Abstract
Aleutian mink disease virus is the type species in the genus Amdoparvovirus, and in mink and other Mustelidae can cause either subclinical disease or fatal chronic immune stimulation and immune complex disease. The authors describe a novel amdoparvovirus in the endangered red panda (Ailurus fulgens), discovered using viral metagenomics. The authors analyzed the prevalence, tissue distribution, and disease association by PCR, in situ hybridization, electron microscopy, and histology in a group of 6 red pandas from a single zoological collection. The study incorporates a fecal shedding survey and analysis of tissues from 4 necropsied animals over a 12-year span. The tentatively named red panda amdoparvovirus (RpAPV) was detected in the feces and/or tissues of all animals tested. At necropsy of 1 geriatric animal, infection was associated with pyogranulomatous peritonitis, pancreatitis, and myocarditis. Other animals had detectable low-level viral nucleic acid in lymph nodes and both oral and intestinal epithelium at the time of necropsy. Full-length genome sequences of RpAPV strains from 2 animals had 12% sequence divergence, demonstrating genetic diversity even among in-contact animals. RpAPV is a persistent infection in this cohort of red pandas, and has variable clinical expression.
Keywords
Amdoparvovirus is 1 of 8 genera currently recognized within the Parvoviridae. 11 Amdoparvoviruses are among the smallest known viruses, with a single-stranded DNA genome of ∼5 kb. The prototype virus, Aleutian mink disease virus (AMDV), is the causative agent of Aleutian disease, first described in the 1950s in commercial mink. 17 The species name, Carnivore amdoparvovirus 1, reflects an extensive host range, with natural or experimental infections observed in other mustelids (ferrets, badgers, weasels, otters) and many small carnivores (eg, skunk, bobcat, dog, and cat). 1,2,8,9,14,18,21,24,26,28,30,32 AMDV was the sole member of the genus until recent discoveries of distinct amdoparvoviruses in gray fox, raccoon dog and Arctic fox, red fox, and skunks (Carnivore amdoparvoviruses 2, 3, 4, and 5, respectively). 5,7,23,37 In mink, the most typical outcome of AMDV infection is subclinical disease, but a systemic disease syndrome characterized by chronic plasmacytosis, immune complex deposition, and progressive wasting is well-recognized. 4 The sequelae of amdoparvovirus infection in other animals are less well-characterized, but collective studies suggest that, like AMDV in mink, outcomes of infection can range from innocuous to fatal.
Recent studies using sensitive molecular detection methods have demonstrated that long-term viral persistence is an important clinical outcome of some parvovirus infections, including AMDV and the human parvovirus B19. 4,19,25,27,33,34 There are multiple potential outcomes of persistent infections. These include innocuous infection, reactivation and subsequent disease, and/or reactivation of infection in (rapidly dividing) cancer cells, which might contribute to host immunity and cancer surveillance. 4,19,25,27,34 These are not mutually exclusive hypotheses, and host response further complicates outcome of infection. For AMDV, persistence is prerequisite to disease in adult animals, but the significance of viral persistence for many other parvoviruses is unknown.
Red pandas (Ailurus fulgens) are native to central and eastern Asia, and are currently listed as endangered by the International Union for Conservation of Nature and as Appendix 1 species within the Convention on International Trade in Endangered Species. 15 Approximately 200 red pandas are in human care in the United States, distributed among numerous zoological institutions and managed by an Association of Zoos and Aquariums Species Survival Plan. 12 Zoo placement and breeding programs have a key role in conservation efforts for the endangered red panda—a role that includes maintaining vigilant surveillance for potential pathogens.
The purpose of this study was to report a newly discovered amdoparvovirus in the red panda and describe its association with disease in a zoological population. The objectives were to (1) characterize the genetic sequences and tissue distribution of the virus, (2) illustrate the association between detected viral nucleic acid and histologic lesions, and (3) determine the fecal prevalence and shedding pattern of the virus through prospective testing of feces.
Materials and Methods
Animals and Sample Collection
The 6 animals in this study were members of a cohort of red pandas (Ailurus fulgens fulgens) housed at the Sacramento Zoo. The group included 4 females and 2 males, ranging in age from 5 to 19 years during the time of the study. All were offspring of red pandas that had been in captivity at various zoological institutions dating back at least 2 generations, and all 6 animals were distantly related, sharing at least 1 common ancestor within the previous 4 generations. The indoor housing allowed for potential direct or indirect contact among 5 red pandas (cases 1-5) over a 3-year period. Outdoor exhibit space was also exchanged between the groups at various times during their stay. Case 6, who died in 2003, was in direct contact with cases 1 and 3 for 6 months prior to her death.
This study included complete routine necropsies of 3 geriatric red pandas (cases 1, 2, and 3—aged 19, 16, and 19 years, respectively) who died or were euthanized over a period of approximately 14 months between 2015 and 2016. Necropsies were performed within 24 hours of death at the William R. Pritchard Veterinary Medical Teaching Hospital at the University of California, Davis. Tissue samples were fixed in 10% neutral buffered formalin, and select fresh tissues were stored at −80°C. For case 6, limited archived formalin-fixed, paraffin-embedded (FFPE) tissues were available for inclusion in this study.
Approximately 8 months after the death of case 1, serial fecal samples were collected from each of the 4 remaining red pandas in the collection (cases 2-5). Animals were housed individually at night, and fecal samples for each animal were collected from floors of their individual dens in the morning approximately once a week for 6 weeks. Several additional fecal samples opportunistically collected from 2 cases (cases 2 and 3) in the months prior to or following routine sample collection were also included. Fecal samples were stored at –80°C until use. In total, 6 to 11 fecal samples were collected for each individual.
