Abstract
Mucociliary clearance is a main defense mechanism of the respiratory tract, which can be inherently impaired in primary ciliary dyskinesia (PCD) or reversibly altered in secondary ciliary dyskinesia (SCD). Limited diagnostic test availability likely leads to misdiagnosis or underdiagnosis of PCD in animals. This study evaluated the light and transmission electron microscopy (TEM) changes in the respiratory mucosa of 15 dogs with chronic respiratory disease suspected of PCD. Necropsy was performed in 1 case and 2 dogs were used as negative controls. PCD was confirmed in 1 dog, which presented with chronic otitis, bronchopneumonia, hydrocephalus and ultrastructural abnormalities in 84% of the assessed cilia, including absence of dynein arms and microtubular changes. The 14 other cases showed only nonspecific alterations, such as ciliary disorientation, compound cilia, ciliary edema, and axoneme bubbles in a minority of the evaluated cilia and were classified as SCD. Ciliary ultrastructural analysis can confirm a diagnosis of PCD if specific abnormalities exist. TEM remains an important investigational tool in veterinary medicine, as no other specific test for PCD in dogs has been standardized yet.
Keywords
Motile cilia in the respiratory epithelium are essential for mucociliary clearance. The ciliated cells have a microtubule-organizing center known as the centrosome that gives rise to the basal bodies, which are composed of 9 triplet microtubules anchored in the surface of the cell. The microtubules of each motile cilium consist of 9 peripheral doublets (PDs) surrounding 2 central singlets (CSs), an arrangement known as 9 + 2, except for the motile cilia in the embryonic node that are 9 + 0, lacking the pair of CSs. Associated structures are present along the length of each PD, including inner dynein arms (IDAs), outer dynein arms (ODAs), radial spokes and nexin links, which provide structural support by adding mechanical strength. Each ciliated cell of the respiratory system has approximately 200 to 300 cilia that are capable of continuous and coordinated motion. 10,16
Alterations in the structure and/or function of respiratory cilia result in defective clearance of infectious agents and particles from the respiratory epithelium and lead to chronic respiratory disease. Whether these changes are primary or secondary to other disease processes is sometimes difficult to differentiate. Primary ciliary dyskinesia (PCD) is a rare, autosomal, recessive, genetic disorder in humans that has occasionally been reported in domestic animals. 19,35 Secondary ciliary dyskinesia (SCD) describes acquired and reversible ultrastructural alterations to the microtubules in the ciliary orientation and in function of the cilia that results from insult to a previously normal mucosa. 6 SCD occurs in many chronic respiratory diseases, in which the impairment of mucociliary clearance is exacerbated by excessive mucus production due to goblet cell metaplasia and hyperplasia, as well as submucosal gland hypertrophy. This results in increased susceptibility to recurrent or persistent infections, bronchiectasis, and airway obstruction, 11,33 as occurs in PCD. Thus, SCD and PCD are difficult to differentiate clinically, although dogs with PCD may present with a triad of bronchiectasis, rhinosinusitis, and situs inversus (Kartagener syndrome), as well as infertility/subfertility and hydrocephalus. 1,15 The ciliary ultrastructure in SCD is affected in only a small subset of cilia, 24 and the return to normal morphology and function depends on the severity of the lesions, persistence of predisposing factors and proper treatment.
In patients with a suggestive clinical phenotype, finding specific ciliary ultrastructural changes can confirm the diagnosis of PCD. These consist mainly of shortening or absence of dynein arms and microtubular changes in all or the majority of the cilia. However, the absence of ultrastructural ciliary changes does not rule out the diagnosis of PCD, since at least 30% of human patients with PCD have normal ciliary ultrastructure. 16 Although only a small number of dogs have been reported to have PCD, this might be an underestimate, considering the complexity of diagnosis and the current lack of information in the literature. Conversely, presumptive PCD may be overdiagnosed given the limited documentation of alternate causes of chronic respiratory disease. The differentiation between PCD and SCD has implications for prognosis and thus warrants the diagnostic effort. The objective of this study was to describe the histologic and ultrastructural changes in the respiratory tract mucosa of dogs with suspected ciliary dyskinesia, including the differences between a dog with PCD and nonconfirmed patients.
Materials and Methods
All histopathologic and transmission electron microscopy (TEM) samples submitted to the Veterinary Diagnostic Ultrastructural Pathology Service of the University of Minnesota Veterinary Diagnostic Laboratory in suspicion of PCD from 2007 to 2016 were reviewed (Table 1). Biopsies from nasal cavity, trachea and/or bronchus from 14 dogs suspected of PCD were fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer (Electron Microscopy Sciences, Hatfield, PA).
Clinical Findings in Dogs Suspected to Have Ciliary Dyskinesia.
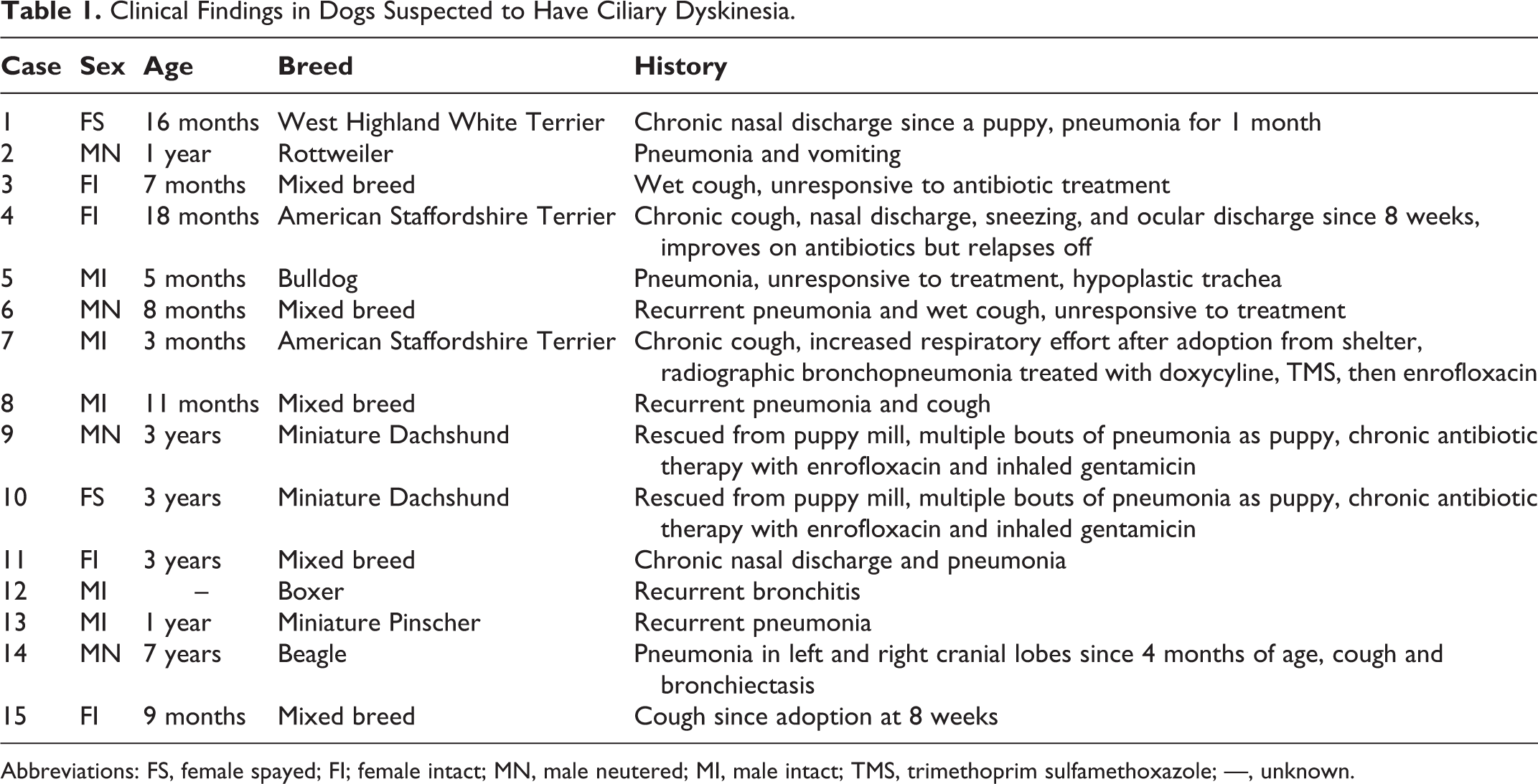
Abbreviations: FS, female spayed; FI; female intact; MN, male neutered; MI, male intact; TMS, trimethoprim sulfamethoxazole; —, unknown.
