD was taken from previous study, p = 0.17. 6
Sampling procedure
Non-probability purposive sampling technique was used, where researchers select the site based on their research objectives. Average number of follow-up patients per day was found out from medical records. It was on average four patients per day. The researcher entertained all three patients who are previously diagnosed with NAFLD.
Data collection procedure
The data was collected from the patients who fulfilled the exclusion and inclusion criteria through face-to-face interview by using a proforma and semi-structured questionnaire. Semi-structured questionnaire included: data related to socio-demographic characteristics, clinical characteristics, lab values, non-pharmacological and pharmacological management and patient compliance. Semi-Structured questionnaire was prepared with the help of expert consultation along with supportive literatures.22,23 Further, the questionnaire was verified by hepatologist to enhance the validity. As per the suggestions and recommendations of the experts, necessary modifications were made to the questionnaire. All the questions were translated into the Nepali language and back-translated into the English language by a language expert to maintain translation validity. The questionnaire translated into Nepali language was pretested among 10% of the total sample, that is, 17 (n = 177), and those individuals were excluded from the main study.
Study parameters
Patients’ baseline parameters (age, sex and comorbidities), anthropometrics measurement (height, weight and body mass index (BMI)) and biochemical parameters (liver function test, Lipid profile and fasting blood glucose) were recorded using specially designed proforma. Height and body weight were measured with the participants wearing light clothing and no footwear. The patient’s BMI was calculated by obtaining their body weight in kilograms and dividing it by the square of their height in meters. These parameters were again reassessed after 3 months to determine the change in baseline characteristics after the treatment.
Ethical consideration
Before data collection, permission was obtained from the centre for liver disease clinic, Kathmandu and the approval was taken from the Institutional Review Committee (IRC-CiST) at the Central Institute of Science and Technology, affiliated with Pokhara University, Nepal (Ref. no: IRC/114/078/079).
Informed consent
Written consent form was given to the participants explaining all the details of the study before data collection. They were given sufficient time to read and understand. After their written agreement, the data collection was carried out. No economic incentives were given nor any coercive measures adopted to collect the data from patients.
Statistical analysis
The analysis was done with IBM-SPSS version 16 (IBM Corporation, Armonk, NY, USA). Descriptive analysis (percentage, mean with standard deviation according to data type and distribution of data) were used to describe the clinical and demographic variables. Student paired t-test was used to assess change in biochemical parameters and Chi-square was used to determine the association between management approach and change in biochemical parameters. p-value < 0.05 was considered statistically significant.
Results
Sociodemographic characteristics of patients
Out of 177 patients, 67.2% were male and 32.8% were female indicating a male predominance. Majority of patients were from Brahmin communities (43.5%) followed by Chhetri (29.94%) and Newar (16.69%). Similarly, more than one-third (35.66%) of the patients were unemployed followed by service (31.6%), and business (29.9%) (Table 1).
Sociodemographic characteristics of the participants (n = 177).

Clinical characteristics of patients
Out of 177 patients, the mean weight (Mean ± SD) of the patients before treatment was 74.88 ± 11.61 which was changed after the treatment (72.37 ± 11.61). Similarly, the mean BMI (Mean ± SD) of the patients before treatment was 28.41 ± 4.05 which was also decreased after the treatment (27.31 ± 4.58) with a p-value of 0.00 (Table 2).
Clinical characteristics of patients before and after treatment (n = 177).

¥: mean ± SD.
p-value significance at <0.05.
Risk factors of NAFLD
Out of 177 patients, more than one-third (61, 34.46%) had obesity and overweight, one-fourth (46, 25.98%) of the patients had hypertension (HTN), Obesity and other factors (hypothyroidism, gilbert syndrome and hepatitis B infection), 35 (19.77%) patients had T2DM + dyslipidemia + obesity and few of them had T2DM + dyslipidemia + HTN (16, 9.03%), dyslipidemia (10, 5.64%) and genetic factors + obesity (9, 5.08%). This indicated that obesity and overweight along with a combination of the above risk factors are the major factors for NAFLD.
