Abstract
Class II major histocompatibility complex (MHCII) is required for the presentation of antigens to CD4 helper T cells. During nephritis, not only primary antigen presenting cells such as histiocytes and lymphocytes, but also cytokine-stimulated tubular epithelial cells express MHCII. Leptospirosis in fattening pigs is characterized by several degrees of nephritis, from absence of lesions to severe multifocal tubulo-interstitial inflammation. Renal tissue from 20 8-month-old pigs with spontaneous nephritis and 6 control pigs without renal lesions were investigated for leptospirosis by indirect immunohistochemistry (IHC) and polymerase chain reaction (PCR). IHC for MHCII also was performed on renal samples. Serum samples were tested for different serovars of Leptospira interrogans. Control pigs were free of interstitial nephritis and negative for leptospirosis by all tests. In pigs with nephritis, serology was positive for serovar Pomona in 19/20 pigs. In 16 of these 19 pigs, leptospiral renal infection was confirmed by PCR and/or indirect IHC. Nephritic lesions were classified histologically into perivascular lymphocytic (4 pigs), lymphofollicular (6 pigs), lymphohistiocytic (8 pigs), and neutrophilic (2 pigs) pattern. MHCII expression by histiocytes and lymphocytes was observed in all lesions. Prominent MHCII expression in regenerating tubular epithelium was observed in lymphofollicular and lymphohistiocytic nephritis. No tubular colocalization between leptospiral and MHCII antigen was observed. Results suggest that during leptospiral nephritis, MHCII contributes to the intensity of the inflammatory response. Furthermore de novo MHCII expression in regenerating tubules may play a role in the defence mechanism against leptospiral tubular colonization.
Keywords
Introduction
Leptospirosis causes consistent economic losses in the pork industry worldwide and represents a zoonotic hazard for animal handlers (caretakers), abattoir workers, veterinarians, and meat inspectors. 23,28 Only a small proportion of infected animals develop clinical signs such as fever, anorexia, and depression. 28 Multifocal interstitial nephritis is the most frequently observed lesion in chronically infected pigs at slaughter. The most important route of natural infection has not been determined, but following a period of bacteremia, leptospires localize in the proximal convoluted renal tubules. 6,12 In swine, leptospiral colonization of renal tubules can lead to varying degrees of inflammation, from absence of lesions to severe multifocal nephritis characterized by interstitial infiltrates of lymphocytes, plasma cells, histiocytes, and, in the most severe cases, neutrophils. 11 The reason for such variability is unknown. A previous immunohistochemical study indicated that the presence of leptospires within the tubular lumen is associated with slight-to-absent interstitial inflammatory reaction, while this reaction is prominent when leptospiral antigen is detectable within the cytoplasm of tubular cells. 31 However, how the infected tubules with intracytoplasmic leptospiral antigen are able to trigger an interstitial inflammatory response is still unclear.
Class II major histocompatibility complex (MHCII) is an integral transmembrane molecule required for the presentation of antigens to CD4 helper T cells. 10 The constitutive expression of this molecule has been reported in dendritic cells, macrophages, and B- cells. 9 Antigen-capturing cells (dendritic cells, macrophages, B cells) are able to endocytose foreign antigens and to mature as antigen-presenting cells (APCs) by proteolytically cleaving complex antigens into small peptides that are complexed with MHCII and displayed on the cell surface. In certain situations epithelial cells may express MHCII and may function as nonprimary APCs. This has been demonstrated in a wide range of epithelial cells including renal tubular cells. In humans and dogs, MHCII expression has been reported in proximal tubular cells, 1,5,35 while in vitro studies showed that de novo expression of this antigen class stimulates a classical epitope-specific immune response. 2 Indirect IHC has been widely used to detect MHCII in formalin-fixed and paraffin-embedded human and animal tissues including tissues from swine. 25,26
The aim of this study was to investigate the expression of MHCII within cases of interstitial nephritis associated with spontaneous leptospiral infection in swine by indirect IHC. We hypothesized that during porcine leptospiral renal infection, different profiles of MHCII expression could account for the wide spectrum of nephritic lesions as well as the various degrees of leptospiral renal colonization.
Materials and Methods
At slaughter, 1 kidney and a serum sample were collected from each of 20 pigs (Nos. 1–20). These pigs were 8-months-old and weighed between 310 and 350 lbs. Animals were randomly selected from a group in which there was high prevalence (approximately 50%) of grossly detectable lesions of interstitial nephritis (white spotted kidneys). Kidneys and sera were also sampled at slaughter in the same period from 6 pigs (Nos. 21–26) randomly selected from animals of the same age and weight, but belonging to a group without renal lesions. These latter pigs were used as weight- and age-matched negative controls. All the animals considered in this study came from swine herds in Northern Italy.
For each kidney a wedge-shaped tissue sample was collected, from both cranial and caudal poles and fixed in 10% buffered formalin, progressively dehydrated in graded alcohols and xylene, and embedded in paraffin. Four-µm serial sections were stained with HE or used for indirect IHC. From each pig 2 wedge-shaped renal sections with a total cortical area of about 150 mm2 were evaluated. Nephritic lesions were classified histologically into the following 4 patterns:
Perivascular lymphocytic pattern: chronic interstitial nephritis characterized by cortical perivascular lymphocytic infiltrates not surrounding tubules.
Lymphofollicular pattern: chronic interstitial nephritis characterized by lymphocytic peritubular infiltrates often forming lymphoid follicles randomly distributed within the cortex. Lymphoid follicles sometimes surround tubular structures.
Lymphohistiocytic pattern: chronic linear corticomedullary lymphocytic and histiocytic interstitial nephritis with various degrees of fibrosis. Inflammatory infiltrates surround tubules often with atrophy, degeneration, and regeneration.
Neutrophilic pattern: lesion characterized by severe subacute lymphocytic, histiocytic, and neutrophilic tubulo-interstitial nephritis. Multifocal and confluent areas are replaced by dense interstitial inflammatory infiltrates comprising lymphocytes, plasma cells, histiocytes, and numerous granulocytes (predominantly neutrophils and a few eosinophils). Degenerate neutrophils are often detectable in the lumen of tubules forming casts. Lesions have linear corticomedullary distribution and affected degenerate and necrotic tubules.
All tissue sections for characterization of lesions and evaluation of immunohistochemical staining were examined in a blinded fashion by board-certified pathologists. The presence and severity of nephritic lesions were semiquantitatively scored as follows: − = absent; + = slight (less than 5% of the tissue examined involved); ++ = mild (from 5 to 25%); +++ = severe (more than 25%).
