Abstract
Primary colorectal follicular lymphomas are rare indolent lymphoid neoplasms in humans that have not been reported in dogs. We describe 3 cases of primary colorectal follicular lymphoma in dogs with histologic and immunohistochemical features similar to their human counterpart. Initial clinical signs in all dogs included tenesmus, hematochezia, and a palpable rectal mass. Two dogs were castrated males and 1 an intact female, between 9 months and 2 years of age, and of varied breeds. All 3 cases of colorectal follicular lymphoma were characterized by proliferation of follicular germinal centers with no polarity or mantle zone and were composed of centrocytes admixed with fewer centroblasts. By immunohistochemistry, lymphoid cells expressed CD20, BCL2, and BCL6 and lacked expression of CD3, CD5, and cyclin D1. Polymerase chain reaction for rearrangements of the immunoglobulin heavy chain confirmed a monoclonal population in all cases. In 2 of the 3 cases, a solitary nodular colorectal mass was excised and appeared curative; however, the third case had multiple colorectal masses and the animal developed multicentric lymphoma. This case series immunohistochemically characterizes and distinguishes colorectal follicular lymphoma from atypical lymphoid hyperplasia.
Keywords
In humans, primary gastrointestinal lymphomas are most commonly B-cell lymphomas, with diffuse large B-cell lymphoma (DLBCL) and mucosa-associated lymphoid tissue lymphoma (MALTOMA) representing the majority of cases. 17 These lymphomas most commonly involve the duodenum or jejunum, with the colon/rectum representing an uncommon primary location. Primary colorectal follicular lymphoma (FL) in humans represents a rare entity that must be differentiated from atypical lymphoid hyperplasia and from other types of nodular lymphoma for appropriate prognostication and treatment. Histologically, FLs, regardless of their site of origin, are characterized by well-circumscribed expansions of follicular germinal centers that do not exhibit polarity and have lost their mantle zone. Within the neoplastic germinal centers, intermediate-sized centrocytes with indented, heterochromatic nuclei and indistinct nucleoli predominate. Mixed within this population are centroblasts that contain large euchromatic nuclei with multiple large amphophilic nucleoli, often marginalized along the nucleolemma. 11 The loss of polarity and loss of the mantle zone are important features in distinguishing this entity from atypical lymphoid hyperplasia. Polarity refers to the 2 morphologically distinct zones within the germinal center, the light zone composed of centrocytes and the dark zone composed of centroblasts that are proliferative and undergo somatic hypermutation. In humans, primary FLs are immunohistochemically characterized by immunoreactivity for CD20, BCL2, and BCL6. 15 In humans, FLs can be differentiated from T-cell lymphomas and mantle zone lymphomas by their lack of immunoreactivity for CD3, CD5, and cyclin D1. 15,18 Primary colorectal FLs tend to have a good response to surgical resection or monoclonal antibody therapy; 11 however, they have been shown to recur and can transform to DLBCL in the later stages of disease. 4,7,8
In dogs, most primary gastrointestinal lymphomas involve the small intestine or stomach, while the colon or rectum is rarely affected. 10 Enteropathy-associated T-cell lymphoma (EATL) large cell (type 1) is the most common primary form of gastrointestinal canine lymphoma; however, diffuse large B-cell lymphoma also occurs as primary gastric and less frequently presents as small intestinal lymphoma. 2,9,10,20,21 FL in dogs is an uncommon nodular lymphoma that typically affects lymph nodes or spleen. 9,21 Like humans, FLs in dogs are B-cell lymphomas that must be distinguished from atypical lymphoid hyperplasia. There are too few studies of canine FLs to accurately predict survival times or therapeutic response. Interestingly, the few cases of FL in dogs with clinical follow-up data had survival times of greater than 1 year, the same survival time for dogs diagnosed with atypical lymphoid hyperplasia. 20,21 Primary colorectal FLs have not been previously described in dogs, and previous cases may have been misdiagnosed as atypical lymphoid hyperplasia. Therefore, there is a need to accurately characterize this specific entity not only for diagnostic purposes but also to correctly predict the biologic behavior and to develop optimal therapeutic options.