Viral Discovery
Spleen, liver, lung, kidney, and small intestine from case 1 were pooled, frozen and thawed on dry ice 3 times, and homogenized with a hand-held rotor in approximately 2X volume of PBS. After centrifugation for 10 minutes in a table-top microfuge (15 000 x g), supernatant (400 µl) was collected and filtered through a 0.45 µm filter (Millipore) to remove eukaryotic and bacterial cell-sized particles. The filtrates were treated with a mixture of DNases (Turbo DNase [Ambion], Baseline-ZERO [Epicentre], benzonase [Novagen]) and RNase (Fermentas) at 37°C for 90 minutes to enrich for viral capsid-protected nucleic acids. 39 Nucleic acids were then extracted using magnetic beads of the MagMAX Viral RNA Isolation kit (Ambion) according to the manufacturer’s instructions. An Illumina MiSeq library was constructed using random RT-PCR followed by use of the Illumina Nextera kit, and sequenced on an Ilumina MiSeq platform using 250 base paired ends. 22 Sequence reads were assembled de novo using the Ensemble program, and contigs and singletons were then translated into hypothetical protein sequences which were compared to all viral proteins in the NCBI virus RefSeq database using BLASTx. 13
RpAPV Specific PCR
A conventional PCR assay was developed to screen feces from cases 2-5 and tissues from cases 1-3. The target was a 154 bp sequence from the nonstructural gene (NS-1) of the RpAPV genome. The 25 μL reactions contained HotStarTaq Master Mix (Qiagen), 0.5 μM forward primer RPAmdoNS_F2 (5’- CGCCAAAACCAACCGACCAA-3’) and reverse primer RPAmdoNS_R2 (5’- AACACGCCCTTAGCTGTGCTT -3’) (Integrated DNA Technologies), and approximately 100 ng of purified DNA. The reaction took place in an MJ Research PCT-200 thermal cycler with an initial denaturation step at 95°C for 5 minutes, followed by 40 cycles of denaturation at 94°C for 30 seconds, annealing at 54°C for 30 seconds, and elongation at 72°C for 30 seconds, with a final elongation step at 72°C for 10 minutes. Amplicons were evaluated by 2% agarose gel electrophoresis, and confirmed by Sanger sequencing (DNA sequencing facility, UC Davis).
Histopathology
Tissues from cases 1-3 and 6 were collected at necropsy, immersion-fixed in 10% neutral buffered formalin (pH 7.2) for at least 24 hours, then processed using standard histologic techniques. Four-micron-thick sections were cut and stained with hematoxylin and eosin or other staining and immunohistochemical techniques for histologic, histochemical, and immunohistochemical evaluation.
In Situ Hybridization
In conjunction with Advanced Cell Diagnostics (ACD) we designed an in situ hybridization (ISH) diagnostic test comprising 30 probe pairs (20-25 oligomer probes within VP1) that would hybridize to the capsid portion of the RpAPV-CA1 genome (ACD catalog No. 453991, V-RPAmdoV-VP-custom). The ISH test was designed to recognize RpAPV DNA (genome) and/or VP1 transcript. Chromogenic ISH was carried out according to the manufacturer’s instructions for nucleic acid detection (RNAscope 2.5 HD Detection Kit - Red). 40 Briefly, FFPE slides were baked at 60°C for 1 hour, then deparaffinized with exposure to xylene twice, 10 minutes each time, followed by stirring in 100% ethanol twice and air-drying, then rehydration with dH2O for 2 minutes. “Pretreatment solution 1” was applied to the slides for 10 minutes at room temperature. The slides were then boiled in “Pretreatment solution 2” at 100°C for 15 minutes, followed by protease digestion in “Pretreatment solution 3” for 30 minutes at 40°C to allow target accessibility. Specific or control probes were applied and the slides were incubated at 40°C for 2 hours. Slides were washed twice with 1X wash buffer for 2 min at room temperature. A series of 6 signal amplification steps was performed per manufacturer’s instructions. Incubation with RED A and B solution was performed at room temperature for 10 minutes and the reaction was quenched with dH2O. Gill’s hematoxylin was applied for 2 minutes, and slides were then rinsed in water, decolorized in 0.02% ammonium hydroxide, and rinsed in water again. Slides were passed through 100% ethanol, 70% ethanol, and twice in xylene before coverslipping with xylene-based mounting medium.
Thin-Section Transmission Electron Microscopy
For transmission electron miscroscopy (TEM), sections of formalin-fixed tongue tissue from case 2 were immersed in modified Karnovsky’s fixative (50% strength) and postfixed in 1% osmium tetroxide. After osmification, the tissue was rinsed in 0.1 M sodium cacodylate, dehydrated through a graded ethanol series, transitioned through propylene oxide, and infiltrated and embedded in Eponate-12 epoxy formulation (Eponate-12; Ted Pella Inc., Redding, CA). Thick sections were cut, mounted on glass slides, stained with toluidine blue, and examined by light microscopy. Thin sections were mounted on 200-mesh copper grids and stained in 4% uranyl acetate in 75% ethanol, followed by poststaining in lead citrate. The grids were examined in a JEOL 1400+ transmission electron microscope at 80 kV accelerating voltage (JEOL USA, Peabody, MA).
Direct Electron Microscopy
Two fecal samples from 1 animal (case 2) were pooled for direct electron microscopy (DEM). Four mL of fecal sample was diluted with water to a final volume of 13 mL and clarified by centrifuging at 2400 x g for 20 minutes at 30°C. The supernatant was filtered sequentially through 5.0, 0.8, 0.45, and 0.2 micron syringe filters. Seven milliliters of the filtered supernatant were placed in a 13.5 mL Beckman polycarbonate thick wall centrifuge tube (Beckman Instruments, Part number 355630) and centrifuged at 55 000 rpm in a Beckman 70.1 Ti rotor for 45 minutes at 5°C. The protein pellet on the bottom of the centrifuge tube was suspended for approximately 20-30 minutes in 1.0 mL water and placed into a 1.5 mL labeled freezing vial. The pellet suspension was mixed with 2% neutralized phosphotungstic acid (PTA) at an approximate ratio of 1:10 to form a slurry that was applied to a carbon coated Formvar grid and excess slurry was wicked away with Whatman #1 filter paper wedges. Grids were examined on an FEI Talos transmission electron microscope at 80 KeV accelerating voltage (FEI, Hillsboro, OR).