Serial tissue sections were stained with the indirect immunohistochemical avidin-biotin peroxidase technique. Mouse monoclonal antibody against the α-chain of human leukocyte antigen (HLA)-DR (Dako, Glostrup, Denmark) and a rabbit polyclonal antibody against Leptospira interrogans serovar Pomona (L. Pomona) were used as primary antibodies. 30,31 Tissue sections were mounted on glass slides coated with L-polylysine and dried. Sections were then deparaffinized in xylene and rehydrated in graded alcohols. Endogenous peroxidases were inhibited with 3% H2O2 in distilled water for 15 minutes. Antigens were retrieved by heating sections in 10 mM citrate buffer pH 6, in microwave. Blocking of nonspecific binding was achieved by incubating slides in 10% horse serum (for MHCII) and goat serum (for L. Pomona) in phosphate-buffered saline (PBS) for 25 minutes. Primary antibody against MHCII (working dilution 1 : 500) was applied overnight at 4°C. Primary antibody against L. Pomona (working dilution 1 : 10,000) was applied for 35 minutes at 37°C. After rinsing, slides were incubated for 30 minutes with biotinylated horse anti-mouse (for MHCII) or biotinylated goat anti-rabbit (for L. Pomona) antibodies (Vector Laboratories, Burlingame, CA, USA) followed by 30 minutes incubation with avidin-biotinylated complex (ABC, Vectastain Elite, Vector). Specific peroxidase activity was demonstrated with diaminobenzidine solution (DAB, Vector), and slides were lightly counterstained with hematoxylin for 1 minute before rinsing, dehydrating, clearing, and coverslipping.
Histopathology, IHC, PCR, and serology findings in pigs with various morphologic patterns of leptospiral nephritis.
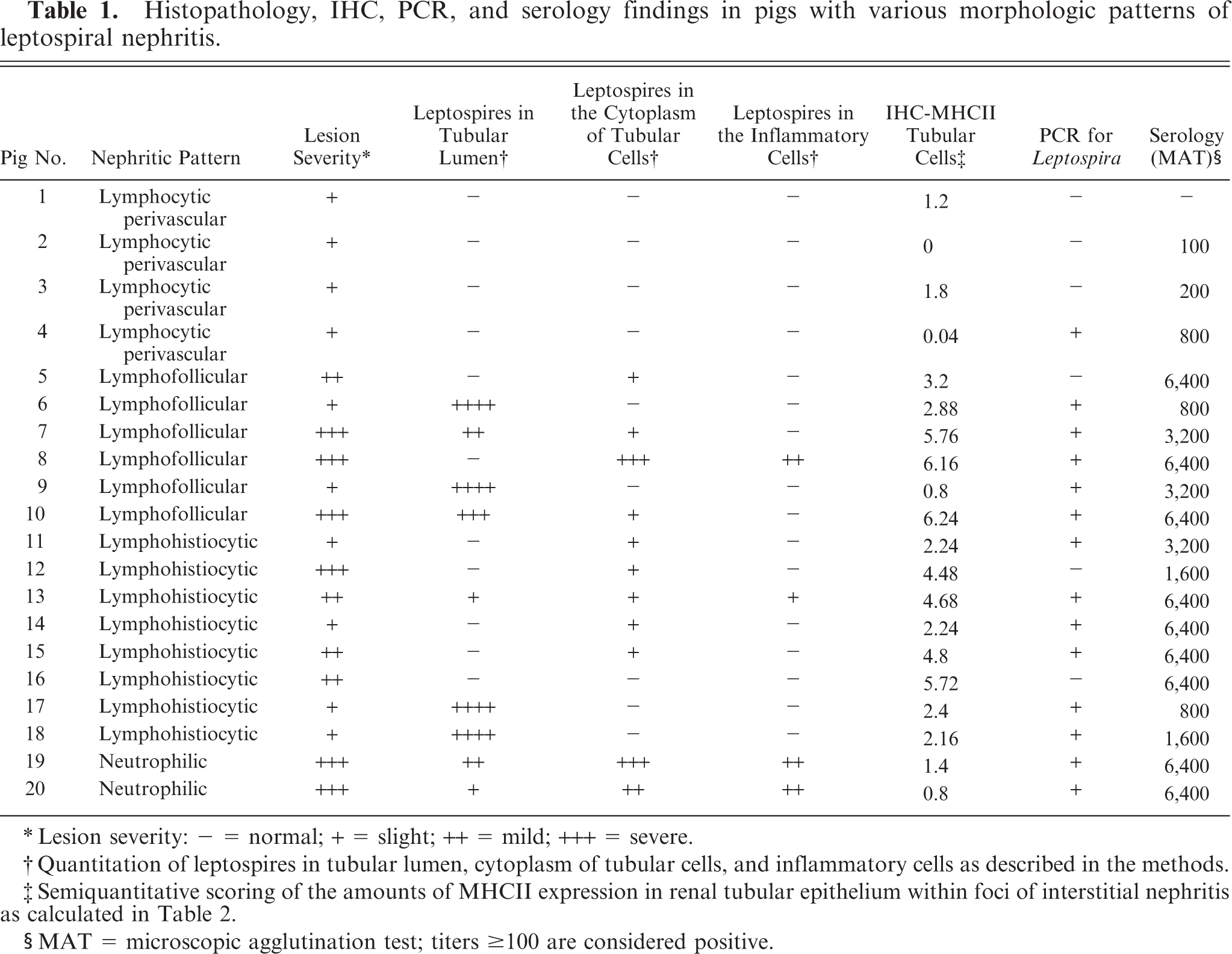
∗Lesion severity: = normal; + = slight; ++ = mild; +++ = severe.
†Quantitation of leptospires in tubular lumen, cytoplasm of tubular cells, and inflammatory cells as described in the methods.
‡Semiquantitative scoring of the amounts of MHCII expression in renal tubular epithelium within foci of interstitial nephritis as calculated in Table 2.
§MAT = microscopic agglutination test; titers ≥ 100 are considered positive.
On 7 pigs (Nos. 6, 8, 10, 12, 17, 19, and 20), MHCII and L. Pomona double antigen-immunoperoxidase labeling was performed as reported in the Guide to Multiple Antigen Labelling (Vector). 13 Representative cases for double antigen immunolabeling were selected among the L. Pomona-immunohistochemical positive lesions, including pigs with different nephritic patterns and different degrees of MHCII and leptospiral antigen expression/distribution (Table 1). Peroxidase activity was demonstrated first by DAB+Ni2+ (DAB, Vector), which resulted in a black chromogen reaction for MHCII, and second by AEC (AEC, Vector), which resulted in a red chromogen reaction for L. Pomona antigen.