Three dogs were included in this study. Full-thickness colorectal biopsies were taken from each dog and submitted for histologic evaluation. The 3 surgical biopsy specimens were routinely fixed in 10% neutral buffered formalin and embedded in paraffin. From each paraffin block, 5-μm serial sections were routinely stained with hematoxylin and eosin or immunohistochemistry (IHC) for CD3, 2,6 CD20, 2,6 BCL2, 3,16 BCL6, 14 and cyclin D1 12 was performed as previously described (see Table 1). Polymerase chain reaction (PCR) for antigen receptor rearrangement (PARR) of the immunoglobulin heavy chain (IGH) was performed on each case as previously described. 1,13
Antibodies and Detection Methods Used for Immunohistochemistry.
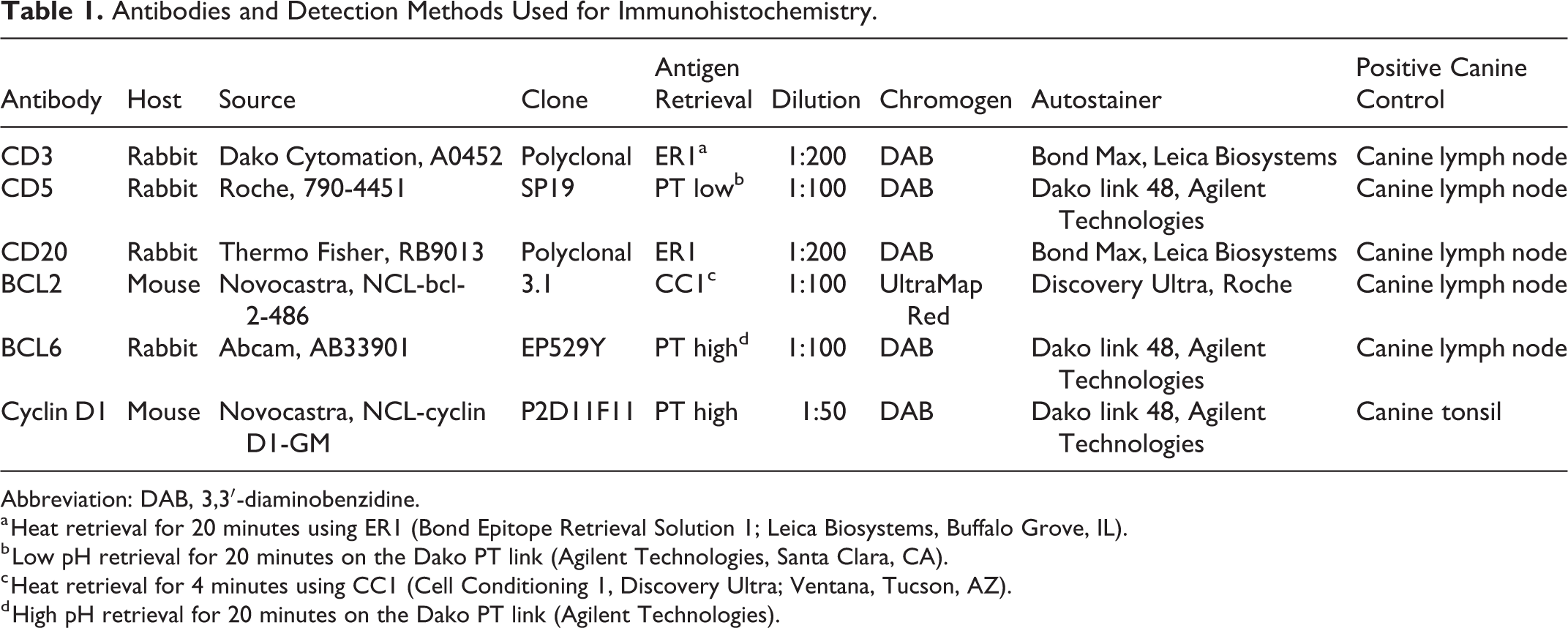
Abbreviation: DAB, 3,3′-diaminobenzidine.
