Abstract
Chlamydia pneumoniae is a ubiquitous pathogen causing disease in humans, mammals, birds, reptiles, and amphibians. Since 2012, C. pneumoniae infection has caused neurologic disease and mortality in a breeding colony of endangered Houston toads (Anaxyrus houstonensis) at the Houston Zoo. The purpose of this report is to present the histopathologic and ultrastructural characteristics of C. pneumoniae infection in Houston toads. Fourteen cases were evaluated by histopathology and 1 case was evaluated by electron microscopy. The major histopathologic finding was necrotizing and histiocytic polioencephalomyelitis and ganglionitis. Bacteria formed intracytoplasmic inclusions within neurons but frequently extended into the surrounding tissue from necrotic cells. Ultrastructural evaluation showed the bacteria formed reticulate and elementary bodies characteristic of Chlamydia spp.
Chlamydia spp cause disease in humans, mammals, birds, reptiles, and amphibians. 1 –4,8,12,13,17 Chlamydia pneumoniae (previously Chlamydophila pneumoniae) was originally discovered as a cause of respiratory infection in humans and has since been isolated from horses, koalas, reptiles, and amphibians. 2,13 Reports in animals include nonpathologic infections in the respiratory tracts of horses, pulmonary and systemic disease in reptiles, and renal and systemic disease in amphibians. 1 –3,8,11,13,16,17 C. pneumoniae–induced encephalomyelitis has not been reported in veterinary species, but a case of meningitis in a royal python (Python regius) and encephalitis secondary to respiratory infection in humans have been reported. 7,13,18
The Houston toad (Anaxyrus houstonensis, family Bufonidae) is endemic to the central coast region of Texas. Houston toads are currently on the International Union for Conservation of Nature (IUCN) endangered species list due to loss of habitat, deaths from periodic droughts, and predation from the red imported fire ant (Solenopsis invicta). 9,19 A collaborative captive breeding program between the Houston Zoo, Texas State University, and the US Fish and Wildlife Service is currently working to bolster this species’ numbers in the wild. A closed population of approximately 400 toads are housed in an indoor facility at the Houston Zoo to maintain a captive assurance colony and produce fertilized eggs for release into suitable habitats.
Beginning in 2012, several toads developed neurologic signs characterized by ataxia, anisocoria, and decreased righting reflex. Affected toads were euthanized and tissues were submitted for histopathology revealing histiocytic inflammation in the central nervous system (CNS) with intracytoplasmic, basophilic inclusions. Bacterial culture of the spinal cord isolated Chlamydia sp. Swabs of the meninges collected postmortem were submitted for Chlamydia-specific polymerase chain reaction (PCR) at the University of Georgia Infectious Disease Laboratory using prototypical real-time fluorescence resonance energy transfer PCR. 14,20 PCR products from several prior cases were extracted and sequenced, and the amplicons were homologous to C. pneumoniae. 20
Between 2012 and 2015, 175 cases of confirmed or suspected chlamydiosis occurred in the Houston Zoo breeding colony, accounting for 29.3% of all mortalities. Antibiotic therapy was unsuccessful or associated with long-term neurologic deficits. Currently, depopulation of tanks housing infected animals is used to control the disease. The presence of C. pneumoniae infection in free-ranging Houston toads is unknown, and reducing the risk of introducing this disease to native populations is essential. In this report, we describe the histologic lesions and ultrastructural characteristics of C. pneumoniae infection in Houston toads.
