Abstract
Eighty-five male Han-Wistar rats were arranged into three groups: CCl4-exposed rats, CCl4 + betaine-exposed rats, and control rats. To see the effect of betaine alone, five rats of the control and of the CCl4 + betaine groups were sacrificed after 7 days, before exposure to CCl4. After that, two of the groups (the CCl4 and CCl4 + betaine groups) were exposed to CCl4 (1 ml/kg per day subcutaneously [SC] for 4 consecutive days), and one of the groups (control group) was given olive oil (1 ml/kg per day SC for 4 consecutive days). At the start of the study (day 0), day 1, day 2, day 3, day 4, and 3 days after the last CCl4 and olive oil injections (day 7), samples of five rats per group were sacrificed, and the livers were taken for chemical analyses and histological examination. Oral betaine, after the acclimation period of a week, increased the number of mitochondria but not mitochondria size (day 0), compared with the case in control rats. Exposure to CCl4 resulted in centrilobular hepatic steatosis, and the administration of betaine significantly reduced this. Morphometric analyses also revealed that the addition of betaine increased the volume density of rough endoplasmic reticulum (RER) in the perinuclear areas of liver cell cytoplasm (day 7). Additionally, the administration of betaine prevented the reduction of Golgi complexes and mitochondrial figures in the cytoplasm observed after the exposures to CCI4. Also, the volume density of mitochondria was smallest in the CCI4-group, but the difference was not statistically significant. The results indicate that oral betaine either improves recovery or reduces the toxic effects of CCl4 on cell organelles in liver cells of male Han-Wistar rats.
Carbon tetrachloride (CCl4) is a highly toxic chemical agent. The toxic effects of CCl4 on the liver have been known for years and studied extensively. 11 , 32 Furthermore, CCl4 treatment has been used as a model to induce fatty liver for studying possible interacting effects of a compound or a treatment. The effects of CCl4 on hepatocytes, depending on dose and exposure time, are manifested histologically as hepatic steatosis (e.g., fatty infiltration), centrilobular necrosis, and ultimately, cirrhosis. Hepatic steatosis of the liver is a multifactorial phenomenon thought to be caused by a blockage of lipoprotein secretion; 31 impaired synthesis or peroxidation of phospholipids, or both; 38 , 40 the toxic effects of free alkyl radicals on cell membranes; 18 , 41 and disturbances in methylation reactions. 42 The endoplasmic reticulum 27 and mitochondria 30 have been shown to be involved in cell damage. The metabolic effects of CCl4 inside mitochondria have been described, 8 and it has been found that damage to the calcium pump in mitochondria 1 is dependent upon haloalkylation. However, the profound accumulation of fat following CCl4 poisoning is considered to be independent of mitochondrial damage. 2 , 33 The fatty infiltration of the liver is thought to develop as a result of the action of free alkyl radicals on biomembranes that in turn cause haloalkylation-dependent blocking at the exit of the lipoprotein micelles from the Golgi apparatus. 28
Betaine is a metabolite of choline. Choline is oxidized in mitochondria to betaine aldehyde and further to betaine, part of which diffuses to the cytoplasm. 29 , 35 The transmethylation reactions promote phosphatidylcholine synthesis, which is vital for the integrity of cell membranes. Betaine is one of the lipotropes and contributes significantly to the transmethylation pathway, the formation of S-adenosylmethionine (SAM), and phospholipid biosynthesis. 4 , 16 , 19 SAM is the key physiological methylating agent in the body, and it has been shown convincingly that exogenous SAM prevents CCl4-induced DNA hypomethylation in rat liver that otherwise could lead to cirrhosis. 42 Betaine has been used to alleviate dietary cirrhosis in rats on a hypolipotropic diet. 7 It has been found to reduce alcohol-induced liver fattiness in rats. 4 , 5 Murakami et al. (1998) 26 have reported a reduction in liver necrosis induced by CCl4 in mice after oral treatment with betaine. Choline has been reported to have a regenerative effect on hepatic tissue damaged by CCl4 in dogs. 25 In rats, betaine has been shown to reduce CCl4-induced necrosis in liver. 20 A choline deficiency causes hepatic lipidosis, which can be prevented by intravenous choline supplementation. 9 Choline oxidation to betaine in humans is less efficient than in rats, 39 which means that choline may be less effective in the medical treatment of CCl4-induced liver injury in humans. Recent research has shown that in addition to transmethylation reactions, betaine serves as an osmolyte in rat liver macrophages 44 and thus improves the function of Kupffer cells.
