Abstract
Three dogs under 12 months old were diagnosed with atypical multiple myeloma (MM), having an aggressive multifocal anaplastic round cell sarcoma in bone marrow, viscera, and/or peripheral blood, which were confirmed by cytology and immunohistochemistry to be of plasma cell origin. The intramedullary sarcomas caused myelophthisis, osteolysis, and hypercalcemia. Complete or free light chain monoclonal gammopathy in the serum and/or urine was demonstrated by protein electrophoresis and immunofixation. The polymerase chain reaction for antigen receptor rearrangement assay performed on 2 cases identified a clonally rearranged immunoglobulin gene. Neoplastic cells lacked expression of CD45, CD3, CD18, CD21, CD34, and MHCII by flow cytometry. Immunohistochemistry revealed MUM1 immunoreactivity of the neoplastic cells. Combining all data, the diagnosis was MM. An aggressive form of MM in young dogs should be a differential diagnosis for patients with an immunoglobulin-productive, B cell-clonal, CD45-negative, MUM1-positive discrete cell neoplasm arising from the bone marrow.
Keywords
Multiple myeloma (MM) is a plasma cell tumor representing roughly 1% of human and dog tumors and 8% of all hematopoietic tumors in dogs.1,7,10 Typically, MM affects middle-aged to older dogs and responds well to chemotherapy, with a reported median survival time of 930 days. 2 In dogs, diagnosis of MM has traditionally involved fulfilling at least two of the following criteria: monoclonal gammopathy, osteolytic bone lesions, >5% neoplastic cells or >20% plasma cells in bone marrow, or Bence-Jones proteinuria. 7 MM typically occurs in geriatric patients and can rarely occur in young dogs.18,19 This work describes clinicopathologic and diagnostic features of atypical MM in 3 young dogs identified in a multimodal approach.16,17
A 10-month-old male intact English bulldog (case 1) and a 10-month old male intact German shepherd dog (case 2) were presented to the Colorado State University Veterinary Teaching Hospital. A 9-month-old, female spayed West Highland white terrier (case 3) was presented to the Purdue University Veterinary Hospital. All had a history of lethargy, hyporexia, and emesis and were diffusely painful and with prominent superficial cervical lymph nodes on physical exam. Neurologically, the patients were mentally dull and case 1 demonstrated paresis with general proprioceptive ataxia affecting all 4 limbs.
Two cases had hypercalcemia characterized by elevated ionized calcium on venous blood gas analysis (1.49–1.9 mmol/1000 cm3, reference interval [RI]: 1.25–1.45 mmol/1000 cm3) and total calcium on serum biochemistry analysis (12.2 to >14.0 mg/100 cm3, RI: 9.7–12.3 mg/100 cm3). Two cases had anemia (hematocrit: 22%–33%, RI: 35%–55%). Two cases had thrombocytopenia with platelet clumps present and estimated platelet count of <50,000/cm3 based on review of the blood smear (RI: 150,000–500,000/cm3). Two cases with a clonal immunoglobulin heavy chain had hyperproteinemia (7.9 and 10.6 g/100 cm3, RI: 4.8–6.9 g/100 cm3) and hyperglobulinemia (4.4 and 6.8 g/100 cm3, RI: 1.7–3.8 g/100 cm3) (Table 1). Serum proteins were within normal limits for one case, which only displayed clonal serum free light chains and Bence-Jones proteinuria (total protein: 5.9 g/100 cm3, globulin: 1.8 g/100 cm3). Blood smear evaluation of one case revealed poorly differentiated round cells (approximately 10–15 μm diameter) with a single eccentrically located round nucleus, lacey or clumped chromatin, high nucleus:cytoplasm (N:C) ratio, and deeply basophilic cytoplasm containing numerous colorless vacuoles. The presence of ionized hypercalcemia, hyperglobulinemia, and finding of atypical round cells on blood smear were suspicious for neoplasia and prompted additional testing.
