Abstract
Canine myocarditis is a rare but serious health concern, potentially causing heart failure and death. Antemortem diagnosis is hampered by the numerous causes, nonspecific course, and dearth of diagnostic criteria. Currently, definitive diagnosis can only be made after death. The current human diagnostic gold standard is endomyocardial biopsy pairing cardiac histopathology with immunohistology to enhance detection of often-multifocal disease. We evaluated immune response markers in the canine heart to establish similar immunohistologic criteria. We hypothesized that myocardial major histocompatibility complex class II (MHCII), cluster of differentiation 3 (CD3), and ionized calcium binding adapter molecule 1 (Iba1), markers increased in human myocarditis, would be increased in canine myocarditis cases. Archived paraffin-embedded myocardial tissue from 22 histopathologically confirmed cases of adult and juvenile myocarditis and 23 controls was analyzed by immunohistochemistry for MHCII, CD3, and Iba1, and the fraction of myocardium with labeling was determined. All 3 markers were significantly increased compared with controls across the entire section: Iba1, 10.1× (P < .0001, Mann-Whitney U test); MHCII, 3.04× (P = .0019); and CD3, 4.4× (P = .0104). To mimic off-target biopsy, samples from 2 mm2 outside of inflammatory foci were analyzed, and these showed significant increases in Iba1 by 3.2× (P = .0036, Mann-Whitney U test) and CD3 by 1.2× (P = .0026). These data show diffusely increased immune response markers with canine myocarditis, with detection potentially independent of tissue sampling. Thus, endomyocardial biopsy and immunohistochemical detection of MHCII, CD3, and Iba1 may permit sensitive antemortem diagnosis of canine myocarditis.
Keywords
There have been few recent advances in our ability to diagnose and treat myocarditis in companion animals. Despite its substantial adverse health impact, canine myocarditis research is underrepresented, and thus the causes and pathogenesis remain elusive. This impairs our ability to diagnose, treat, and prognosticate this underdiagnosed disease. 15
The clinical diagnosis of canine myocarditis is presumptive and the underlying cause is often not identified. Our recent retrospective study screened archived cases with a comprehensive polymerase chain reaction (PCR) panel and demonstrated the limitations of current targeted diagnostic tests: Agents were not detected in 53% of cases, particularly in adult cases of myocarditis. 17 Pathogen-negative canine myocarditis cases suggest immune-mediated myocarditis, a diagnosis made by exclusion in humans. Canine myocarditis is characterized by nonspecific clinical signs, many of which overlap with noninfectious and degenerative cardiac disease. 19 Noninvasive major and minor clinicopathologic criteria have recently been proposed for the antemortem diagnosis of myocarditis in dogs, but screening for the multitude of possible pathogens is expensive and often unrewarding in identifying a causative agent.15,17,25 Considering that infectious, immune-mediated, noninfectious, and noninflammatory canine cardiac disease requires different treatment strategies, knowledge and testing gaps could lead to erroneous or delayed treatment, and thus negatively impact health. This study aimed to improve methods of antemortem detection of myocarditis and aid in the differentiation between the broad categories of inflammatory and noninflammatory heart disease.
In human patients, routine unequivocal diagnosis of myocarditis is via endomyocardial biopsy (EMB) samples evaluated using immunohistologic criteria along with PCR for viral nucleic acid as established by the European Society of Cardiology Working Group and World Health Organization Task Force.3,9,20 Immunohistochemistry (IHC) to detect focal to diffuse macrophage and T cell infiltrates and expression of human leukocyte antigen (HLA) class II molecules has been demonstrated to enhance detection of myocarditis across all stages of the disease, reduce interobserver variability, and provide prognostic information compared with histopathology.2,3,14EMB is feasible in dogs but has not been utilized as a standard diagnostic test due to technical expertise and cost.12,21,22 Furthermore, the absence of standardized evaluation criteria is a significant barrier to performing EMB in dogs. While testing for specific agents has largely not been fruitful, having immunohistologic guidelines for the clinical diagnosis of canine myocarditis will form the basis for improving our diagnostic tool kit, advance the standard of care to the level of human medicine, and inform treatment of dogs with myocarditis.17,25
With this in mind, the aim of this study was to investigate the myocardial immune response marker profile in the canine heart as well as establish immunohistologic criteria for the antemortem detection of myocarditis, similar to that available for humans. We hypothesized that major histocompatibility complex class II (MHCII), cluster of differentiation 3 (CD3), and ionized calcium binding adapter molecule 1 (Iba1) are diffusely increased in the myocardium of both adult and juvenile dogs with myocarditis. We further predicted that these markers are increased in tissue outside of distinct inflammatory foci, thus relevant to off-target EMB.
