Abstract
Naked mole-rats (NMRs;
Keywords
Naked mole-rats (NMRs;
In the first case, a 22-year-old male captive-born worker NMR presented to the zoo veterinary service (Brookfield Zoo, Chicago Zoological Society, Brookfield, IL) for a mass in the right axillary region. The animal was in an established zoo colony and had an otherwise unremarkable clinical history. On physical examination, the animal had a 1.5-cm diameter, mottled dark red-to-purple, firm, and movable subcutaneous mass surrounded by fluctuant material. Aspiration of the mass recovered blood along with a small amount of clear, colorless, tacky fluid. The mass was surgically removed, placed in 10% neutral buffered formalin, and submitted with the cytologic preparations for pathological evaluation. Romanowsky (Diff-Quik) stained impression smears of the aspirated fluid contained numerous individual and variably sized cohesive aggregates of polygonal to round cells with abundant pale basophilic, granular cytoplasm on a background of erythrocytes, and proteinaceous material (Fig. 1). Some cells contained individual or multiple discrete, colorless, cytoplasmic vacuoles (Fig. 1). Histologically, sections were comprised almost exclusively of neoplastic tissue, namely, a densely cellular, multi-nodular neoplasm composed of epithelial cells arranged in variably sized lobules, nests, and rare disorganized acinar-like structures supported by scant fibrovascular stroma and intervening fibrous septa (Fig. 2). Neoplastic