Clinical, hematology, and serum biochemistry findings in 3 dogs with atypical multiple myeloma.

Abbreviations: MI, male intact; MC, castrated male; FS, spayed female; PL, peripheral lymphadenopathy; AO: abdominal organomegaly.
12- to 15-μm cells with single eccentrically placed nucleus and moderate amount of deeply basophilic cytoplasm, presence of cryoglobulin.
All cases were negative for Ehrlichia canis, Borrelia burgdorferi, and Anaplasma phagocytophilum antibody, and Dirofilaria immitis antigen (SNAP-4Dx, IDEXX Laboratories, Westbrook, ME, USA). In addition, cases 1 and 2 were negative by quantitative polymerase chain reaction (qPCR) for Toxoplasma, Neospora, Bartonella, Rickettsia, Ehrlichia, Anaplasma, Neorickettsia, and Wolbachia.
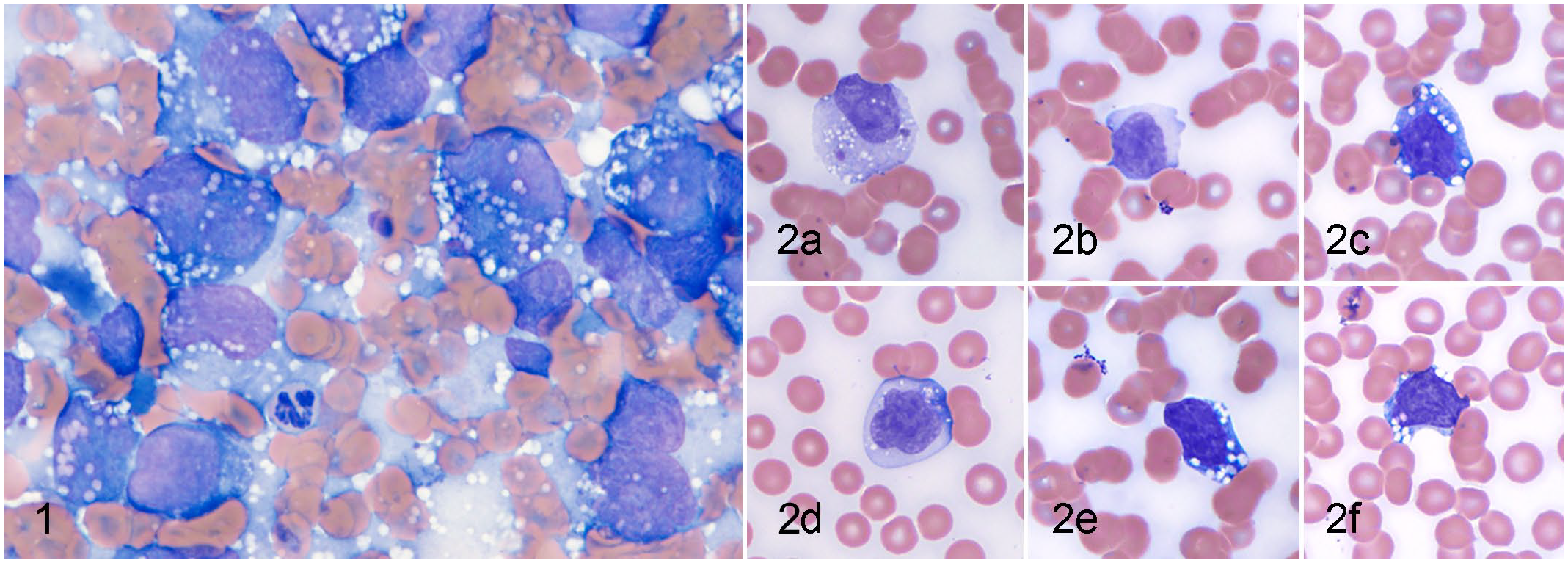
Wright-Giemsa-stained fine needle aspirates from the ilium and peripheral blood (case 1), spleen (case 2), and peripheral blood and bone marrow (case 3) revealed an anaplastic round cell neoplasm (Figs. 1, 2). Neoplastic cells were typically large discrete cells with a high N:C ratio and large round nucleus which ranged from 1.5 to 6 times the size of a neutrophil. The cells had deeply basophilic cytoplasm and clear cytoplasmic vacuoles.