Results
Cases
Case 1 died in 2015 with systemic pyogranulomatous inflammation in multiple tissues. Histochemical stains to rule out potential infectious causes included Gomori methenamine silver, Warthin-Starry silver, periodic acid Schiff, Brown and Brenn modified Gram stain, Giemsa, and Ziehl-Neelsen and Fites acid-fast stains. Immunohistochemistry for canine distemper virus, canine coronavirus 2 (with known cross reactivity to feline infectious peritonitis virus), and West Nile virus were performed, and all were negative. Case 2 was euthanized in 2016 for age- and cancer-related deterioration. Significant lesions included intestinal and biliary carcinomas and chronic cholangiohepatitis. No significant infectious organisms were detectable using a battery of histochemical stains similar to those used for case 1. Tissues from cases 1 and 2 were submitted on separate occasions, and in separate runs, for viral metagenomic analyses. Case 3 was a geriatric animal that died naturally in 2016, with a constellation of age-related degenerative changes affecting the axial and appendicular skeleton. Cases 4 and 5 are alive at the time of this study, with no major clinical concerns. Case 6 was a 7-year-old red panda that died in 2003. The most significant lesions in this case included segmental enteritis and transmural colitis, both of which were attributed to a history of colonic impaction. Limited FFPE tissues were available from this case.
Viral Discovery
For case 1, colon, small intestine, spleen, kidney, and liver were pooled and processed for viral metagenomics, with generation of 1.98 million reads. Of the total reads, 1.66% (n = 32 946) showed BLASTx E score <10−5 to amdoparvovirus proteins. The entire protein coding sequence of the parvovirus genome was assembled and deposited in GenBank (NC_031751.1) as red panda amdoparvovirus strain CA1 (RpAPV-CA1). Also detected were 617 reads (0.033%) related to polyomaviruses. After using PCR and Sanger sequencing to fill gaps, the complete polyomavirus genome was assembled, and was deposited in GenBank (KT878838.1) as red panda polyomavirus strain CA1 (RpPyV-CA1). In case 1, a low number of reads were also identified with closest similarity to anelloviruses (n = 5) and papillomaviruses (n = 6). Although read numbers do not necessarily indicate significance, in this instance the latter viruses were considered either contaminants or commensal infections and less significant to the case.
Feces and a tissue pool (lung, liver, pharyngeal tonsil) from case 2 were analyzed separately by viral metagenomics. The number of RpAPV reads derived from feces was 2% (n = 24 700 out of 1 229 703) and from the tissue pool 6% (n = 524 997 out of 8 658 526). Reads were assembled into a separate full coding sequence parvovirus genome (RpAPV-CA2, KY564173). RpAPV-CA2 is 88% identical at a nucleotide level to RpAPV-CA1. No other viral sequences were detected in this case.
Phylogenetic analyses were performed to classify the red panda amdoparvovirus (Figs. 1 and 2) and polyomavirus. The current criteria for the family Parvoviridae proposed by the International Committee on the Taxonomy of Viruses (ICTV) requires that the NS1 region of distinct virus species in the same Parvoviridae genus should be <85% identical at the amino acid sequence level. 11 RpAPV CA1 and CA2 both share approximately 75% NS1 amino acid sequence identity with their closest phylogenetic neighbors (AMDV), warranting classification as a novel viral species. Surprisingly, given that the 2 in-contact source animals died approximately a year apart, the 2 RpAPV sequences were 12% divergent in both whole genome alignment and in NS1 amino acid sequence. Reported skunk and mink amdoparvoviruses also show a relatively high degree of sequence diversity among isolates (from 88-98% by full sequence and 84-98% by NS1 amino acid sequence).
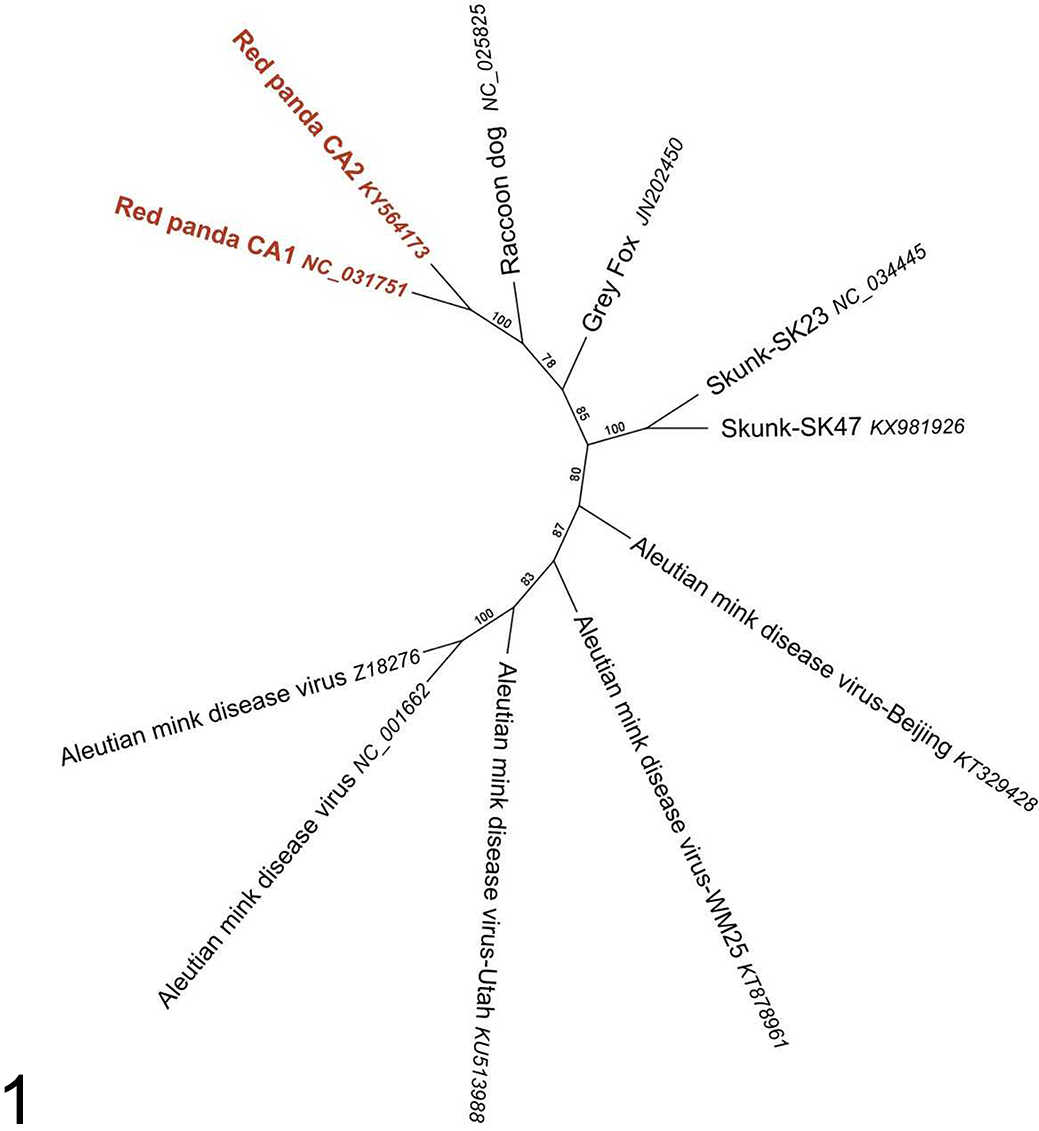
Phylogenetic analysis of red panda amdoparvovirus (RpAPV) and other amdoparvoviruses using predicted amino acid alignment of NS-1 protein.