cells were cuboidal to polygonal with moderate amounts of pale eosinophilic cytoplasm ranging from granular to globular to foamy (Figs. 3, 4) with distinct cell borders. Nuclei were central and round, with finely dispersed chromatin and 1 to 2 large nucleoli (Figs. 3, 5). Anisocytosis and anisokaryosis were moderate, with infrequent karyomegaly, binucleate cells (Figs. 3, 4), and chromatin clearing interpreted as pseudoinclusions (Fig. 4). Mitoses were generally infrequent (0–1 per 400× field); however, some sections had a high mitotic index (4 per 400× field, Figs. 3a, 4) with bizarre mitotic figures. Throughout the mass, blood vessels were engorged, lined by plump (reactive) endothelium. Scattered throughout the mass were individual necrotic neoplastic cells, low numbers of neutrophils, mild to moderate lymphoplasmacytic infiltrates, and hemorrhage with rare erythrophagocytosis. Neoplastic tissue extended to all surgical margins; thus, local invasion could not be determined.
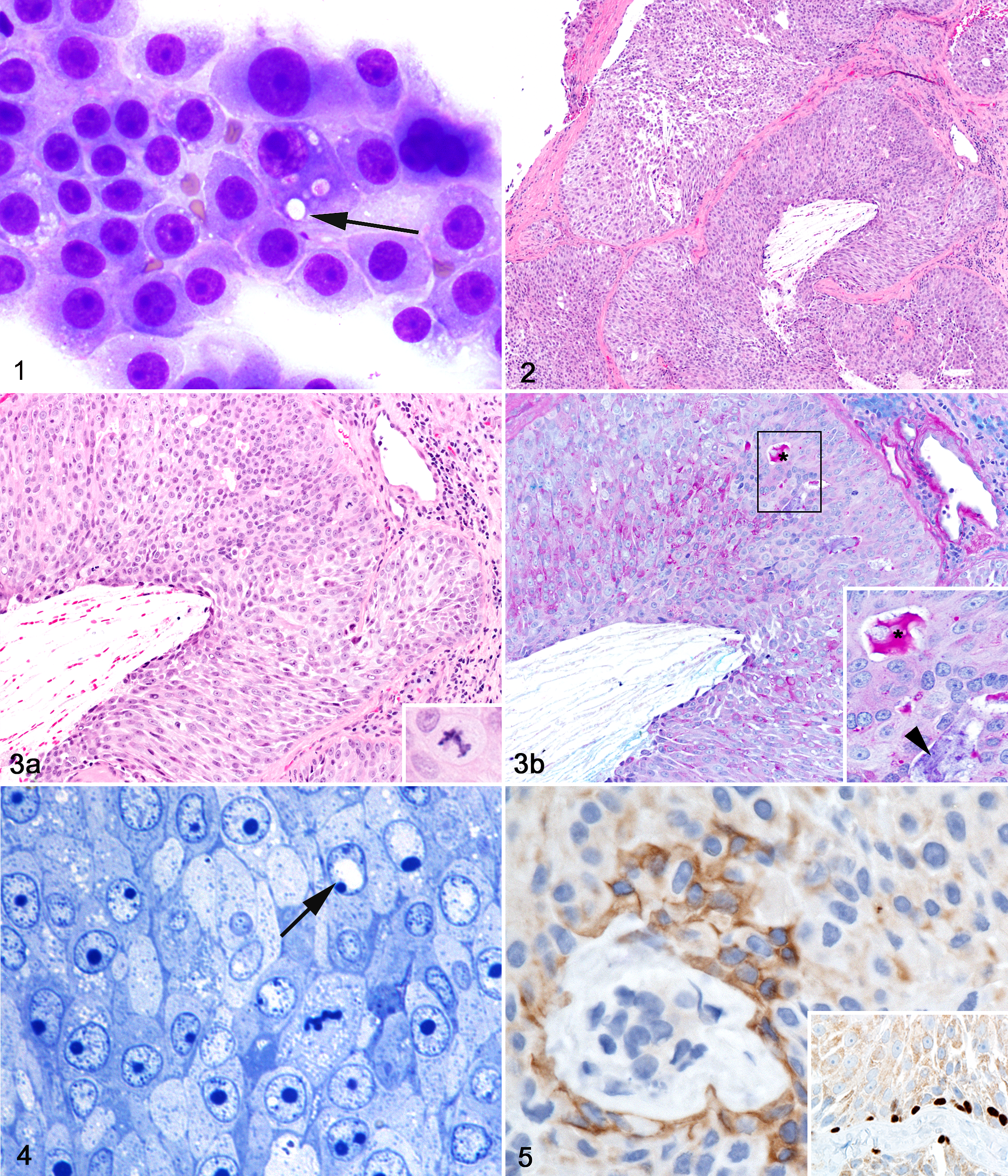
Axillary adenocarcinoma, naked mole-rat.
The histochemical stains Periodic Acid Schiff (PAS) and Alcian Blue (AB) were utilized in attempts to distinguish the cytoplasmic vacuoles of the neoplastic cells. PAS-AB-stained sections of the mass revealed intense PAS positive granular cytoplasmic staining in scattered cells (Fig. 3b) and extracellular accumulation of positive stained mucoid material within the lumens of poorly formed acini. Antibodies against (pan)-cytokeratin (CK; Z0662, rabbit polyclonal, Dako),
