Abstract
Melanosis coli is a well-described condition in humans, characterized by the accumulation of lipofuscin-laden macrophages in the lamina propria of the colon, giving it a dark tone. An increased apoptosis rate of colonic epithelial cells appears to be the underlying pathogenesis. In pigs, oxidative damage has been proposed as one of the causes for melanosis coli. In this article, we report a series of cases of melanosis coli in pigs affecting several finishing units in the south of Spain. Large intestines had dark green to brown pigmentation of the mucosa. Histological, histochemical, and ultrastructural studies confirmed a high number of lipofuscin-laden macrophages in the lamina propria of the rectum and colon, which additionally stained positive for the apoptosis marker cleaved caspase-3. Of note, all affected finishing units utilized water supply with a high content of sulfates, which may be one of the causes for melanosis coli development in pigs.
Melanosis coli alludes to the abnormal dark pigmentation of the colonic mucosa, owing to a high accumulation of lipofuscin-laden macrophages in the lamina propria. 2,3,5,9,11,17,21 In humans, this condition has been widely associated with patients suffering from chronic constipation and using laxatives for long periods, especially anthranoid purgatives. 3,7,8,10,17 However, subjects with chronic diarrhea and no history of laxative use also displayed melanosis coli histologically, 5,12 and it has been diagnosed as a concomitant process in cases of inflammatory bowel disease. 13 This condition has been experimentally reproduced in a guinea pig model after the administration of anthranoid laxatives to study the underlying pathogenesis. These purgatives induced the apoptosis of colonic epithelial cells, which are phagocytosed and degraded by macrophages, leading to the formation of lipofuscin. 6,19 In the pig, melanosis coli was first described in 2015 with evidence of protein nitration and oxidative damage, thought to be due to hypovitaminosis E. 21 At present, there is little knowledge of the causes of melanosis coli in the pig. In this report, we characterize a series of cases of melanosis coli and consider high levels of sulfates in drinking water as a possible cause in pigs.
The study was based on samples of descending colon and rectum from 15 pigs that showed an abnormal pigmentation of the gut mucosa, identified during slaughtering. These were crossbreed pigs ([Landrace × Large White] × Pietrain) coming from 4 different finishing units in the provinces of Granada and Málaga in the south of Spain with different origins and genetics. Three pigs each were from finishing units 1 to 3, and 6 pigs were from finishing unit 4. On average, over 75% of reported cases for the 4 finishing units occurred between June and September (2017), with fewer animals being affected before and after this period. The number of affected animals was roughly 1600 out of 2000 in finishing unit 1, 2200 out of 2400 pigs in finishing unit 2, 2600 out of 3000 in finishing unit 3, and 6000 out of 6500 in finishing unit 4. Both males and females were equally affected and no signs of disease were reported either in the farm or prior to slaughter.
Tissue samples from all 15 animals were fixed in 10% neutral buffered formaldehyde for histochemical and immunohistochemical examination, and selected samples from 3 animals were fixed in 2.5% glutaraldehyde for transmission electron microscopy. Affected herds were surveyed about health and management parameters to investigate possible causes.
Grossly, all submitted sections showed a diffuse but not uniform dark green to brown pigmentation of the colonic and rectal mucosa without affecting the other layers of the intestine (Fig. 1). Microscopically, the simple columnar epithelium of the mucosa was multifocally discontinuous and a high number of pigment-laden macrophages as well as free granular brown pigment of varying size were found in the whole thickness of the lamina propria between the colonic crypts (Fig. 2). Individual differences were found between animals, not only in regard to the degree of mucosal pigmentation but also in the number of lipofuscin-laden macrophages. The submucosa, muscularis, and serosa did not show any histopathological changes.