The Polyomaviridae taxonomy was revised in 2016 to accommodate a rapidly growing number of newly discovered viruses in diverse animal species. 6 A new criterion for creation of novel polyomavirus species is based on >15% difference in sequence identity of the nonstructural T antigen (T Ag) coding sequence compared to the most closely related species. 6 The red panda polyomavirus LT protein is closest to that of Mus musculus polyomavirus 2 (ALN69895.1) with 65% identity, indicating that the RpPyV is the first representative of a new species in the Betapolyomavirus genus. A complete set of tissues from case 1 was tested for RpPyV by ISH (not shown), and viral genome was detected only in rare renal tubule epithelial cells, with no histologic lesions. This is similar to the histologic appearance of low-productive, clinically inconsequential infections of other animals (humans, raccoons) by other members of this PyV virus family. In cases 2 and 3, tissues (spleen, bone marrow, kidney, lung) were negative for RpPyV by PCR and ISH (not shown).
Detection of RpAPV Genome Among Cohort Red Pandas
A primer set was designed to amplify a 154 bp segment of the RpAPV NS1 gene that was 100% conserved between the CA1 and CA2 isolates. To establish whether other animals in the cohort were infected with RpAPV, following the death of case 1 serial fecal samples were collected from all 4 in-contact red pandas (cases 2-5). Two of these animals subsequently died (cases 2 and 3). The collection period ended 4 weeks prior to the death of case 2 and 16 weeks prior to the death of case 3. All 4 in-contact animals had detectable virus in their feces. In 3 of the 4 cases (cases 3-5), all fecal samples were positive. In case 2, virus was detected in all but 2 fecal samples (Supplemental Table S1, Supplemental Fig. S1).
The same NS1 primer set was used to analyze tissue distribution of the virus in select frozen tissues of necropsied animals (cases 1-3). Size- and sequence-specific amplicons were detected in all tissues tested, including spleen, liver, lung, small intestine, and kidney from case 1, in kidney, spleen, liver, lung from case 2, and kidney, liver, lung, and a mesenteric lymph node from case 3 (Table 1).
Pathologic Findings and Detection of Red Panda Amdoparvovirus (RpAPV) in Tissues of 4 red Pandas (cases 1-3 and 6) by PCR and In Situ Hybridization.
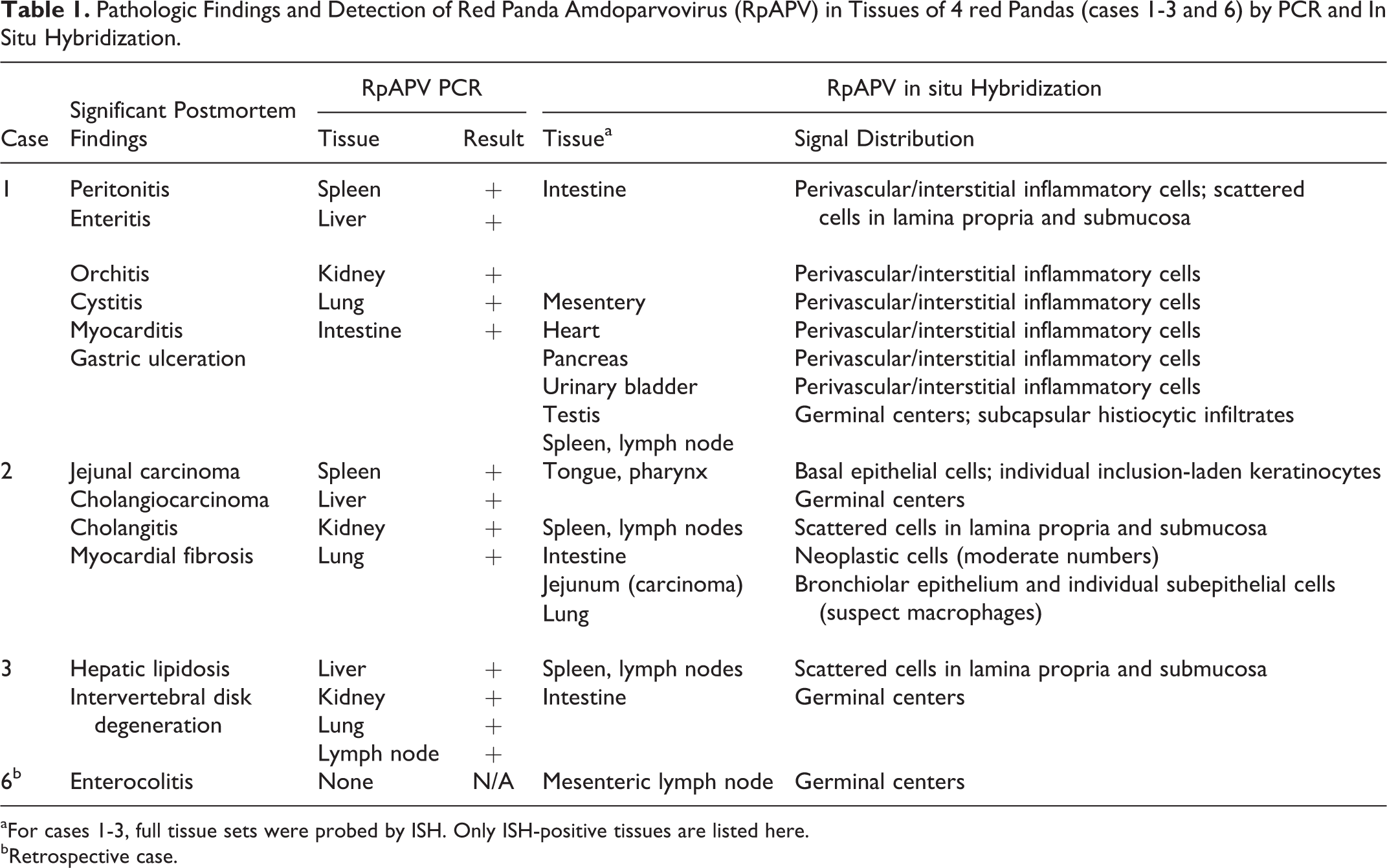
aFor cases 1-3, full tissue sets were probed by ISH. Only ISH-positive tissues are listed here.
bRetrospective case.