Medical and necropsy records from the Houston Zoo and Texas Veterinary Medical Diagnostic Laboratory were reviewed to identify cases previously diagnosed with C. pneumoniae infection. Histologic sections stained with hematoxylin and eosin from 27 cases diagnosed with chlamydiosis were reviewed for the following criteria to be included in the study: (1) inflammation within the CNS, (2) the presence of intracytoplasmic inclusions characteristic of Chlamydia spp., and (3) positive PCR for Chlamydia sp. or presence of Chlamydia sp. organisms on electron microscopy. Histologic sections, including brain, spinal cord, heart, lung, liver, spleen, kidneys, gastrointestinal tract, gonads, and hindlimb, were evaluated for C. pneumoniae infection and any comorbidities. Pierce–van der Kamp modified Gimenez stain was used to highlight Chlamydia inclusions. Where indicated, Twort’s Gram, Grocott’s methenamine silver, and Fite’s acid fast stains were used to evaluate comorbidities. In case No. 14, Luna stain and myeloperoxidase immunohistochemistry were performed to characterize the inflammatory cells within the CNS. 4
Case No. 4 was submitted for electron microscopy. For electron microscopy, 1.0-mm3 tissue blocks from the spinal cord were postfixed in 0.166 M cacodylate buffered, 3% glutaraldehyde solution, and then in 1% osmium tetroxide. Using a graded series of ethyl alcohol, tissue blocks were dehydrated and embedded in Embed (Electron Microscopy Sciences, Hatfield, PA). One-micron, toluidine blue–stained sections were used to visualize areas of interest. Ultrathin sections (60–90 nm) were examined under a JEOL transmission electron microscope (JEOL Ltd., Tokyo, Japan). Dimensions of microorganism structures were taken using iTEM software (Olympus SIS, Munster, Germany).
Fourteen of the 27 examined cases exhibited intracytoplasmic bacteria and inflammation within the CNS (Supplemental Table S1). Twelve cases were excluded due to lack of organisms on histopathology. One case was excluded due to lack of intact sections of brain on histopathology slides. Thirteen of the 14 cases included in the study were positive for Chlamydia sp. on meningeal swab by PCR. The only case negative on PCR was case No. 4, which showed elementary bodies (EBs) and reticulate bodies (RBs) characteristic of Chlamydia sp. on electron microscopy of the CNS. Of the 14 included cases, 8 were female and 6 were male.
The distribution of the lesions ranged from multifocal nodules within the gray matter of the brain and spinal cord in mild infections (3/14) to coalescing areas of inflammation that obliterated the gray matter and extended into the white matter and meninges (11/14). Foci of inflammation were within the cranial nerve and spinal root ganglia in all cases that contained visible ganglia (11/14 and 9/14 cases, respectively). Mild cases were composed of histiocytic nodules with few lymphocytes. Moderate to severe infections were predominantly histiocytic with variable numbers of lymphocytes, plasma cells, and eosinophils (Fig. 1). Luna stain (case No. 14) confirmed eosinophils were among the inflammatory cells. Myeloperoxidase immunohistochemistry (case No. 14) for neutrophils was inconclusive. Clusters of pleiomorphic bacteria moderately to markedly expanded the cytoplasm of neurons and frequently extended into the tissue from necrotic neurons (Fig. 2). Secondary changes included edema (10/14), dilated myelin sheaths (8/14), digestion chambers (6/14), central chromatolysis (5/14), and spheroids (2/14). Secondary changes were more common in moderate to severe cases; however, these were often obscured by the marked inflammatory infiltrate. Case No. 3 showed histiocytic inflammation within the liver, and hepatocytes contained intracytoplasmic Chlamydia inclusions (Fig. 3). Pierce–van der Kamp modified Gimenez stain of the CNS and liver showed positive staining of the intracytoplasmic bacteria (Fig. 4). At the ultrastructural level, numerous 290 × 500 nm EBs and 500 × 750 nm RBs were within the cytoplasm of neurons and free within cell debris (Figs. 5, 6). EBs were round, electron dense, and lined by a double membrane, whereas RBs had a granular cytoplasm with diffuse, fibrillar nucleic acid (Figs. 7, 8).