Materials and Methods
Case Selection
Formalin-fixed, paraffin-embedded (FFPE) canine myocardial tissue was identified from the archives of the Cornell University Animal Health Diagnostic Center (AHDC), including tissues from previous studies of dogs with diagnoses of “myocardial fibrosis,” “myocarditis,” “cardiac fibrosis,” “chronic myocarditis,” “fibrosing cardiomyopathy,” “cardiac failure,” “restrictive cardiomyopathy,” or “end-stage heart” and several myocarditis cases collected after November 2015.8,17 Animals without myocardial tissue available in blocks were excluded. Cardiac tissue was characterized for myocarditis based on myocardial inflammation, necrosis, and degeneration as previously described. 8
Controls included dogs with ostensibly healthy hearts from previous studies8,17 of similar age range. Additional controls with nonmyocarditis cardiac disease were identified using a database search for arrhythmogenic right ventricular cardiomyopathy, pulmonic stenosis, and mitral valve degeneration.
Immunohistochemistry
IHC for MHCII (mouse antihuman MHCII antibody #F0817, Dako, Carpinteria, CA, USA) was first tested in canine splenic tissues; this antibody has been used for canine flow cytometry applications. 18 A dilution series with combinations of varying primary and secondary antibody concentrations was performed to optimize labeling and a dilution of primary 1:2000 and secondary 1:200 was selected based on producing an appropriate level of labeling on expected cell types (cells with macrophage, dendritic cell, or B cell morphology) without off-target labeling of other cells. As MHCII is expected to be increased during an infectious process, optimized antibody dilutions were tested on canine myocardial tissue with confirmed parvoviral myocarditis (IHC and PCR). Splenic tissue processed with omission of primary antibody served as negative control.
IHC for MHCII was performed on rehydrated and deparaffinized 5-µm-thick myocardial sections. Endogenous peroxidase was blocked (0.3% hydrogen peroxide in phosphate buffered saline). Heat antigen retrieval (microwaving for 20 minutes) was performed in antigen unmasking solution (Vector Laboratories, Burlingame, CA, USA) with Tris-buffered saline. Sections were blocked with avidin, biotin, and protein (Dako) prior to incubation with primary antibody (mouse antihuman MHCII antibody, #F0817, Dako) at 1:2000 dilution at 4°C for 24 hours, then were incubated in secondary antibody (biotinylated goat antimouse IgG) at 1:200 dilution at room temperature for 30 minutes. Sections were incubated with Vectastain ABC Elite reagent (Vector Laboratories) for 30 minutes at room temperature followed by 3,3′-Diaminobenzidine (DAB) chromogen (Dako) and counterstained with hematoxylin. Canine myocardial tissue with PCR-confirmed parvoviral myocarditis was used as a positive control. Sections incubated without primary antibody served as negative controls as indicated above.
Iba1 (polyclonal rabbit antirat Iba1 antibody, #019-19741, Wako, Richmond, VA, USA) and CD3 (monoclonal mouse antihuman antibody, #PA00553, Leica, Wetzlar, Germany) IHC was performed by the AHDC Histopathology Laboratory on an automated immunohistochemical stainer, the Leica Bond Max, using a Bond Polymer Refine Detection Kit (DAB, #D59800). Appropriate positive and negative controls were used. Images were scanned using a Roche Ventana iScan HT (Rotkreuz, Switzerland) slide scanner. Slides with significant staining artifacts, such as pronounced striping, were excluded from subsequent image analysis.
Image Analysis
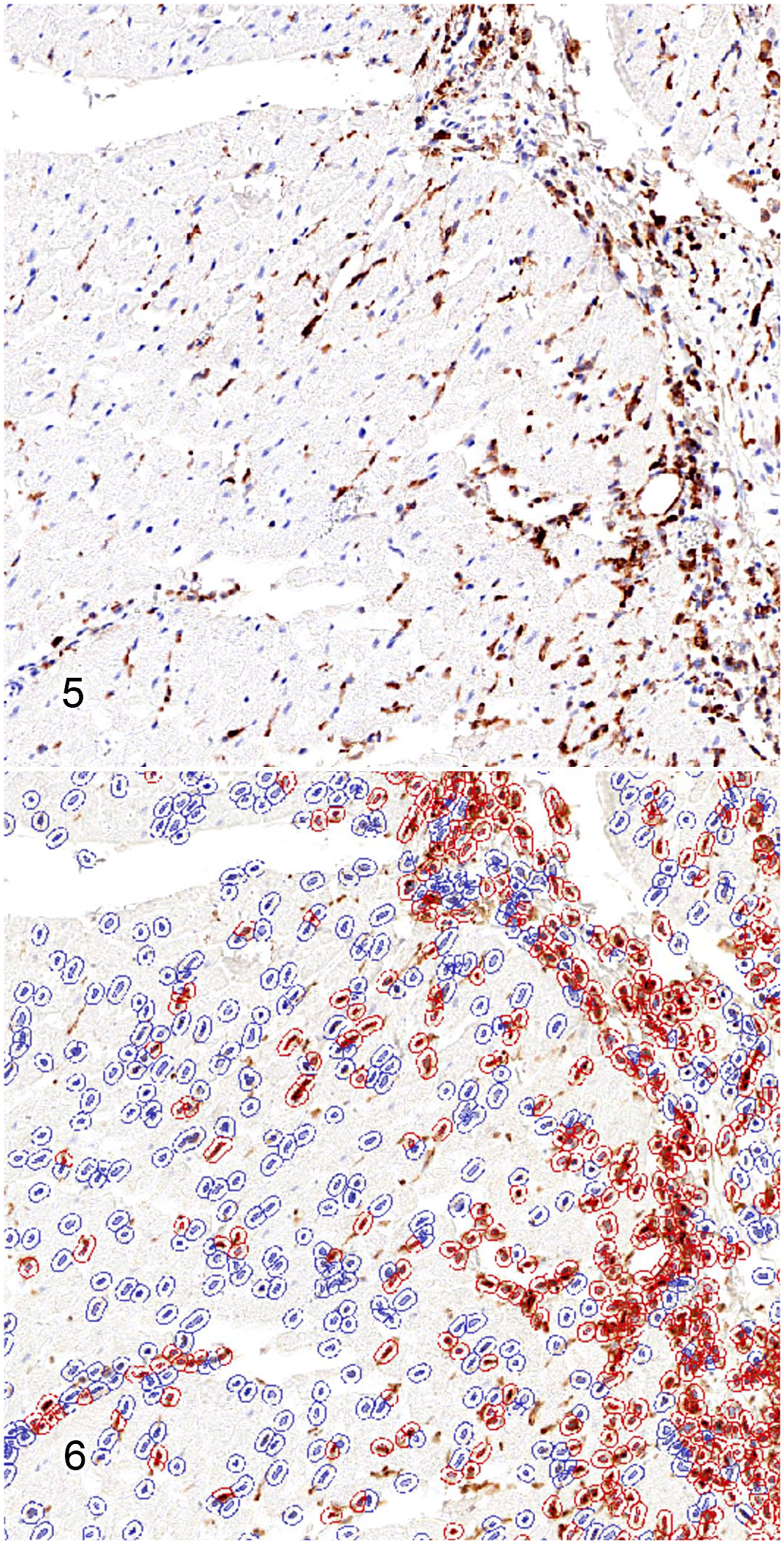
