Abstract
Multiple myeloma oncogene 1/interferon regulatory factor 4 (MUM1/IRF-4) immunohistochemistry (IHC) is mainly used for diagnostic confirmation of plasma cell tumors (PCTs) in dogs and cats. This article describes MUM1/IRF-4 IHC expression in 20 cases of canine cutaneous histiocytoma (CH) and compares it with 10 cutaneous or mucocutaneous PCTs and 5 cutaneous histiocytic sarcomas (HSs) submitted to the same IHC protocol. All histiocytomas had strong nuclear and variable cytoplasmic immunolabeling for MUM1/IRF-4, whereas all PCTs had strong nuclear and moderate cytoplasmic immunolabeling for MUM1/IRF-4. No MUM1/IRF-4 immunolabeling was detected in the HSs. Although not typically a diagnostic challenge, MUM1/IRF-4 expression may have to be used with caution or in conjunction with additional immunomarkers to differentiate among poorly differentiated round cell tumors, especially when a histiocytic or plasma cell origin is suspected.
The diagnosis of canine cutaneous round cell tumors may be difficult, and immunohistochemistry (IHC) is often required for diagnostic confirmation, particularly in situations where tumor morphology does not suffice for a final diagnosis based on histopathology. 1,10,12 For this reason, a wide variety of antibodies and IHC panels have been developed and are currently available for the diagnosis of round cell tumors in dogs. 1,9,10,12 Cutaneous histiocytoma (CH) and cutaneous or mucocutaneous plasma cell tumor (PCT) are common round cell tumors of dogs. 5,7 Histiocytomas arise from the cutaneous Langerhans cells and are usually solitary, occurring mainly on the head, ears, and limbs. 5 Plasma cell tumors can be single or multiple and affect mainly the ears, digits, lips, and oral cavity. 7 Distinction between CHs and PCTs is not often a diagnostic conundrum, but IHC may be needed in less clear-cut cases. 1,12 We have come across cases of canine cutaneous round cell tumors that, while morphologically consistent with histiocytoma, were concomitantly immunoreactive for multiple myeloma oncogene 1/interferon regulatory factor 4 (MUM1/IRF-4) and ionized calcium-binding adapter molecule 1 (Iba1), suggesting that MUM1/IRF-4 expression is not specific for PCTs.
MUM1/IRF-4 is a transcription factor associated with cell growth, especially B-cell maturation, but is also expressed by plasma cells, activated T-cells, and subsets of macrophages and dendritic cells. 3,6,10,11,13,14 For this reason, MUM1/IRF-4 IHC has been routinely used for diagnostic confirmation of plasma cell neoplasia in dogs. 10 Different than what we have experienced in our diagnostic cases, MUM1/IRF-4 expression has not been shown to occur in canine histiocytic tumors, including histiocytomas. 10 In this article, we demonstrate consistent MUM1/IRF-4 immunoreactivity in a set of canine CHs and compare it with MUM1/IRF-4 IHC results in a group of PCTs and cutaneous histiocytic sarcomas (CHSs) subject to the same IHC protocol.
Cases were randomly selected from the biopsy service of the University of Georgia Athens Veterinary Diagnostic Laboratory (Athens, GA). A total of 20 CHs, 10 cutaneous or mucocutaneous PCTs, and 5 CHSs were examined (Suppl. Table S1). All neoplasms had been diagnosed during the routine diagnostic service based on histopathology. Tissue sections from all CHs, PCTs, and CHSs were subjected to MUM1/IRF-4 and Iba1 IHC, with MUM1/IRF-4 positivity and Iba1 negativity considered diagnostic for PCTs and Iba1 positivity diagnostic for CHs and CHSs. The IHC was performed on an automated stainer (Nemesis 3600, Biocare Medical, Concord, CA). For the Iba1 protocol, a rabbit polyclonal antibody directed against Iba1 (Wako, Neuss, Germany) at a dilution of 1:8000 for 60 minutes was used. Antigen retrieval was performed using Antigen Retrieval Citra Solution 10X (BioGenex) at a dilution of 1:10 for 15 minutes at 110°C. A biotinylated rabbit antibody (Vector Laboratories, Burlingame, CA) was used to detect the target, and the immunoreaction was visualized using 3,3-diaminobenzidine (DAB; Dako, Santa Clara, CA) substrate for 12 minutes counterstained with hematoxylin. For the MUM1/IRF-4 protocol, a rabbit monoclonal antibody against MUM1/IRF-4 (Biocare Medical) at a dilution of 1:50 for 60 minutes was used. Antigen retrieval on tissue sections was conducted using Reveal Decloaker 10X (Biocare Medical) at a dilution of 1:10 for 15 minutes at 110°C. A biotinylated rabbit antibody (Vector Laboratories) was used to detect the target, and the immunoreaction was visualized using DAB (Dako) substrate for 12 minutes counterstained with hematoxylin. Control tissues consisted of normal lymph node for MUM1/IRF-4 and normal-haired skin for Iba1. In addition, MUM1/IRF-4 immunoreactivity was scored visually within a tumor across all CHs and PCTs as follows: 3 = >60% positive cells; 2 = 30%–59% positive cells; 1 = 10%–29% positive cells; and 0 = <10% positive cells. 10 Results were assessed by Fisher exact test.