The following scoring system for the presence and amount of leptospiral antigen was used. Absence of staining was considered as negative (−); presence of <5 positive tubules or focal infiltrates was scored as (+); presence of 5 to 50 positive tubules or focal infiltrates was scored as (++); presence of >50 positive tubules or focal infiltrates was scored as (+++). Pigs with more than 50% of infected tubules were scored (++++). The following scoring system was used for the quantification of tubular epithelial cells expressing MHCII within foci of interstitial nephritis. In each pig, 5 microscopic fields were examined (magnification 400×), selected from areas characterized by the most severe inflammatory lesions. The percentage of tubules with MHCII-positive epithelial cells was scored as follows: 0 = absence of positive tubules; 1 = ≤20% positive tubules; 2 = 20–50% positive tubules; 3 = ≥50% positive tubules. The percentage of immunostained epithelial cells within positive tubules was then scored as follows: 1 = ≤20% of positive cells; 2 = 20–50% positive cells; 3 = ≥50% positive cells. Two measurements were made, and a combined score was obtained by multiplying the values of both MCHII expression scores (Table 2).
Quantitation of MHCII expression in renal tubular epithelial cells of pigs with various morphologic patterns of leptospiral nephritis.
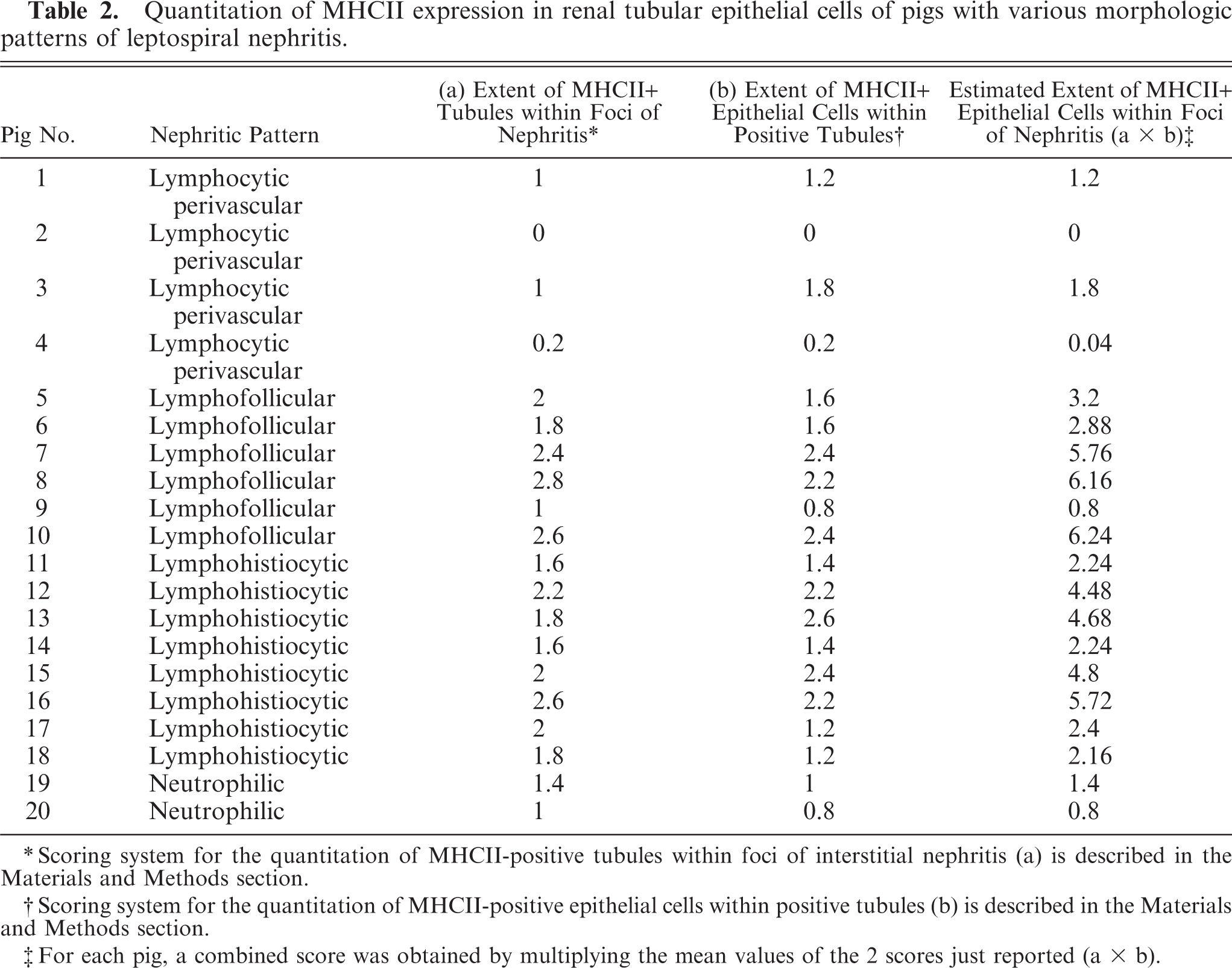
∗Scoring system for the quantitation of MHCII-positive tubules within foci of interstitial nephritis (a) is described in the Materials and Methods section.
†Scoring system for the quantitation of MHCII-positive epithelial cells within positive tubules (b) is described in the Materials and Methods section.
‡For each pig, a combined score was obtained by multiplying the mean values of the 2 scores just reported (a x b).
Serum samples were tested for antibodies to the following serovars of L. interrogans representing the 8 serogroups found most frequently in domestic animals in Italy: Bratislava (serogroup Australis), Castellonis (serogroup Ballum), Canicola (serogroup Canicola), Grippotyphosa (serogroup Grippotyphosa), Icterohaemorrhagiae (serogroup Icterohaemorrhagiae), Pomona (serogroup Pomona), Hardjo (serogroup Sejroe), and Tarassovi (serogroup Tarassovi). Antibody titers were determined using a microtechnique adapted to the microscopic agglutination test (MAT). 7 MAT titers ≥ 1 : 100 were considered positive. 21
Total DNA was extracted from kidney samples using the QIAamp blood and tissue kit (Qiagen Gmbh, Hilden, Germany) according to the manufacturer's instructions. The polymerase chain reaction (PCR) assay for the identification of pathogenic leptospires was performed using previously described primers targeting a specific fragment of the hemolysis-associated protein-1 gene (hap1). 4 PCR reactions were performed in a 25-µl volume containing 2 µl of DNA sample, 0.4 µM of each primer (Adia214 and Adia215), standard amounts of amplification reagents (200 µM dNTPs [each], 10 mM Tris/HCl, 50 mM KCl, 1.5 mM MgCl2, [pH 8,3]), and 1 U of Taq DNA polymerase (Roche, Manheim, Germany). Amplification was carried out in a GeneAmp 9700 Thermal Cycler (Applied Biosystems, Foster City, CA) under the following conditions: 94°C for 2 minutes, followed by 45 cycles at 94°C for 20 seconds, annealing at 56°C for 30 seconds, 72°C for 40 seconds; and final extension at 72°C for 5 minutes. Negative and positive controls were included in each PCR experiment. Amplification products (262 base pair [bp]) were analyzed by electrophoresis in a 2% agarose gel stained with ethidium bromide.