The fraction of cells per slide with optically detectable DAB chromogen signal was determined for each marker by a blinded researcher using QuPath image analysis (version 0.1.2, Edinburgh, Scotland, UK). 1 Using the program cell detection algorithm, cells were identified based on measurements of nuclear intensity and morphology, including area, circularity, area ratio of nucleus: cell, and staining intensity of hematoxylin and DAB. 1 The default positive cell detection function was then used to measure mean cytoplasmic DAB optical densities for each cell. Intensity threshold settings for positive cell classification were determined manually and then applied across all slides.
Marker labeling differences between inflammatory foci and sites distant from inflammatory foci were compared with the same analysis protocol using 2.0 × 2.0 mm2 sample regions, an area similar in size to an EMB sample. Inflammatory foci were defined as collections of leukocytes aggregating in numbers of greater than 20 per 900 µm 2 . Samples taken within areas of inflammation represented an “on-target” EMB. Based on the often multifocal nature of myocarditis, to mimic an “off-target” EMB, samples were taken at least 1 cm distant from the edge of an inflammatory focus. Three off-target samples were taken per slide to simulate sampling and their positive cell percentage values were averaged. Some but not all “off-target” samples included subendothelial myocardium, an area targeted by in vivo EMB that includes fibrous tissue, hemorrhage, or artifacts, thus reducing the overall cells per unit area.
Statistical analyses were performed using JMP Pro 15. Case and control IHC labeling was compared using Mann-Whitney (MW) test with significance defined as a P < .05. Candidate threshold or cutoff values for each marker were determined by receiver operating characteristic (ROC) analysis and the sensitivity and specificity determined from the ROC curves. For markers that had value with sensitivity and specificity >75%, the value that generated the highest sensitivity and specificity was selected as the threshold value; positive predictive value (PPV) and negative predictive value (NPV) for selected threshold values were then calculated from the study data. 23 To analyze the usefulness of a combination of markers for detecting myocarditis, predictive modeling was performed using decision tree partitioning, with a minimum group size set at 5.
Results
Case Demographics
Adult (n = 6) and juvenile (n = 6) myocardial tissue blocks from the previous canine studies were selected at random to be included in this study with an additional 4 adult and 6 juvenile archived cases collected after November 2015. In total, histopathologically confirmed cases of myocarditis in 10 adult dogs (4–14 years of age, 6 archived, 4 new) and 12 juvenile dogs (less than 2 years of age, 6 archived, 6 new) were selected. The demographic characteristics of this cohort are detailed in Supplemental Table S1. Etiologies included viral (n = 3), bacterial (n = 5), trypanosomal (n = 1), and undetermined causes (n = 13). The median age of the adult cohort was 84 months (range = 48–168 months), and the median age of the juvenile cohort was 2.4 months (range = 1–24 months). Breeds in the adult cohort consisted of 3 mixed breed dogs and one each of Labrador Retriever, Greyhound, Weimaraner, Shetland Sheepdog, Dachshund, Yorkshire Terrier, and Bichon Frise. Breeds of the juvenile cohort included French Bulldog (3), German Shepherd (2), and one each of Siberian Husky, Saint Bernard, Vizla, Plott Hound, Labrador Retriever, Golden Retriever, and Labradoodle. In the adult cohort, there were 1 intact male, 1 intact female, 3 castrated males, and 5 spayed females. In the juvenile cohort, there were 4 intact males, 6 intact females, and 2 castrated males.