Because of lack of data on the effects of betaine in the CCl4 hepatotoxic model in rats, we ran an experimental study to quantitate these effects at the ultrastructural level in liver. Mitochondria were accorded special interest as the site of choline oxidation.
Materials and Methods
Animals
Male Han-Wistar rats (85 specific pathogen–free animals), aged 8 weeks (265 g mean biological weight [BW]) were used in the study (see group sizes below) and housed separately (12 hours light:12 hours dark; temperature, 21 ± 0.5°C; relative humidity, 49±3%). All the rats received humane care in compliance with the National Laboratory Animal Center's guidelines (Kuopio, Finland).
Groups
The 85 rats were arranged into three groups as follows: 1) control group (30 rats); this group was given olive oil SC at 1 ml per kilogram BW for 4 consecutive days and given mock treatment with water intragastrically (IG) at 0.7 ml per day. 2) Carbon tetrachloride group (25 rats); this group was given 1 ml CCl4 per kilogram BW SC for 4 consecutive days and given mock treatment with water at 0.7 ml per day, IG. 3) Carbon tetrachloride + betaine group (30 rats); this group was given CCl4 at 1 ml per kilogram BW SC for 4 consecutive days and given IG treatment with betaine (830 mg per kilogram per day in 0.7 ml concentrated solution).
The animals were given a commercial rat food (R36, Lactamin AB, Stockholm, Sweden) and local tap water for drinking (analyzed and controlled for heavy metals, chlorophenols, pesticides and PCB compounds, Kuopio, Finland) on an ad libitum basis. The chemicals used were carbon tetrachloride (Riedel-Haen, Algol, Helsinki, Finland), olive oil (Spanish olivae oleum, Oriola Oy, Espoo, Finland), and anhydrous betaine (Betafin BP, Cultor Ltd. Finnsugar Bioproducts, Helsinki, Finland).
All the animals were acclimatized to the diet and the intragastric administration of water and betaine liquid for 7 days before the CCl4 and olive oil injections were initiated. During that period and throughout the study, betaine was administered intragastrically to rats in the CCl4 + betaine group, while those in the control and CCl4 groups were given equal volumes (0.7 ml) of water as a mock treatment. Samples of five rats each of the control animals and of the betaine-receiving animals were taken on day 0 to study the effect of betaine before the onset of CCl4 injections. The daily CCl4 and olive oil injections were started on day 0 and lasted for 4 days. Samples of five rats were killed from all of the groups on the day following each injection (days 1, 2, 3, and 4) and 3 days after the fourth CCl4 and olive oil injection (day 7). The rats were individually euthanatized by CO2 exposure, immediately followed by decapitation. Their livers were excised, weighed, and directly processed for the chemical and histological analyses. Feed consumption and body weight were recorded throughout the study. Fat content of the fresh livers was chemically analyzed by the technique described by Frings and Dunn (1970). 13
Light microscopy
Samples for light microscopy (LM) were taken from the left and right lobes and were placed in buffered 10% formalin and, after fixation, were processed and stained according to the conventional techniques. 3 , 23 , 36 The samples for LM were histochemically examined for fat using Sudan IV–stained frozen sections. Objective (10×) and ocular (10×) lenses with a graticule Leica E10A grid (100 squares; side lengths, 0.5 mm) were used. One thousand squares per rat liver (5,000 squares per treatment) were chosen blindly and at random and were used for calculating fat globules. Every square with noticeable fat was calculated as one point.