a Heat retrieval for 20 minutes using ER1 (Bond Epitope Retrieval Solution 1; Leica Biosystems, Buffalo Grove, IL).
b Low pH retrieval for 20 minutes on the Dako PT link (Agilent Technologies, Santa Clara, CA).
c Heat retrieval for 4 minutes using CC1 (Cell Conditioning 1, Discovery Ultra; Ventana, Tucson, AZ).
d High pH retrieval for 20 minutes on the Dako PT link (Agilent Technologies).
Dog No. 1 was a 2-year-old male castrated Dachshund that had a palpable rectal mass and mild hematochezia. Surgical exploration identified a solitary 6-mm × 2-mm mass expanding the rectal mucosa. The mass was completely excised. Dog No. 2 was a 9-month-old female intact English Bulldog that presented with tenesmus and hematochezia. The dog was treated with metronidazole and amoxicillin. A month after initial presentation, a solitary 2-cm × 3-cm rectal mass was found expanding the dorsolateral rectal mucosa. The mass was completely excised. Dog No. 3 was a 2-year-old male castrated Shih Tzu that presented with tenesmus, hematochezia, and a palpable rectal mass. Surgical exploration identified 2 masses, 2 cm and 0.6 cm in diameter, that expanded the rectal mucosa. In all 3 cases, there was no peripheral lymphadenopathy or clinical signs to suggest systemic disease.
Histologically, all 3 cases appeared similar and were characterized by well-circumscribed nodular masses within the submucosa that elevated the overlying rectal mucosa but did not extend into it (Fig. 1). The masses were composed of prominent, often confluent, lymphoid follicles with expanded germinal centers that had lost the morphologically distinct light and dark zones consistent