Necropsy was performed in 1 case, in which bacterial culture from the lungs was conducted. Tissues from various organs obtained at necropsy were formalin fixed within 2 hours after euthanasia. Tissue fragments were paraffin embedded, sectioned at 5 μm and stained with hematoxylin and eosin. Additional samples from respiratory and reproductive organs and ventricles of the central nervous system were glutaraldehyde fixed for TEM for ultrastructural analysis.
For ultrastructure, all tissue samples were washed 3 times in 0.1 M sodium cacodylate buffer and postfixed in 1% osmium tetroxide (Electron Microscopy Sciences) in 0.1 M sodium cacodylate buffer for 1 hour, followed by 3 more washes in distilled water. Samples were dehydrated using a 25% to 100% acetone gradient. Samples were infiltrated with 2:1 acetone/embed 812 resin (Electron Microscopy Sciences) for 1 hour and subsequently transferred to a 1:2 acetone/embed 812 resin mixture for 1 hour. Thereafter, samples were infiltrated with 100% resin (Electron Microscopy Sciences) and incubated at 58°C for 24 hours to polymerize the resin. Then, 1-μm-thick sections were mounted on glass slides and stained with toluidine blue, and light microscopy of the respiratory mucosa was examined for preserved ciliated epithelium. Areas with intact ciliated cells were sectioned at 60 to 70 nm with a diamond knife on a Leica UC6 Ultramicrotome (Leica Microsystems, Vienna, Austria) and mounted on a 200-mesh copper grid (Electron Microscopy Sciences). Grids were stained with 5% uranyl acetate for 20 minutes and Sato’s lead citrate for 6 minutes, as well as observed under a JEOL 1200 EX II transmission electron microscope (JEOL, Tokyo, Japan). Images were obtained using a Veleta 2 K × 2 K camera with iTEM software (Olympus SIS, Munster, Germany). Nasal, tracheal and bronchial mucosa obtained approximately within 2 hours after death from 2 dogs without a history of respiratory disease were used as negative controls.
For light microscopy, the morphology of ciliated epithelial cells and lamina propria of respiratory epithelium were evaluated. Histological lesions were graded as mild, moderate or marked, according to the intensity of alterations, density, integrity, and uniformity of cilia and basal body lamina; hyperplasia or metaplasia of respiratory epithelial cells; and the estimated number of infiltrating leukocytes. For TEM, the percentage of ciliary defects was determined by examination of cilia cross sections at different levels (Figs. 1, 2). The ultrastructural morphology of at least 300 cilia from each sample was systematically evaluated, avoiding ciliary tips, repeated analysis or unclear