Transmission electron microscopy
Samples for transmission electron microscopy (TEM) were taken on days 0 and 7. Two freshly cut liver sections (size, about 1 mm3 each) from the left lobes of the livers were taken. The liver samples were immersed in buffered 2% glutaraldehyde, dehydrated in ethanol, and embedded in propylene oxide–epoxy resin. The embedded liver samples were processed further by the conventional techniques used for TEM analyses.
For the preliminary LM survey of the TEM samples, 1-µm sections were cut from the resin-embedded tissues and stained with toluidine blue. Centrilobular areas close to the surface of those sections were selected randomly for the TEM grids.
TEM samples were examined with a JEM 100 S-electron microscope (Jeol, Tokyo, Japan). Micrographs were taken (original magnification, 8000×) of perinuclear areas at random and on a blind basis, starting from the upper-left corner of the grids. Five micrographs per rat (25 micrographs per group) from the livers of day 0 samples and 20 micrographs per rat (100 micrographs per group) from the livers of day 7 samples were taken. The micrographs were examined by the method of Weibel et al. 43 A lattice with a 100-point graticule was used, and the volume densities of cell organelles elaborated by the lattice were recorded; thus, the quantitation was relative, neglecting possible absolute volume changes. The following cell structures were selected for counting: mitochondria, lipid globules, Golgi complexes, smooth endoplasmic reticulum (SER), rough endoplasmic reticulum (RER), glycogen, peroxisomes, other microbodies, lysosomes, myelin, cytoplasmic vacuoles, and undefined cytoplasm. The sum of 2,500 points per group was calculated from the day 0 samples; 10,000 points per group was calculated from the day 7 samples. Additionally, the total number of mitochondria was calculated from every micrograph, and the relative sizes of the mitochondria were calculated as elaborated mitochondrial points/total number of mitochondria. The resulting data were analyzed for statistical significance by ANOVA; Tukey's test was employed for pairwise comparison of the groups with the SAS statistical software package. 37
Results
The exposure to CCl4 significantly reduced the average feed intake and growth rates of the rats. On average, the rats in the CCl4 group had the lowest feed intake and the poorest growth rates during and after CCl4 injections. Liver weights did not differ between the groups (Table 1).
Average daily feed consumption, average weight gain, liver weight, and cumulative feed consumption. ∗
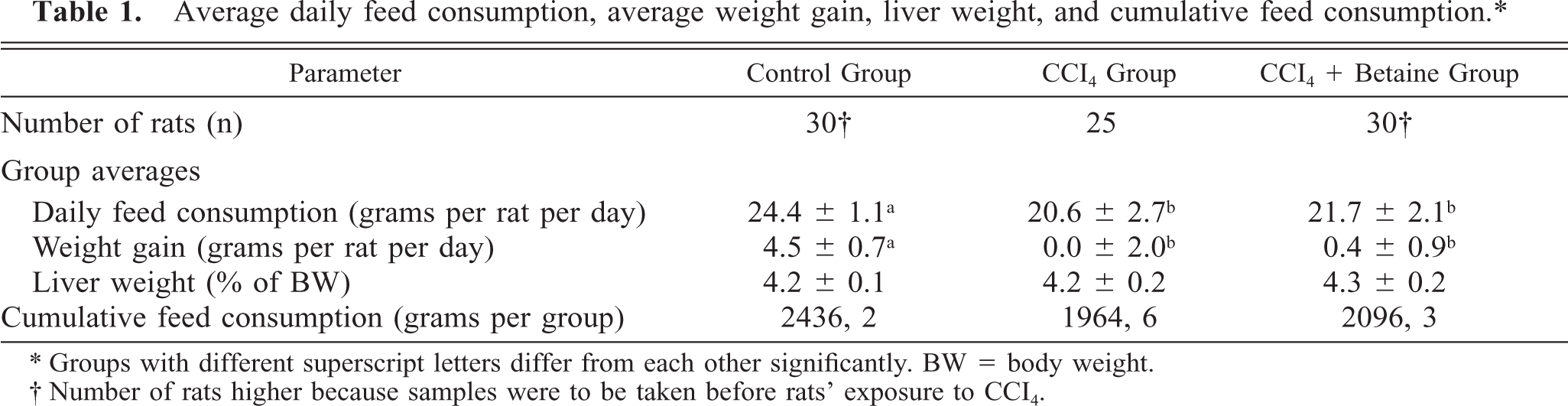
∗ Groups with different superscript letters differ from each other significantly. BW = body weight.