All CHs and CHSs exhibited strong cytoplasmic immunoreactivity for Iba1, and all PCTs had strong nuclear and moderate cytoplasmic immunoreactivity for MUM1/IRF-4 and no immunoreactivity with Iba1, confirming the routine diagnoses in all cases (Suppl. Table S1; Figs. 1–6). All CHs exhibited immunoreactivity for MUM1/IRF-4, including nuclear immunoreactivity that was patchy to diffuse and weak to strong and cytoplasmic immunoreactivity that was variable and weak to moderate. All CHSs were consistently negative for MUM1/IRF-4 IHC. There were no statistically significant differences in scoring (P = .14) between CHs and PCTs (Suppl. Table S1). Histiocytomas with scores of 1 or 2 (<60% of cells positive) had most of the positive cells distributed along the superficial aspect of the tumor.
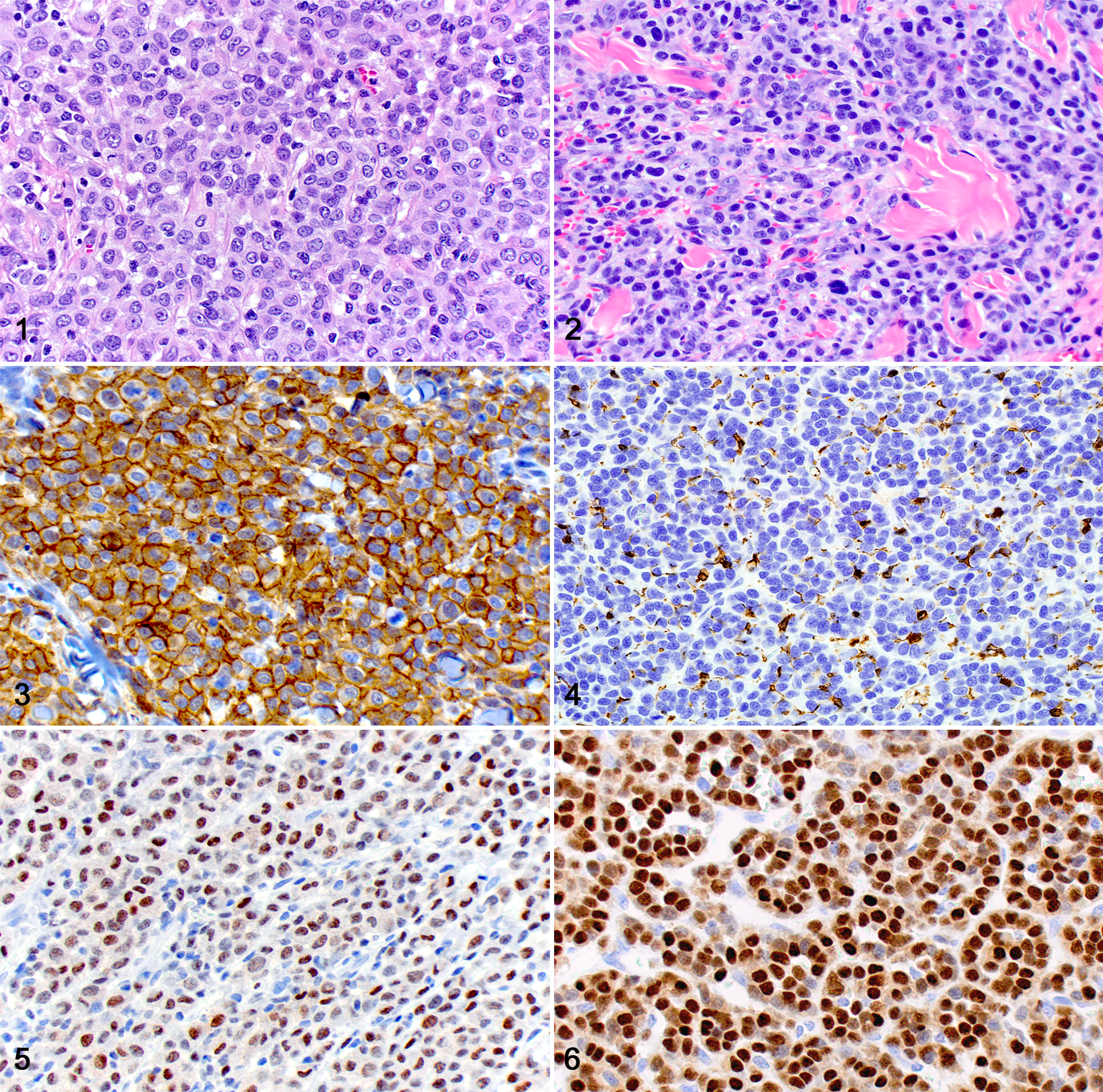
In this study, we describe 20 canine CHs with consistent immunolabeling MUM1/IRF-4 by IHC. To our knowledge, this is the first report describing MUM1/IRF-4 expression in canine CH. These results are different from current information in veterinary medicine, which indicates that MUM1/IRF-4 is not expressed in canine histiocytic tumors, including CHs. 10 MUM1/IRF-4 expression has been demonstrated in human and murine macrophages, where its overexpression activates interleukin (IL)–1β and represses PU.1-independent H-2 L promoter. 4 In combination with interferon consensus sequence binding protein, MUM1/IRF-4 also represses interferon-stimulated gene-15 in macrophages. 11 Finally, MUM1/IRF-4 expression is upregulated and plays an important role in human monocytic cells undergoing dendritic cell differentiation. 3 Dendritic cells further express MUM1/IRF-4 to modulate IL-10 and IL-33 cytokine production and specifically promote Th2 differentiation and inflammation. 14 These studies illustrate the expression of MUM1/IRF-4 by normal monocyte-derived macrophages and dendritic cells and their molecular functions. Normal human epidermal Langerhans cells and neoplastic cells in Langerhans cell histiocytosis express high levels of MUM1/IRF-4, similar to what is observed in cases of multiple myeloma. 2 For these reasons, MUM1/IRF-4 is often not used for the diagnosis of human round cell tumors as it lacks sufficient specificity for plasma cell origin compared with other markers, such as CD138 and VS38. 6 MUM1/IRF-4 expression in canine CHs is likely due to its Langerhans cell origin, and this is further supported by the lack of MUM1/IRF-4 immunoreactivity in our CHSs, as these originate from interstitial dendritic cells and not from Langerhans cells. 5,10
Although PCTs had strong cytoplasmic immunolabeling with MUM1/IRF-4, cytoplasmic immunolabeling was weak or moderate in the CHs, which may be reflective of lower expression of MUM1/IRF-4 in monocytic cells when compared with plasma cells. In fact, normal murine macrophages have been shown to express 65%–75% lower levels of MUM1/IRF-4 transcripts compared with B-lymphocytes. 4 In addition, neoplastic transformation can result in upregulation or downregulation of normally produced target antigens. 8