with normal polarity and had no mantle zone. These follicles were composed predominantly of intermediate-sized (10–13 μM) centrocytes with characteristic indented, heterochromatic nuclei and indistinct nucleoli. Mixed within this population and comprising less than 10% of the whole population were centroblasts that had large euchromatic nuclei with multiple large (14–20 μM) amphophilic nucleoli that often marginalized along the nucleolemma (Fig. 2). In all cases, these neoplastic lymphocytes did not infiltrate into the overlying lamina propria mucosa or muscularis mucosae and did not invade into the underlying circumferential tunica muscularis.
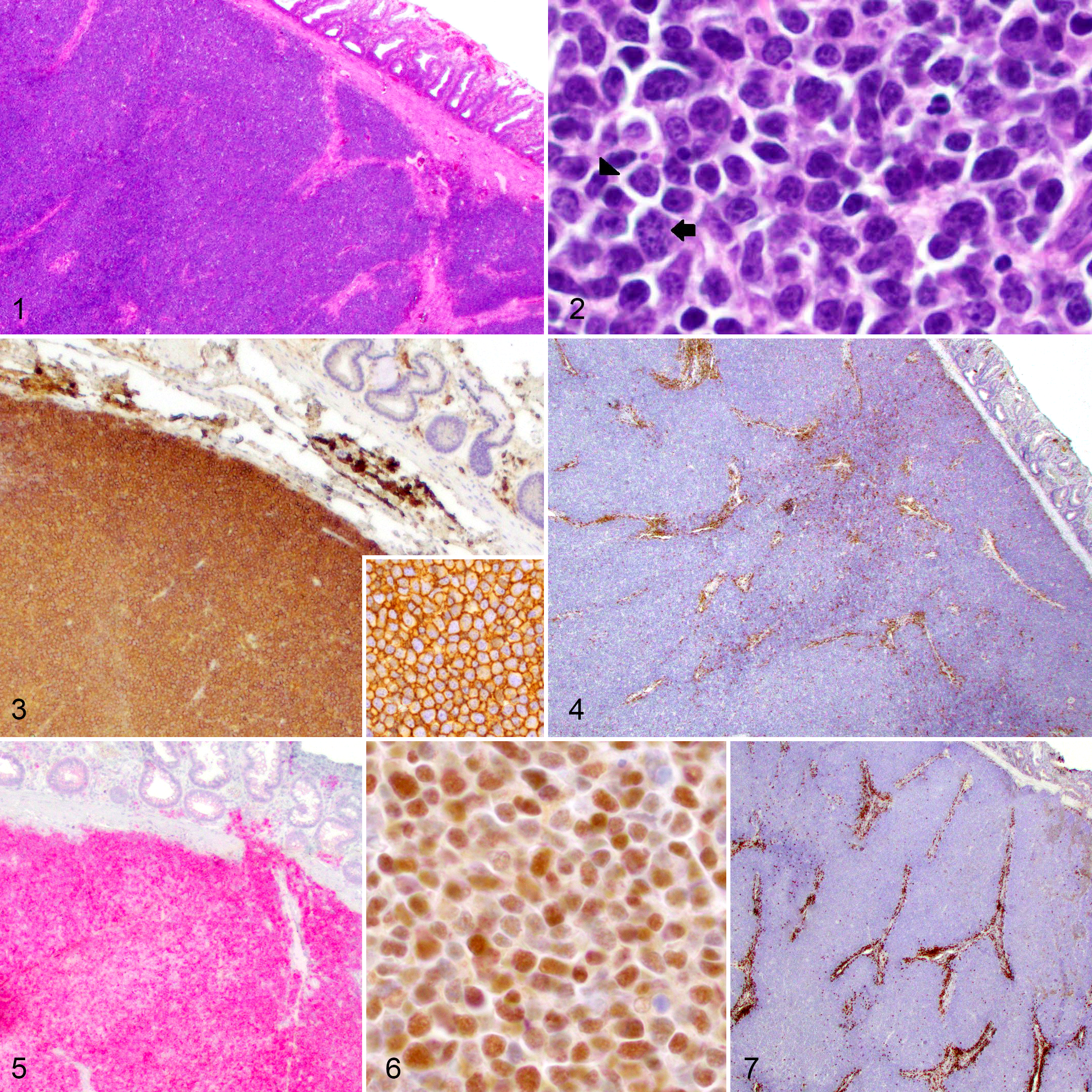
Follicular lymphoma, dog; submucosa, large intestine.
The centrocytes and centroblasts forming confluent follicles had strong membranous labeling for CD20 confirming B-cell lineage (Fig. 3). These cells also exhibited strong intracytoplasmic labeling for BCL2 (Fig. 5) and strong nuclear labeling for BCL6 (Fig. 6), confirming germinal center origin. The neoplastic lymphocytes did not label with CD3, CD5, or cyclin D1; however, control slides run in parallel showed positive labeling (Figs. 4, 7; cyclin D1 and controls not shown). The morphologic immunophenotypic findings exclude T-cell lymphoma, marginal zone lymphoma, and mantle cell lymphoma as differential diagnoses. PARR testing demonstrated monoclonal rearrangement of the IGH gene for all 3 cases, further confirming a B-cell lymphoma (Fig. 8). The clinical presentation and histologic, immunohistochemical, and PARR results were consistent with a diagnosis of primary colorectal FL in all 3 cases.