Of the “healthy heart” control animals identified, the 5 juveniles had a median age of 3 months (range = 2–6 months) and the 5 adults had a median age of 3 years (range = 2–12 years). Juvenile breeds included one each of Chihuahua, Labrador Retriever, Bulldog (not otherwise specified), German Shepherd, and mixed breed. Adult breeds included one each of Bulldog (not otherwise specified), Rat Terrier, Boston Terrier, Cocker Spaniel, and Cane Corso. There were 3 intact males and 2 intact females in the juvenile group. In the adult group, there were 1 intact male, 1 intact female, 2 castrated males, and 1 spayed female.
The animals selected for the nonmyocarditis cardiac diseases control group had a median age of 84 months (range = 1–132 months). These animals included 4 arrhythmogenic right ventricular cardiomyopathy (7–11 years of age), 5 pulmonic stenosis (4 weeks–7 years of age), and 4 mitral valve degeneration (4–12 years of age). There were 5 intact males, 1 intact female, 5 castrated males, and 2 spayed females. Breeds in the group consisted of Boxer (2), Bulldog not otherwise specified (2), and one each of Airedale Terrier, Portuguese Water Dog, Cavalier King Charles Spaniel, Staffordshire Bull Terrier, English Shepherd, Chihuahua, Maltese, mixed breed, and Bichon Frise.
Image Analysis
All 3 immunostains were available for the majority of cases and controls: MHCII (cases, n = 22; controls, n = 23), Iba1 (cases, n = 22; controls, n = 23), and CD3 (cases, n = 22; controls, n = 22) were analyzed via image analysis. Animals with myocarditis had an average of 3474 (±1192) total cells per mm2, whereas controls had 