Spearman correlation ranks were calculated among the following variables: number of MHCII positive tubules, number of MHCII positive tubular cells within foci of interstitial nephritis, severity of inflammation, presence of leptospiral antigen within the cytoplasm of tubular cells, and serologic titers against L. Pomona.
Results
Pigs of the control group (Nos. 21–26) were negative for Leptospira infection when tested by PCR, IHC, and serology and their kidneys were histologically normal. MHCII expression was consistently detected in interstitial stellate cells and endothelium lining interstitial blood vessels. Capillary endothelium of glomerular tufts mildly expressed MHCII. No MHCII was detected within tubular epithelial cells (Fig. 1).
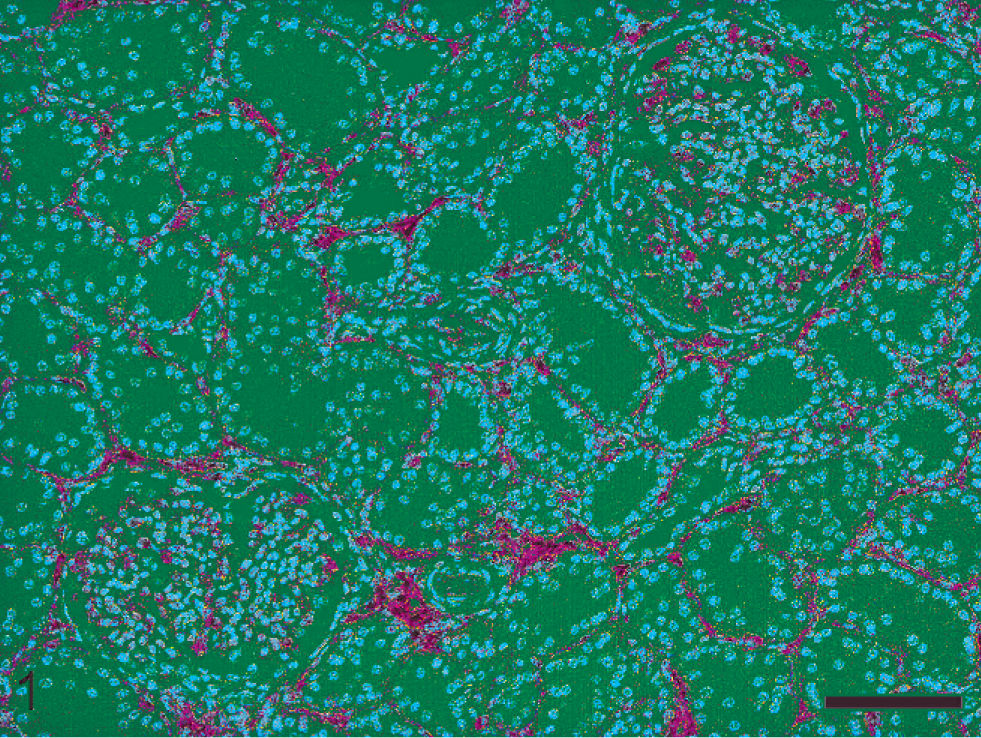
Porcine kidney; pig No. 22. Distribution of MHCII expression in normal renal tissue. MHCII is constitutively expressed by interstitial stellate cells and endothelium lining interstitial blood vessels and glomerular capillaries. Immunohistochemical staining for MHCII, hematoxylin counterstain. Bar = 100 µm.
Interstitial nephritis was histologically identified in all kidneys from the pigs (Nos. 1–20) coming from the group with high prevalence of grossly detectable lesions (white spotted kidneys); although, severity and type of lesion varied between different pigs (Table 1). In this group of swine, the presence of leptospiral infection was confirmed by PCR (14 pigs), IHC (15 pigs), and/or serology (19 positive pigs for serovar Pomona) (Table 1). Each pig was histologically classified in one of the 4 categories defined in Materials and Methods (Table 1). Often, more than one histologic pattern was present in a single pig. In these pigs, the prevailing nephritic pattern was considered for classification purposes.
Perivascular lymphocytic pattern was observed in 4 nephritic lesions (Nos. 1–4). No leptospiral antigen was detected, while bacterial DNA was detected via PCR in one pig (No. 4; Table 1). Low antibody titers (ranging from 100 to 800 MAT titers) were detected in 3 pigs (Nos. 2–4). A few tubules with low level MHCII expression in epithelial cells were observed in these pigs (Table 2).
Lymphofollicular pattern was observed in 6 nephritic lesions (Nos. 5–10). All cases contained bacterial DNA and antigen. All pigs had very high serologic titers ranging from 1 : 3,200 to 1 : 6,400, except one, which had a 1 : 800 MAT titer. In 2 pigs (Nos. 6 and 9) the lesions were mild despite more than 50% of the tubular lumina being heavily colonized by leptospires (Table 1). In these 2 pigs, tubular epithelial MHCII expression within foci of interstitial nephritis was observed with a low or intermediate level of expression (Table 2). Four pigs (Nos. 5, 7, 8, and 10) were characterized by severe lesions and presence of intracytoplasmic leptospiral antigen within tubular epithelial cells and, in a single pig, also within interstitial inflammatory cells (pig No. 8, Table 1, Fig. 2). Intracytoplasmic leptospiral antigen was morphologically characterized by coarse granular fragments (Fig. 2). Within foci of interstitial nephritis, a high number of MHCII-positive tubules with high level of expression in tubular epithelial cells were also observed (Table 2).

Porcine kidney; pig No. 8. Interstitial nephritis characterized by lymphofollicular pattern. Abundant leptospiral antigen is present within the epithelium of scattered tubules. Bar = 100 µm. Inset: Scant signal is also associated with the surrounding interstitial inflammatory cell infiltrate. Bar = 50 µm. Immunohistochemical staining for leptospiral organisms, hematoxylin counterstain.