All cases had evidence of an immunoglobulin paraprotein. Cases 2 and 3 had a monoclonal gammopathy in the serum and case 1 had Bence-Jones proteinuria documented by routine electrophoresis (Supplemental Table S1, Supplemental Methods S1). 3 Immunofixation performed on case 1 documented the presence of serum and urine clonal free light chains (Supplemental Fig. S1) and identified case 2 as a complete IgG4 gammopathy (Supplemental Table S1, Supplemental Methods S2). 4 Immunofixation was not performed on case 3, but there were protein aggregates on the blood smear when the sample was cooled to 20°C for 30 minutes which were not present after heating the sample to 37°C for 30 min, consistent with cryoglobulinemia.
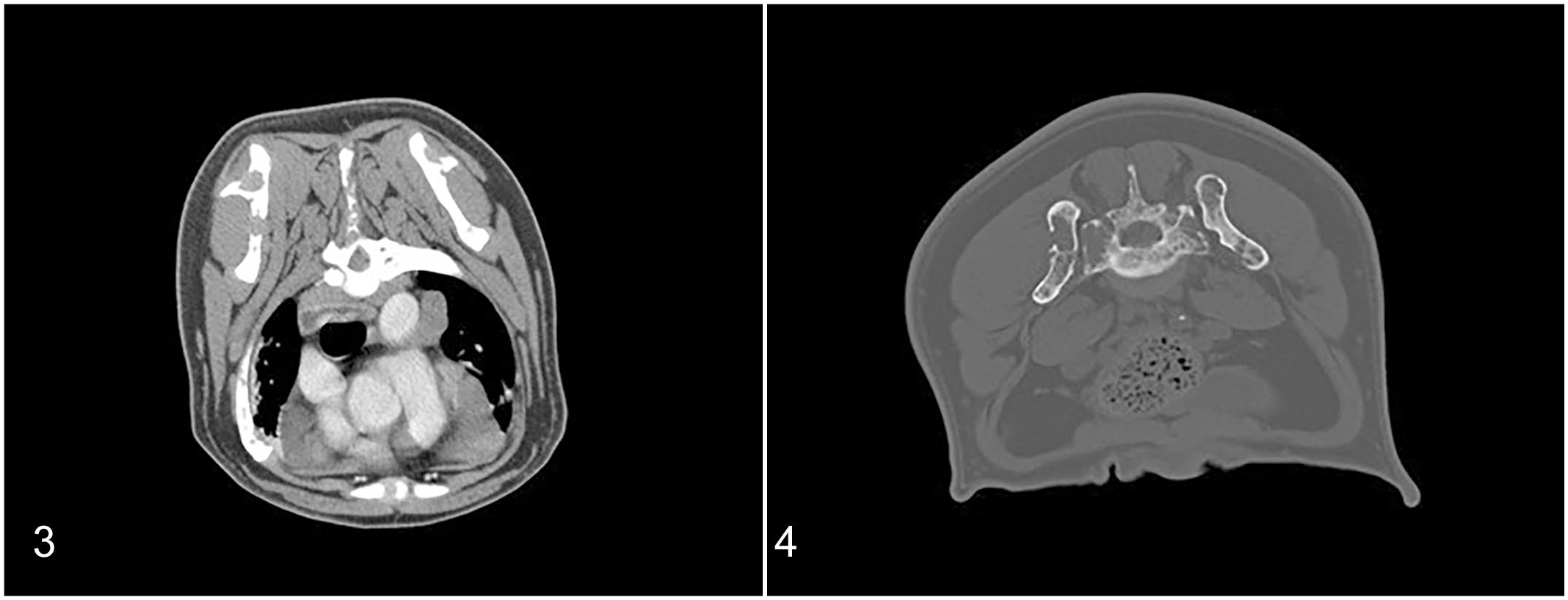
Cervical radiographs of case 1 revealed multifocal lucent osseous lesions of several cervical vertebral bodies and pedicles, proximal humeri, and scapulae. Computed tomography (CT) studies of the thorax and abdomen (pre- and 3-phase postcontrast) were performed in cases 1 and 3 using 64 slice Helical CT scanners with 1.25-mm slice thickness. CT studies showed disseminated marked cortical and medullary bone lysis. Lesions were identified in the skull, spine, ribs, humerus, and femur, with the most severe regions of lysis in the scapulae, thoracic spinous processes, and pelvic bones (Figs. 3, 4). Generalized peripheral, thoracic, and abdominal lymphadenomegaly (up to 4.0 cm) was present in cases 1 and 3. There was evidence of fractured ribs with associated pleural thickening in cases 1 and 3 and soft tissue-attenuating, moderately contrast-enhancing material, causing moderate to marked extradural spinal cord compression in the cervical spinal canal in case 1. Case 1 also had multiple large, lobulated, soft tissue-attenuating, mildly contrast-enhancing masses within the cranial mediastinum and liver, and mild splenomegaly with innumerable iso-attenuating contrast-enhancing nodules.