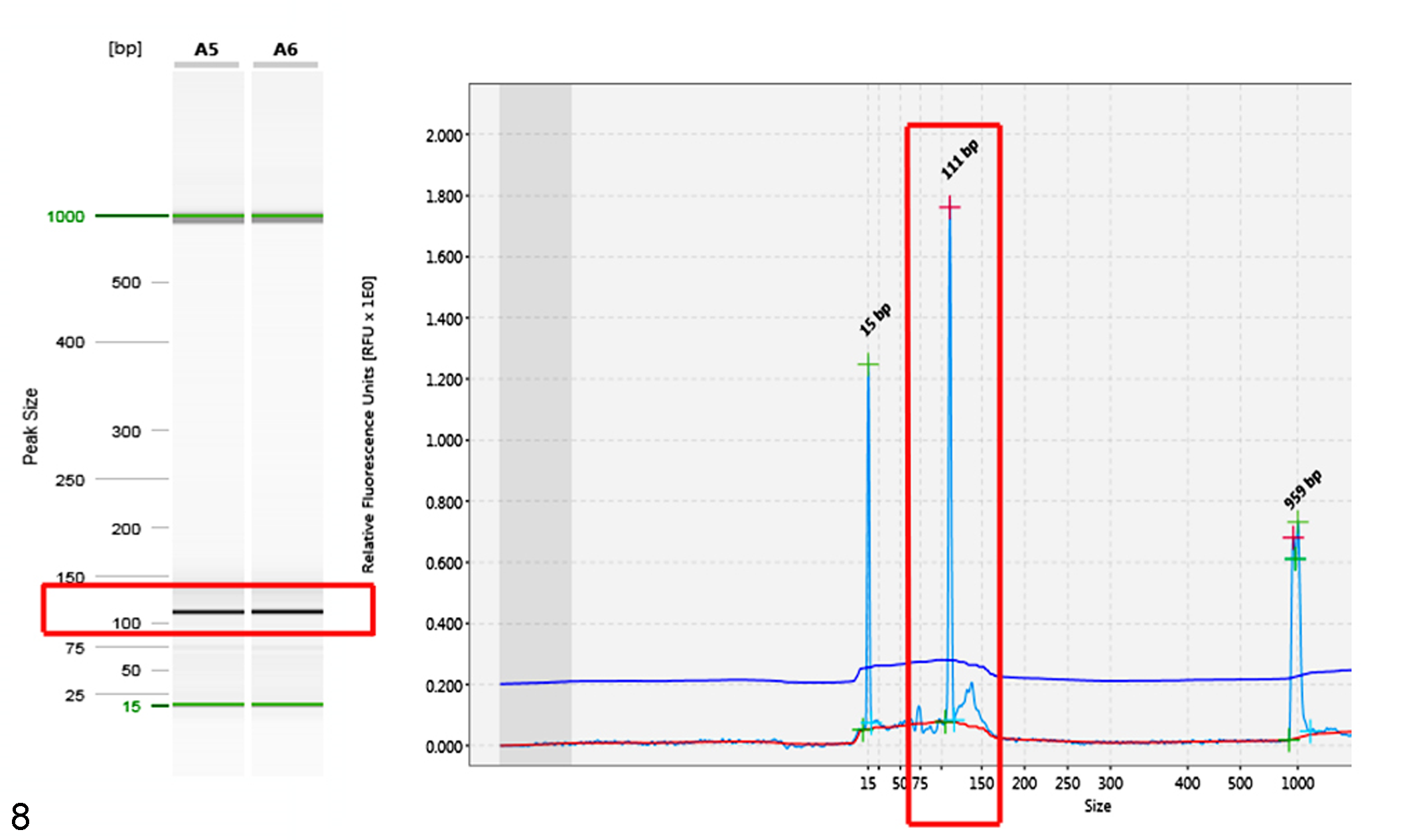
Polymerase chain reaction for antigen receptor rearrangements (PARRs) for the immunoglobulin heavy chain (IGH) gene assessed by capillary gel electrophoresis. The red boxes depict a single strong band at 111 bp run in duplicate within the gel image (left) and a single tall narrow peak at 111 bp within the electropherogram (right) confirming clonal rearrangement of the IGH gene. On either side of the red box within the electropherogram are smaller alignment markers.
For dog No. 1, the solitary nodular rectal mass was completely excised. At 8 years after surgery, there has been no local recurrence of the mass and no indication of systemic lymphoma. For dog No. 2, the solitary nodular rectal mass was also completely excised. At 2 years after surgery, there has been no reported recurrence of the mass and no indication of systemic lymphoma. For dog No. 3, 2 masses had expanded the rectal mucosa, and only 1 mass was excised. There was no indication of systemic disease at the time of surgical resection. Two months after the initial presentation, the dog was diagnosed with multicentric lymphoma that was not further characterized. A multiagent chemotherapy protocol of cyclophosphamide, doxorubicin, vincristine, and prednisolone was started. The dog went into complete remission and was followed for 2 years. Repeated complete blood counts remained within reference intervals. Since completion of chemotherapy, there has been no indication of lymphadenopathy or clinical recurrence in this animal.
