Abstract
Giraffe skin disease (GSD) is an emerging disease of free-ranging giraffe recognized in the last 25 years in several species, including the critically endangered Nubian giraffe (Giraffa camelopardalis camelopardalis) of Uganda. Identifying the cause of GSD and understanding its impact on health were deemed paramount to supporting these vulnerable populations. Sixty-four giraffes were immobilized in Murchison Falls National Park, Uganda, from 2017 to 2019, and GSD lesions were opportunistically biopsied. Fifty-five giraffes (86%) had GSD lesions on the neck, axilla, chest, and cranial trunk. Lesions were categorized into early, intermediary, and dormant stages based on gross and histological characteristics. Early lesions were smaller, crusted nodules with eosinophilic and pyogranulomatous dermatitis and furunculosis. Intermediary lesions were thick plaques of proliferative and fissured hyperkeratosis and acanthosis with dense dermal granulation tissue and severe eosinophilic and granulomatous dermatitis. Lesions appeared to resolve to dormancy, with dormant lesions consisting of hairless plaques of hyperkeratosis with dermal scarring and residual inflammation. The periphery of early and intermediary lesions included follicular granulomas containing adult filarid nematodes, with myriad encysted microfilariae in the superficial dermis. Stage L3 larvae were common in early and intermediary lesions, and dormant lesions had remnant encysted microfilariae with no adult or stage L3 larvae. Nematodes were morphologically and genetically novel with close identity to Stephanofilaria spp. and Brugia malayi, which cause infectious filariasis. Identification of potential insect vectors, long-term monitoring of GSD lesions, and evaluating response to therapy is ongoing in the efforts to help conserve the Nubian giraffe.
Keywords
Giraffe (Giraffa spp.) is one of the most iconic taxa of wildlife, currently extending across 21 countries (Giraffe Conservation Foundation 2020 assessment). In the last 30 years, it is estimated that populations of giraffe have dramatically decreased by approximately 30% due to habitat loss, habitat fragmentation with loss of critical habitat diversity, poaching, and a number of other key factors, including climate change.31,35 Giraffes were formerly recognized as a single species designated as least concern but are now elevated to vulnerable on the International Union for Conservation of Nature’s (IUCN) Red List.31,35 Multilocus population genetic analyses of distinct giraffe populations throughout Africa were performed in recent years, and 4 highly distinct lineages, representing discrete species with certain subspecies, were identified.6,10 The species and subspecies identified include Masai giraffe (Giraffa tippelskirchi) with the Luangwa giraffe (Giraffa tippelskirchi thornicrofti) as a subspecies; Northern giraffe (Giraffa camelopardalis) with 3 subspecies that include the Kordofan (Giraffa camelopardalis antiquorum), Nubian (Giraffa camelopardalis camelopardalis), and West African giraffe (Giraffa camelopardalis peralta); the reticulated giraffe (Giraffa reticulata); and the Southern giraffe (Giraffa giraffa) with 2 subspecies that include the Angolan (Genetta angolensis) and South African giraffe (Giraffa giraffa). The Nubian giraffe number an estimated 3000 individuals confined to isolated groups in progressively fragmented habitat in Ethiopia, Kenya, South Sudan, and Uganda. As a result of their critically endangered status, targeted conservation efforts within Uganda have increased to protect, monitor, and reestablish the Nubian giraffe. The largest population of Nubian giraffe resides in northern Murchison Falls National Park (MFNP), Uganda, consisting of approximately 1550 individuals. This population is critical for the genetic preservation of the species.
Roughly 45% of adult and subadult giraffes in MFNP are afflicted with skin lesions that manifest as patches of hairless, hyperkeratotic skin, coated with sheets of small flies; the condition is dubbed giraffe skin disease (GSD).20,33,51 Lesions were first noted on giraffe in Uganda in 1995, 20 with similar GSD-like lesions recognized on the pinna and forelimbs of Masai giraffe in Tanzania, and the forelimbs of Angolan and South African giraffe. 33 A current report of GSD-like lesions identified affected herds in 13 giraffe populations in 7 countries, showing the relatively wide distribution of this condition. 33 A recent report identified a filarid nematode morphologically similar to Stephanofilaria spp. as the cause of the GSD lesion in 7 giraffes from MFNP. 51 Filarid nematodes were also identified within GSD lesions affecting the forelimbs and ears of Masai giraffe in Tanzania.8,27
Thorough understanding of GSD is of paramount interest in maintaining healthy populations of Uganda’s Nubian giraffe. MFNP is under pressure by impending oil exploration and drilling, intense infrastructure development, as well as poaching for bushmeat. It is therefore crucial to reestablish giraffe populations in their former historic ranges throughout Uganda. With a high prevalence of GSD lesions in MFNP giraffe, it is of potential concern to translocate giraffe with GSD to areas with no previous reports of GSD-like lesions in wild hoofstock and domestic livestock. The goal of the current project was to expand our understanding of the underlying cause of GSD in giraffe from MFNP, with clinical and pathologic characterization of GSD, and to determine the impact of GSD on the overall health of these Nubian giraffes.