images of the cilium. The abnormalities were recorded and the mean percentage for each abnormality was calculated. Changes in the dynein arms include absence or shortening of IDAs and/or ODAs. Alterations of the CSs include absence or displacement of the pair, presence of only 1 singlet and supernumerary singlets. Defects of the PDs include absence, displacement, transposition to the center and supernumerary doublets. Other abnormalities include the presence of compound cilia, ciliary edema, axoneme bubbles and electron-dense inclusions in the basal bodies.
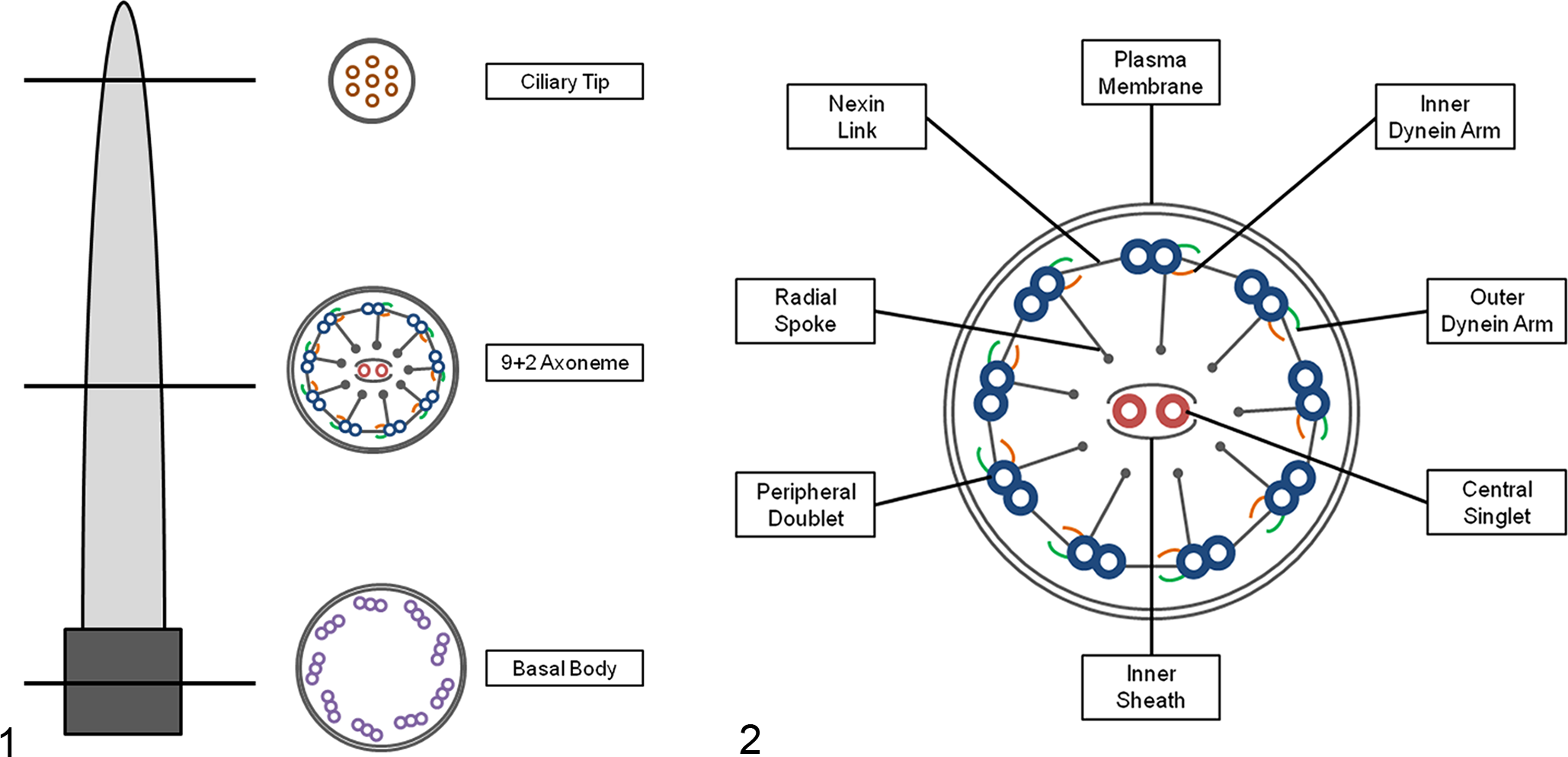
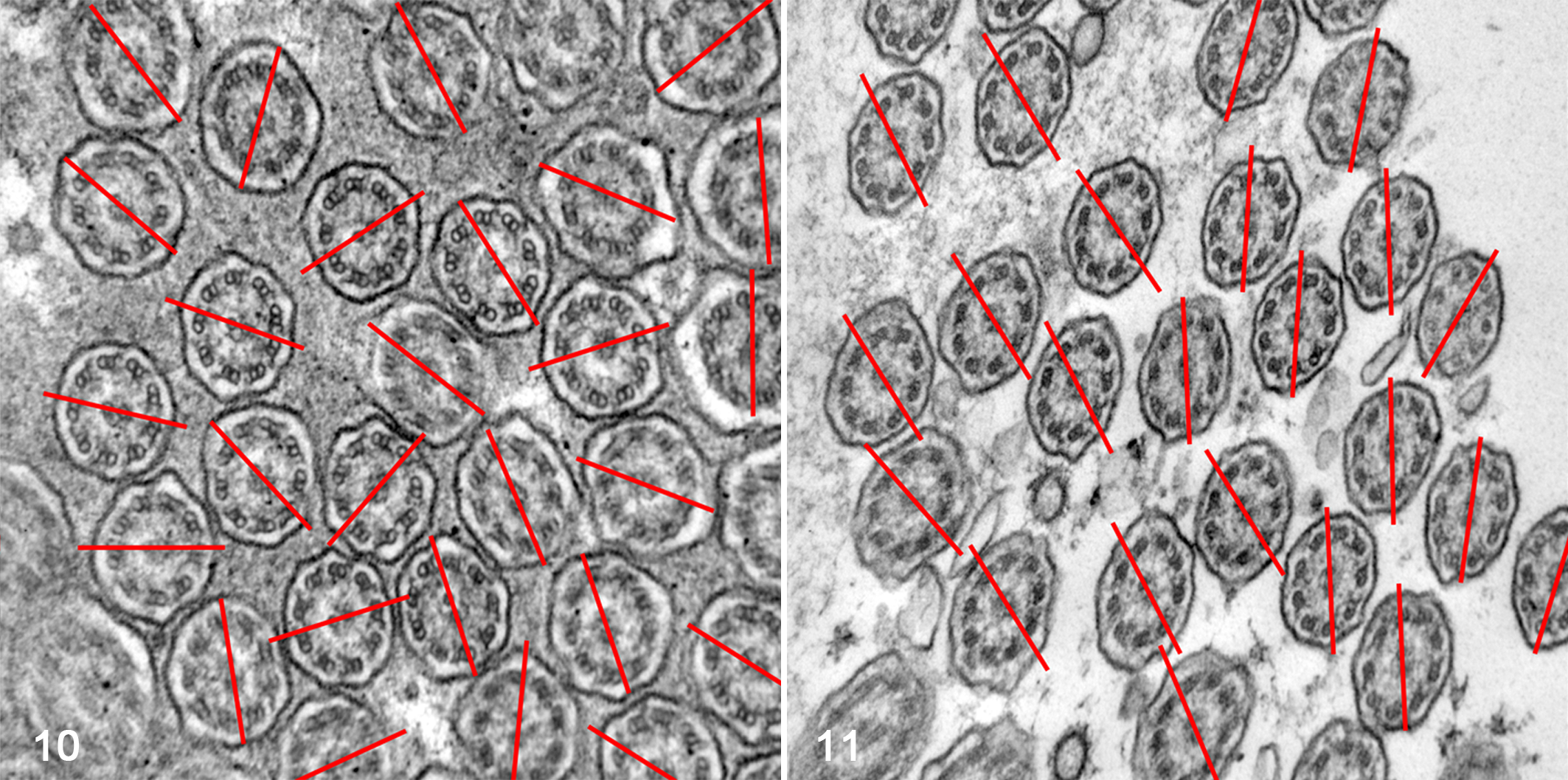
Finally, ciliary orientation was measured in all cilia cross sections in which the pair of central microtubules could be seen on TEM, as previously described. 14,24 Briefly, on one 100 000× magnification photograph from each patient (with an average number of 50 cilia per patient), a line was drawn through the CSs of each cilium, and the angle between this line and a reference line was established. A line perpendicular to the ciliary axis, transecting the central microtubules, points in the direction of ciliary beat and therefore can measure ciliary orientation. Disorientation was considered when the line through the CS differed from a reference line by more than 20°, in the majority of cilia.
The medical records of 6 dogs in the study, which were patients of the University of Minnesota Veterinary Medical Center, were examined in detail. Data collected included signalment (age, breed, sex), history and presenting clinical signs, respiratory bacterial culture results, airway cytology and imaging study interpretations by board-certified radiologists.
Results
Clinical Presentation
The 6 dogs for which the detailed clinical findings were available (see Suppl. Table S1 for details) presented with a chronic cough and/or chronic nasal discharge and sneezing and had radiographic evidence of pneumonia. Airway cytology findings were available for 5 dogs, and neutrophilic inflammation was found. Bacteria were seen cytologically in 2 of 5 airway wash samples; aerobic culture was negative, although
The only dog with confirmed PCD had a cough at the time of adoption as a puppy (between 4 and 8 weeks of age). Medical records prior to referral to the University of Minnesota Veterinary Medical Center were limited but suggested that the dog was treated empirically with doxycycline for presumed
Pathology
Samples from 15 dogs (14 biopsies and 1 necropsy) with chronic respiratory disease suspected of PCD and from 2 control dogs were examined. The patients’ mean age was 1 year and 8 months (range, 5 months–7 years).