In humans, colorectal FL is differentiated from atypical lymphoid hyperplasia, T-cell lymphoma, and mantle zone lymphoma through an array of immunohistochemical markers and PARR testing that all serve to increase specificity and improve the selection of therapeutic strategies. 11,17 In veterinary medicine, such thorough characterization is not typically pursued, and nodular rectal masses may be mistaken for atypical lymphoid hyperplasia on histologic evaluation alone. Therefore, this study outlines the need for additional characterization in cases of primary colorectal FL in dogs, particularly in cases with multiple masses where transformation to an aggressive phenotype, such as DLBCL, may be of concern.
In this study, PARR testing was performed on all 3 cases and demonstrated a monoclonal rearrangement in the IGH gene, further supporting the diagnosis of a neoplastic rather than a hyperplastic lymphoid proliferation. Using PARR testing is essential in accurately differentiating nodular type B-cell lymphomas from hyperplastic lesions. 5 All samples were strongly immunoreactive for CD20, BCL2, and BCL6. These results are all consistent with a B-cell phenotype and germinal center origin. 14 In humans, BCL2 and BCL6 are commonly used to diagnose FL and to differentiate it from other nodular type lymphomas such as marginal zone lymphoma or mantle cell lymphoma. 4,7,19
Most commonly, canine intestinal lymphomas are enteropathy-associated T-cell lymphomas of the large cell type (EATL type 2) and are associated with poor survival. 8,11 In humans, primary colorectal FLs tend to have a good prognosis following complete surgical resection. 5 –7 Based on the limited number of cases described here, dogs with colorectal FL appear to have prolonged survival times following complete surgical resection. It has been suggested that approximately 30% of FLs in humans undergo transformation from an indolent disease to a more aggressive phenotype, such as DLBCL. 7 In dogs, recent studies have suggested that manifestations of DLBCL represent de novo growth, 19 rather than transformation of an indolent lymphoma precursor; however, a more thorough examination including molecular markers of transformation, variability of specific nodal and extranodal locations, and identification of genetic mechanisms is necessary. One of the dogs diagnosed with colorectal FL in this series developed multicentric lymphoma following resection of 1 of the 2 reported colorectal masses. The dog responded well to cyclophosphamide, hydroxydaunorubicin, vincristine and prednisone (CHOP) therapy and has remained in remission for at least 2 years. Unfortunately, no further biopsies were taken to further characterize the multicentric lymphoma, and it is unclear whether malignant transformation from an indolent phenotype to a more aggressive phenotype, such as DLBCL, may have occurred. Furthermore, FL in dogs may go unnoticed until transformation to DLBCL and more severe clinical signs are evident, which increases the difficulty in understanding the role of malignant transformation in colorectal FL in dogs.
This study characterizes the previously unrecognized entity of colorectal FL in dogs and highlights the importance of combining histomorphology, clinical presentation, immunophenotyping, and PARR testing to distinguish colorectal FL from atypical lymphoid hyperplasia. An array of immunohistochemical markers that includes CD3, CD20, BCL2, BCL6, and PARR testing is recommended to accurately differentiate colorectal FL from atypical lymphoid hyperplasia or other forms of indolent lymphoma. This differentiation may be important in cases where surgical resection is not elected, as such cases have the potential to transform to an aggressive phenotype over time. In addition, the results of this case series suggest that colorectal FL in dogs may follow a similar clinical course to that of humans.
Footnotes
Acknowledgements
We thank the histology team at the Michigan State University Veterinary Diagnostic Lab, the multiple clinicians involved in each of these cases, and the owners of each animal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