Materials and Methods
Giraffe and Sample Collection
Sixty-four free-ranging Nubian giraffes in MFNP were immobilized and examined, and biopsies were collected. Subadult (1–5 years of age) giraffes were preferentially selected for immobilization to avoid translocating older or pregnant adult giraffe and there was no bias for giraffe with or without GSD lesions. Adult giraffes in the study (≥6 years of age) were opportunistically rescued from snare traps with similar study specimens collected. Giraffes were identified in sequential order of capture as cases 1 through 64 during the following capture events: August 2017 (cases 1–10), April 2018 (cases 11–28), August 2018 (cases 29–46), and October 2019 (cases 47–64). Study animals included 17 males and 47 females.
Giraffes were immobilized in the field primarily with etorphine hydrochloride (7–15 mg; M99, Wildlife Pharmaceuticals, South Africa), and occasionally in combination with the tranquilizer azaperone (40 mg; Wildlife Pharmaceuticals). All immobilization drugs were administered intramuscularly via dart (Dan-Inject, USA) by Uganda Wildlife Authority veterinarians. Giraffes were brought into lateral recumbency with ropes to reduce induction time and aid in quick reversal administration. Once giraffes were in lateral recumbency, the etorphine hydrochloride was immediately reversed with either diprenorphine hydrochloride (administered at 2× the mg dosage of opioid used as a partial antagonist; M5050, Wildlife Pharmaceuticals) or naltrexone hydrochloride (10–30× mg of opioid used as a full antagonist; Wildlife Pharmaceuticals). Reversal drugs were administered intravenously in the jugular vein, and the giraffes were manually restrained in lateral recumbency for examination and sample collection. Physical examination included gross characterization of GSD lesions with location and size of the lesions recorded on the exposed lateral aspect of the body. Skin biopsies and jugular venous blood were collected from all giraffes. Blood was divided into EDTA (ethylenediamene tetraacetic acid) tubes for complete blood cell count (CBC) and serum separator tubes for biochemistry analysis. The giraffes were closely monitored throughout the procedure with continuous calculation of respiration rate and depth, heart rate, and rectal temperature every 5 minutes. Blood gas analysis was measured patient side on cases 11–46 to further monitor patient stability, with all procedures lasting 15–20 minutes. While restrained, giraffes were given either ivermectin (0.1–0.2 mg/kg subcutanesous; cases 1–10; Noromectin®; Norbrook Laboratories Limited, Northern Ireland), eprinomectin (1.0–1.5 mg/kg subcutanesous; cases 11–20, 29–64; Long-Range, Merial, USA), or no treatment (untreated controls, cases 20–28). Global positioning system (GPS) satellite ossi-units were mounted to 1 ossicone on select giraffes from each capture to monitor spatial movement, posttranslocation movements, and health. Select giraffes were loaded into a field recovery chariot and transported to a temporary boma before subsequent loading onto a truck for translocation to new habitats. Five of the study giraffes were found and released from snare traps and were opportunistically sampled at that time (cases 31, 38, 46, 48, 59), with handling as described previously.
An 8-mm punch biopsy instrument (Integra Miltex®, Japan) was used for skin biopsies. Grossly normal skin punch biopsies were obtained from all 64 Nubian giraffes, from either the neck, shoulders, or pinna. When present, GSD lesions were biopsied at both the border and center of the lesion. Biopsies were hemisected with half placed in 5 ml of 10% formalin for histopathology and half placed in a storage reagent for freezing, transport, and molecular work (RNAlater® Sigma-Aldrich, USA, or PrimeStore Molecular Transport Medium® [MTM], Longhorn Vaccines and Diagnostics LLC, USA). Specimens in storage reagent were chilled on ice before freezing at −80°C. Formalin-fixed specimens at ambient temperature and molecular specimens in medium frozen on dry ice were exported using certified permits to Plum Island Animal Disease Center, USA, for infectious disease screening, and then to the Diagnostic Medicine Center at Colorado State University, USA, with shipment occurring over 2–6 months’ time following collection. GSD and normal skin biopsies from cases 47 to 64 could not be exported from Uganda during the writing of this manuscript, due to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic.
CBC (cases 11–46), blood gas (cases 1–28, 47–64), and serum biochemistry (cases 1–28) were analyzed on subsets of giraffe depending on equipment and handler availability. Jugular blood was collected into EDTA tubes or lithium heparin tubes and serum separator tubes, respectively, using standard procedures. CBCs were performed manually in the field at the end of each capture day while blood chemistries were evaluated with either CG8+ iStat cartridges (Abbott, USA) or a portable Abaxis analyzer (Abaxis, Inc., USA).
All research was approved by the Uganda Wildlife Authority and conducted collaboratively under a Memorandum of Understanding between the Giraffe Conservation Foundation and the Uganda Wildlife Authority. Animal care and use for this study (anesthesia, capture, sampling, and release) was performed and overseen by the Uganda Wildlife Authority veterinary department in accordance with their official protocols and guidelines.
Histopathology
Formalin-fixed skin biopsies from cases 1 to 46 were processed using standard methods, sectioned at 5 µm, and stained with hematoxylin and eosin. Additional special stains were performed on representative biopsies to further examine areas of inflammation and included Fite’s acid-fast, 43 Grocott’s methenamine silver (GMS), 11 and Brown and Brenn Gram stains. 26 Slides were interpreted by light microscopy, and lesions were separated into 3 categories based on chronicity and parasite burden: early (mostly haired skin with hyperkeratosis, dermatitis, and low parasite burden), intermediary (chronic hyperkeratosis with extensive inflammation and abundant parasites), and dormant (end-stage lesions with remnant inflammation and microfilariae).