Biochemical parameters of patients
Out of the total patients, the mean of liver enzymes (ALT, AST, and AP) before treatment was 87.80 ± 58.75, 51.48 ± 33.96, 109.68 ± 64.5 IU/L respectively which reduced to 51.96 ± 36.04, 36.44 ± 24.45, 83.38 ± 36.09 IU/L after the treatment (p-value: 0.00). Similarly, there was a significant reduction (p-value: 0.00) in mean Gamma-Glutamyl transferase (GGT; 71.23 ± 47.63 IU/L to 48.35 ± 29.10 IU/L). Total bilirubin also reduced from (0.91 ± 0.75) mg/dL to (0.79 ± 0.70) mg/dL after the treatment. Identically, before-treatment mean value of direct bilirubin (0.29 ± 0.40) mg/dL was reduced to (0.23 ± 0.30) mg/dL after the treatment which was not found to be significant.
Similarly, there was a significant change in the mean value of fasting blood glucose before and after treatment which dropped from 99.43 ± 20.90 mg/dL to 94.43 ± 15.47 mg/dL with a p-value of 0.00. Consistently, the mean lipid profile (TC, LDL, TG) dropped significantly except for HDL. In parallel, the mean HDL value increased significantly with a p-value of 0.01, from 40.52 ± 9.12 mg/dL to 44.74 ± 9.15 mg/dL after the treatment (Table 3).
Biochemical parameters of patients (n = 177).

p-value significance at <0.05.
Pharmacotherapeutic management of NAFLD
Out of the total 177 patients, more than one-third of patients (35.02%) were given no medicine (only non-pharmacological treatment) and around one-fourth (26.55%) of the patients were given Vitamin E + Ursodeoxycholic acid + Antilipidemic for pharmacological management of NAFLD, one-fourth (25.98%) were given Vitamin E, 15.81% were given antilipidemic, 13.55% were given other medication, 11.86% were given Vitamin E + Ursodeoxycholic acid and only 6.21% were given Ursodeoxycholic acid alone (Table 4).
Pharmaco-therapeutic management of NAFLD.

Patient compliance regarding treatment
Out of the total 177 patients, nearly half (49.7%) of the patients reported undertaking exercise regularly followed by irregular exercise (39.54%). Only a few patients did not do the exercise (10.76%). The overall effect is reported in the study as ‘positive changes after the treatment’ that is, both pharmacological and non-pharmacological which was detected in near about three-fourths (88.70%) of the patients (Table 5).
Patient compliance regarding exercise and positive changes towards therapy (n = 177).

Association between management approach and biochemical parameters
The present study shows a significant association between biochemical parameters (ALT, AST and AP) and management approach (pharmacological and non-pharmacological). For all the biochemical parameters the p-value observed was less than 0.05 (Table 6).
Association between management approach and biochemical parameters.
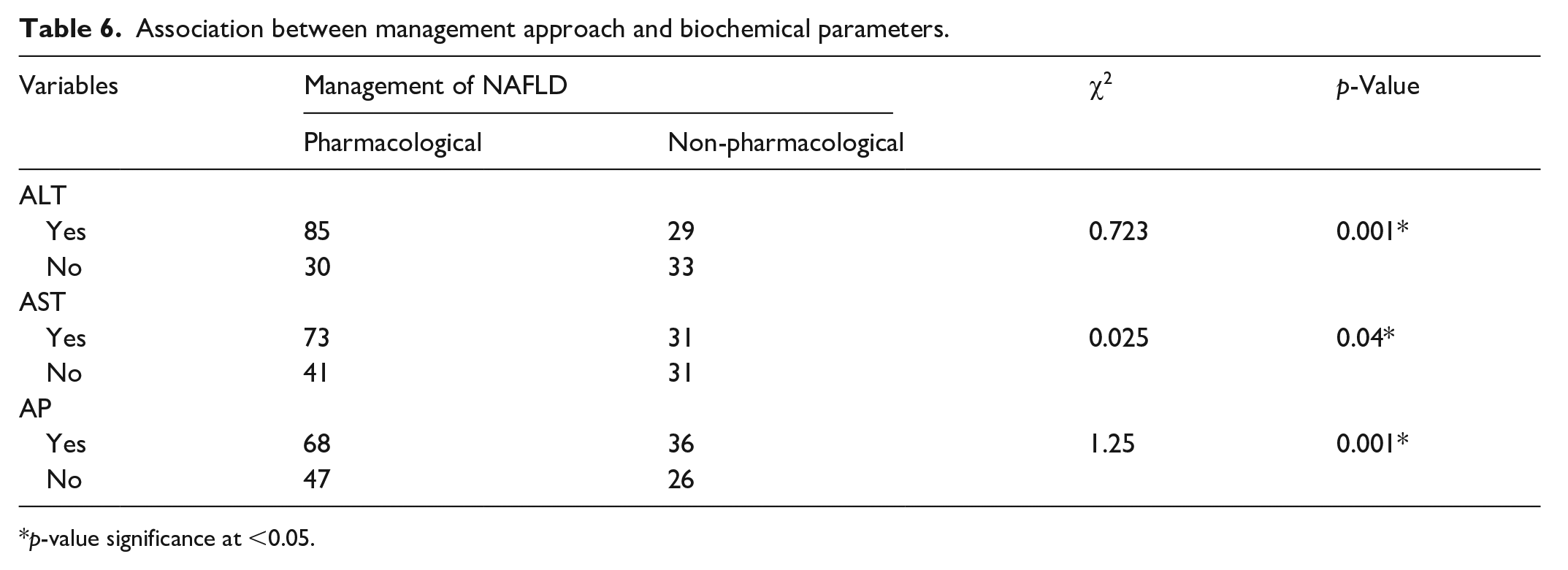
p-value significance at <0.05.