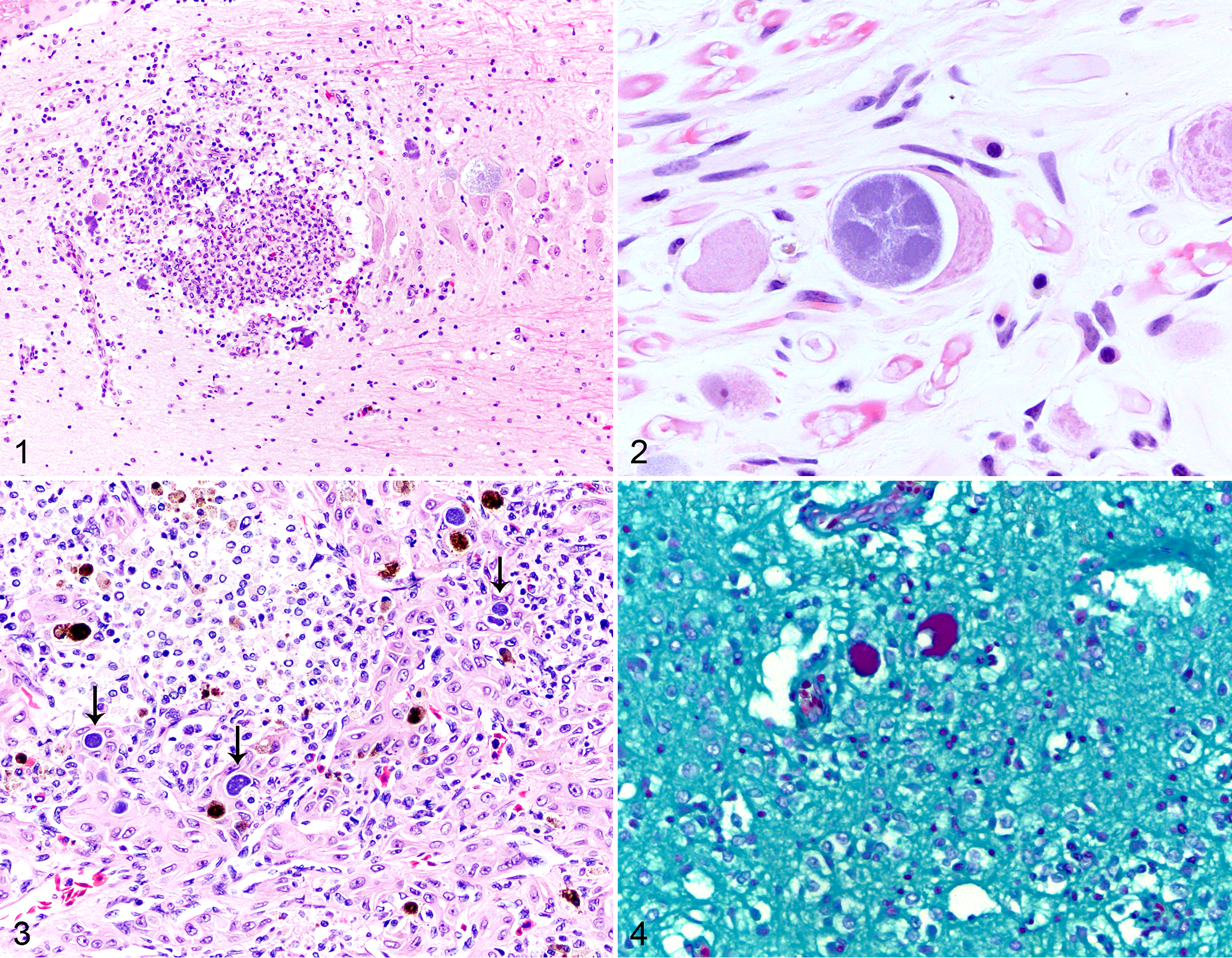
Chlamydia pneumoniae infection, Houston toad.