† Number of rats higher because samples were to be taken before rats’ exposure to CCI4.
The fat contents of the livers, analyzed chemically, increased in the CCl4 and CCl4 + betaine groups after the CCl4 injections (Table 2). This did not happen in the control group, which received injections of olive oil instead of CCl4. The treatment with betaine simultaneously with CCl4 exposure reduced the fat content of day 7 liver samples by 25%; however, this was still more than twice as high as that in the control livers (Table 2).
Hepatic lipids and day 7 analysis of the lipid content of livers. ∗
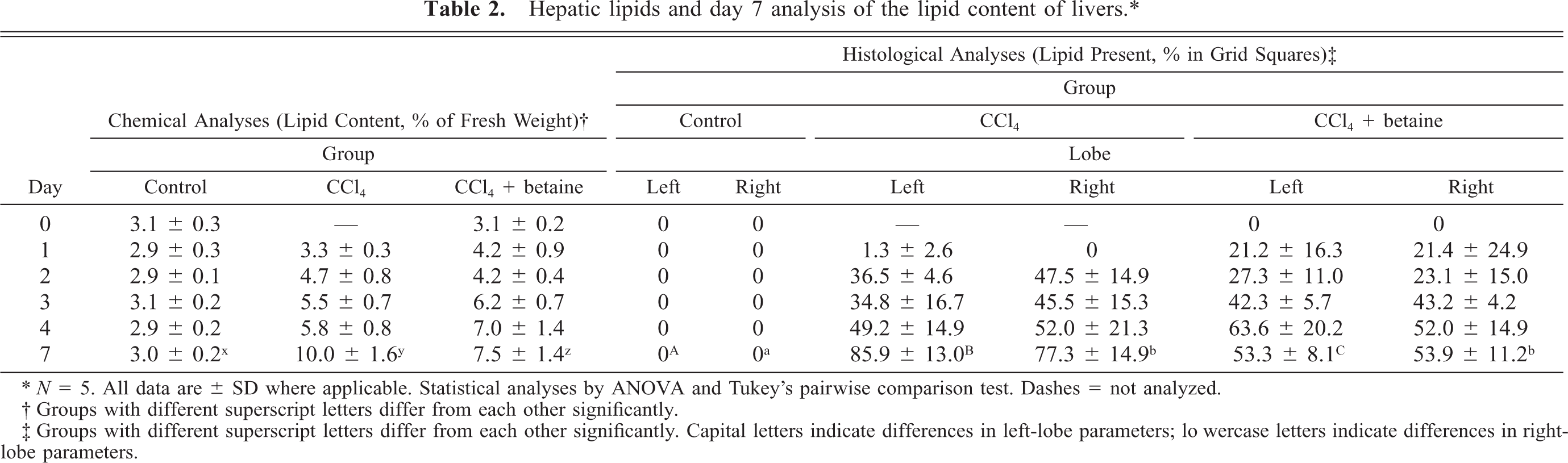
∗ N = 5. All data are ± SD where applicable. Statistical analyses by ANOVA and Tukey's pairwise comparison test. Dashes = not analyzed.
† Groups with different superscript letters differ from each other significantly.
‡ Groups with different superscript letters differ from each other significantly. Capital letters indicate differences in left-lobe parameters; lowercase letters indicate differences in right-lobe parameters.