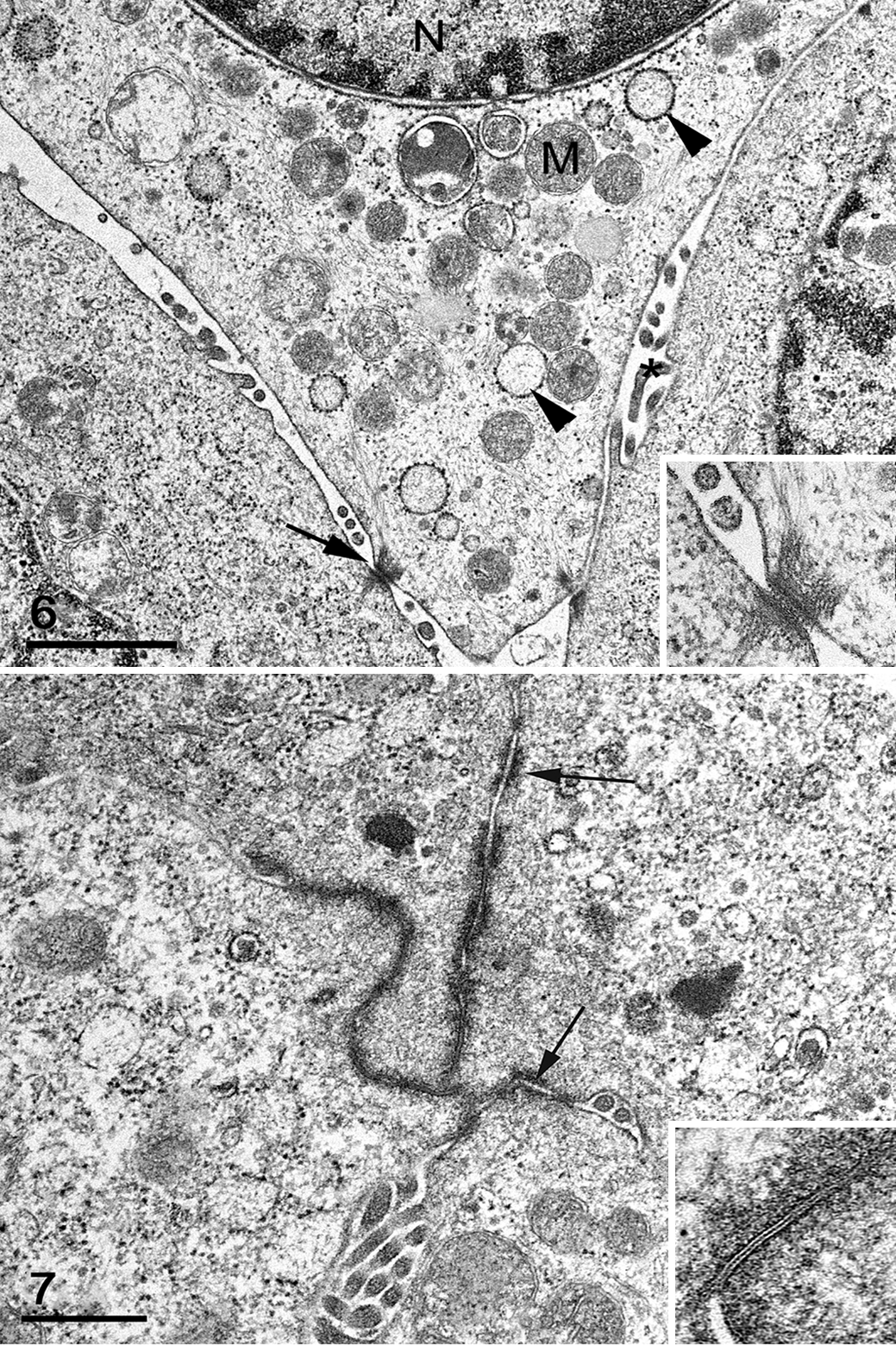
Subsamples of the mass were evaluated and imaged using a JEOL JEM 1400 transmission electron microscope. Ultrastructurally, cells had features common to neoplasms such as prominent mitochondria and vesiculated rough endoplasmic reticulum (Fig. 6). 14 Nuclear findings included aggregation and margination of chromatin and marginated nucleoli (Fig. 6 and data not shown). 14 Some nuclei contained large electron-lucent pseudoinclusions, as noted on HE and thin sections (Fig. 4). Multifocally, neoplastic cells were bound by intercellular junctions, including tight and gap junctions and desmosomes (Figs. 6, 7), a finding common in normal epithelial cells and carcinomas. 14 Several characteristics distinguished the mass as an adenocarcinoma: intercellular canaliculi and lumina formed by apices of adjacent cells linked by terminal bars and numerous apical (and intercellular) microvilli and cell projections (Figs. 6, 7). 14 Based on cytologic, histologic, histochemical, immunohistochemical, and ultrastructural evaluation, the mass was diagnosed as an undifferentiated adenocarcinoma of unknown origin. Mammary or salivary gland origin was considered most likely based on mass location, cytomorphology, and staining characteristics, although metastasis from another neoplasm or poorly differentiated carcinoma could not be ruled out.
Axillary adenocarcinoma, naked mole-rat.