Biopsies
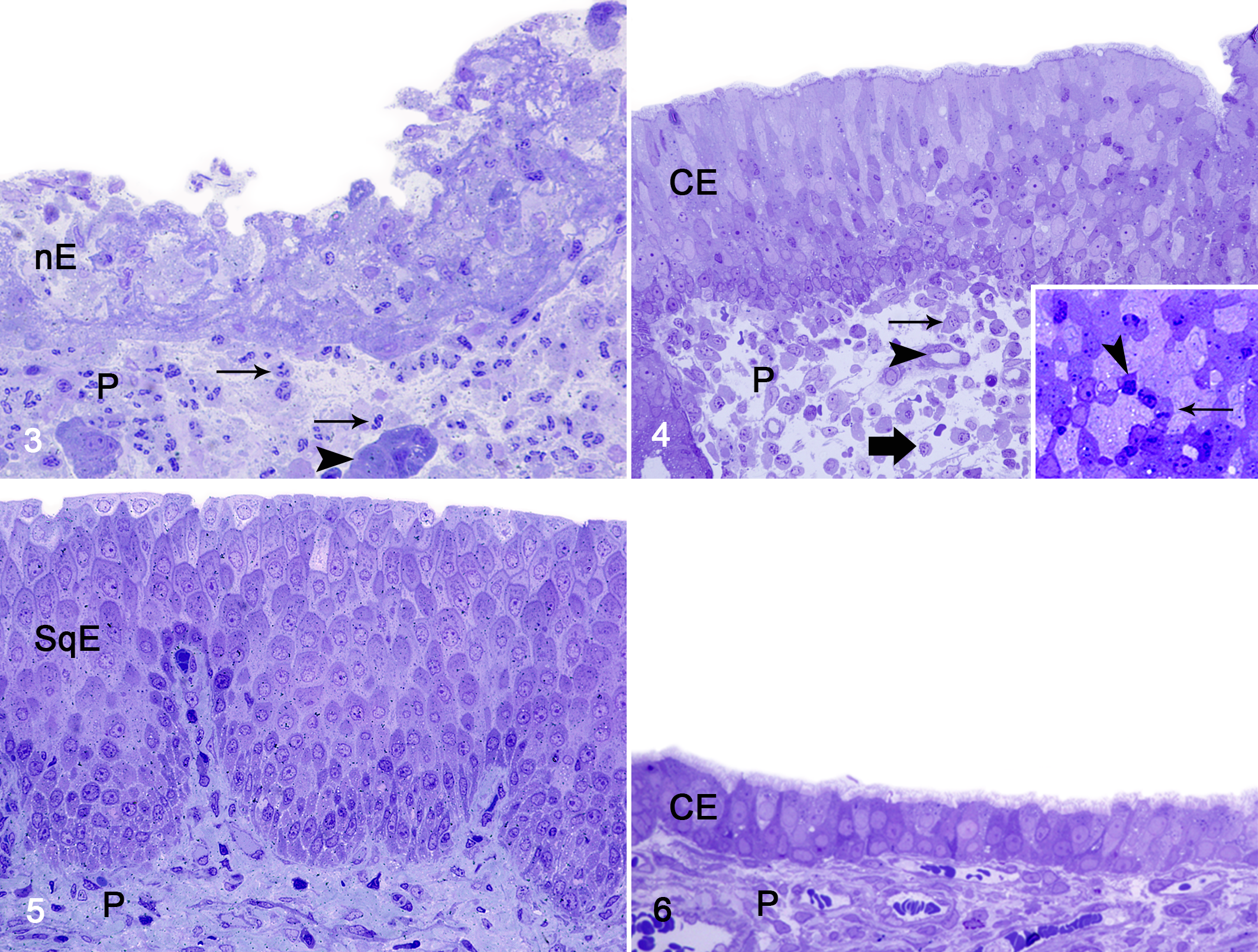
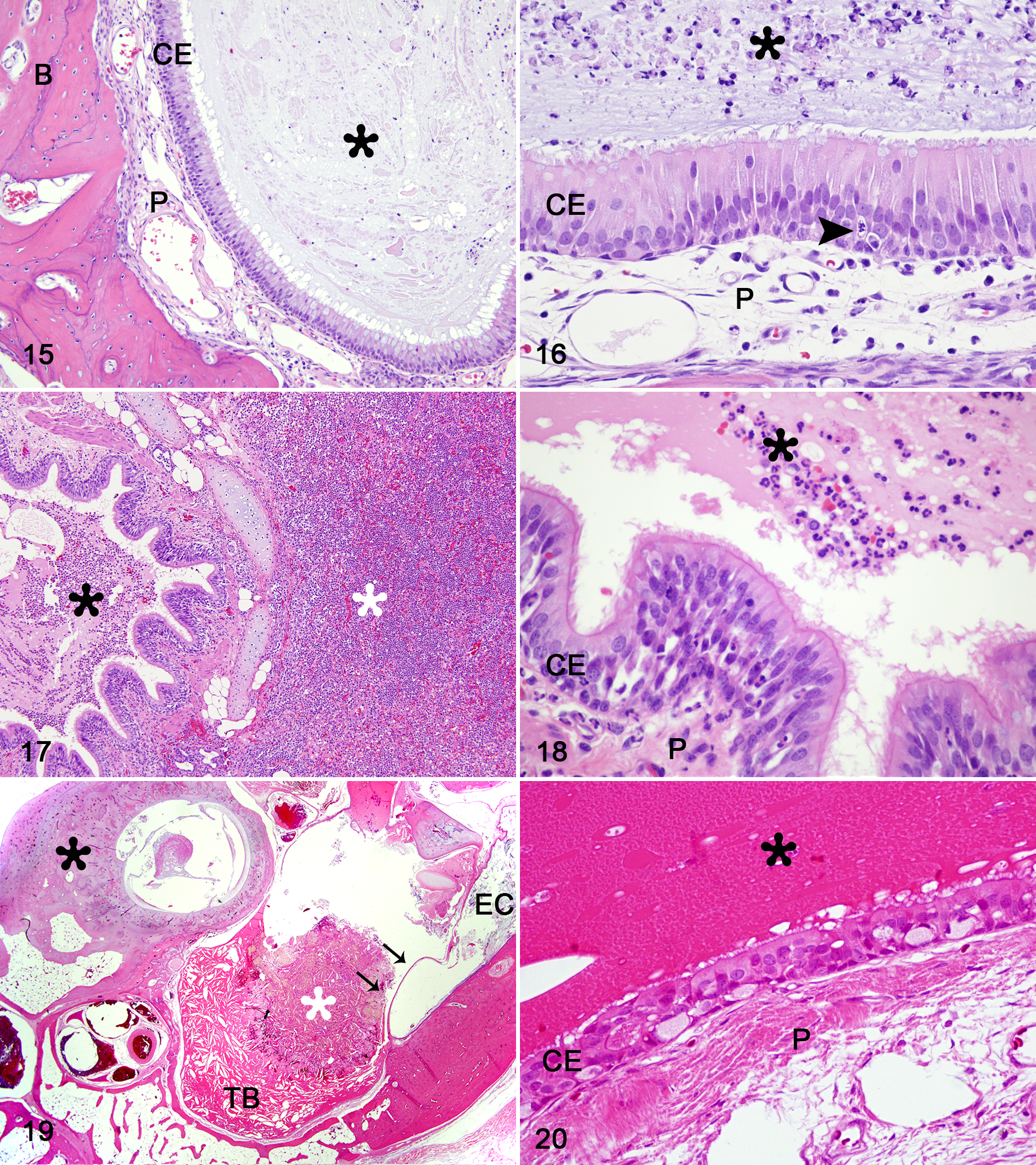
By light microscopy, all the clinical samples contained different degrees of epithelial disorganization, decreased density of cilia, respiratory epithelial hyperplasia, transepithelial migration of leukocytes, goblet cell hyperplasia, squamous metaplasia, edema of lamina propria and mild to severe increases in the number of lymphocytes, plasma cells, eosinophils, and mast cells (Figs. 3–5; see Suppl. Table S2 for details). The respiratory mucosa from control animals showed a uniform pseudostratified columnar ciliated epithelium reposing over a lamina propria with seromucous glands (Fig. 6).