The histological observations with LM (hematoxylin-eosin– and toluidine blue–stained sections) revealed a typical centrilobular steatosis in the CCl4 and CCl4 + betaine groups but not in the control group. Necrotic changes were not observed. In the sections that had been fat stained with Sudan IV, steatosis was most prevalent in the CCl4 group, and lipid globules from hardly visible droplets up to droplets the size of cell nuclei were present both in the left and right lobes of the livers. The administration of betaine reduced steatosis significantly, by about 30% (day 7, Table 2).
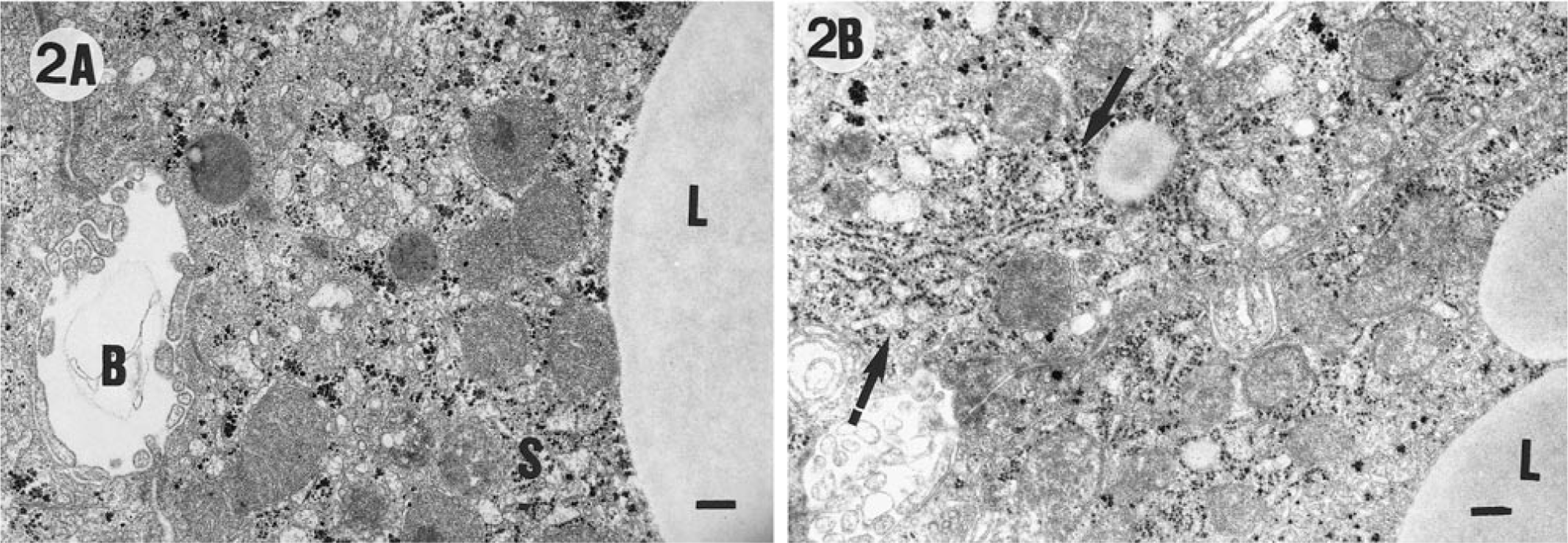
In the morphometric analyses using TEM after an acclimation period of 7 days prior to CCl4 injections (day 0), the control group and the CCl4 + betaine group differed slightly from each other only in the total number of mitochondria (Fig. 1A, B, Table 3). The cytoplasm of hepatocytes in both groups was visually sharp and easy to interpret.