Following removal of the mass, the NMR was successfully returned to the colony and has continued to maintain its place in the social structure. Three months postoperatively, there is a small amount of presumed scar tissue at the site of surgical excision but no clinical signs or recurrence of the mass.
The second case of NMR neoplasia came from a different zoological institution (National Zoological Park, Washington, DC). A confirmed 16-year-old, though estimated at least 20-year-old, male captive-born worker NMR was humanely euthanized due to unresolving severe facial dermatitis and progressive weight loss. Upon histologic examination of the stomach, a densely cellular neoplasm was detected, expanding and effacing the gastric submucosa and mucosa (Figs. 8, 9). The mass was composed of neoplastic epithelial cells arranged in tightly packed nests and trabeculae among a fine fibrovascular stroma (Fig. 10). Neoplastic cells were polygonal with small to moderate amounts of finely granular to foamy eosinophilic cytoplasm and variably distinct cell borders (Fig. 11). Nuclei were round to oval with coarse to vesicular chromatin and 1–2 variably apparent nucleoli. Anisokaryosis and anisocytosis were mild with infrequent binucleate cells, karyomegaly, and <1 mitosis per ten 400× fields. Neoplastic cells focally invaded the gastric tunica muscularis, and adjacent thin-walled vessels (likely lymphatics) contained luminal rafts of clumped neoplastic cells; however, no metastases were detected. Scattered throughout the mass were mild lymphoplasmacytic infiltrates. Immunohistochemical