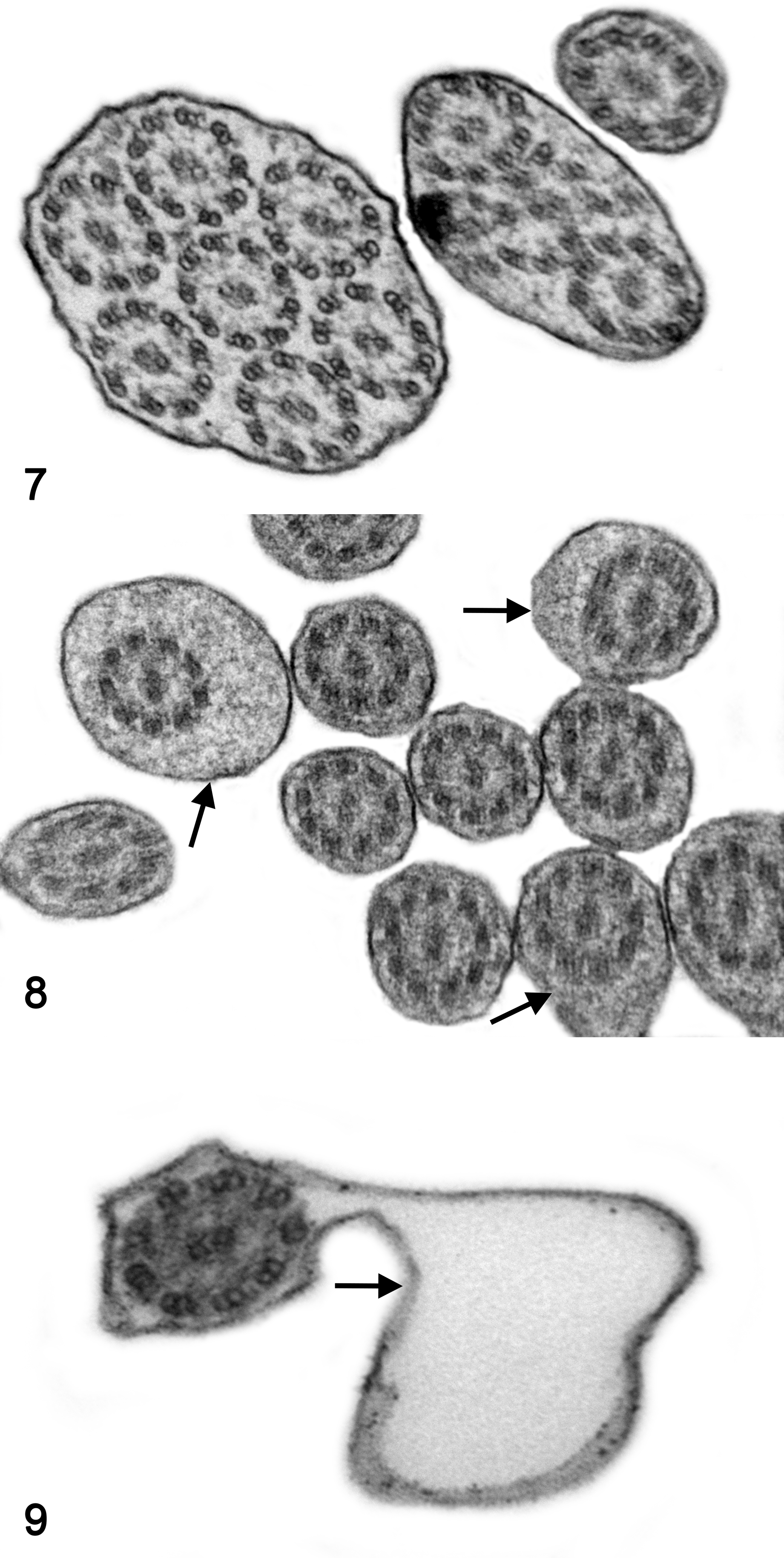
By TEM, 13 of 14 biopsies did not show specific ultrastructural findings compatible with PCD and were classified as SCD. The ultrastructural findings are summarized in Table 2. Abnormalities were present in the central apparatus of up to 13% of the analyzed cilia; these consisted of absence of CSs in 8%, displacement of CSs in 2%, only 1 singlet in 2%, and supernumerary singlets in 3%. Abnormalities were present in the peripheral apparatus of 11% of the analyzed cilia, including absence of PDs in 4%, displacement of PDs in 2%, transposition of PDs in 7% and supernumerary doublets in 3%. Nonspecific abnormalities were found in 18% of the evaluated cilia, such as compound cilia (Fig. 7), ciliary edema (Fig. 8), axoneme bubble (Fig. 9), electron-dense basal body inclusions and ciliary disorientation defined as misalignment of the CS with an angle >20° (Figs. 10, 11). In the 2 dogs used as normal controls, 8% of cilia had nonspecific irregularities, including 6% compound cilia and 2% with electron-dense inclusions in the basal bodies. In 4 cases, the respiratory mucosa exhibited multifocal areas of transition from ciliated to squamous epithelium, with formation of microvillus-like extensions in the apical domain of the cell and markedly low density of the cilia and stereocilia compared to the control cases. The TEM evaluation of cilia was not performed in 1 case due to marked squamous metaplasia in the bronchial mucosa, in which ciliated cells were completely replaced by stratified squamous epithelium. In 1 case, the bronchial epithelium had severe necrosis with intralesional bacteria. In another case, there was marked collagenolysis and infiltration of eosinophils in the wall of the bronchus.
Ultrastructural Findings in Dogs Suspected to Have Ciliary Dyskinesia.