Volume densities of perinuclear cytoplasmic components within centrilobular hepatocytes of rat liver after acclimation to betaine (day 0) and after exposure to CCI4 (day 7) based on TEM analyses. ∗
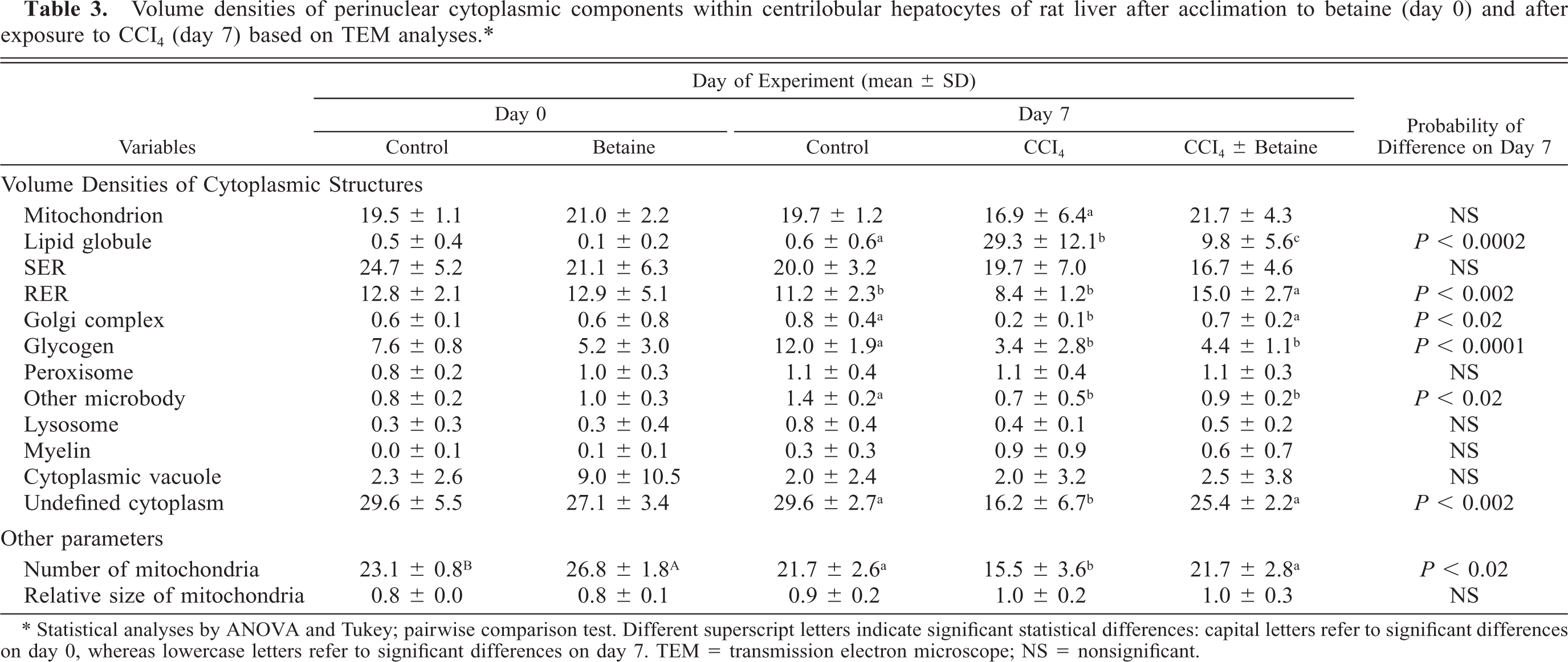
∗ Statistical analyses by ANOVA and Tukey; pairwise comparison test. Different superscript letters indicate significant statistical differences: capital letters refer to significant differences on day 0, whereas lowercase letters refer to significant differences on day 7. TEM = transmission electron microscope; NS = nonsignificant.