Discussion
As per the finding of our study, male (67.2%) showed predominance in cases of NAFLD over female (32.8%). A similar result was reported by various studies24,25 where male was more prevalent than female. The mean age of a patient with NAFLD was found to be 46.80 ± 12.06 years, which showed consistency to that of a study 26 in which the mean age reported was 48 ± 12 years. A cross-sectional study performed in South Korea showed a positive association between prolonged sitting, decreased physical activity and the prevalence of NAFLD in a middle-aged population. 27
Weight reduction is the most established treatment for NAFLD. In the present study, we found that there was significant weight loss in patients, the mean reduction in weight was 2.60 kg (p = 0.000). Similarly, there was a significant change in mean BMI as well. The main reason may be due to exercise as advised by the doctor (Brisk walking 4 km over 40 min/day). Exercise reduces cardiovascular-related risk thereby reducing liver fat. And, ATC guidelines also suggest a regular physical activity for patients with NAFLD, with a target of 150–300 min of moderate-intensity or 75–150 min of vigorous-intensity aerobic exercise per week as it plays an important role in weight loss and also in improvement in liver function. 28
Out of 177 patients, the majority of patients had obesity and overweight along with the combination of multiple risk factors that is, HTN, T2DM, dyslipidemia and genetic factors. As per the literature, the major risk factors that play a role in the pathogenesis of NAFLD include Obesity, DM, HTN, hypertriglyceridemia, dyslipidemia and genetic factors.29,30 This more likely supported the finding of this study. The dietary pattern of people is changing rapidly; instead of consuming more vegetables and fish and less red meat and high-fat dairy products, the use of energy derived from fat has increased over time. This is associated with IR, postprandial lipid metabolism dysfunction, and NAFLD development or progression. 31 In addition, the consumption of soft drinks with dietary fructose has increased, which increases obesity and the risk of NAFLD in people. 32 Similar to this finding is the finding of the study from Japan and Hong Kong, where 15.2% and 19.3% of overweight and obese subjects (BMI > 25 kg/m2) were found to have NAFLD. 33
The finding from previous study illustrated that there was a significant rise in the liver enzymes and lipid profiles except for HDL (HDL: 44.74 ± 9.15 mg/dL) which was significantly reduced after the treatment. 34 This result more or less corresponds to that of our study.