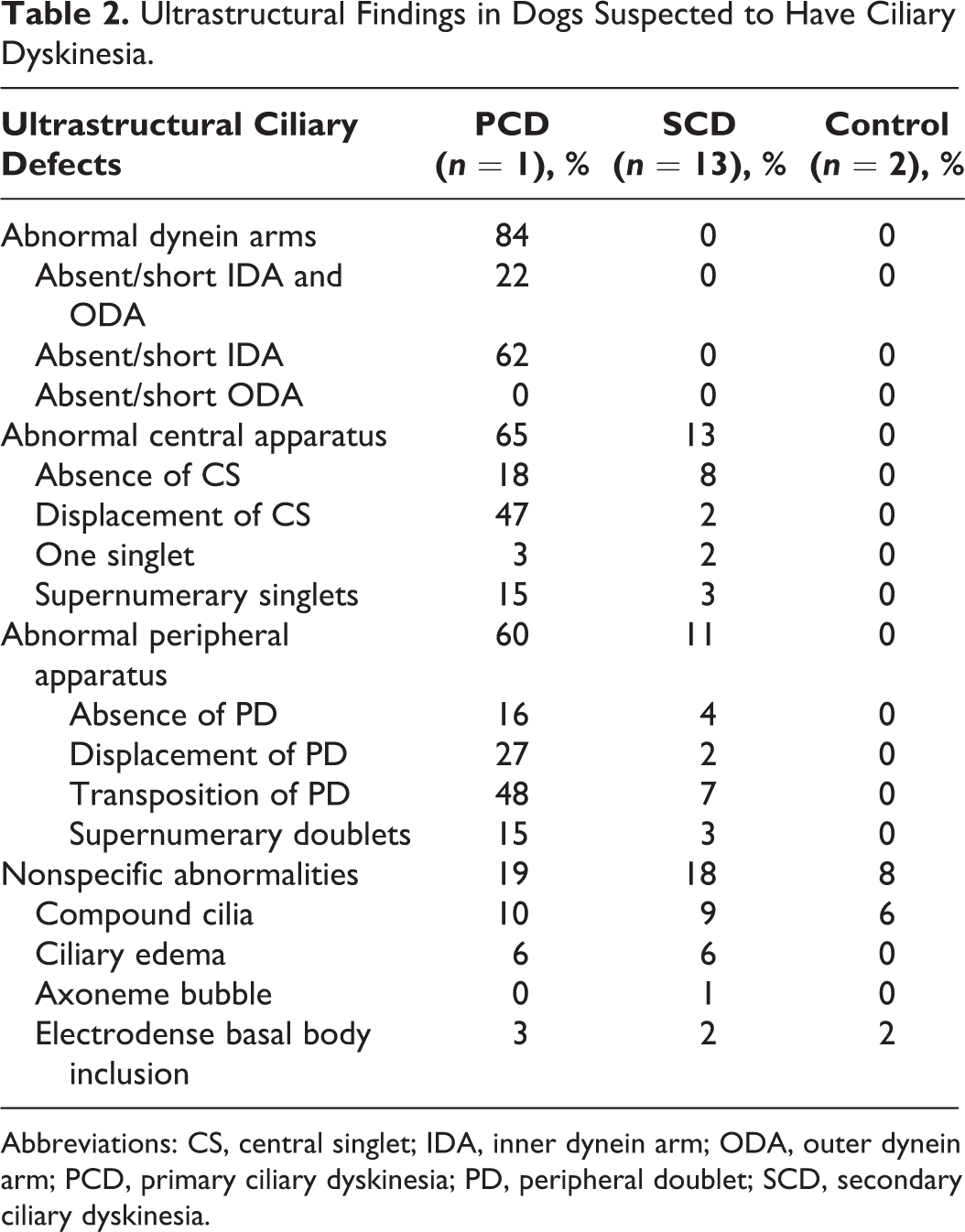
Abbreviations: CS, central singlet; IDA, inner dynein arm; ODA, outer dynein arm; PCD, primary ciliary dyskinesia; PD, peripheral doublet; SCD, secondary ciliary dyskinesia.
Necropsy
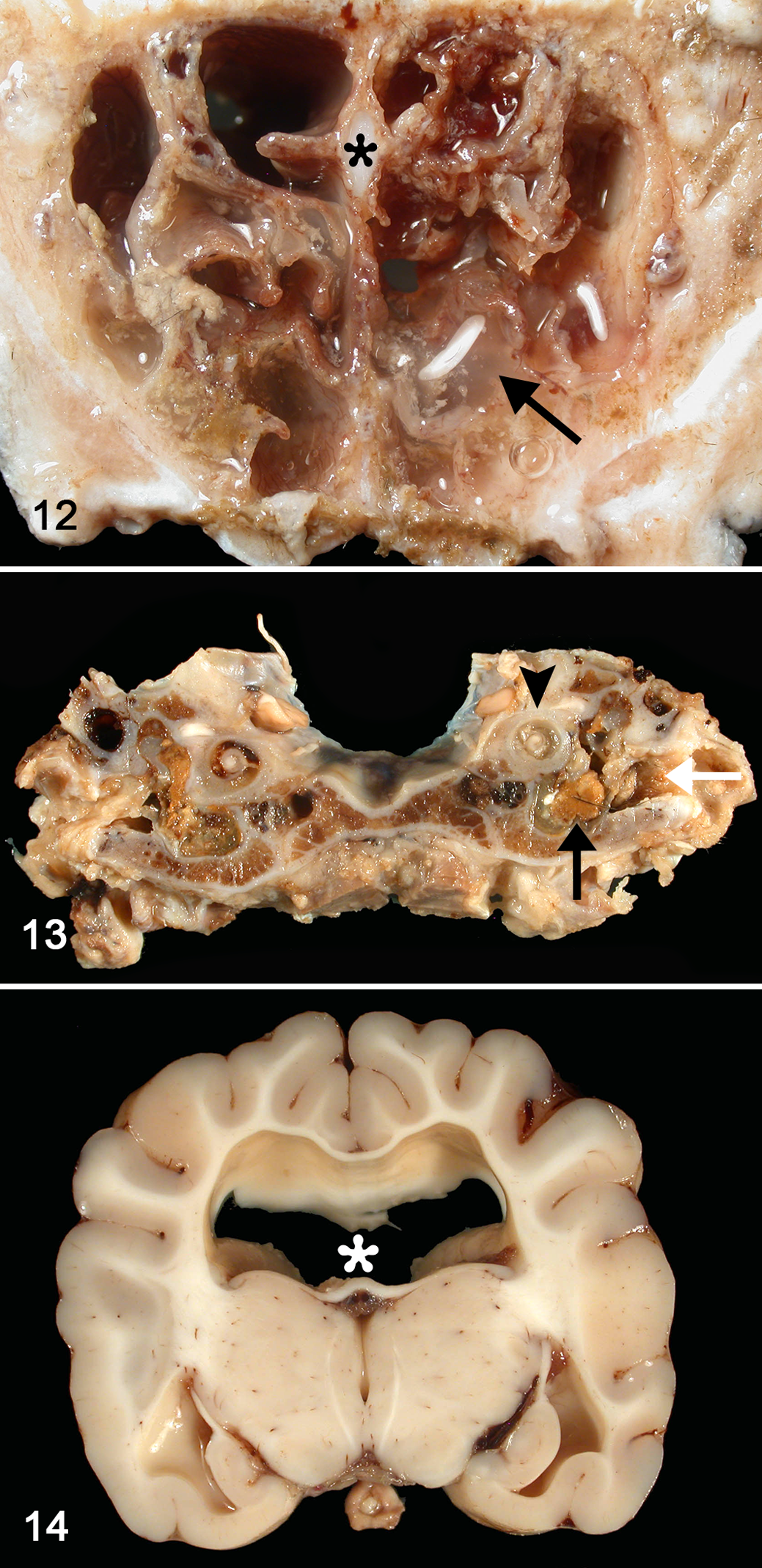
An 8-month-old female, mixed-breed dog with a history of bacterial pneumonia secondary to bronchointerstitial lung disease was examined at necropsy. Grossly, the nasal and paranasal sinuses were filled with large amounts of catarrhal-purulent and hemorrhagic exudate, and the mucosa was congested (Fig. 12). The middle ear (tympanic bulla) was filled with a large amount of catarrhal exudate and waxy material (Fig. 13). The trachea, bronchi and bronchioles contained catarrhal-purulent exudate. The lungs were markedly congested with multifocal, firm and dark red areas randomly distributed throughout all lobes. In the brain, there was marked, bilateral and symmetric dilation of the lateral ventricles (Fig. 14). Other tissues did not show significant macroscopic lesions. Aerobic culture of the lungs revealed beta-hemolytic
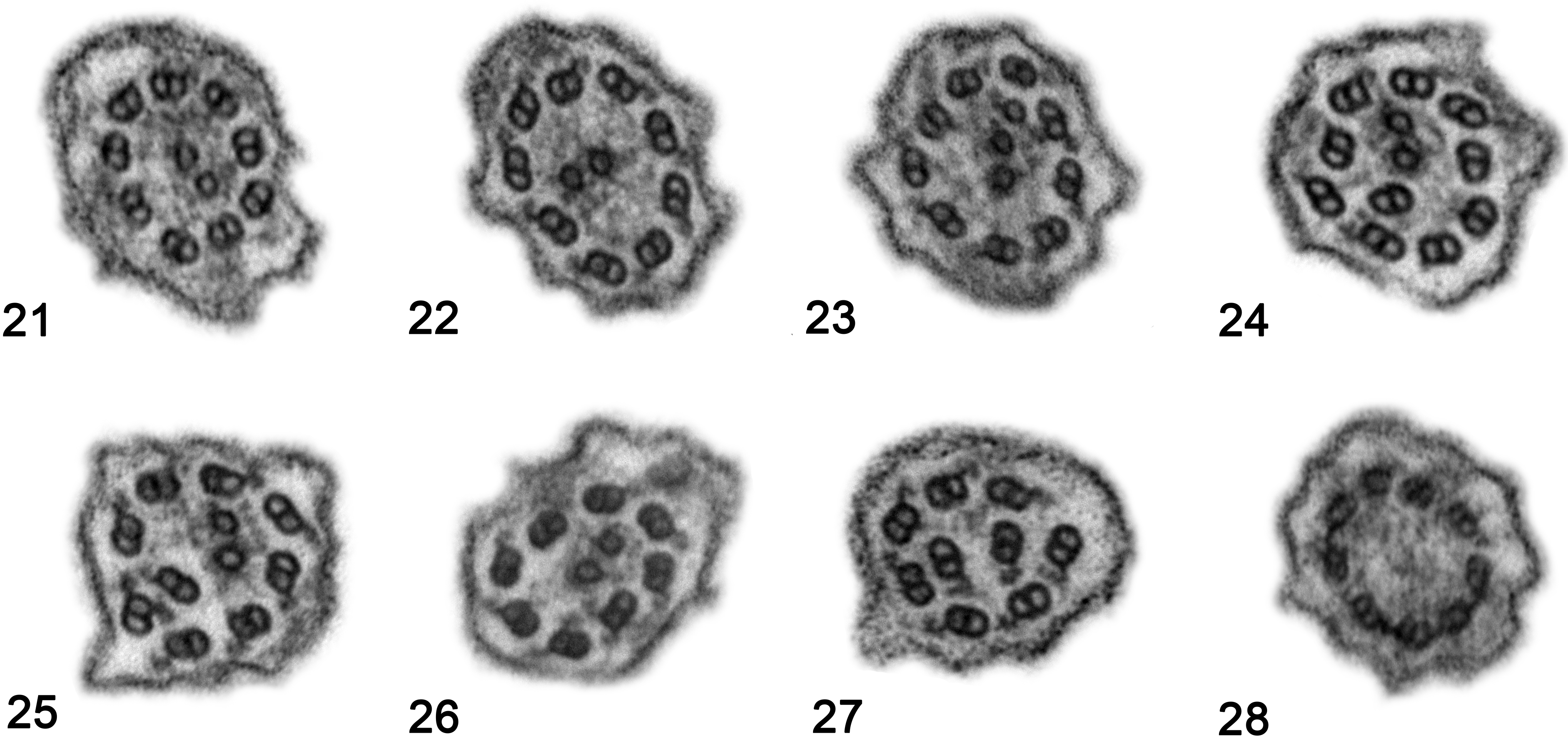
TEM of the nasal epithelium showed abnormalities in the majority of cilia, including deficiencies in the dynein arms in 84% of cilia and absence or shortening of both IDAs and ODAs in 22% or only IDAs in 62%. Abnormalities in the central apparatus were found in 65% of the assessed cilia, including absence of CSs in 