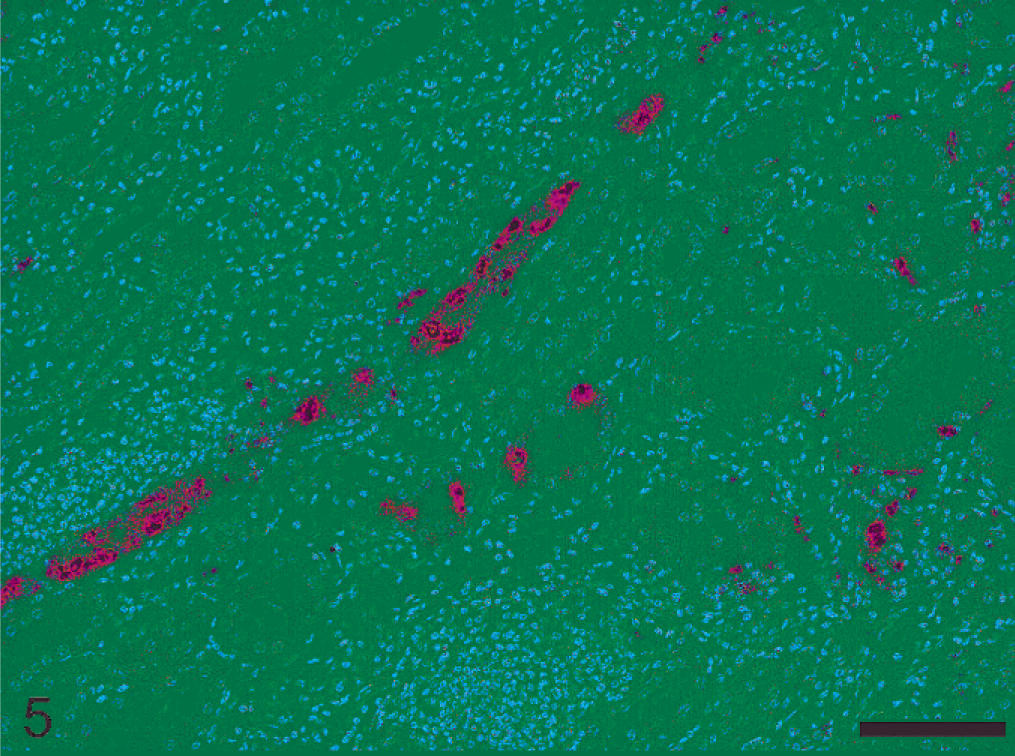
Lymphohistiocytic pattern was observed in 8 nephritic lesions (Nos. 11–18). All cases except one (No. 16) contained leptospiral antigen and DNA. All but one pig (No. 17) displaying a 1 : 800 MAT titer, had very high serologic titers ranging from 1 : 1,600 to 1 : 6,400. In 4 pigs (Nos. 11, 14, 17, and 18) lesions were mild despite the fact that in 2 of these pigs (Nos. 17 and 18) more than 50% of the tubular lumina were heavily colonized by leptospires (Table 1, Fig. 3). In these 4 pigs, MHCII intermediate expression was observed in few tubules within foci of interstitial inflammation (Table 2). Four pigs with moderate-to-severe lesions (Nos. 12, 13, 15, and 16) had numerous tubules expressing high level of MHCII (Table 2, Fig. 4). Three pigs (Nos. 12, 13, and 15) had intracytoplasmic intratubular leptospiral antigen, and also within interstitial inflammatory cells in one pig (No. 13), (Table 1, Fig. 5).
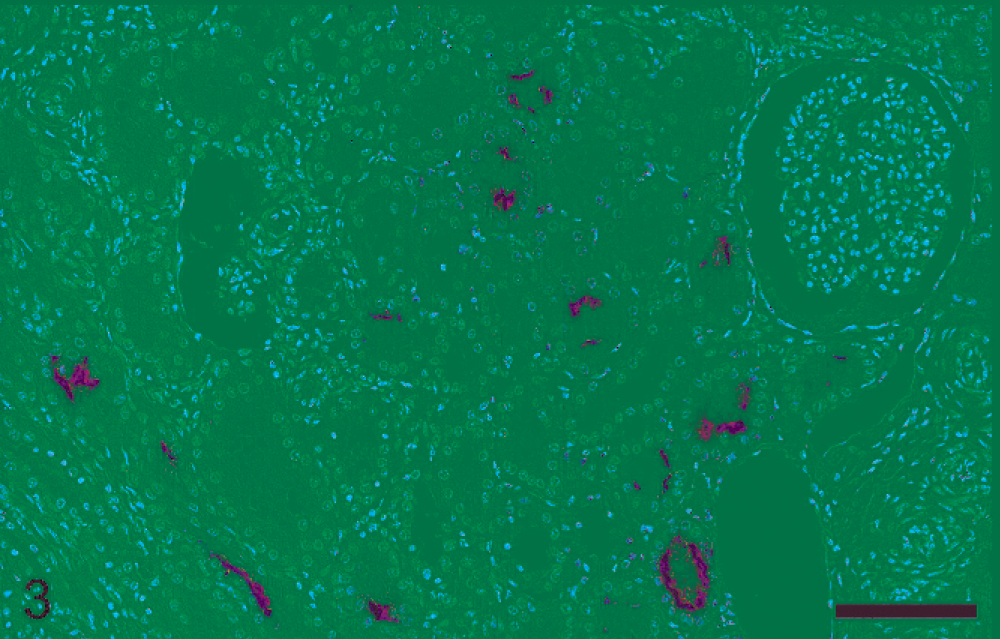
Porcine kidney; pig No. 17. Interstitial nephritis characterized by lymphohistiocytic pattern. Minimal interstitial inflammatory changes are associated with heavy intraluminal colonization of renal tubules by leptospires. Immunohistochemical staining for leptospiral organisms, hematoxylin counterstain. Bar = 100 µm.