2501 (±733) total cells per mm2; notably, the number of cells is expected to vary widely based on factors including overall cell orientation (ie, cross vs longitudinal cardiomyocytes) and patient age. The average positive cell detection of all 3 markers via whole-slide image analysis was significantly increased in dogs with myocarditis compared with all controls (Table 1 and Figs. 1–6). Increased detection of Iba1 and CD3 corresponded to increased numbers of infiltrating leukocytes. MHCII endothelial immunoreactivity along with interstitial leukocyte and cardiomyocyte labeling was observed in the hearts of dogs with myocarditis. Iba1 detection was highest (Mann-Whitney U test; P < .0001) and was 10.1× that of controls, followed by MHCII (3.04×, MW P = .0019) and CD3 (4.4×, MW P = .0104). This trend was similar regardless of age group. Whole-slide positive cell detection results are summarized in Table 1. Marker detection ratios compared by groups are shown in Supplemental Table S2.
Comparison of canine myocarditis case and control whole-slide myocardial inflammatory marker percent immunoreactive cells.
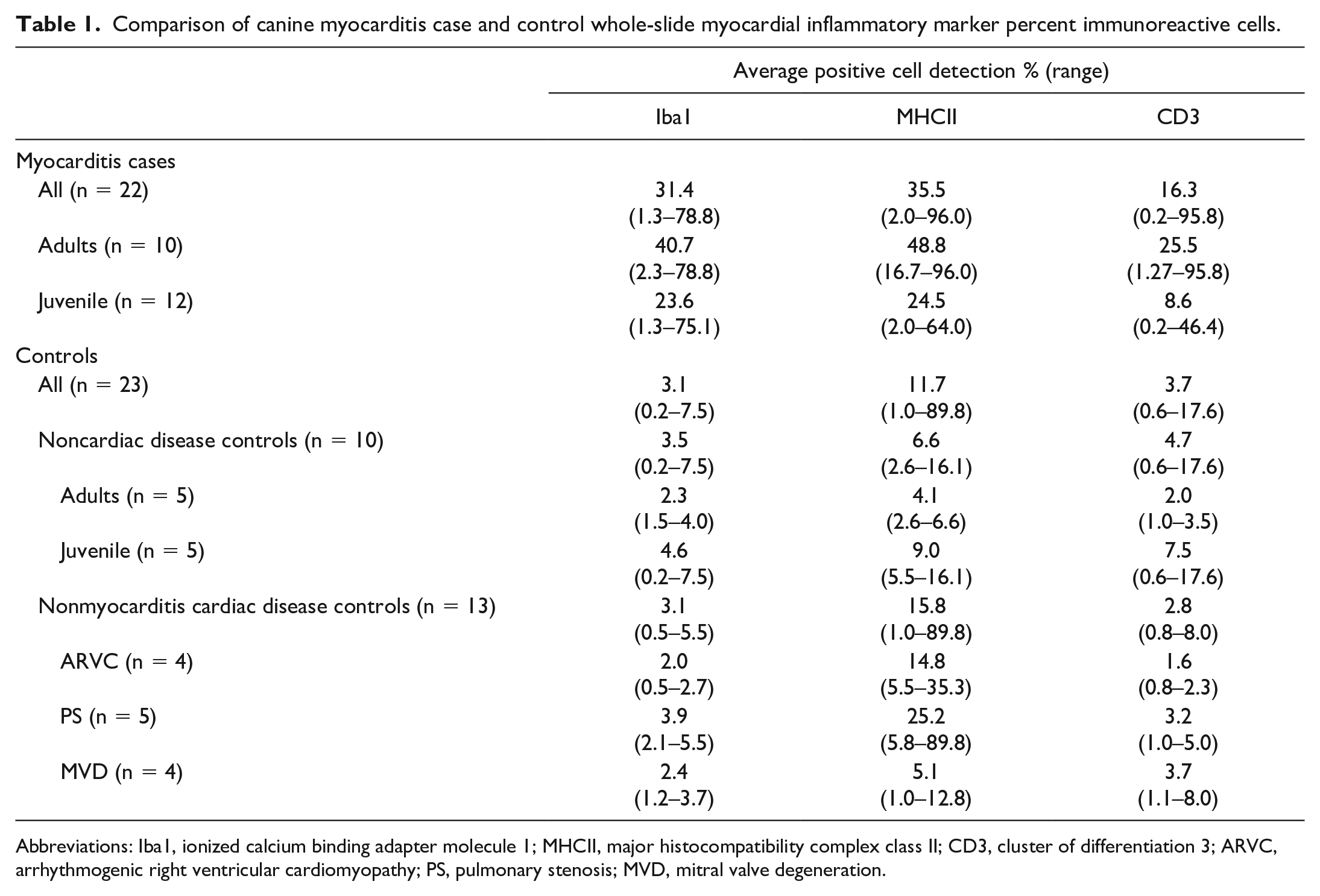
Abbreviations: Iba1, ionized calcium binding adapter molecule 1; MHCII, major histocompatibility complex class II; CD3, cluster of differentiation 3; ARVC, arrhythmogenic right ventricular cardiomyopathy; PS, pulmonary stenosis; MVD, mitral valve degeneration.
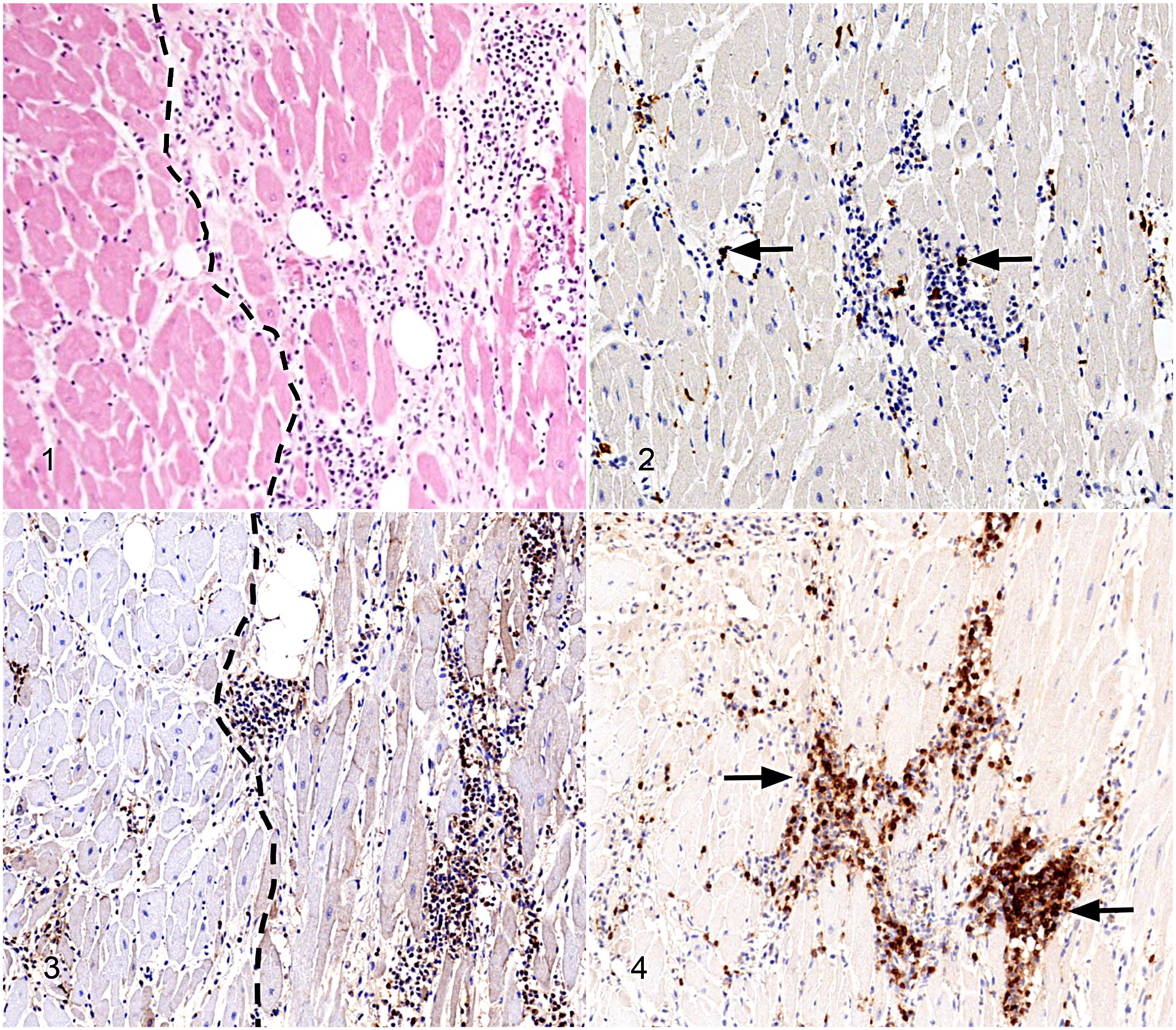
Myocarditis, myocardium, adult dog, case 1. Representative images of inflammatory markers in an adult dog with myocarditis. Approximately the same field is shown in each figure.