18%, displacement of CSs in 47%, only 1 singlet in 3% and supernumerary singlets in 15%. Abnormalities in the peripheral apparatus were found in 60% of the evaluated cilia, including absence of PDs in 16%, displacement of PDs in 27%, transposition of PDs in 48% and supernumerary doublets in 15%. Nonspecific abnormalities were seen in 19% of the analyzed cilia, such as compound cilia (10% of cilia), ciliary edema (6%), electron-dense inclusions in the basal bodies (3%) and ciliary disorientation (Table 2, Figs. 21–28).
Discussion
In the respiratory epithelium, ciliary ultrastructural abnormalities can lead to chronic respiratory disease. Although PCD and SCD are functionally similar, the secondary forms predominantly show nonspecific ciliary microtubular abnormalities acquired from airway damage, which tend to return to functional and anatomical normality after proper treatment. 6 Thus, differentiating PCD from SCD has implications for patient prognosis. Although there is no consensus on diagnostic criteria of PCD, the analysis of ciliary ultrastructure by TEM remains important in veterinary medicine, as no other test for dogs has yet been standardized. Because most reported cases of PCD in dogs were clinically diagnosed with Kartagener syndrome, few reports include TEM evaluation. Thus, this study aimed to characterize the light microscopic and TEM characteristics of respiratory epithelial samples from dogs suspected of having PCD.