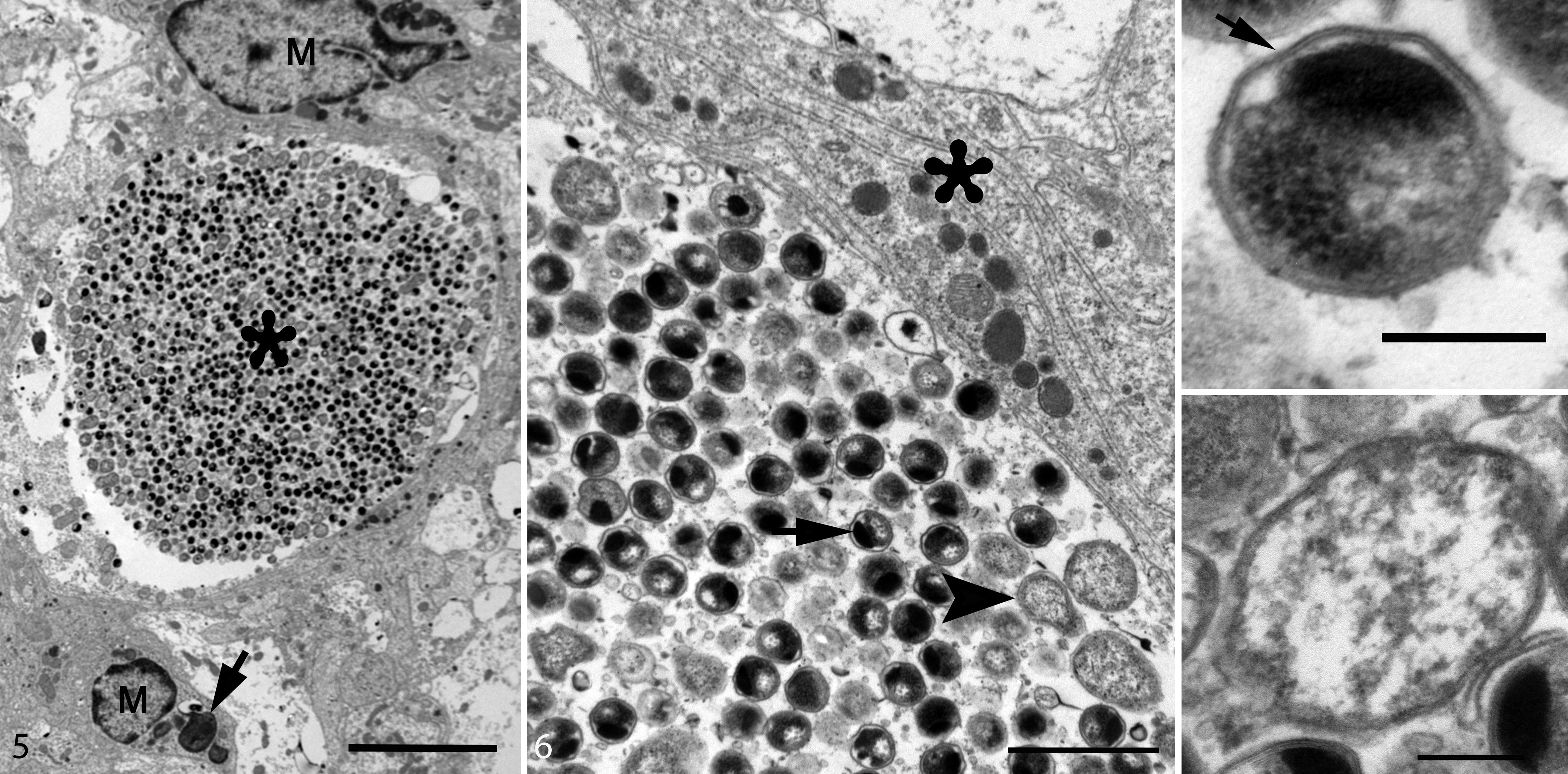
Chlamydia pneumoniae infection, brain, Houston toad. Case 4. Transmission electron microscopy.
Seven of the 14 cases evaluated (case Nos. 3, 4, 6, 8, 9, 10, 14) had 1 or more concurrent lesions in other organs. The most frequent comorbidity was pulmonary chromomycosis (case Nos. 6, 8, 14). Other lesions included peripheral nerve sheath tumor (case No. 4), fungal dermatitis (case No. 6), parasitic dermatitis (case No. 6), cutaneous mycobacteriosis (case No. 9), interstitial nephritis (case No. 10), and endocarditis (case No. 14).
C. pneumoniae causes pulmonary, renal, and systemic disease in many species, including humans. 2,13 Like other Chlamydia sp., C. pneumoniae has a biphasic life cycle composed of an infectious EB for attaching to and entering host cells and a noninfectious RB that is metabolically active and replicates by binary fission. C. pneumoniae is capable of infecting epithelial, endothelial, smooth muscle, and white blood cells such as monocytes and lymphocytes; the latter may facilitate disseminated infections. 6,13 The primary histologic features of C. pneumoniae polioencephalomyelitis in Houston toads were multifocal to coalescing areas of histiocytic inflammation in the gray matter with extension into the surrounding white matter. More severe infections showed mixed inflammation and marked destruction of the neural tissue. Bacterial inclusions were in the cytoplasm of neurons in the brain, spinal cord, and ganglia. Ultrastructurally, the bacterial inclusions were composed of EBs and RBs. 15 This severe inflammation and extensive damage within the CNS explains the neurologic signs observed clinically and the persistent neurologic deficits despite treatment and apparent recovery from overt infection.