A cisternal cerebrospinal fluid analysis performed on case 1 (Supplemental Methods S3) documented increased protein (69 mg/100 cm3, RI < 25 mg/100 cm3) and pleocytosis (13 nucleated cells/cm3, RI 0-5 cells/cm3) consisting of 30% non-degenerate neutrophils and no neoplastic cells, with >16,000/cm3 red blood cells, suggesting blood contribution.
A CT-guided bone marrow core biopsy of the right scapula in case 1 and left humerus in case 3 were performed. Histological examination revealed marked myelophthisis with megakaryocytic and myeloid hypoplasia caused by monomorphic sheets of plasmacytoid round cells; this population had moderate amounts of amphophilic to eosinophilic cytoplasm with occasional perinuclear clear zones, one eccentrically placed round to indented nucleus with open reticular chromatin, and one to multiple prominent nucleoli. Anisocytosis and anisokaryosis were moderate with occasional binucleation. Case 1 had 7 mitotic figures in 10 representative 400× high-power fields. Approximately 90%–95% of the neoplastic cells had a strong positive nuclear labeling for MUM1 and no immunolabeling for CD79a.
Flow cytometry (antibodies and methods described in 14 ) of aspirates from lytic ilium (case 1) and humeral bone marrow (case 3) revealed 75% of the cells present were large, with a linear forward scatter value twice that of the neutrophils in the sample (Supplemental Fig. S2). Neutrophils were identified by expression of CD4 and absence of CD5 in a separate labeling tube. The large, neoplastic cells did not express CD3, CD18, CD21, CD34, CD45, or MHCII.
The PCR for antigen receptor rearrangement (PARR) assay was performed with primers that amplify a complete immunoglobulin heavy chain rearrangement (V-J), an incomplete immunoglobulin heavy chain rearrangement (D-J), the lambda light chain, and the kappa deleting element (Kde) in order to increase sensitivity of the assay (Supplemental Fig. S3). 12 Amplification of the incomplete heavy chain rearrangement and the lambda light chain genes demonstrated clonality in case 1. Amplification of the complete heavy chain gene revealed a clonal rearrangement in case 3 (Supplemental Fig. S3). 11 PARR and flow cytometry were not performed on Case 2.