Molecular Diagnostics
Skin biopsies were preserved in RNAlater® and PrimeStore MTM®. Representative biopsies from each capture with the highest numbers of parasites were processed by maceration and proteinase K digestion. DNA was extracted from the samples using a commercial kit (Qiagen DNeasy Blood and Tissue Kit; Qiagen, USA) according to the manufacturer’s instructions. DNA was additionally extracted from a formalin-fixed and paraffin-embedded skin biopsy derived from a North American domestic cow with confirmed stephanofilariasis using similar technique. Parallel sequencing was performed using the Oxford Nanopore MinION with the 9.4.1 flow cell and ligation kit (SQK-106). Reads were base-called using Guppy 3.4.5 using the computer processing unit (CPU) algorithm. Reads were filtered based on quality score using Nanofilt 2.7.1 and classified using Centrifuge 1.0.4 and the National Center for Biotechnology Information (NCBI) nucleotide nonredundant sequences database (3 March 2018). Reads of interest were extracted and analyzed by Basic Local Alignment Search Tool (BLAST) using the discontinuous megablast against the NCBI nucleotide collection to confirm their similarity to known organisms.
Results
Clinical Findings
The summary and raw data from 64 Nubian giraffes from MFNP, Uganda, that were captured in the “short rains” seasons of August 2017, August 2018, and October 2019, and the “long rains” season of April 2018, are presented in Supplemental Tables S1 and S2. All giraffes were visually determined to be healthy with no abnormal physical examination findings other than GSD lesions. Giraffe had normal and stable vital parameters during handling, and blood gas values were considered acceptable for field capture of giraffe.
Of 64 giraffes, 55 (86%) had grossly evident GSD lesions on the exposed lateral aspect while in lateral recumbency, with 1–6 lesions per affected giraffe. Location of lesions included the neck, cranial aspect of the chest, axilla, and shoulders. There were no lesions detected on the head and ears, dorsum, lateral or ventral abdomen, flanks, hips, or limbs. The size of the lesions varied from 1 to 23 cm diameter. Gross GSD lesions had a spectrum of progression categorized as early, intermediary, or dormant based on diameter, presence of hair, amount of scar tissue, and contraction of the scar tissue. Specifically, early lesions (n = 8 cases) were 1–5 cm diameter, raised, firm, and edematous nodules, which were haired with oozing serum crusts (Fig. 1). Intermediary lesions (n = 53 cases) varied from 2 to 23 cm diameter and were hairless crusted plaques of fissured hyperkeratosis, approximately 1.5 cm thick, often oozing serum, and overlying a dense bed of granulation tissue (Figs. 2, 3). Intermediary lesions progressed in some cases to dormant lesions that were hairless and depigmented. Dormant lesions (n = 15 cases) were similarly sized, 2–16 cm in diameter, and consisted of dry, contracted granulation tissue with ragged and flaking hyperkeratosis (Fig. 4). Adjacent skin was grossly normal, and all categories of lesions involved both pigmented and nonpigmented skin. Large numbers of flies were feeding on all phases of the lesions, with high numbers of flies most notable during the “long rains” seasons of April through June. Affected giraffe in all seasons exhibited intermediary lesions with a combination of early and/or dormant lesions.
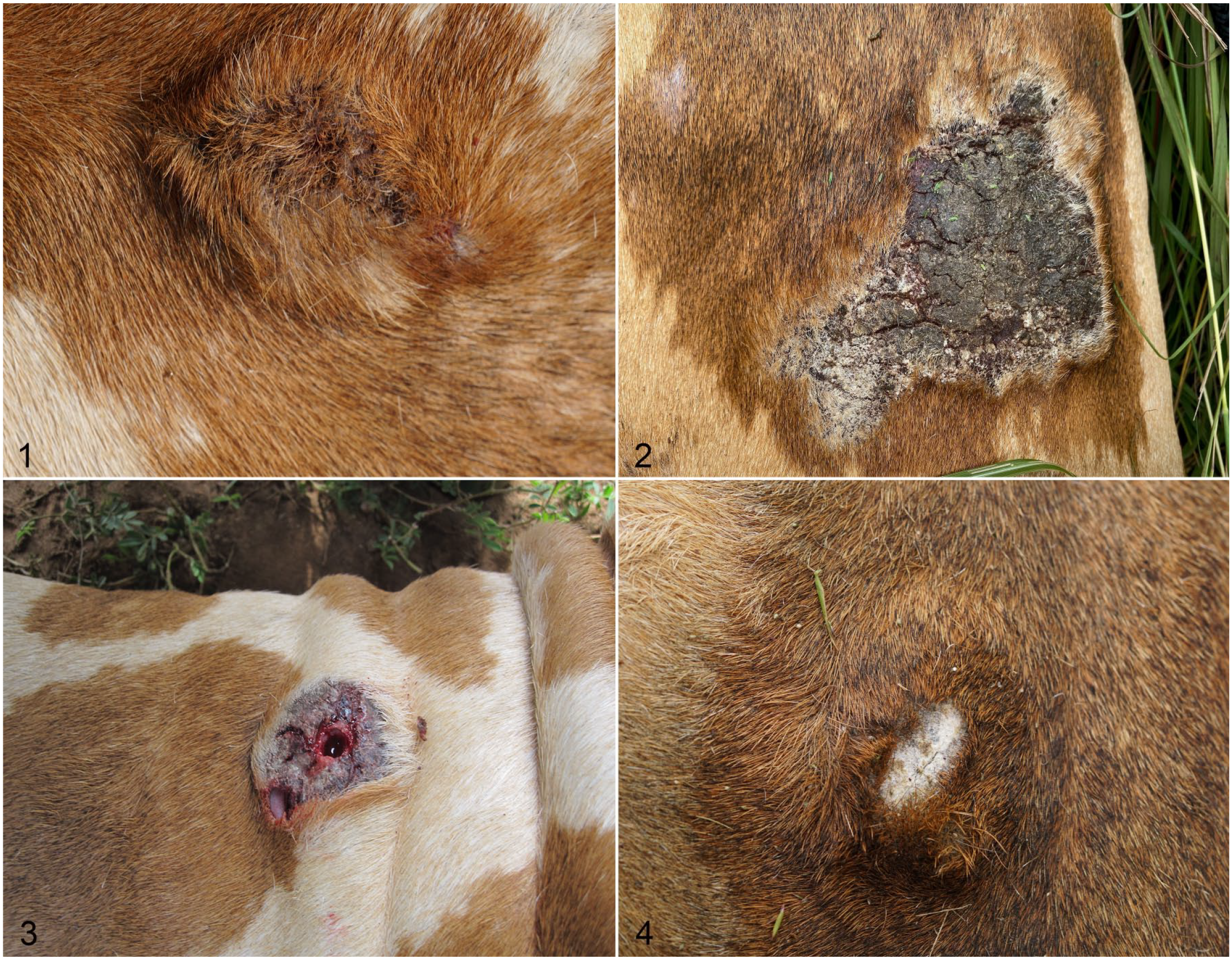
Giraffe skin disease (GSD), skin, Nubian giraffe.