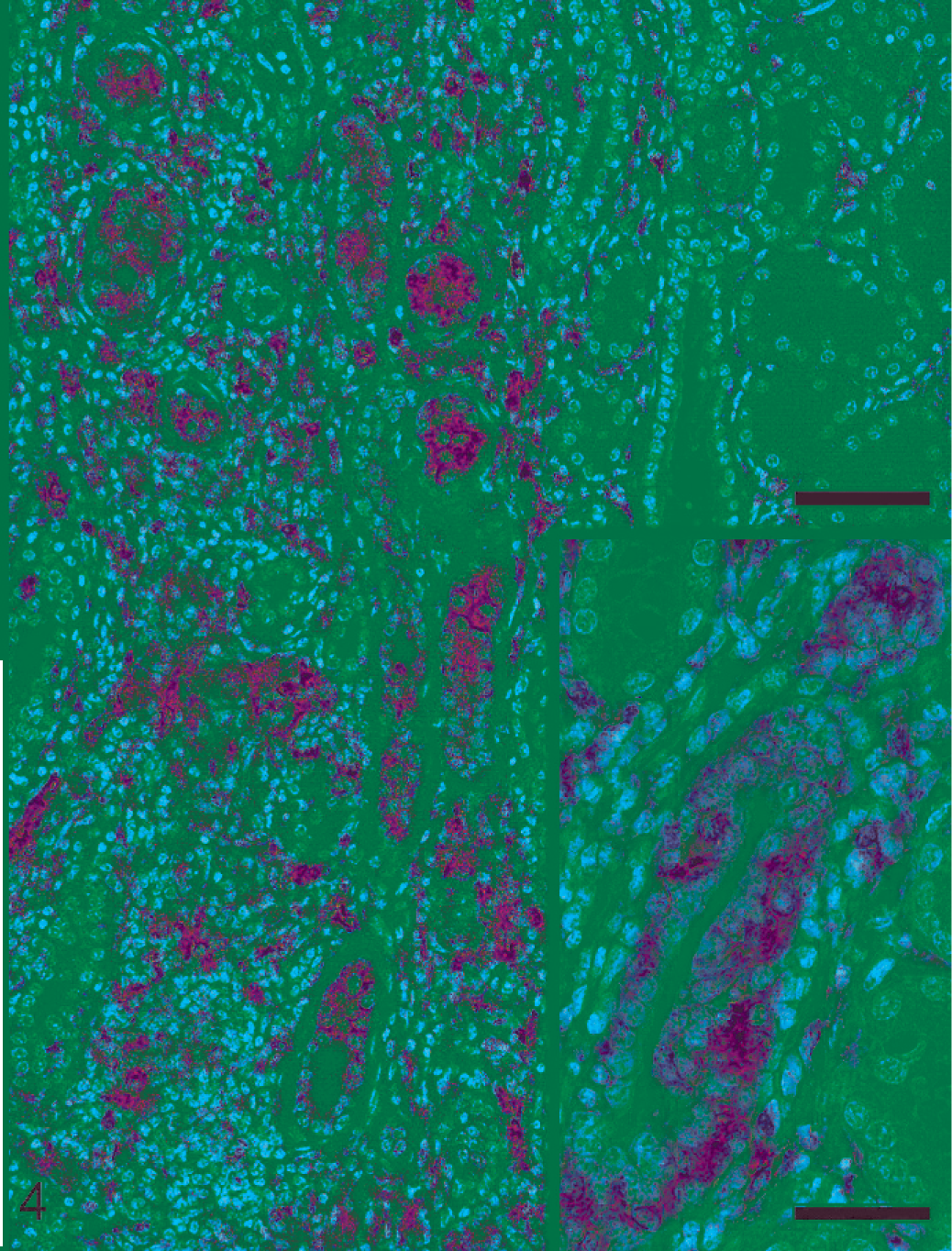
Porcine kidney; pig No. 12. Interstitial nephritis characterized by lymphohistiocytic pattern. Marked and diffuse MHCII-tubular expression within foci of interstitial nephritis is evident. Bar = 70 µm. Inset: Note the regenerative morphology of MHCII-positive tubules. Bar = 35 µm. Immunohistochemical staining for MHCII, hematoxylin counterstain.
Porcine kidney; pig No. 13. Interstitial nephritis characterized by lymphohistiocytic pattern. Severe tubulointerstitial inflammatory changes are associated with abundant intracytoplasmic leptospiral antigen in tubular epithelial cells and interstitial phagocytes. Immunohistochemical staining leptospiral organisms, hematoxylin counterstain. Bar = 100 µm.
Neutrophilic pattern was observed in 2 nephritic lesions (Nos. 19 and 20). In both pigs, leptospires were detected via PCR and IHC, and there was abundant antigen in tubular lumina and cytoplasm of tubular and inflammatory cells (Table 1, Fig. 6). Both animals had very high antibody titers. The expression of MHCII in atrophic/degenerating tubular epithelial cells within foci of interstitial inflammation was low in both pigs (Table 2, Fig. 7).

Porcine kidney; pig No. 20. Interstitial nephritis characterized by neutrophilic pattern. Abundant leptospiral antigen is present within tubular epithelium and surrounding infiltrating granulocytes. Bar = 100 µm. Inset: Leptospiral antigen is also present within leukocytic casts. Bar = 50 µm. Immunohistochemical staining for leptospiral organisms, hematoxylin counterstain.
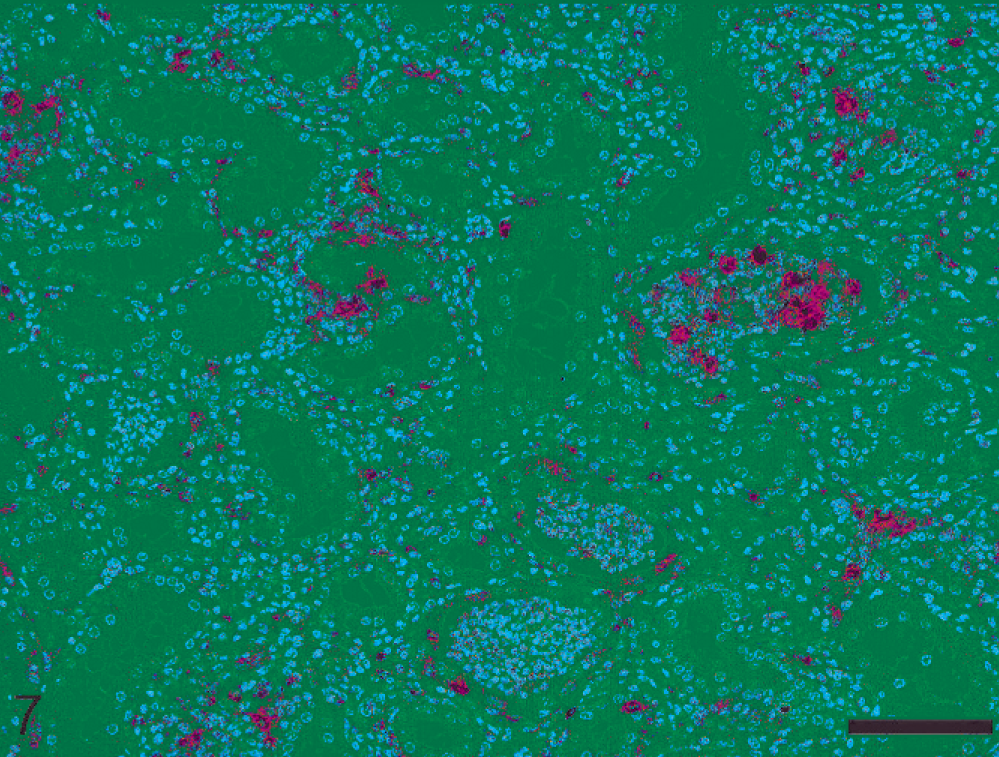
Porcine kidney; pig No. 19. Interstitial nephritis characterized by neutrophilic pattern. Scant MHCII expression is evident in atrophic degenerating tubules. Variable MHCII expression is detectable within leukocytic casts. Immunohistochemical staining for MHCII, hematoxylin counterstain. Bar = 100 µm.