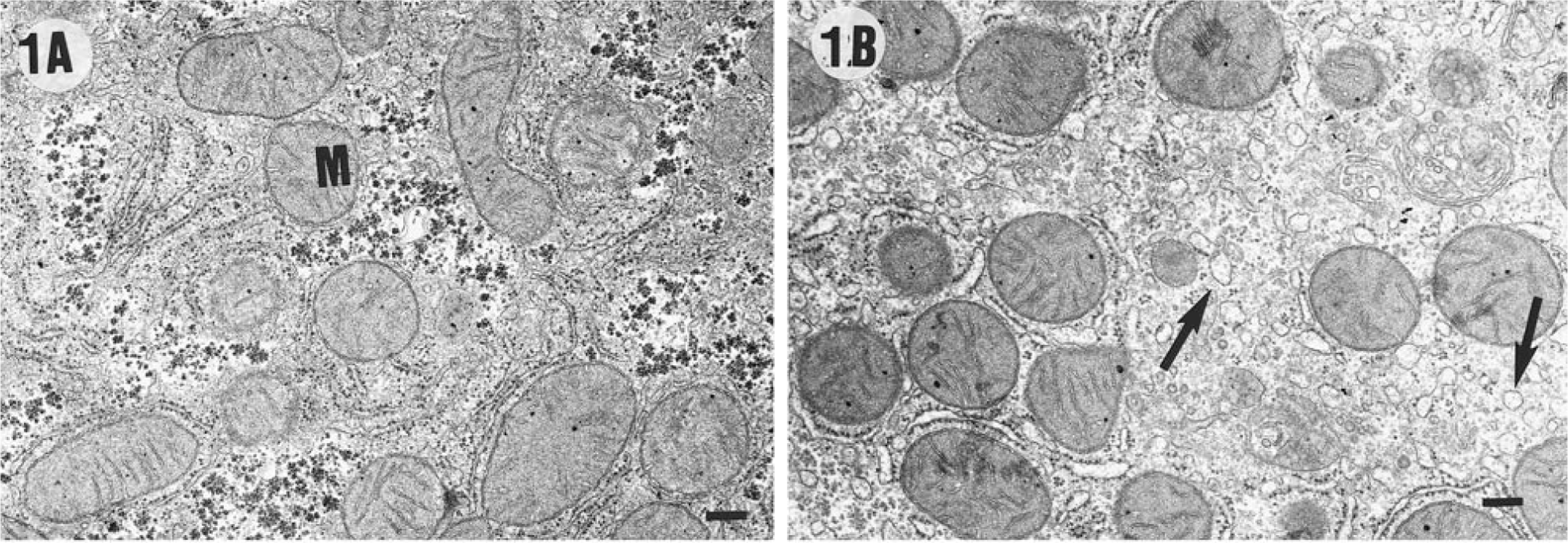
The examination of the livers by TEM after the CCl4 and olive oil treatments (day 7) revealed significant differences in several cell organelles among the groups (Fig. 2A, B, Table 3). The cytoplasm in the micrographs of the CCl4-treated groups was often cloudy, unlike that in the control group. The exposure to CCl4 treatment reduced the number of mitochondria in the CCl4 group, but the administration of betaine prevented this. Furthermore, the volume density of mitochondria was lowest in the CCI4-group, but the difference was not significant. The perinuclear cytoplasm of the livers of the CCl4-treated rats contained lipid globules of various sizes that were almost lacking in the control group. In the CCl4 + betaine group, the number of lipid globules in the cytoplasm was significantly reduced of the CCI4-group but were more numerous than the control group. It is also noteworthy that the amount of Golgi complexes in the CCl4 + betaine group stayed at the level of the control livers and that the amount of rough endoplasmic reticulum (RER) increased despite the exposure to CCl4 (Table 3).