Necropsy, Histopathology, and in situ Hybridization
Causes of death or reasons for euthanasia were distinct in all 3 cases (cases 1-3) that were necropsied within a 14-month period of time. Complete histologic examinations were performed on these 3 cases, and full sets of tissues were probed by ISH designed to detect RpAPV VP1 genome or transcript. Significant gross and histopathologic findings and RpAPV ISH results from these cases are briefly described below. Consistent findings included positive ISH probe hybridization in lymphoid germinal centers in lymph nodes and/or spleen, and positive hybridization in scattered individual cells in the mucosa and lamina propria of the intestines. ISH signal was associated with severe inflammatory lesions in 1 case (case 1), and with evidence of cytolysis and intranuclear inclusions in another (case 2) (Table 1). In case 1, inflammatory lesions colocalized with RpAPV nucleic acid as determined by ISH. Gross findings included peritoneal and pericardial effusions, duodenal serosal petechiation, multifocal segmental thickening of the small intestine, and mesenteric lymphadenomegaly. Histologically, there were scattered aggregates and perivascular cuffs of histiocytes, lymphocytes, plasma cells, and neutrophils within the mesentery and under serosal surfaces of the intestines (Fig. 3) and urinary bladder. In the intestines, inflammation multifocally extended into the longitudinal muscularis, with associated degeneration and necrosis of individual or clustered myocytes. In the pancreas, similar inflammatory aggregates surrounded vessels in the interlobular septa, occasionally extending to surround and separate exocrine glands (Fig. 4). Similar inflammatory foci were scattered throughout the myocardium, with associated cardiomyocyte degeneration and necrosis. An enlarged mesenteric lymph node contained large subcapsular aggregates of macrophages, presumed to have drained from the peritoneal cavity (Fig. 5). RpAPV-CA1 nucleic acid (punctate cytoplasmic and nuclear staining) was consistently detected by ISH within all regions of inflammation. The hybridization was interpreted to be in macrophages based on the cell size, shape, and location, but specific cell identity could not be assigned definitively. Positive hybridization was also seen in germinal centers of lymph nodes and spleen (presumed follicular dendritic cells), but without evidence of significant lymphocytolysis, lymphoid depletion, or other possible consequences of viral infection. Small numbers of scattered individual cells in the mucosa and lamina propria of the intestines were also positive by ISH.
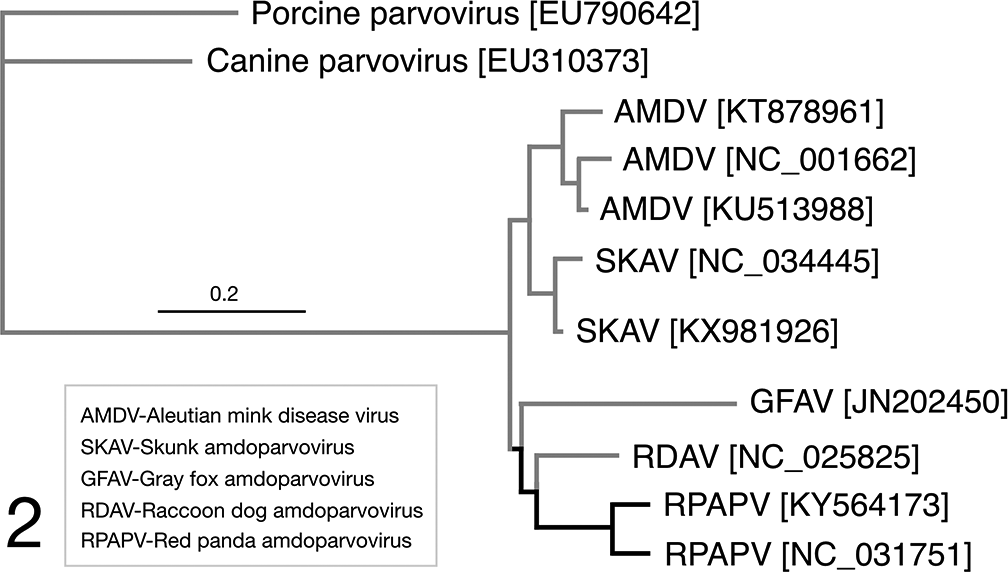
Alignment of the whole-genome sequences, demonstrating the closest phylogenetic neighbors for RpAPV, with canine parvovirus 2 and porcine parvovirus outgroups. Sequence comparisons were performed using the Clustal Omega(1) plugin of Geneious version 9.1.8 (2). Phylogenetic reconstruction of maximum likelihood (ML) trees was performed in Geneious using PhyML(3) plugin with 100 bootstrap replicates. 16,20,38
Major pathologic findings in case 2 included carcinoma (liver, small intestine) and chronic cholangiohepatitis. Sections of tongue and pharynx revealed scattered hypereosinophilic, shrunken epithelial cells (interpreted as apoptotic) in the basal and spinous epithelial layers (Figs. 6-8). Rare epithelial cell nuclei had peripheralized chromatin with central, variably distinct, eosinophilic intranuclear inclusions. There was minimal associated inflammation in the subjacent submucosa. By ISH, signal was localized to the basal epithelial layers, including inclusion-laden and apoptotic cells. This case also exhibited positive probe hybridization in presumed dendritic cells in many germinal centers of the spleen and lymph nodes, with approximately 75% of germinal centers in examined sections displaying at least small amounts of hybridization (Supplemental Fig. S2). In the lungs, positive hybridization was seen segmentally at the level of the bronchiolar epithelium (Supplemental Fig. S3). This was suspected to represent intraepithelial hybridization, but could not be definitively distinguished from signal in subepithelial inflammatory cells. Similar to case 1, individual intestinal mucosal and lamina proprial cells were ISH-positive. Scattered neoplastic epithelial cells in the jejunal carcinoma were also positive (Supplemental Fig. S4).