CBCs were performed on cases 1–28. Of these, 4/28 giraffes had elevated total white blood cell counts that ranged from 25.75 to 69.25 cells × 109/L. White blood cell counts of other giraffes were less than 17.75 cells × 109/L (total leukocyte count reference range = 12.6 ± 4.8 × 109/L). 46 Leukocytosis consisted of neutrophilia and lymphocytosis. Eosinophilia was common in giraffes with leukocytosis, with 12 giraffes showing eosinophilia of 0.96–5.04 × 109/L (eosinophil reference range = 0.40 ± 0.40 × 109/L) 46 (Supplemental Table S1). Biochemistry panels were performed on this same subset of giraffes (cases 1–28), with no underlying abnormalities detected (Supplemental Table S2). 39
Histopathology
Histologic lesions of GSD were categorized as early, intermediary, and dormant. Early nodular lesions (n = 8 cases) had epidermal acanthosis with erosions, long and irregular rete ridges, compact orthokeratotic hyperkeratosis, and corneal pustules with serocellular crusts. The epidermal-dermal interface was thickened with fibrosis and abundant eosinophilic, granulomatous, and lymphoplasmacytic inflammation, which extended along adnexa, occasionally effacing hair follicles with eosinophilic furunculosis (Fig. 5). Interspersed throughout the superficial dermis were numerous clear vacuoles, 30–80 µm diameter, that contained eosinophilic globular debris (vitelline membranes) and portions of distinct microfilariae (Fig. 5). Microfilariae were 5–8 µm diameter, with tapered ends and containing several 1–2 µm basophilic nuclei (Fig. 5, inset). Microfilariae varied from 46 to 87 µm in length (average ± SD of 63 ± 16 µm). Early lesions sometimes included small numbers of stage L3 nematode larvae that burrowed along follicular infundibula, with smaller numbers of microfilariae. Stage L3 larvae were 50-µm diameter, with a thin 1-µm cuticle, coelomyarian musculature, with prominent hypodermal lateral cords, and a pseudocoelom containing a digestive tract lined by cuboidal epithelial cells and no reproductive tract (Fig. 6).
All of the giraffes with GSD lesions in this study (n = 55 cases) had intermediary lesions, characterized by erosions, severe proliferative acanthosis, and ragged orthokeratotic hyperkeratosis, with corneal pustules and serocellular crusts, overlying a dense bed of organized granulation tissue that replaced the dermis, with dropout of hair follicles. Granulation tissue consisted of severe eosinophilic and granulomatous, necrotizing inflammation with myriad superficial encysted microfilariae and frequent stage L3 larvae (Fig. 7). In several intermediary lesions, there were eosinophilic granulomas at the base of hair follicles that contained adult male and female nematodes (Figs. 7–9). Adult nematodes varied from 75 to 100 µm diameter, with a 5-µm-thick cuticle, polymyarian-coelomyarian musculature, with prominent vacuolated lateral cords. The central pseudocoelom contained a digestive tract lined by cuboidal epithelial cells with a brush border, granular eosinophilic cytoplasm, and small round nuclei. The reproductive tract of male nematodes included testes consisting of numerous small basophilic nuclei. Female nematodes had paired uteri containing abundant eosinophilic disc-shaped material (vitelline membranes) encasing numerous small developing microfilariae (Figs. 7, 8). Histomorphologic features were consistent with a filarid nematode closely resembling Stephanofilaria stilesi as described in domestic cattle.5,14,25 Granulomas and adult nematodes were surrounded by dense fibrosis and lymphoplasmacytic inflammation. Biopsies from the center of lesions tended to have chronic organized dermal fibrosis with most adult nematodes and microfilariae clustered at the periphery of the lesions.