All dogs were euthanized. Post-mortem gross findings were disseminated bone lysis that included osteolytic masses disrupting the ribs in cases 1 and 3; multifocal lysis and fractures of cervical spine (Fig. 5), mandible, maxilla, pelvis, and rib in case 1; and severe meningeal hemorrhage in cases 1 and 3. There was widespread lymphadenomegaly in all cases, and multifocal soft, pale, tan-to-white, discrete hepatic and splenic nodules in cases 1 and 2, and similar pancreatic nodules in case 1. Histopathologically, in all grossly affected tissues, neoplastic cells replaced normal tissue in the bone marrow (Figs. 6, 7), spleen, liver (Supplemental Fig. S4b), and visceral lymph nodes (Supplemental Fig. S4c). Case 1 had neoplastic cells in the epaxial muscles (Fig. 8) and rib (Supplemental Fig. S4a, Supplemental Table S2).
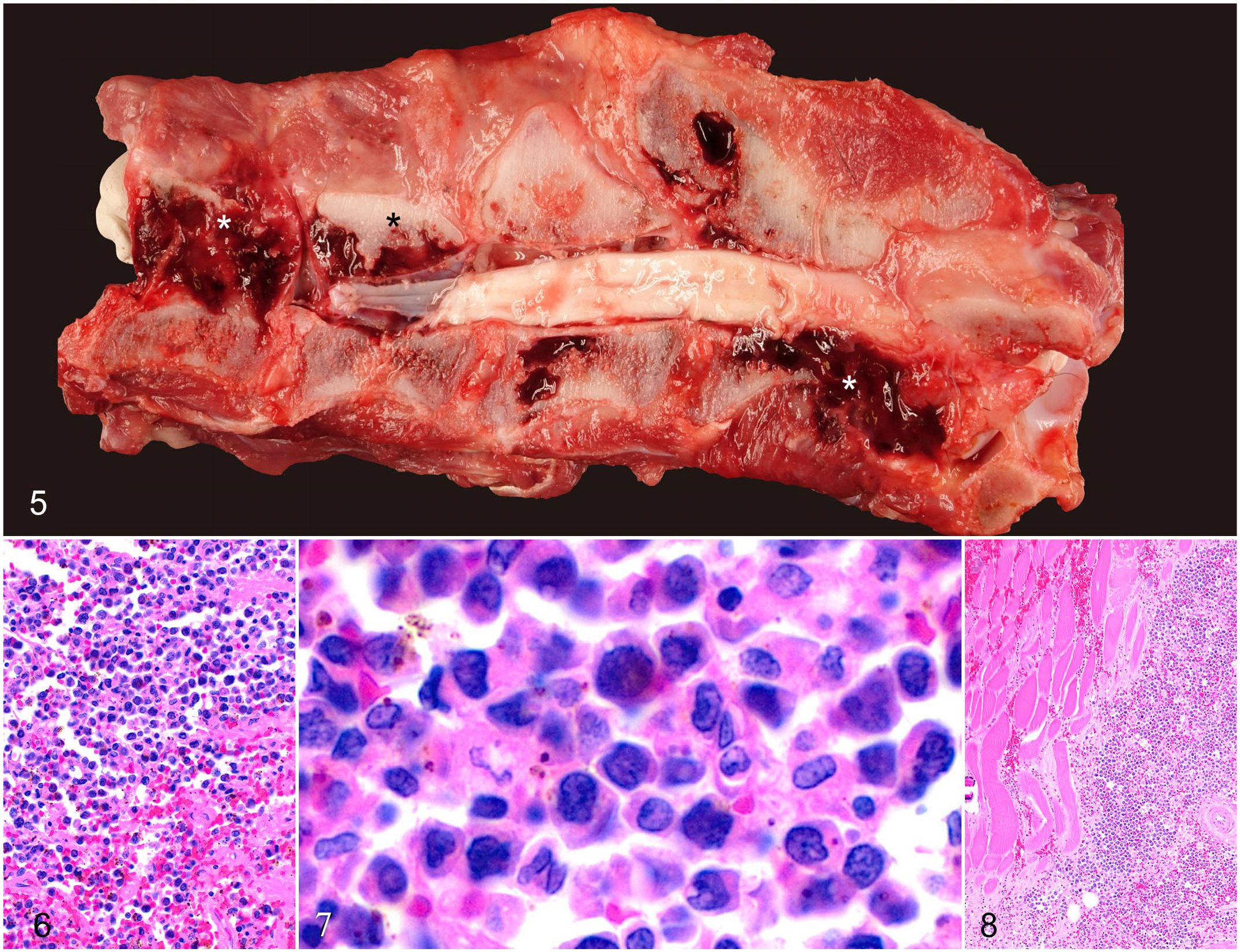
Multiple myeloma, dog, case 1.