In the present study, of 15 chronic respiratory disease cases with suspected PCD, it was possible to confirm the diagnosis in 1 by combining macroscopic and ultrastructural findings. In addition to the alterations to the respiratory epithelium, no cilia were found in the oviduct epithelium from the dog with PCD in the present study. There was also clear evidence of hydrocephalus in this dog due to defects in the ependymal cilia of the ventricular system. 12 Hydrocephalus has been previously reported in at least 12 dogs with PCD. 3,7 –9,20,21,26,27
In most of the other 14 cases, neither TEM nor light microscopy permitted specific diagnosis. The histopathologic and ultrastructural changes were heterogeneous among the cases of SCD in this study. For the only case in which a sample of lung was also submitted for ultrastructural analysis (dog No. 14), the patient clinically recovered after undergoing a lung lobectomy, decreasing the suspicion of PCD or another diffuse interstitial lung disease. In 7 cases, the respiratory mucosa contained intact and degranulated mast cells, eosinophils, lymphocytes, and plasma cells, which is suggestive of an allergic process. In 1 dog examined here, the bronchial mucosa revealed marked infiltration of eosinophils, collagenolysis and fibrosis, raising suspicion of eosinophilic bronchopneumopathy (dog No. 11). Culture-confirmed
Although the histologic changes in the SCD patients in this study were varied, the number of abnormal cilia in these patients was intermediate between that of the PCD patient and the healthy controls: 8% of cilia were affected in the control cases, up to 18% in the SCD cases, and 84% in the dog with confirmed PCD. This mirrors what has been reported in humans, in which 4% to 10% of cilia may be abnormal in healthy populations, up to 20% in SCD, and most or all cilia in PCD. 24 In the present study, 84% of the dynein arms were abnormal in the cilia of the dog with confirmed PCD. This is consistent with human PCD, in which most patients have ultrastructural ciliary defects in the dynein arms, including isolated defects of ODAs, isolated defects of IDAs, or both. 4,6,16,17,22,24,30,31,35 Studies in humans and in animal models of PCD suggest that the ODAs are primarily responsible for ciliary beat frequency and force, whereas IDAs appear responsible for the ciliary bending pattern. 35 The PCD case of the present study had IDA abnormalities in 62% of cilia, transposition of PDs in 48% and absence of CSs in 18%. The transposition of PDs in the absence of CSs (8 + 1 arrangement) is attributed to defects in radial spokes, central sheath or nexin links. 30 The dog with PCD in this study had microtubular changes in 65% of cilia, while the nonconfirmed cases had only 13%. In humans, although microtubular abnormalities may be present in patients with PCD as well as those with other chronic diseases, 30,32 it has been concluded that patients with acquired ciliary alterations have only a minority of cilia with ultrastructural defects. 24 No microtubular irregularities were identified in the control cases in the present study, although microtubular abnormalities may be present in about 2% of the cilia in healthy dogs 34 and 2% to 5% in healthy humans. 29 This suggests that ciliary ultrastructural changes are present in the absence of disease, which is important in the interpretation of diseased epithelium.
PCD in dogs has been described in at least 19 different breeds,
19
but it is often reported in a single individual or among littermates. Because most reported cases were clinically diagnosed with Kartagener syndrome, few reports of PCD in dogs include TEM evaluation. The dog with PCD in the present study primarily showed loss of IDAs and microtubular defects. Mutations in
Several of the ultrastructural findings in this study were considered nonspecific. Ciliary disorientation, defined as misalignment of the CSs with an angle >20°, was found in all cases of the present study but not in the controls and is a secondary change in humans. 14,24 Thus, ciliary disorientation should not be used as a diagnostic criterion for PCD. In addition, compound cilia, consisting of multiple axonemes surrounded by a mutual cell membrane, are considered a nonspecific finding that can be present in a small subset of cilia in patients with any chronic respiratory disease, as well as in healthy individuals. 18,34 The present study revealed compound cilia in 10% of the assessed cilia in the PCD case, 9% in SCD and 6% in the controls and thus was not considered a discriminatory finding. The loss of cilia, observed on multiple biopsies with no evidence of squamous metaplasia, could be suggestive of ciliary aplasia or hypociliosis. A genetic etiology has not been identified for these lesions, and instead they are suggested to be secondary to another disease process. 16 Other nonspecific abnormalities described in the literature are the presence of electron-dense basal body inclusions, 23 ciliary edema, membranous bubbles and squamous metaplasia. 6 Electron-dense basal body inclusions are suggested to be a technique artifact and were found in 3% of the assessed cilia in the PCD case, 2% in SCD cases and 2% in the control cases. Membranous bubbles were found in 1% of the assessed cilia from 1 case with SCD and ciliary edema in 6% from both PCD and SCD cases. Squamous metaplasia of the ciliated epithelium is an important pathologic change that occurs secondary to damage of the respiratory mucosa. It was found in different degrees in the specimens of this study but completely hindered the ultrastructural evaluation in 1 case. If the normal epithelium is entirely replaced by squamous epithelium, no cilia can be evaluated by TEM, emphasizing that biopsies for ultrastructural ciliary evaluation should be obtained from intact mucosa, avoiding secondary lesions. Light microscopic examination should be performed to verify sample suitability for TEM.
It is sometimes difficult to distinguish primary and acquired defects using TEM. The ultrastructural examination requires technical acuity, as well as expertise in interpretation to identify normal variations and nonspecific changes. Multiple factors hamper the TEM analysis as a diagnostic test for PCD, including secondary damage to the respiratory epithelium and poor fixation that makes the sample unsuitable for evaluation. A suitable specimen should be obtained by scrape or brush biopsy from nasal, tracheal, or bronchial ciliated epithelium and fixed in 3% to 4% glutaraldehyde. In the present study, the ciliary ultrastructural findings were similar among samples of nasal, tracheal and bronchial epithelia taken from the same individual. Although a larger number of samples are preferable for PCD evaluation, mucosal specimens from any of these anatomic sites should be suitable.
In dogs, as in humans, the diagnosis of PCD lacks a “gold-standard” test and is instead based on combinations of tests, including measuring nitric oxide in nasal air as a screening test, high-speed video-microscopic analysis, TEM and genotyping. 13 In dogs, other supportive tests for PCD are not standardized yet, but ciliogenesis in culture may be useful and genetic tests are gaining prominence. 5,19 PCD diagnosis can be supported by high-speed video microscopy to examine ciliary beat, inasmuch certain ciliary beat patterns correlate with ultrastructural changes and genetic variants. 4,25 In the present study, the assessment of the ciliary function was not performed, and all the non-PCD cases were classified as SCD based on the lack of ultrastructural evidence for a primary condition, such as dynein arms and microtubular abnormalities in the majority of the cilia.
PCD is a rare and poorly characterized disease in animals. Quantitative and qualitative ultrastructural analysis plays an important role in the diagnosis of ciliary dyskinesia. PCD can be confirmed by TEM if ciliary ultrastructural abnormalities exist, although it is possible that the ciliary defects in dogs with PCD may be functional rather than ultrastructural. Dynein arms and microtubular abnormalities in most or all cilia can confirm the diagnosis, but ciliary disorientation, compound cilia, ciliary edema and axoneme bubbles should not be used as diagnostic criteria for PCD. Even with advances in genetic testing, methodical ultrastructural analysis can provide critical information to help identify candidate genes. Future studies are necessary to identify causes of chronic respiratory disease in dogs and whether ultrastructural analysis will be valuable in defining these diseases. This study provides information regarding ultrastructural changes seen in dogs with primary and secondary ciliary dyskinesia.
Footnotes
Acknowledgements
We thank Dr James Collins for supporting this research and Dean Muldoon and Don Aryiakumar for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