Dormant GSD lesions (n = 15 cases) consisted of severe hyperkeratosis and acanthosis with few corneal pustules. Dermatitis was resolving and superficial with no granulomas, and the dermis was largely replaced by organized granulation tissue with dropout of hair follicles (Fig. 10). Biopsies from the center of dormant GSD lesions consisted predominantly of scar tissue with mixed eosinophilic and granulomatous inflammation, with microfilariae most commonly encysted at the periphery of dormant lesions. No adults or stage L3 larvae were identified in these resolving lesions.
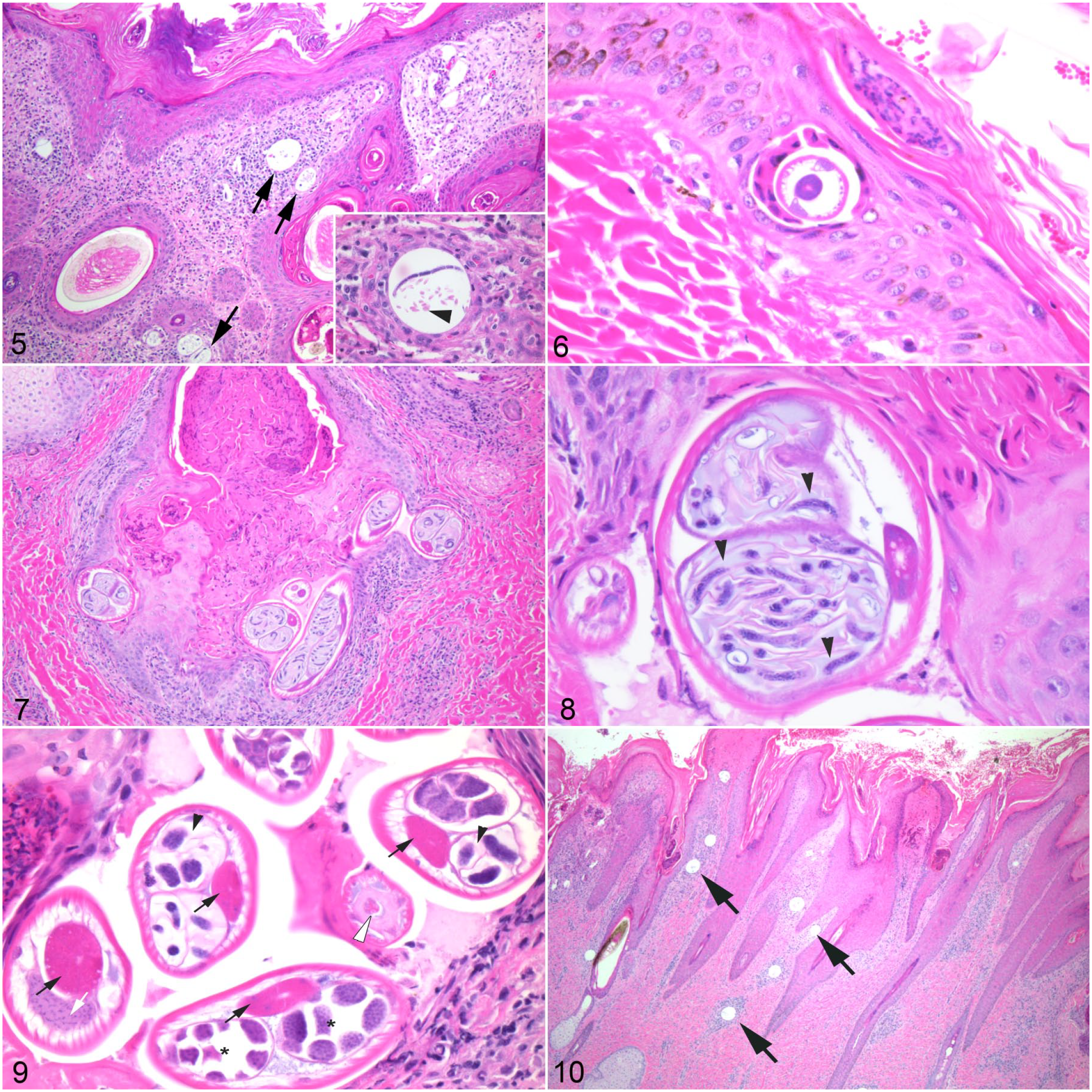
Giraffe skin disease (GSD), skin, Nubian giraffe.
Representative lesions from each stage were examined with Gram, silver, and acid-fast stains, and no additional microorganisms were identified. Grossly normal skin biopsies from the neck, shoulders, and pinna were histologically normal, and none contained parasitic lesions.