Statistical analysis indicated a significant positive correlation between the number of MHCII positive tubules and the severity of inflammation (σ = 0.858; P < .001), the presence of leptospiral antigen within the cytoplasm of tubular cells (σ = 0.653; P < .01) and the serologic titers against serovar Pomona (σ = 0.556; P < .01). The number of MHCII positive tubular cells within foci of interstitial nephritis was significantly correlated with the severity of inflammation (σ = 0.781; P < .001) and the serologic titers (σ = 0.741; P < .001).
MHCII expression in tubular epithelial cells was detected in cortical tubules within areas of interstitial nephritis and often in tubules with regenerative changes (Fig. 4). The immunoreactivity distribution varied from single MHCII-positive epithelial cells within a tubular structure to diffuse MHCII immunoreactivity involving the entire tubule. For the former type of MHCII-labeling, the signal was concentrated in the basal part of the tubular cells, while for the latter MHCII-labeling, a homogeneous immunoreactivity involved the whole cytoplasm of the cells. There was no difference between the immunohistochemical labeling of normal kidney tissue and unaffected areas of nephritic tissue. All the infiltrating histiocytes and the majority of lymphoid cells expressed MHCII regardless of the histologic pattern observed. In pigs with neutrophilic pattern, weak and sporadic MHCII expression was observed in association with neutrophils infiltrating the interstitium and forming intratubular casts.
Comparison of serial sections immunolabeled for MHCII and leptospira as well as double-immunolabeled sections did not reveal colocalization of leptospiral antigen and MHCII in the cytoplasm of epithelial tubular cells (Fig. 8). Double-antigen immunolabeling confirmed that in the 2 pigs with neutrophilic pattern, neutrophils and macrophages containing leptospiral antigen occasionally expressed MHCII.
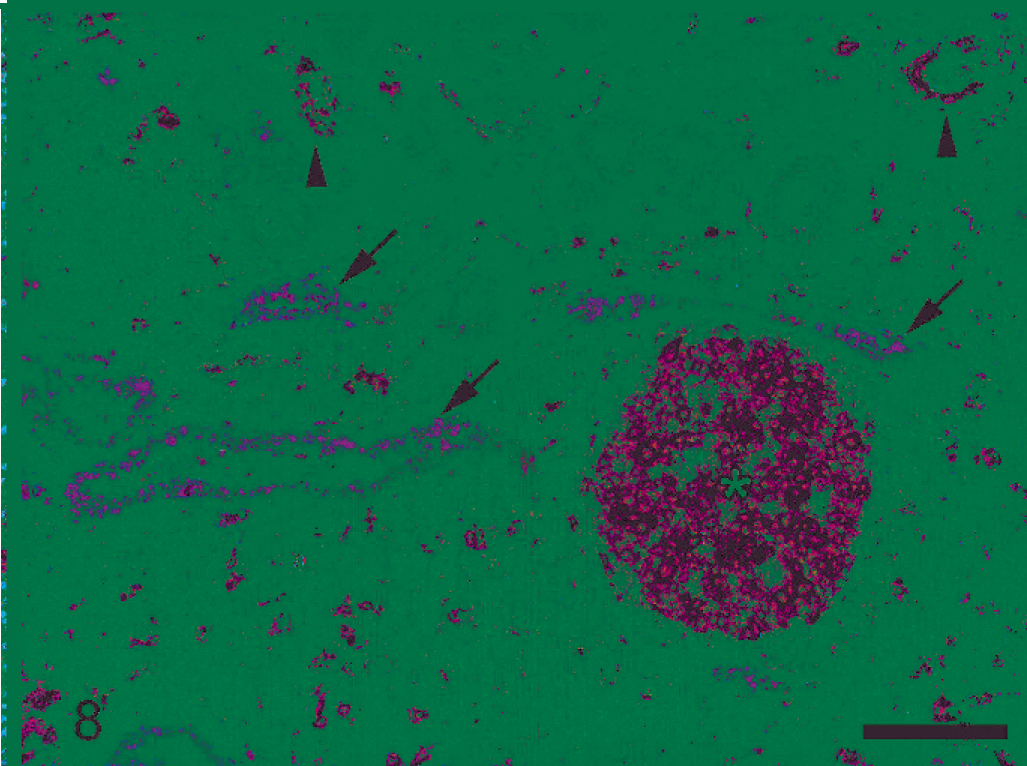
Porcine kidney; pig No. 8. Interstitial nephritis characterized by lymphofollicular pattern. Tubules bearing leptospiral organisms do not express MHCII (arrows). On the contrary, a few adjacent tubules without leptospiral colonization (arrowheads) and an interstitial lymphoid follicle (white asterisk) show variable degrees of MHCII expression. Double-antigen immunolabeling for leptospiral organisms (red signal) and MHCII (black signal). Bar = 100 µm.
Discussion
Compared with the normal renal parenchyma where only stellate interstitial cells (currently considered the resident pool of dendritic cells) and endothelial cells constitutively expressed MHCII, different additional cell populations were MHCII-positive in areas of interstitial nephritis. 15–18 Among all the different infiltrating inflammatory cells, histiocytes and to a lesser extent lymphocytes markedly expressed MHCII. In swine, these inflammatory cell populations represent primary APCs that commonly express MHCII. 25,26,29 Weak but diffuse MHCII expression in intratubular and interstitial neutrophils was observed. This is consistent with the reported possibility that cytokine stimulation may induce neutrophils and their precursors to actively express MHCII and act as APCs. 8,36 Alternatively, recruited neutrophils may express an MHCII signal by passively acquiring MHCII and other membrane molecules from apoptotic or necrotic professional APCs. 36 Furthermore, variable degrees of MHCII expression were also reported in intralesional renal tubules.