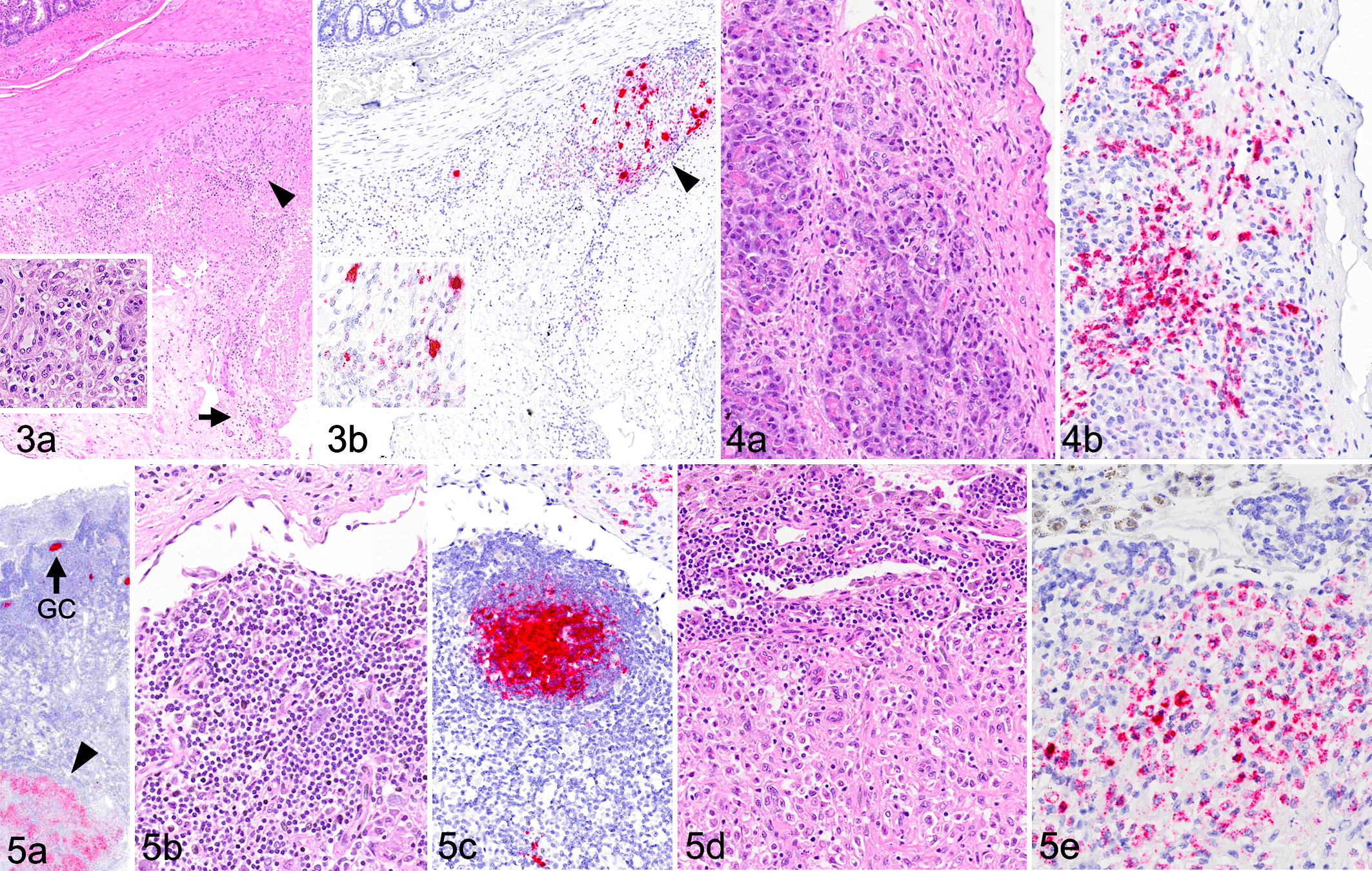
Red panda amdoparvovirus (RpAPV) infection, red panda, case 1.
Case 3 died naturally with a constellation of gross and histologic changes attributable to advanced age, and hybridization was seen in mesenteric lymph node germinal centers and in rare scattered individual cells in the spleen. Cells in the intestines were also ISH-positive, in a distribution similar to that previously described.
Case 6 died with a transmural colitis and segmental ileitis attributed recent colonic impaction (which was reported in the clinical history), for which a cause was not identified. A limited selection of archived FFPE tissues was available. ISH was performed on sections of intestine and mesenteric lymph node, and demonstrated positive probe hybridization in mesenteric lymph node germinal centers.