Molecular Diagnostics
Because 18S and cox-1 sequences were consistent with mammalian sequences (data not shown), nonparallel sequencing was performed. Macerated fresh skin biopsies from select intermediary GSD lesions were sequenced using Oxford Nanopore Technologies MinION; 4592 reads with q score of 18 or greater were classified using Centrifuge database NCBI nucleotide nonredundant sequences database (3 March 2018). Of 1052 sequences uniquely identified, 14 were classified as filarids, including the Loa, Wuchereria, and Brugia genus; Brugia was the most frequent with 6 sequences. Twelve of the 14 sequences were unique; by BLAST (discontinuous megablast), these were most closely related to Brugia malayi (NCBI: txid6279) (92.8% identity and e value of 4.06 e−68) and Loa loa (NCBI: txid7209) (86.0% and e value of 7.84 e−71), both of which are causes of filariasis in humans. B. malayi and L. loa are closely related filarid nematodes presently in GenBank. This unique GSD sequence was deposited in GenBank as a putative partial sequence (NCBI: OL598359). S. stilesi is not currently in GenBank. As an experimental comparison, DNA sequence was derived from a skin biopsy from a domestic North American cow with grossly and histologically confirmed stephanofilariasis. A short 403 base pair (bp) sequence of S. stilesi did not overlap with the GSD sequence but did include a 100 bp sequence with 100% identity to a highly conserved sequence seen in filarid nematodes currently in GenBank. Like the GSD sequence, S. stilesi was most closely related to Brugia sp. and Wuchereria sp. This information confirms GSD nematodes as closely related to other pathologic filarid nematodes and indicates that further genetic characterization is required, with work currently in progress.
Discussion
Critically endangered Nubian giraffes from MFNP, Uganda, 32 have proliferative crusted and hyperkeratotic skin lesions of the cranial body, termed GSD and recognized as an important emerging disease. The study giraffe had an 86% prevalence of GSD with no significant clinical abnormalities, though low numbers of study giraffe exhibited leukocytosis and eosinophilia. These findings suggest that GSD is relatively well tolerated by the giraffe in this population. This study confirms that GSD in the Nubian giraffe is caused by an invasive filarid nematode morphologically similar to S. stilesi as described in domestic cattle and transmitted by a biting insect vector. A novel finding of this study was that gross lesions of GSD started as nodular, haired lesions, with characteristic eosinophilic and granulomatous dermatitis with microfilaria and stage L3 larvae. Intermediary lesions were most common and occurred in all giraffes in the study population from MFNP. Intermediary lesions were larger, hairless plaques of thick, fissured hyperkeratosis, proliferative acanthosis, and intense eosinophilic and granulomatous dermatitis, with dermal granulation tissue and myriad encysted microfilariae, including migrating stage L3 larvae and adult nematodes encysted within eosinophilic granulomas, similar to that described in established stephanofilariasis lesions in cattle. 25 Histopathology of early and intermediary lesions overlapped, though grossly the lesions were distinct. GSD appears to resolve to dormant lesions characterized grossly and histologically as hairless and depigmented scar tissue, with chronic hyperkeratosis, acanthosis, and remnant dermatitis with peripherally encysted microfilariae, which lacked adult or stage L3 larvae, similar to chronic resolving stephanofilariasis described in adult cattle. 25 GSD lesions were grossly and histologically similar to that previously published on an initial subset of Nubian giraffe from MFNP. 51 Establishing the gross and histological stages of GSD is important to better understand and recognize the pathogenesis and progression of GSD, identify the potential spread of GSD to new areas or novel hoofstock, and assist in monitoring long-term health, survival, and response to possible future therapy of individual giraffe in the field.
To date, the Stephanofilaria genus, like many other parasites, has been exclusively described morphologically, and there are currently no published genetic data or sequence information in GenBank, hindering molecular advancement. In addition, overlap of nematode sequences with mammalian host sequences can complicate interpretation of findings, requiring pure nematode material for molecular techniques. Molecular characterization of the GSD nematode was attempted utilizing MinION technology. We compared sequences derived from fresh-frozen macerated GSD biopsies with sequences derived from a formalin-fixed and paraffin-embedded skin lesion from a North American cow with grossly and histologically diagnosed stephanofilariasis. Both specimens yielded nonoverlapping sequences such that specimens could not be directly compared. GSD sequences had 92.8% identity to B. malayi and 86% identity to L. loa, both of which cause filariasis in humans. The bovine stephanofilariasis sequence had regions of 100% identity to Brugia sp. and additionally to Wuchereria sp., also a cause of filariasis in humans. These limited molecular data correlate with initial published efforts at molecular identification of the MFNP nematode derived from GSD lesions, which described a 92% identity to the filarid parasite Onchocerca. 51 These cumulative findings confirm that the nematode causing GSD is a unique filarid nematode, closely related to S. stilesi and other agents of pathogenic filariasis in humans and animals. Molecular diagnostics in this study were limited by the inability to extract nematodes from imported fresh-frozen GSD biopsies, resulting in a shorter nematode sequence of poorer quality. Purification of nematodes from fresh GSD lesions is currently underway.