staining using antibodies against human synaptophysin (697301, mouse monoclonal, MP Biomedicals) (Fig. 12a) and human chromogranin (CM010B, mouse monoclonal, Biocare Medical) (Fig. 12b) confirmed that the neoplastic cells had neuroendocrine origin; thus, a diagnosis of gastric neuroendocrine carcinoma (carcinoid) was made.
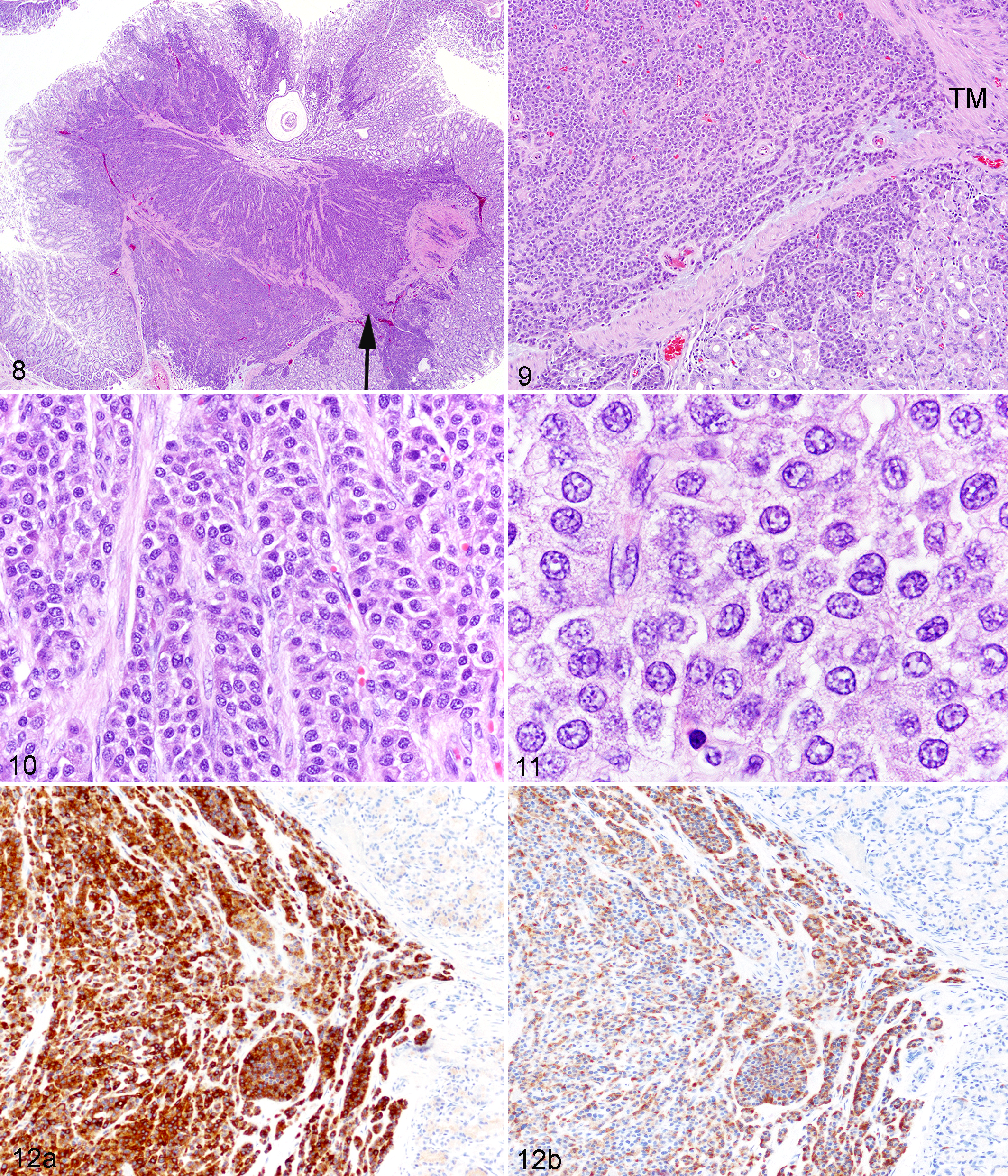
Gastric neuroendocrine carcinoma, naked mole-rat.
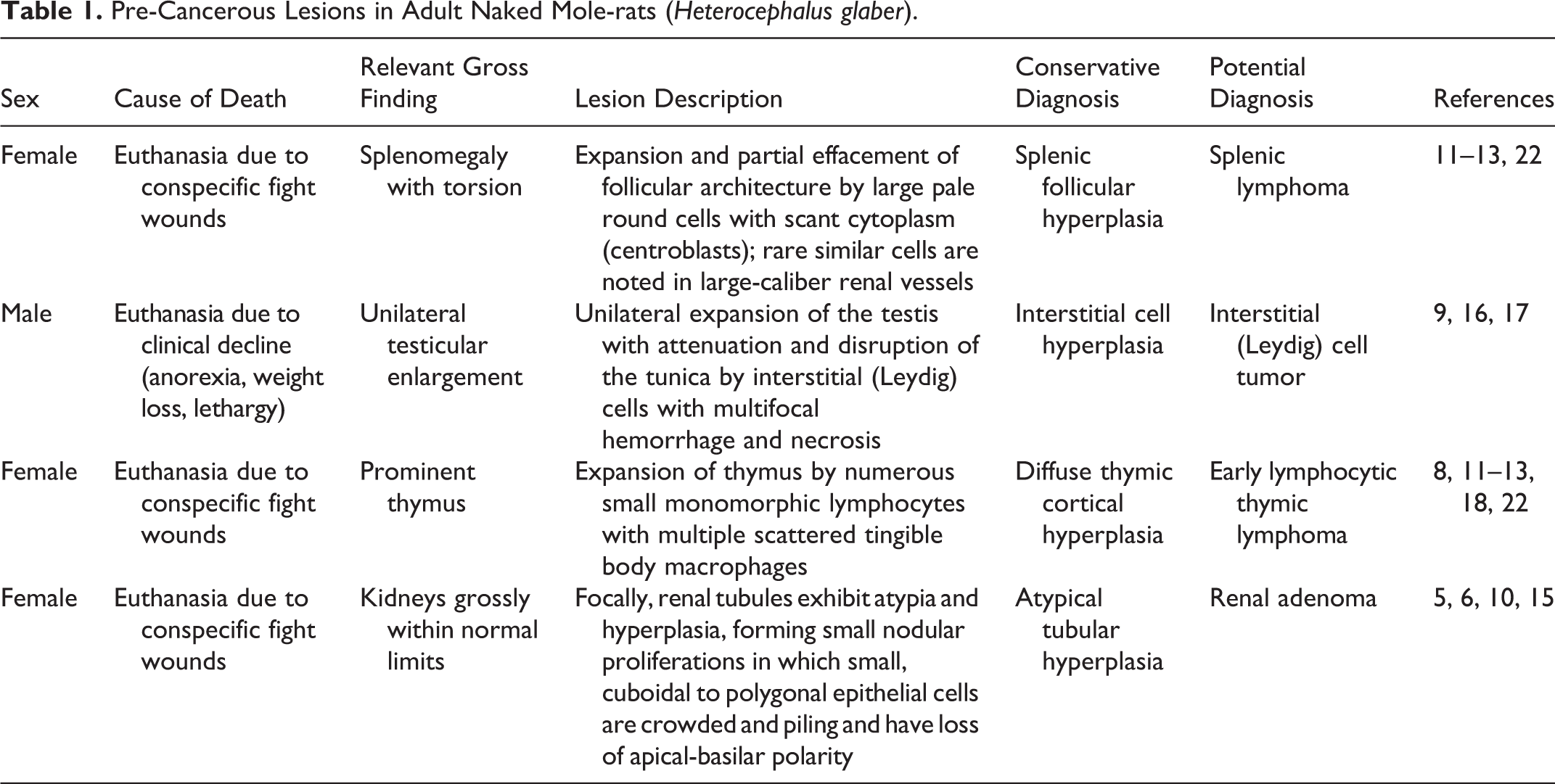
These cases represent the first formal reports of cancer in the NMR, a rodent species best known for its extreme longevity and apparent resilience to typical health span–limiting diseases, including cancer. Our previous investigations of the NMR colony at the Brookfield Zoo found 4 NMRs with lesions considered to be examples of atypical hyperplasia, pre-cancerous changes, or benign neoplasia depending on which diagnostic criteria are followed (Table 1).
4,6,16,22
In 3 of the 4 cases, NMRs were euthanized due to conspecific bite wounds, a common finding in the Brookfield Zoo colony
6
; thus, these pre-cancerous lesions were incidental. In that study,
6
conservative diagnoses were applied to all of these lesions based on their ambiguous morphology and given that no other cases of cancer or pre-cancerous change had been described in NMRs (Table 1). Furthermore, published guidelines developed for laboratory rodents were followed, which include strict criteria for the diagnosis of neoplasia.
4,8,10
–13,15,16,18,22
In contrast, the 2 cases in the current report of poorly differentiated adenocarcinoma and gastric neuroendocrine carcinoma