Using immunohistochemistry all cases had nuclear labeling for MUM1 (Supplemental Fig. S4d), cases 1 and 2 displayed moderate cytoplasmic labeling for vimentin, few neoplastic cells were labeled for CD18, and the neoplastic cells were not labeled for PAX5, CD20, CD3, pancytokeratin, GFAP, Olig2, S100, NSE, desmin, or smooth muscle actin. Internal positive controls were adequate for all immunohistochemical markers tested.
Immunoreactivity for MUM1 is expected in plasma cells and can be seen bimodally in B cell lineage neoplasms prior to expression of PAX-5 and again with terminal differentiation to plasma cells, concurrent with downregulation of PAX-5. 20 An acute B cell lymphocytic leukemia was considered a differential diagnosis. MUM-1 expression has also been reported in T-cell and histiocytic neoplasms.16,17 The MUM1 immunoreactivity without other labeling using immunohistochemistry and flow cytometry, and the lack of CD45 expression and clonal IG PARR results suggested plasma cell origin. This combined with osteolytic bone lesions and paraproteinemia in a <1-year-old dog with widespread disease was consistent with an atypical, anaplastic form of MM.
Strikingly, the two cases evaluated by flow cytometry lacked CD45 labeling. Although normal plasma cells are generally positive for CD45, abnormal human plasma cells are more variable in their expression of CD45. 5 Lack of CD45 expression by neoplastic canine plasma cells has been previously documented. 13 A study of 95 human myeloma cases undergoing high-dose therapy demonstrated a shorter survival for those lacking CD45 expression on plasma cells. 9 Thus, as in these canine cases, CD45-negative neoplastic plasma cells may be associated with greater severity of MM.
The patient with Bence-Jones proteinuria had a serum M-protein detected using an immunofixation protocol even though there was no hyperproteinemia. Previous cases of monoclonal gammopathy without hyperproteinemia have been described in dogs.8,15 Interestingly, both immunofixation and PARR documented clonal light chain rearrangement with an incomplete, and nonproductive, rearrangement of the heavy chain gene. Similar findings have been documented in human free light chain MM. 6 Evaluation of serum and urine electrophoresis and immunofixation is recommended to diagnose and characterize MM.
This report describes 3 cases of atypical aggressive MM in young dogs and highlights the importance of considering plasma cell neoplasia for anaplastic round cell neoplasms even when CD45 labeling is negative. All 3 dogs were severely affected, and 2 cases had widespread osseous involvement, making a prompt diagnosis critical for early intervention. The findings from this report will aid in accurate diagnosis of atypical cases of MM. In addition to common differential diagnoses for hypercalcemia, anaplastic plasma cell tumors should also be considered in young dogs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221087637 – Supplemental material for Atypical multiple myeloma in 3 young dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858221087637 for Atypical multiple myeloma in 3 young dogs by Ian J. Wachowiak, A Russell Moore, Anne Avery, Forgivemore Magunda, Adam Harris, Hannah Laurence, Christopher M. Fulkerson, Caroline V. Fulkerson, Joanne B. Messick, Natalia J. Strandberg and Stephanie McGrath in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