GSD causes proliferative and crusted skin lesions affecting multiple body regions of giraffe and has been recognized as an emerging disease in giraffe for nearly 25 years, affecting populations in Tanzania, Uganda, Kenya, South Africa, Namibia, Botswana, and Zimbabwe.8,20,24,27,30,33,51 Characteristics of GSD vary by region and giraffe species. For instance, Masai giraffes in Tanzania’s Ruaha National Park were reported to have severe proliferative and crusted lesions centered on the caudal and medial aspects of the carpi, with severe lesions being associated with poor body condition and significant lameness.8,24 GSD of the pinna has also been described in giraffe from Ruaha National Park, a condition named giraffe ear disease (GED), which is histologically similar to GSD and has also been associated with filarid nematodes. 27 GED can be progressive with sloughing of the pinna and ill thrift in affected giraffe. Lesions of the pinna and carpi are most prevalent in the wet seasons, first emerging in 1999 following heavy El Niño rains of 1997–1998. 27 GED is associated with myiasis, particularly house flies (Musca domestica) and face flies (Musca sorbens), which are presumed to be important vectors of the filarid nematode. Molecular identification of the nematode isolated from the lesion was attempted with cox-1 and 12S rDNA primers, which resulted in sequence with strongest identity to Onchocerca spp. and Dirofilaria spp., supportive of a primary pathogenic filarid nematode. GED was also associated with ticks of the genus Rhipicephalus appendiculatus and Amblyoma variegatum and oxpecker birds (Buphagus spp.), which may have contributed significantly to chronic trauma and progression of the lesions. 27
Although the etiological agents associated with GSD and GED are relatively poorly understood, similar Stephanofilaria spp. have been widely described in other taxa. Stephanofilariasis was first described as crusted skin lesions in North American beef cattle in 1934, with the parasite named S. stilesi. 5 Lesions in cattle are chronic midventral hyperkeratotic dermatitis lesions, 25 affecting up to 90% of animals in endemic regions. 28 Filarid dermatitis was soon recognized as a worldwide disease of cattle, with similar lesions described on the midventrum in beef cattle in the Soviet Union; 17 the head, neck, and sternum of Bos indicus cattle in Australia; 19 and the scrotum of Charolais bulls in France, 49 all caused by the same nematode. A stephanofilarid nematode was reported as the cause of crusted hyperkeratosis and dermatitis lesions occurring on the withers (“hump-sore”) in zebu cattle (Bos taurus indicus) in Pakistan and India. This nematode had distinct morphological differences from S. stilesi, and was recognized as a novel species, Stephanofilaria assamensis, 36 and was later found to cause skin lesions in a wide range of hoofstock throughout India.1,37,44,45 Stephanofilariasis is now recognized on every major continent as a cause of dermatitis that is often clinically significant and affects not only cattle but other livestock such as pigs and goats as well as elephants. 1 Studies have since recognized additional species including Stephanofilaria kaeli in cattle and goats in Malaysia, 9 and Stephanofilaria okinawaensis causing sores of the teats and muzzle in Japanese black cattle.12,23
Lesions of stephanofilariasis in cattle generally begin at less than 1 year of age, progressing as active but generally nondebilitating lesions until dormancy at approximately 11 years of age.7,14,28 Variation in stephanofilariasis lesion distribution is thought to be due to specific fly vectors, environmental conditions (heat, sunlight, humidity, access to manure, etc), and favorability of the skin for myiasis in different species.1,13,42 S. stilesi in cattle of North America, Europe, Australia, and Russia are transmitted via an intermediate horn fly (Haematobia irritans) vector,7,13,17,42 while Asian hump-sore lesions are associated with Musca conducens.41,45 Vectors feed on crusted lesions, ingesting microfilariae. Microfilariae molt twice within the hemocoel of the vector, migrating as stage L3 larvae to the vectors’ salivary glands by days 18–21. During feeding, larvae pass in the saliva to infect wounds of cattle, burrowing along hair shafts and encysting near the hair bulb, where they molt to adult stage L4 and L5 nematodes with full development of gonads. Adult S. stilesi produce microfilariae circumscribed by vitelline membranes, a unique characteristic in this genus. Microfilariae migrate to the superficial dermis and encyst within open sores, remaining dormant until consumed by feeding intermediate host flies. 13
Stephanofilariasis in Africa was first described in the black rhinoceros (Diceros bicornis) in Kenya and South Africa, recognized as thick encrusted skin lesions of the lateral thorax and extending along the cranial chest, erupting during the wet seasons concurrent with highest vector concentrations, and resolving to dormancy during the dry cool seasons. Filarid nematodes were identified histologically as the cause of these recurrent and seasonal lesions. The filarid nematode was identified as morphologically distinct from Stephanofilaria of cattle and was named Stephanofilaria dinniki.15,38,40,47 Severe and progressive lesions have since been reported in black rhinoceros in Zimbabwe 22 and black and white rhinoceros (Ceratotherium simum) in Kenya. 34 Lesions were most severe in rhinoceros with poor body condition and increased stress from crowding, livestock encroachment, or poor nutrition. This suggests that lesion progression may be associated with systemic stress and immune suppression in rhinoceros. 22 The Eastern black rhinoceros of Uganda was anecdotally described as having cutaneous filariasis, though this species became extirpated within Uganda in 1983. Cutaneous filariasis of the shoulder was described in a Nile hippopotamus (Hippopotamus amphibius) in Kruger National Park, South Africa. The nematode had slight morphological differences, and the parasite was provisionally named Stephanofilaria thelazoides. 4 Stephanofilaria spp. lesions have not been described in African domestic hoofstock with the exception of an outbreak of skin disease on the legs, pinna, and teats of domestic pigs in the former Zaire (current Democratic Republic of the Congo). Lesions were attributed to filarid nematodes and were most similar to but distinct from S. dinniki and S. thelazoides. 2 Microfilariae in GSD of Nubian giraffes appear morphologically distinct from S. dinniki based on the relative length of microfilariae, being 46–87 µm in GSD compared with 120–150 µm in rhinoceros. 18
Other proliferative skin diseases in giraffe and wild hoofstock in Africa include papillomatosis and lumpy skin disease. Papillomatosis affects the muzzle and nares in giraffe and the coronary bands of impala, presenting as small, raised nodules of epidermal hyperplasia and ulceration, caused by a novel deltapapillomavirus, G. camelopardalis papillomavirus-1.21,48 Lumpy skin disease, caused by a capripoxvirus, is a nodular and proliferative, debilitating skin disease of cattle, first described in Zambia in 1929. 29 The disease is endemic in Sub-Saharan Africa and is spread by biting insect vectors Stomoxys calcitrans and Musca confiscata,16,50 with only experimentally induced infection noted in impala and giraffe. 52 Importantly, these diseases were not detected grossly or histologically in any of the MFNP Nubian giraffe.