Myocarditis, myocardium, adult dog, case 2.
Marker detection was also increased in areas of myocardial tissue distant from inflammatory foci (mimicking “off-target” biopsy samples) compared with control animals: Iba1 was elevated by 3.2× (MW P = .0036) and CD3 by 1.2× (MW P = .0026). MHCII was elevated but not significantly over controls. Image analysis average positive cell detection results comparing inflammatory foci and nonfoci are summarized in Table 2.
Comparison of myocardial inflammatory marker percent immunoreactive cells between inflammatory and noninflammatory foci in canine myocarditis cases.
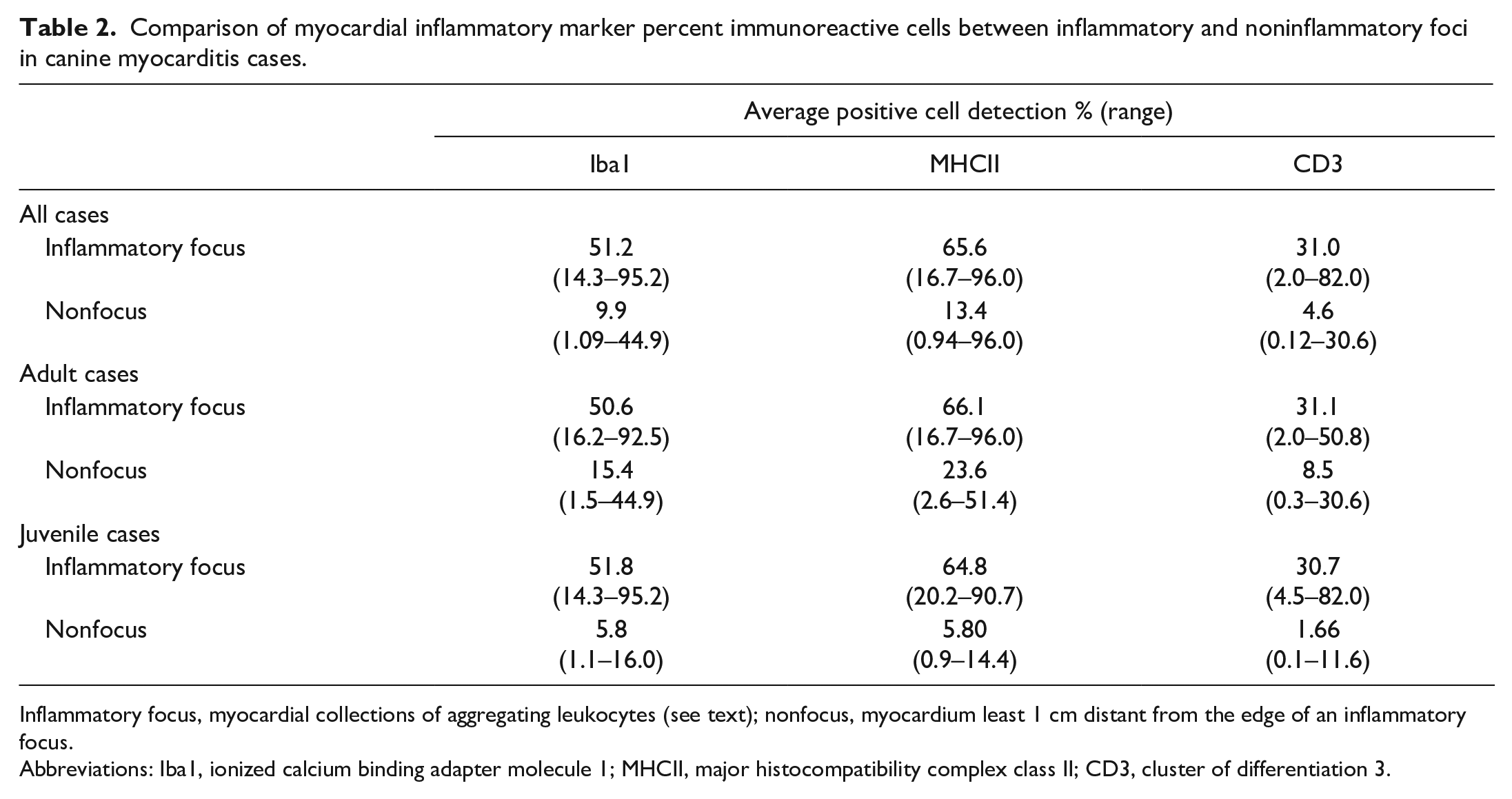
Inflammatory focus, myocardial collections of aggregating leukocytes (see text); nonfocus, myocardium least 1 cm distant from the edge of an inflammatory focus.