Despite differences in cytoplasmic immunolabeling, nuclear immunolabeling with MUM1/IRF-4 IHC is considered the most important feature in determining positive expression within target cells. 10 Histiocytomas and PCTs in this study had prominent nuclear expression of MUM1/IRF-4, making differentiation between poorly differentiated CHs and PCTs solely based on MUM1/IRF-4 IHC difficult. There were no statistically significant differences in scoring MUM1/IRF-4 positivity between the CHs and PCTs, making the percentage of positive cells an unreliable criterion for differentiation. Specific, unidentified mutation(s) and subsequent upregulation of MUM1/IRF-4 expression in neoplastic Langerhans cells of cutaneous canine histiocytoma may explain the strong nuclear MUM1/IRF-4 positivity seen in these cases compared with those in previous reports. The diverse age, breed, sex, and anatomic location of these cases (Suppl. Table S1) suggest that MUM1/IRF-4 is likely expressed at variable levels in all CHs and is not specific for any of those factors.
Our study design contains technical differences from previous work, which may have contributed to conflicting results with the literature. 10 Differences between our studies include the antibody used (rabbit versus mouse monoclonal), method of antigen retrieval (low pH versus high pH), and method of detection (ABC versus ImmPRESS). 10 The antibodies could have differences in binding affinity or avidity for MUM1/IRF-4, allowing the antibody in the current study to bind more favorably to MUM1/IRF-4 and detect it at lower levels of expression. This is consistent with the diffuse, strong nuclear immunolabeling in all our PCTs compared with only 60% of PCTs with a score of 3 in previous work. 10 Similarly, lower-level expression of MUM1/IRF-4 in CHs may be below detectable limits for previous antibodies, making the antibody in this study more sensitive for MUM1/IRF-4 expression and less specific for PCTs.
In this article, we describe a set of canine CH that are consistently immunoreactive for MUM1/IRF-4, an immunomarker typically used for diagnostic confirmation of plasma cell neoplasia in veterinary medicine. Therefore, we recommend MUM1/IRF-4 IHC to be used as part of a round cell tumor panel, including not only Iba1 but also lymphocytic immunomarkers, and urge caution when using MUM1/IRF-4 IHC alone to distinguish between poorly differentiated round cell tumors, especially CHs and PCTs.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818759770 - Immunohistochemical Labeling of Multiple Myeloma Oncogene 1/Interferon Regulatory Factor 4 (MUM1/IRF-4) in Canine Cutaneous Histiocytoma
Supplemental Material, DS1_VET_10.1177_0300985818759770 for Immunohistochemical Labeling of Multiple Myeloma Oncogene 1/Interferon Regulatory Factor 4 (MUM1/IRF-4) in Canine Cutaneous Histiocytoma by Justin M. Stilwell, and Daniel R. Rissi in Veterinary Pathology
Footnotes
Acknowledgments
We thank Nicole Young (Histology Laboratory, Department of Pathology, College of Veterinary Medicine, University of Georgia) for the outstanding support with immunohistochemistry.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