Discussion
The lipotropic effects of betaine, choline, and methionine, as well as their roles in transmethylation reactions are well-known facts in the biochemical literature. However, recent published literature on the mode of action of betaine in CCl4-induced fatty livers is very scanty. On the other hand, an increasing number of scientific data describing the osmoprotective functions of betaine in different biological tissues and cells have been reported. 10 , 14 , 17
The published literature reveals different approaches for preventing the harmful effects of CCl4 to hepatocytes: vitamin E therapy has been used in the treatment of acute CCl4-induced hepatic injury in rats, 22 hepatic stimulator substance from weanling rats used in mice, 24 and adenosine used in rats. 15 Betaine has been shown to reduce CCl4-induced centrilobular necrosis in rats and to increase the activities of cytochrome P-450 enzymes. 20 Murakami et al. (1998) 26 have shown in mice that orally and intraperitoneally administered betaine promoted hepatocytes to recover from CCl4-induced hepatic injury. Our earlier studies have demonstrated that betaine increases S-adenosylmethionine and betaine homocysteine methyltransferase levels in rat liver and additionally reduces hepatic steatosis induced by either ethanol or CCl4. 4 , 19
The results of this study indicate a clear lipotropic and protective effect of betaine in alleviating the hepatotoxic effects of CCl4 in rat liver. There could be several explanations for the observed effects. The reduced hepatic steatosis was expected because of the lipotropic actions of betaine, and it is in line with our earlier preliminary histological observations. 19 The effect is supported by the fact that we clearly see more cytoplasmic lipid globules in hepatocytes of the CCl4-insulted livers than in the livers of rats receiving oral betaine simultaneously with CCl4 injections. The reduced hepatic steatosis could be explained by the lessened damage to the Golgi complex, and the increased volume density of RER noted in TEM analyses (Table 3). This would mean that in the presence of supplementary betaine, there is less damage to lipoprotein synthesis and excretion of very low density lipoproteins, despite the hepatotoxic effects of CCl4. At the same time, because more mitochondrial components were present, maintenance of β-oxidation and the biosynthesis of SAM could take place. It seems obvious that either improved recovery or reduced toxicity stay behind the noticed changes in cell organelles in the CCl4 + betaine group and have led to the reduction of hepatic steatosis.
The decreased volume density of RER in the CCl4 group is in line with previous results, 31 in which the retarding effects of CCl4 on protein synthesis have been observed. It has been observed earlier 34 that the administration of CCl4 to rats transformed the Golgi complex into a cluster of dilated vacuoles. Because a choline deficiency produces fatty liver in rats, 6 , 12 it follows that the marked reduction in hepatic lipidosis in the betaine group is partly due to betaine's improvements of the metabolism of choline.
Choline oxidation takes place in mitochondria and betaine appeared to increase the numerical density of liver mitochondria (day 0, Table 3). Even after the CCl4 injections (day 7), the mean number of mitochondria was not different from those in the control group and were about the same as the levels found in the day 0 samples (about 20%). It is probable that CCl4 treatment damaged mitochondria in the hepatocytes and thus prevented or retarded choline oxidation. Supplementary betaine possibly reduced this damage. As betaine has been shown to promote the generation of SAM in liver, 4 , 19 it is possible that the positive effects of betaine on hepatocytes might be partly explained by the improved cell membrane biosynthesis, in which SAM plays an important role. 16
The reduced glycogen content in the hepatocytes of the CCl4-injected animals might indicate higher energy use in the possible repair processes. The reduction in glycogen content has been reported in rats with CCl4-induced cirrhosis. 21
Another possible explanation for betaine's alleviation of the toxic effects of CCl4 is that it may have a supporting role in repairing the effects of CCl4 in liver by promoting the function of liver macrophages in vivo, just as has been demonstrated to happen in vitro. 44
The relative morphometric TEM method used in these studies neglect possible changes in cell volume. Because a possible increase or decrease in cell volume was not directly estimated (only liver weights were recorded), the results are indicative of relative presence and not of the absolute volumes of the measured cell organelles.
In summary, the data suggest that even though biological variability was relatively high within the groups, betaine spares the hepatotoxic effects of CCl4 in rats. It would appear that betaine either improves recovery or protects, and thus sustains, the function of the vital cell organelles, such as RER, Golgi complexes, and eventually mitochondria under CCl4-induced stress. Also, a marked alleviation of fatty infiltration of hepatocytes can be observed.