MHCII expression by renal tubules has been previously described in renal transplants during graft-versus-host reactions, in the MRL/lpr mouse model of systemic lupus erythematosus and in dogs spontaneously affected by chronic progressive tubulo-interstitial nephritis. 20,22,35 Several studies suggest that MHCII expression by renal tubules can promote an interstitial inflammatory reaction. 2,14,37 In dogs, a significant association was also found between the amount of MHCII-tubular expression within foci of interstitial nephritis and the severity of renal failure-dysfunction. 35 Pigs experimentally exposed to leptospiral antigen activate different phenotypes of immune responses. 24 The marked variability in MHCII expression and distribution during chronic leptospiral nephritis observed in the current study may reflect that reported ability to activate different immune response phenotypes. Thus, it is suggested that MHCII expression by epithelial cells partially accounts for the profound variation in immune competence and disease resistance that commonly characterizes leptospiral swine infection. In the present study, the statistical analysis demonstrated a positive correlation among the degree of MHCII-tubular expression within foci of interstitial nephritis, severity of inflammatory reaction, and serologic titer against serovar Pomona. These correlations may indicate that de novo MHCII-tubular expression during swine leptospiral nephritis plays a role in both the stimulation of a local inflammatory reaction and systemic humoral response. However, examination of the expression of costimulatory molecules by MHCII-positive epithelial cells and functional studies would be necessary to confirm the specific effect of de novo expression of MHCII on the immune response to leptospiral bacteria.
MHCII-signal distribution and particularly MHCII expression by tubular epithelial cells varied according to the severity and distribution of nephritic lesions. Pigs with mild chronic nephritic lesions with a prevalent perivascular lymphocytic pattern (Nos. 1–4) were characterized by very low level MHCII-tubular expression within foci of interstitial nephritis. The severity score of these lesions was always minimal. In this group, leptospiral renal infection was confirmed in pig No. 4 only. However, given the positive serologic titers for serovar Pomona, the implication of leptospiral infection in the nephritic lesions should also be considered for pigs Nos. 2 and 3.
As evidenced by statistical analysis, marked and diffuse MHCII-tubular expression within foci of interstitial nephritis was observed mostly in pigs characterized by moderate-to-severe chronic nephritic lesions with a prevalent lymphofollicular or lymphohistiocytic pattern (Nos. 5, 7, 8, 10, 12, 13, 15, and 16). Conversely, mild nephritic lesions with the same patterns (Nos. 6, 9, 11, 14, 17, and 18) displayed low MHCII-tubular expression within foci of interstitial nephritis. As previously observed in lymphofollicular and lymphohistiocytic patterns, a marked tubulo-interstitial inflammatory response was mainly associated with intratubular intracytoplasmic leptospiral antigen (Nos. 5, 7, 8, 10, 12, 13, and 15). 31 Conversely, pigs with tubular intraluminal leptospires (Nos. 6, 9, 17, and 18), even when characterized by heavy colonization, had very mild inflammatory responses. In these pigs, very mild MHCII-tubular expression within foci of interstitial nephritis and low serologic titers were observed. This discrepancy among heavy leptospiral colonization, scant inflammatory response, and low serologic titers represents a common finding in chronically infected pigs. 3,31 Similar to rats infected with serovar icterohaemorrhagiae, pigs may act as a reservoir species (maintenance host) of leptospiral infection. In reservoir hosts, the prominent intraluminal leptospiral colonization of renal tubules is typically not accompanied by clinical signs or lesions with antibody titers being negative or low. 27 These aspects seem to reflect the capability of Leptospira to select a specific microenvironment segregated from the host response, resulting in long-standing leptospiruria. 6
In pigs of severe, subacute tubulo-interstitial nephritis with a prevalent neutrophilic pattern (Nos. 19 and 20), abundant interstitial leptospiral antigen was associated with infiltrating phagocytes. Here the severe inflammatory reaction elicited by a large number of leptospires within renal tubules may lead to tubulorrhexis with release of bacteria into the surrounding interstitium. During this transition the opsonization of leptospires may play a crucial role in activating a strong phagocytic reaction. The phagocytosis of locally numerous opsonized leptospires by macrophages and neutrophils is expected to produce large amounts of lytic enzymes, reactive oxygen species (ROS), and reactive nitrogen species (RNS), amplifying the severity of tubular damage. 19 Thus, an exaggerated local immune response may account for the inability to express MHCII by injured tubular epithelial cells within foci of tubulo-interstitial nephritis.
No association between leptospiral antigen and MHCII-tubular expression was identified. This aspect was further demonstrated by double-antigen immunolabeling that revealed the lack of colocalization between MHCII expression and L. pomona antigen within tubules. This observation indicates that de novo expression of MHCII by tubular epithelium is not directly involved in the triggering of the inflammatory reaction during the early phase of tubular colonization. The following findings suggest that de novo MHCII-tubular expression represents a late event during leptospiral nephritis: 1) negative or slight MHCII expression by atrophic and degenerating tubules during the subacute phase of the infection (neutrophilic pattern); 2) chronic nature of the inflammatory response always associated with MHCII-tubular expression; and 3) regenerative morphology of the MHCII-positive tubules within foci of interstitial nephritis. As demonstrated in previous studies, during the chronic phase of inflammatory response, regenerating tubules express MHCII under the influence of an IFN-γ-rich cytokines milieu. 32,33 There are several studies suggesting that IFN-γ specifically stimulates de novo MHCII expression by tubular epithelium within foci of interstitial nephritis. 22,34,35 In the current study, MHCII-positive tubules were always associated with a marked peritubular lymphofollicular or lymphohistiocytic inflammatory response. In a self-perpetuating redundant process, MHCII-positive tubular cells may activate peritubular CD4+ T-cells to produce proinflammatory cytokines including IFN-γ. Consequently, the inflammatory signal extends amplifying de novo MHCII expression by tubular epithelial cells. This novel phenotype of tubular cells characterized by MHCII expression may represent a potential defense mechanism against the leptospiral colonization of renal tubules: indeed, leptospires were never detected in MHCII-positive tubules.
In conclusion, MHCII expression is highly modified in renal leptospirosis of swine. De novo MHCII expression in regenerating tubular epithelial cells occurs in the chronic phase of the renal infection. This late event contributes to the inflammatory reaction and may be specifically involved in the defense against leptospiral infection creating an unfavorable condition for tubular colonization by leptospires. Further studies investigating the leukocyte subset and cytokine profile of the different nephritic patterns as well as the intralesional distribution of MHCII costimulatory molecules are needed for a more comprehensive evaluation of MHCII-tubular expression in renal leptospirosis.