Abbreviations: Iba1, ionized calcium binding adapter molecule 1; MHCII, major histocompatibility complex class II; CD3, cluster of differentiation 3.
Markers were evaluated by ROC analysis. For whole-slide analysis, Iba1 had the greatest area under the curve (AUC) value (AUC = 0.8953). Each marker ROC curve was inspected for coordinates that generated the highest sensitivity and specificity (>75%) for use as a threshold or cutoff value; MHCII and CD3 did not have coordinates with sensitivity and specificity both >75%. For Iba1, a value of ≥8.084% cell labeling was selected as an optimum threshold; for this cutoff value, the sensitivity was 77.27% (95% confidence interval [CI] = 55%–92%), specificity was 100% (95% CI = 85%–100%), PPV was 100% (95% CI = 80%–100%), NPV was 82.14% (95% CI = 63%–94%), and the positive likelihood ratio was not calculable due to zero false positives. ROC statistics for myocardial Iba1 are shown in Supplemental Table S3.
Generation of a predictive model decision tree revealed a threshold Iba1 value of 8.084% that differentiated all 17 animals (≥8.084%) as having a histologic diagnosis of myocarditis. For hearts with labeling <8.084%, a secondary split differentiating myocarditis cases from controls was made with MHCII ≥5.516% and an ROC curve with an AUC of 0.9505. Based on the remaining group size, additional splits were not performed. Sensitivity of this model was 0.773, specificity was 1.00. PPV was 1.00 and NPV was 0.821.
For noninflammatory-foci analysis, Iba1 had the best fit among the 3 markers using a logistic regression analysis (P = .0003); MHCII and CD3 were not significant. For Iba1, the AUC value was 0.7578. The ROC coordinate that yielded the highest sensitivity and specificity value was a threshold of ≥3.069. For this cutoff value, sensitivity was 85.71% (95% CI = 64%–97%), specificity was 60.87% (95% CI = 39%–80%), PPV was 66.67% (95% CI = 46%–83%), NPV was 82.35% (95% CI = 57%–96%), and the likelihood ratio was 2.190.
Discussion
MHCII, CD3, and Iba1 were all diffusely increased in the myocardium of dogs with myocarditis. In this study, Iba1 was a highly sensitive and specific marker for canine myocarditis. Similar to the human heart, we observed few Iba1+ macrophages as well as CD3+ T cells scattered within the myocardium and in mild perivascular clusters in the myocardial interstitium of control hearts. In dogs with myocarditis, leukocytes were increased, infiltrating and separating cardiomyocytes and often clustered into inflammatory foci. 16 Thus, the increased detection of Iba1 and CD3 in the myocardium was attributed to increased numbers of infiltrating leukocytes. Similar to expression patterns in the human myocardium, MHCII labeling was weak and limited to endocardial cells in control canine myocardium. Markedly increased MHCII immunoreactivity in endothelial cells along with interstitial cells and cardiomyocytes was observed in the hearts of dogs with myocarditis.
As expected, inflammatory marker detection was highest in whole-slide analysis including inflammatory foci but was also increased in myocardial tissue distant from inflammatory foci in cases of myocarditis. As myocarditis is often a multifocal disease, these results are relevant for diagnosis of myocarditis even with “off-target” biopsy sampling. These findings provide an evaluation framework for myocardial inflammatory markers and are very promising for implementation of IHC as a clinical diagnostic test specific to myocarditis in dogs. Based on current diagnostic limitations, this study could not compare immune marker immunoreactivity in infectious and immune-mediated myocarditis. We also did not specifically investigate marker immunoreactivity in the hearts of dogs with cardiotoxicity.
Iba1 threshold value of 8.084% was highly sensitive and highly specific for myocarditis in our cohort. As the number of cells in any sample will vary widely based on factors including overall cell orientation and patient age, we did not provide threshold criteria based on number of positive cells/mm2. Analysis of larger cohorts of myocarditis and control tissues collected by EMB may define clinically useful cells/area thresholds for the subendocardial myocardium.