C. pneumoniae–associated CNS disease has been reported in a royal python and in several cases in humans. In the royal python, lesions included meningitis and granulomatous enteritis and splenitis, but bacteria were detected only in the spleen. 7 In humans, encephalitis is associated with respiratory infection, but diagnosis is based on C. pneumoniae–specific IgG and IgM in serum or cerebrospinal fluid. 10,18 C. pneumoniae antigen has been detected by PCR and immunohistochemistry in the brains of humans affected by neurodegenerative diseases, including Alzheimer disease and multiple sclerosis. 6
In our study, most cases showed infection only within the CNS, but in 1 of the 14 cases (case No. 3), lesions were also in the liver. Infection of the liver with C. pneumoniae has been reported in several other amphibian and reptile species, including the African clawed frog (Xenopus tropicalis), the puff adder (Bitis arietans), and the flap-necked chameleon (Chamaleo dilepsis), but these cases do not report CNS infection. 8,12,16,17 The presence of C. pneumoniae in both the CNS and the liver suggests a probable hematogenous route of infection in Houston toads, presumably via leukocyte trafficking. 6
Surprisingly, the case submitted for electron microscopy (case No. 4) was negative for Chlamydia sp. on meningeal swab. However, since Chlamydial organisms were evident on ultramicroscopic sections of the CNS lesions, this result is a false negative that may be due to absence of chlamydial DNA in the sample submitted for PCR or improper handling of the swab. Although PCR is a rapid diagnostic test for this infection, results should be corroborated with histopathologic and/or ultrastructural lesions.
Seven of the 14 (50%) cases had concurrent disease in other organs. Whether these lesions occurred independent of or secondary to C. pneumoniae infection is unknown. The most prevalent comorbidity was pulmonary chromomycosis, which is a common cause of dermatitis and systemic infection in amphibians. 5 Other lesions included peripheral nerve sheath tumor, cutaneous mycobacteriosis, parasitic dermatitis, interstitial nephritis, and endocarditis. Further evaluation of the prevalence of these lesions in Houston toads with and without C. pneumoniae infection is needed.
In summary, this report characterized the histologic and electron microscopic lesions of C. pneumoniae infection in Houston toads. The histopathologic findings included multifocal to coalescing, histiocytic polioencephalomyelitis and ganglionitis with intracytoplasmic inclusion bodies. Electron microscopy of the lesions revealed EBs and RBs within the cytoplasm of neurons and free within cellular debris. Although the degree of inflammation varied depending on the severity of infection, foci of inflammation were consistently within the gray matter of the brain and spinal cord and within the ganglia. Further study of C. pneumoniae infection in Houston toads may reveal whether this neurotropic form of infection is due to a characteristic of C. pneumoniae or an inherent susceptibility in this species.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819844818 - Chlamydia pneumoniae Polioencephalomyelitis and Ganglionitis in Captive Houston Toads (Anaxyrus houstonensis)
Supplemental Material, DS1_VET_10.1177_0300985819844818 for Chlamydia pneumoniae Polioencephalomyelitis and Ganglionitis in Captive Houston Toads (Anaxyrus houstonensis) by Alycia Fratzke, Lauren L. Howard, Maryanne E. Tocidlowski, Anibal Armién, Fabiano Oliveira, Branson Ritchie, Erin Berlin and Eric Snook in Veterinary Pathology
Footnotes
Acknowledgements
We thank Dr. Allan Pessier of Washington Animal Disease Diagnostic Laboratory for initial molecular identification of the Chlamydia spp. isolates, the Houston Zoo Veterinary Department and Herpetology Department for assistance in sample collection, and the Dean Muldoon of Minnesota Veterinary Diagnostic Laboratory for the electron microscopy preparations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