Confirmation of Viral Shedding
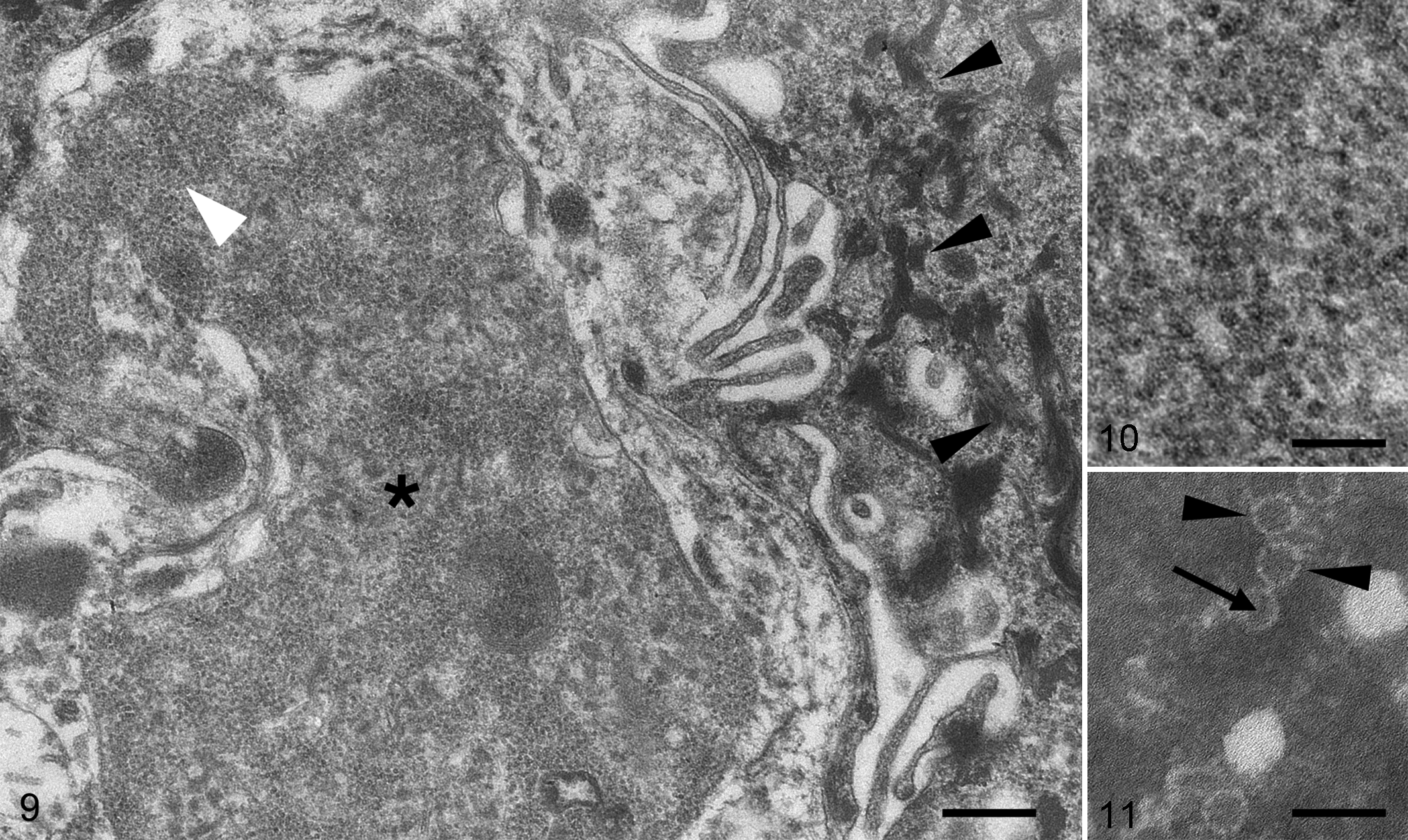
Tissue from the lingual epithelium from case 2 was examined by TEM. This demonstrated 22-nm-diameter capsids (consistent with parvovirus) densely packed in pseudo-crystalline fashion, with associated degenerative changes in the infected cells (Figs. 9-10). The intranuclear virions were tightly associated with nucleoli, in many cases packed so densely as to make them indistinguishable from the nucleolus.
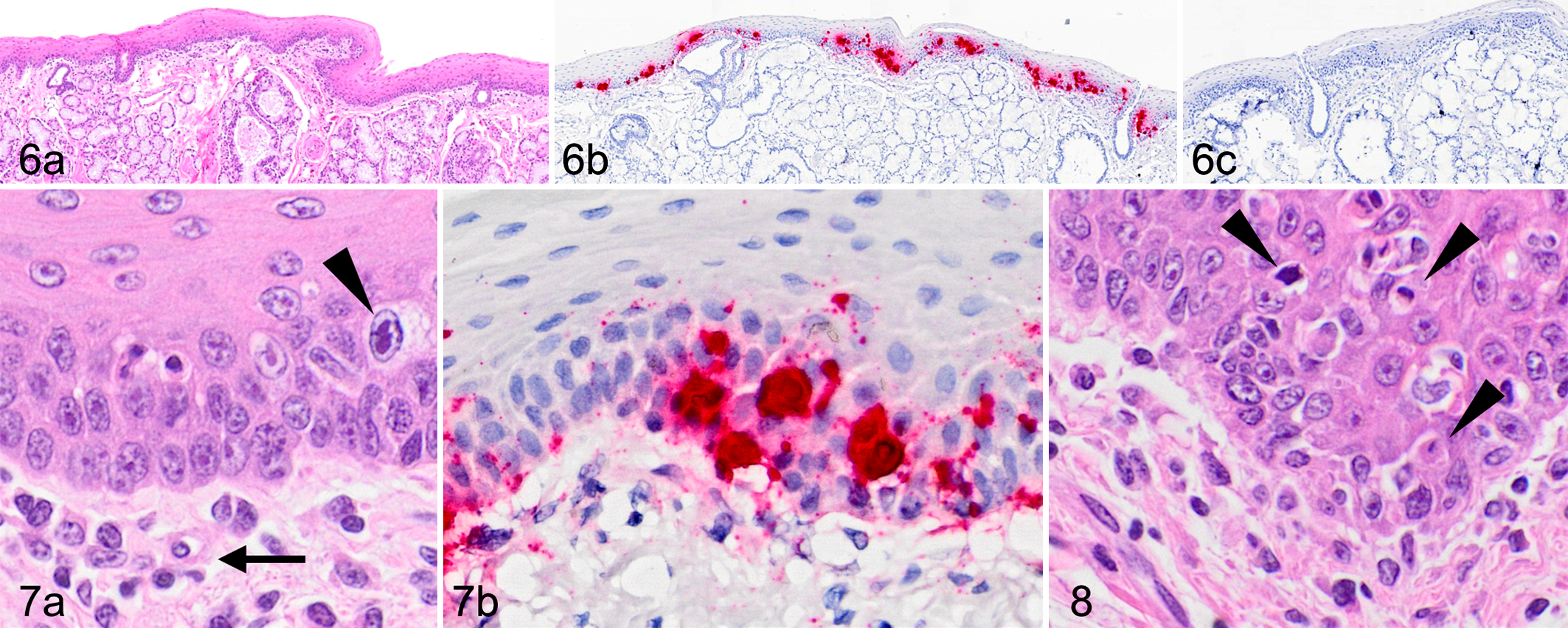
Red panda amdoparvovirus (RpAPV) infection, red panda, case 2. Tongue.
Red panda amdoparvovirus (RpAPV) infection, red panda, case 2.