EMB with IHC and viral PCR is now an integral part of the diagnosis of myocarditis in human patients. The European Society of Cardiology recommends EMB in all human patients with suspected myocarditis. 10 With routine stains, a variety of cells within the myocardial interstitium resemble leukocytes but can be confidently identified by IHC. Immunohistology to detect focal to diffuse mononuclear cell infiltrates (macrophages and T cells) and expression of HLA class II molecules has been demonstrated to enhance detection of myocarditis, reduce interobserver variability, and provide prognostic information compared with histopathology.3,14 The presence of myocardial inflammation in an EMB sample is determined by established histologic criteria (Dallas criteria) and IHC detection of focal or diffuse mononuclear inflitrates.3,21
MHC molecules are fundamental to the adaptive immune response due to MHC restriction of the presentation of antigens to T lymphocytes. As normal muscle tissue uniquely does not express detectable sarcolemmal MHC, MHCI or MHCII has been used in the diagnosis of inflammatory muscle disorders in humans, dogs, and horses.6,7 Enhanced myocardial expression of HLA class II, identifying tissue inflammation, has been used to identify human patients with acute or chronic myocarditis.11,24 Myocarditis associated with canine Leishmania infection is associated with T lymphocyte-dominant inflammation and increased MHC labeling of vascular tissue, cell infiltrates, and also cardiomyocytes in the absence of detectable Leishmania amastigotes which raised concern for immune-mediated disease. 5
To inform pathogen testing strategies, we recently screened archived tissues from adult and pediatric/juvenile myocarditis cases for causes of myocarditis. The screen included a comprehensive panel of conventional and real-time PCR assays, which included canine parvovirus 2 (CPV-2), canine distemper virus (CDV), canine herpesvirus (CHV), Borrelia spp., West Nile virus, adenovirus, parainfluenza virus, pneumovirus, respiratory coronavirus, influenza virus, Bartonella spp., Rickettsia spp., Mycoplasma spp., and Neospora caninum. This panel did not detect agents in 53% of cases. 17 Although several agents (CDV, CHV, adenovirus, coronavirus, parainfluenza virus, Mycoplasma haemocanis, and Neospora caninum) were occasionally detected in both cases and controls, we did not consider PCR detection to indicate causation as many of the detected agents are associated with subclinical, latent, or inapparent infection or prophylaxis using modified live vaccines. The pathogen-negative canine myocarditis cases may indicate immune-mediated disease, a diagnosis made by exclusion in humans. 17
Others investigating the prevalence of cardiotropic agents in dogs with unexplained rhythm disturbances (including dilated cardiomyopathy, atrioventricular block, and nonfamilial arrhythmia) have reported a similar prevalence of myocardial pathogen detection: At least one potential pathogen was detected in 57% of EMB samples from dogs with rhythm disturbance. Detection of a potential pathogen was more likely in the dogs with rhythm disturbance compared with a small control group of dogs with congenital heart disease. Similar to our study, canine coronavirus, CDV, and CPV-2 were the most frequently detected potential pathogens; Bartonella, not detected in our cohort, was also a frequently detected agent in their study. 22 While our CDV PCR+ cases did not have convincing evidence of CDV myocardial infection (by IHC), myocarditis with naturally acquired systemic CDV infections and myocardial CDV confirmed by IHC in 4- to 7-week-old puppies has recently been reported. 13
Our previous study using archived samples was limited in the evaluation for bacterial causes. Bacterial myocarditis, resulting from sepsis or extension of endocarditis, was the most frequent cause in dogs with antemortem presumptive clinical diagnosis of myocarditis; other causes included CPV-2, Neospora, Toxoplasma, as well as isolated cases of Bartonella, Trypanosoma, Leptospira, and Dirofilaria. 15 Data from this cohort were used to propose noninvasive major (cardiac troponin I >1 ng/mL and positive blood/fluid culture or infectious disease testing) and minor diagnostic (fever, new heart murmur, inflammatory complete blood count (CBC), ventricular arrhythmia, echocardiographic changes, or pericardial effusion) criteria for the antemortem diagnosis of myocarditis in dogs. 15 The authors proposed “definitive” diagnosis of myocarditis on EMB, presence of 2 major criteria, or presence of 1 major and 3 minor noninvasive criteria. Although the latter are compelling evidence in lieu of invasive myocardial sampling, these clinicopathologic criteria may not be definitive in the unequivocal diagnosis of nonbacterial myocarditis. Particularly in human adults, serology is considered of limited value to determining myocarditis causation due to prevalence of positive titers to often ubiquitous viruses. 4 Prospective serologic-pathology studies would be required to determine the relevance, particularly of positive antibody serology, in identifying the causes of canine myocarditis.
Both logistic regression and partition tree analyses of whole-slide marker values supported the use of Iba1 as a sensitive and highly specific marker. Iba1 may be considered as stand-alone marker, especially where cost is limiting factor, with MHCII and CD3 less sensitive and specific but still valuable confirmatory markers. Altogether, these data provide criteria for the evaluation of the canine myocardium to help bring the standard of care on par with that of humans.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221098433 – Supplemental material for Myocardial Iba1, MHC class II, and CD3 are diffusely increased in canine myocarditis: A step toward antemortem myocarditis diagnostics
Supplemental material, sj-pdf-1-vet-10.1177_03009858221098433 for Myocardial Iba1, MHC class II, and CD3 are diffusely increased in canine myocarditis: A step toward antemortem myocarditis diagnostics by Kristina Vu, Vanessa Wikel, Alex Molesan, Erika Mudrak and Kathleen Kelly in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank the Cornell University College of Veterinary Medicine for program support and the Animal Health Diagnostic Center histology lab for assistance with ionized calcium binding adapter molecule 1 (Iba1) and cluster of differentiation 3 (CD3) immunohistochemistry. We would also like to extend a heartfelt thanks to Pete Bankhead for creating QuPath and providing timely support with the program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Please refer to the online version of the manuscript for all supplemental materials referenced in the text. The data analyzed in this study are available as supplemental materials upon request to the author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