In the current study, more than one-third (35.02%) of the patients were under the non-pharmacological treatment approach, which showed significant positive changes in biochemical parameters, and weight loss after the treatment. This might be due to the effectiveness of the treatment approach used, and compliance of patients towards regular physical exercise (49.72%), This finding is well supported by a study from the USA, where 152 patients with NAFLD and elevated transaminases achieved significant improvements in liver function tests with subsequent lifestyle changes, counselling interventions targeting physical activity, changes to nutrition behaviours, and modest weight loss. 35
In the case of pharmacological management, the finding of the study illustrated Vitamin E + Ursodeoxycholic acid + Antilipidemic (26.55%) to be the commonly prescribed regimen for the pharmacological management of NAFLD patients followed by vitamin E alone (25.98%). The rationale for ursodeoxycholic acid therapy in fatty livers is because of its role in decreasing cholesterol secretions into bile. Moreover, it has hepatoprotective effects. 36 Antihyperlipidemic drugs have been widely studied for the treatment of fatty liver because of the simple rationale of preventing the accumulation of free fatty acid and triglycerides in the hepatocytes. 37 Vitamin E is a common drug used alone and in combination therapy too. A systemic review from Pakistan supported the use of Vitamin E in the treatment of NAFLD. 24
Another study reported on the beneficial use of statin with Vitamin E in NAFLD patients with (60%) or without dyslipidemia. The findings in total show antihyperlipidemic + vitamin E to be a potential therapy in NAFLD patients. 38 Likewise, another study from France reported on the use of Ursodeoxycholic acid + Vitamin E in NAFLD patients. The finding obtained was a significant decrease in mean serum AST, ALT and GGT levels (1.39 ± 0.74 to 0.78 ± 0.34 for AST, 1.72 ± 0.92 to 0.91 ± 0.69 for AST and 3.25 ± 2.85 to 1.30 ± 1.30 for GGT) and AST, ALT and GGT had reached normal range in 80%, 70% and 65% of the patients, respectively. 39 A similar trend was seen in this recent study after the treatment, that is, AST, ALT and GGG along with AP were significantly reduced in NAFLD patients after the treatment.
There was a significant association seen between the management approach of NAFLD and biochemical parameters (AST, ALT, AP, TP) with a p-value < 0.05 in this recent study. All of these findings and supportive material suggest the potentiality of a combination of Vitamin E + Ursodeoxycholic acid + Antilipidemic along with a non-pharmacological approach to be the ideal therapeutic management for NAFLD.
Limitation of the study
This study has several limitations. First, the study was limited to a small sample size (n = 177) since the data was collected from a single hepatology clinic in short study duration. Second, the study may have had variation in patients’ compliance towards regular physical exercise which can affect the results. More studies in large samples are required to further examine the relationship between physical activity intensity and biochemical parameters in individuals with NAFLD.
Conclusion
The major risk factors of NAFLD were found to be obesity, HTN, T2DM and dyslipidemia. Management through both pharmacological and non-pharmacological approaches shows significant improvement in biochemical parameters along with weight reduction through exercise indicating that either alone or as a combination of both approaches plays an effective role in treating NAFLD. Also, Vitamin E alone or in combination with other drugs shows promising results in improving liver function. To ensure therapeutic effects, patients’ compliance with exercise and achievement of exercise goals should be monitored. For improved evidence-based lifestyle modification guidelines, more rigorous, controlled studies of longer duration are required.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241227090 – Supplemental material for Effect of exercise and pharmacotherapy on non-alcoholic fatty liver disease
Supplemental material, sj-docx-1-smo-10.1177_20503121241227090 for Effect of exercise and pharmacotherapy on non-alcoholic fatty liver disease by Amrita Kandel, Poonam Pant, Sushila Todi, Sudhamshu KC and Sudip Pandey in SAGE Open Medicine
Supplemental Material
sj-pdf-2-smo-10.1177_20503121241227090 – Supplemental material for Effect of exercise and pharmacotherapy on non-alcoholic fatty liver disease
Supplemental material, sj-pdf-2-smo-10.1177_20503121241227090 for Effect of exercise and pharmacotherapy on non-alcoholic fatty liver disease by Amrita Kandel, Poonam Pant, Sushila Todi, Sudhamshu KC and Sudip Pandey in SAGE Open Medicine
Footnotes
Acknowledgements
The authors express their sincere thanks to the department of pharmacy, CiST College and liver clinic for their kind support and for providing research facilities.
Author contributions
AK, PP and SK designed the study. AK collected the data. AK, PP and SP analysed the data. AK, PP, ST and SK drafted the original manuscript. All authors revised and edited the manuscript. All authors approved the final version of the manuscript.
Data availability statement
Data are available on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Before data collection, permission was obtained from the centre for liver disease clinic, Kathmandu and the approval was taken from the Institutional Review Committee (IRC-CiST) at the Central Institute of Science and Technology, affiliated with Pokhara University, Nepal (Ref. no: IRC/114/078/079).
Informed consent
Written consent form was given to the participants explaining all the details of the study before data collection. They were given sufficient time to read and understand. After their written agreement the data collection was carried out. No economic incentives were given nor any coercive measures adopted to collect the data from patients.
Patient consent for publication
The research participants were assured that the data were completely confidential and information will be published without mentioning their name. Written consent from participants was obtained for data publication.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