Pre-Cancerous Lesions in Adult Naked Mole-rats (
In instances of proliferative lesions in species without established criteria for neoplasia or malignancy, such as NMRs, it is difficult to distinguish benign, pre-cancerous, and malignant processes.
4,10
–13,16,22
However, with additional investigation and documentation of these lesions in available populations, a basis for future diagnostic criteria may be defined. NMRs clearly have a very low incidence of cancer when compared to laboratory rodents and other mammal species with similar life spans. However, our report documents for the first time that NMRs
Footnotes
Acknowledgments
We would like to thank the veterinarians and animal care staff of the Brookfield Zoo and National Zoological Park, Stephanie Rhodes, and the Fragile Kingdom staff for providing case materials; Renee Walker and staff from the Histology Lab of University of Illinois Veterinary Diagnostic Laboratory for slide preparation. We thank the University of Washington (UW) Histology and Imaging Core and Comparative Pathology Program staff, Mac Durning of the Washington National Primate Research Center, Lena Hart of UW Anatomic Pathology Lab, the staff at University of Iowa Comparative Pathology Laboratory, and Bobbie Schneider of the Electron Microscopy Resource at Fred Hutchinson Cancer Research Center for technical assistance. We appreciate the ultrastructural expertise of Vince Meador, DVM, PhD, DACVP. Finally, we thank Kathleen S. Montine, PhD, ELS, for critical review and copyediting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