The overall health impact of GSD on giraffe in MFNP appears to be limited, as giraffes in this study were in good body condition with no evidence of more advanced systemic disease. This concurs with preliminary analysis of ongoing field studies, which suggest that GSD lesions do not significantly impair giraffe survival, though this is a key topic of ongoing studies.3,33
Identification of novel therapies for GSD is additionally ongoing, and long-acting anthelminthics are of interest for experimental treatment. Connected to this study, several Nubian giraffes from MFNP were translocated to either the south bank of MFNP, Kidepo Valley National Park in North Eastern Uganda, or Pian Upe Wildlife Reserve in eastern Uganda on the border with Kenya as part of the national conservation effort for this species. GSD has not been identified in giraffe, wild hoofstock, or livestock around Kidepo Valley National Park or Pian Upe Wildlife Reserve, and discussions were held around the potential for transmission of a novel disease into a naïve region. Select translocated giraffes were treated with varying dosages of ivermectin or long-acting topical eprinomectin and are to be monitored using GPS satellite ossi-units and photographic surveys over the coming years. Monitoring for GSD-like lesions in other wild ruminants and domestic cattle in the host and release areas is proposed to better understand the breadth and distribution of GSD in Uganda. A detailed tolerance and pharmacokinetics study of eprinomectin in captive giraffe has been initiated, as well as morphological identification of fly vectors associated with GSD lesions in Nubian giraffe. It is hopeful that the results of these studies will advise future conservation management of Nubian giraffe in MFNP, and in the regions within Uganda in which this critical population have been translocated.
Conclusion
GSD is a recently emerging disease in wild giraffe, including Uganda’s Nubian giraffe. Proliferative dermatitis lesions were generally active and progressive but were not associated with significant systemic disease or debilitation. GSD is caused by a filarid nematode morphologically similar to Stephanofilaria spp. and transmitted by biting flies. GSD in Nubian giraffe had gross and histological similarities to severe skin disease described on the pinna (GED) and limbs of Masai giraffe in Tanzania, the black rhinoceros of Zimbabwe, and the black and white rhinoceros of Kenya.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221082606 – Supplemental material for Giraffe skin disease: Clinicopathologic characterization of cutaneous filariasis in the critically endangered Nubian giraffe (Giraffa camelopardalis camelopardalis)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221082606 for Giraffe skin disease: Clinicopathologic characterization of cutaneous filariasis in the critically endangered Nubian giraffe (Giraffa camelopardalis camelopardalis) by Sushan Han, Liza Dadone, Sara Ferguson, Priya Bapodra-Villaverde, Patricia M. Dennis, Robert Aruho, Miranda J. Sadar, Julian Fennessy, Margaret Driciru, Arthur B. Muneza, Michael B. Brown, Matthew Johnston and Kevin Lahmers in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858221082606 – Supplemental material for Giraffe skin disease: Clinicopathologic characterization of cutaneous filariasis in the critically endangered Nubian giraffe (Giraffa camelopardalis camelopardalis)
Supplemental material, sj-xlsx-1-vet-10.1177_03009858221082606 for Giraffe skin disease: Clinicopathologic characterization of cutaneous filariasis in the critically endangered Nubian giraffe (Giraffa camelopardalis camelopardalis) by Sushan Han, Liza Dadone, Sara Ferguson, Priya Bapodra-Villaverde, Patricia M. Dennis, Robert Aruho, Miranda J. Sadar, Julian Fennessy, Margaret Driciru, Arthur B. Muneza, Michael B. Brown, Matthew Johnston and Kevin Lahmers in Veterinary Pathology
Footnotes
Acknowledgements
We would like to sincerely thank the Uganda Wildlife Authority and Giraffe Conservation Foundation through which the research was conducted, and permits obtained. Also we thank the many supporters of Operation Twiga II, III, & IV through the Giraffe Conservation Foundation and the Metzger Foundation, Ivan Carter Wildlife Conservation Alliance, Cheyenne Mountain Zoo, Cleveland Metroparks Zoo, Columbus Zoo and Aquarium, Mrs. Rae Memorial Fund, and World Giraffe Day 2018.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Association of Zoos and Aquariums Conservation Grants Fund/Disney Conservation Fund 2018.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