Direct electron microscopy was performed in 1 case (case 2) using a pool of 2 fecal samples. Numerous 22-nm-diameter viral capsids were identified, with round silhouettes decorated with evenly spaced turrets around their peripheries. Most particles exhibited a central dark (presumably empty) core (Fig. 11). This morphology is consistent with parvoviral virions, and capsid size is similar to that reported for AMDV virions. 10,29
Discussion
The recent, rapid expansion of the genus Amdoparvovirus has been driven in large part by highly sensitive metagenomics techniques, as were used in the present study and are increasingly used in virus discovery. Here, the discovery of a novel amdoparvovirus was combined with diagnostic methods (ISH, PCR) that provide information about the natural history and pathogenic potential of RpAPV in a cohort of red pandas. The combined results, which include a fecal shedding survey, 3 necropsied animals, and one 12-year-old retrospective case, demonstrate that all tested animals were infected. Among all 3 red pandas that died during this study, RpAPV was present in lymphoid tissues and on mucosal gastrointestinal surfaces. In 1 case, virus distribution was more widespread and correlated tightly with regions of inflammation. In the absence of other identified causes, infection in this case was considered likely to have been a significant factor in the animal’s death, although causation is difficult to definitively prove.
Manifestations of RpAPV infection are likely dependent on some combination of viral and host factors, although the specific factors responsible for variation in clinical outcomes are not known. Three of the 4 red pandas that died were geriatric. It is possible that the immunosenescence that accompanies old age is a major factor in case 1. RpPyV was also detected in this animal and demonstrated within scattered renal tubular epithelial cells (data not shown). While the distribution of polyomavirus in this case did not overlap with that of RpAPV, its presence could also be consistent with viral reactivation postimmunosuppression. Potential contribution of the coinfection(s) to disease outcome is not known. Both viral genetics and other host-related genetic or husbandry factors might be relevant in explaining the different clinical features of RpAPV infections in this group.
Parvoviruses appear to be relatively widely distributed in small carnivore species and undergo frequent cross species transmission among carnivores. 3 The predicted evolutionary mutation rate for parvoviruses is high, but may be accelerated by evolutionary pressure such as infection of a novel host species, making true evolutionary rate difficult to predict. 36 Recombination among parvoviruses is also rampant. 35 There is remarkable sequence diversity within the expanding Amdoparvovirus genus. Novel species are defined by ICTV according to amino acid sequence identity in the NS1 protein. The NS1 regions of RpAPV CA1 and CA2, which were derived from the same cohort of animals approximately a year apart, were 11.8% divergent. The closest genetic relatives of RpAPV (skunk amdoparvovirus, raccoon dog and arctic fox amdoparvovirus, Aleutian mink disease virus isolates) are 25 and 29% nonidentical to corresponding RpAPV NS1 sequences. We therefore speculate that the divergence between RpAPV CA1 and CA2 represents natural sequence variation within this population, rather than 2 different sources of infection. Further surveillance and sequence analyses for RpAPV in other collections, and for regional amdoparvoviruses in free-ranging mesopredator carnivore species, would help to clarify the dynamics of parvoviral transmission among red pandas and other carnivores.
Evidence that this virus has successfully adapted to the red panda host includes detection in all tested animals and the demonstration that infection can persist in the absence of clinical disease. PCR results from serial fecal samples demonstrated shedding of viral nucleic acid in all tested animals, including clinically normal animals. Direct electron microscopy in 1 case demonstrated that molecular detection corresponded with the presence of intact virions with characteristic features of parvoviruses, suggesting a productive infection. Given that viral nucleic acid was present within gastrointestinal epithelia, that inclusions and cytolysis were evident, and that virions were detected in feces, we propose that the gastrointestinal tract is a site of persistence, although we cannot rule out repeated infections. In 1 case, an intestinal carcinoma contained viral nucleic acid within neoplastic cells, and, subjectively, the number of infected cells was higher in the neoplasm than in the surrounding normal mucosa. There is a sensible and intriguing correlation of parvoviruses with infection of rapidly dividing cell populations, such as the neoplastic cells and inflammatory cells in this region, but the validity of this association in the present cases is entirely speculative. In humans, parvoviruses infecting tumor cells are well-documented. They have been proposed to contribute to the development of cancer, or, alternatively, to be oncolytic, serving a protective function by cytolysis or clearance of (rapidly dividing) neoplastic cells. 4,19,25,27,31,34
While this study was limited to samples from a single zoo, we hypothesize that RpAPV will be more widespread. The identification of a novel and potentially pathogenic amdoparvovirus in red pandas may have implications for conservation and management of the species. Red pandas in the wild are imperiled by illegal poaching and habitat destruction. Those in human care, as demonstrated in the cohort in this study, tend to live much longer. Age-related immunosenescence, proximity to other species, and husbandry practices can all contribute to new infectious disease dynamics, with implications for management practices. Wild animals are increasingly exposed to humans, urban and suburban wildlife, and domestic animals, creating unique opportunities for interspecies transmission of infectious agents. An understanding of natural host ranges of RpAPV and other pathogens of animals in and around zoological collections is critical to global conservation efforts.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818758470 - Amdoparvovirus Infection in Red Pandas (Ailurus fulgens)
Supplemental Material, DS1_VET_10.1177_0300985818758470 for Amdoparvovirus Infection in Red Pandas (Ailurus fulgens) by Charles E. Alex, Steven V. Kubiski, Linlin Li, Mohammadreza Sadeghi, Raymund F. Wack, Megan A. McCarthy, Joseph B. Pesavento, Eric Delwart, and Patricia A. Pesavento in Veterinary Pathology
Footnotes
Acknowledgements
We thank Ken Jackson for technical assistance and mentorship. Drs Sebastian Carrasco and Sarah Stevens generously provided retrospective cases. We thank the keeper and curatorial staff of the Sacramento Zoo for making animals, samples, and records available. This study was supported in the Pesavento laboratory by Boehringer Ingelheim Vetmedica (for viral discovery) and the Bernice Barbour Foundation (research in naturally occurring infectious disease). Linlin Li, Mohammadreza Sadeghi, and Eric Delwart are supported by funds from Blood Systems Research Institute. Mohammadreza Sadeghi also supported by The Sigrid Jusélius Foundation (grant WBS 4703764).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