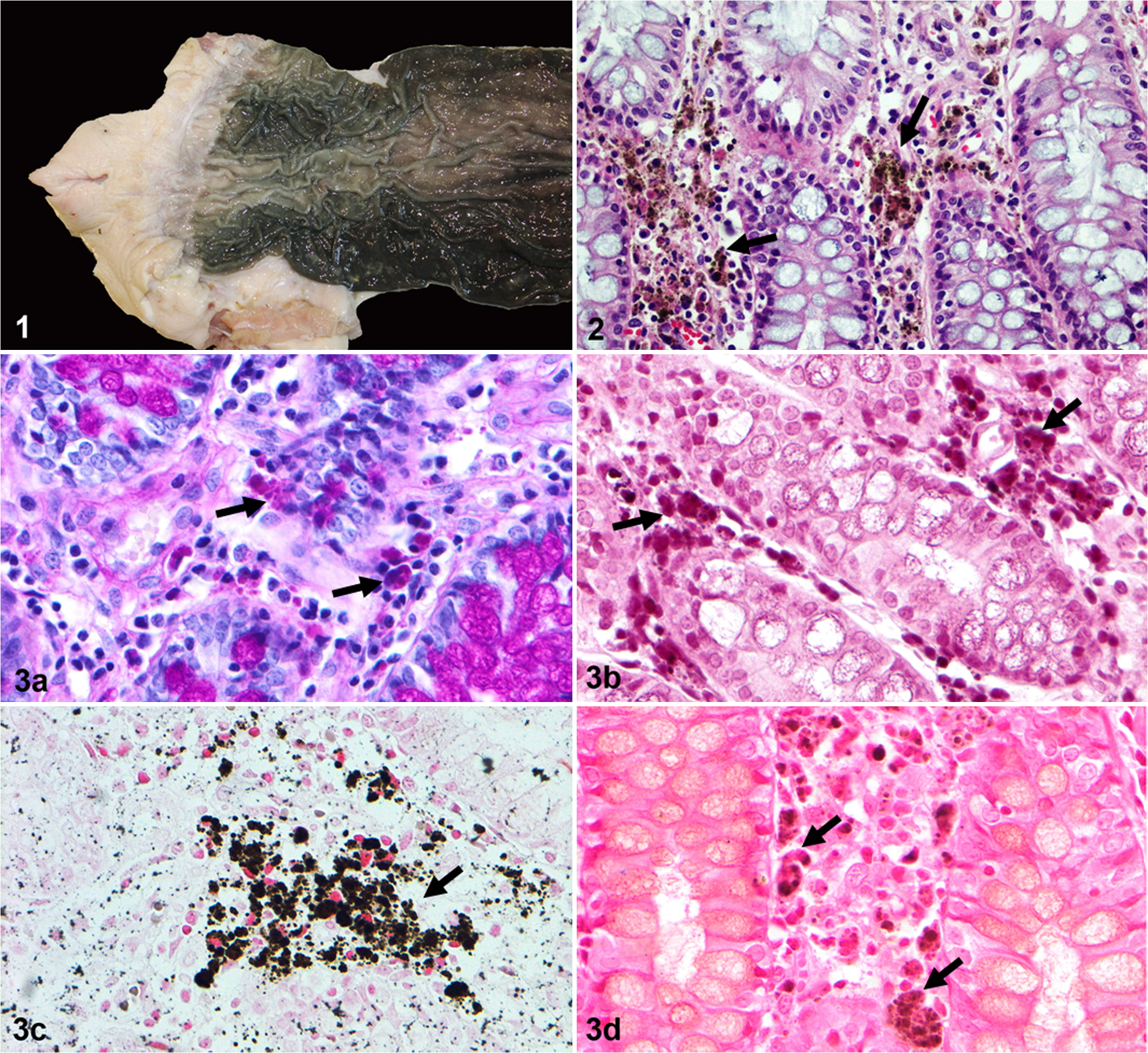
Melanosis coli, colon, pig.
Histochemical techniques to characterize this pigment were Prussian blue for ferric iron, Masson-Fontana for melanin, and periodic acid-Schiff (PAS) and long Ziehl-Neelsen for lipofuscin. Similar results were obtained for each technique in all 15 examined animals. Bleaching with 10% hydrogen peroxide was also performed. In addition, Masson’s trichrome and luxol fast blue methods were also included to detect other associated lesions. For all above-mentioned histochemical techniques, appropriate known positive controls were included. With PAS and long Ziehl-Neelsen, these pigment granules were a strong magenta color (Fig. 3a and b). With Masson-Fontana, the pigment was a charcoal-dark color and negative for Prussian blue (Fig. 3c and d). With Masson’s trichrome and luxol fast blue, the pigment was unstained and no other lesions were revealed. Bleaching of sections caused a moderate loss of the pigment. The findings were interpreted as compatible with lipofuscin.
Immunohistochemistry for cleaved caspase-3 was used as a marker of apoptosis, according to manufacturer instructions (SignalStain Apoptosis Cleaved Caspase-3 IHC Detection Kit #12692, Cell Signaling Technology, Inc). A section of lymph node exhibiting abundant apoptosis was included as positive control. As negative controls, an age-matched genetically related pig without discoloration of the mucosa and belonging to the same finishing unit (control animal) and sections affected by melanosis coli incubated with an isotype control antibody were used to validate the technique. Pigment-laden macrophages and free pigment in the lamina propria were immunolabeled for cleaved caspase-3 (Fig. 4). Isolated cells from the lining epithelium and colonic glands as well as scattered lymphocytes and plasma cells from the lamina propria were also positive for cleaved caspase-3. A scarce number of cleaved caspase-3+ cells from the lining epithelium of the colon and within the lamina propria were observed in the control animal. Transmission electron microscopy identified pigment-laden macrophages in the lamina propria of the colon. The cytoplasm of these cells was electron-lucent containing some mitochondria, lysosomes, heterolysosomes, residual bodies, and lipofuscin granules that were characterized by electron-dense material and electron-lucent lipid droplets (Fig. 5). Thus, based on gross examination, histology, histochemical stainings, and transmission electron microscopy, a diagnosis of melanosis coli in pigs was performed. In addition, positivity of the pigment to cleaved caspase-3 indicated the presence of apoptosis.
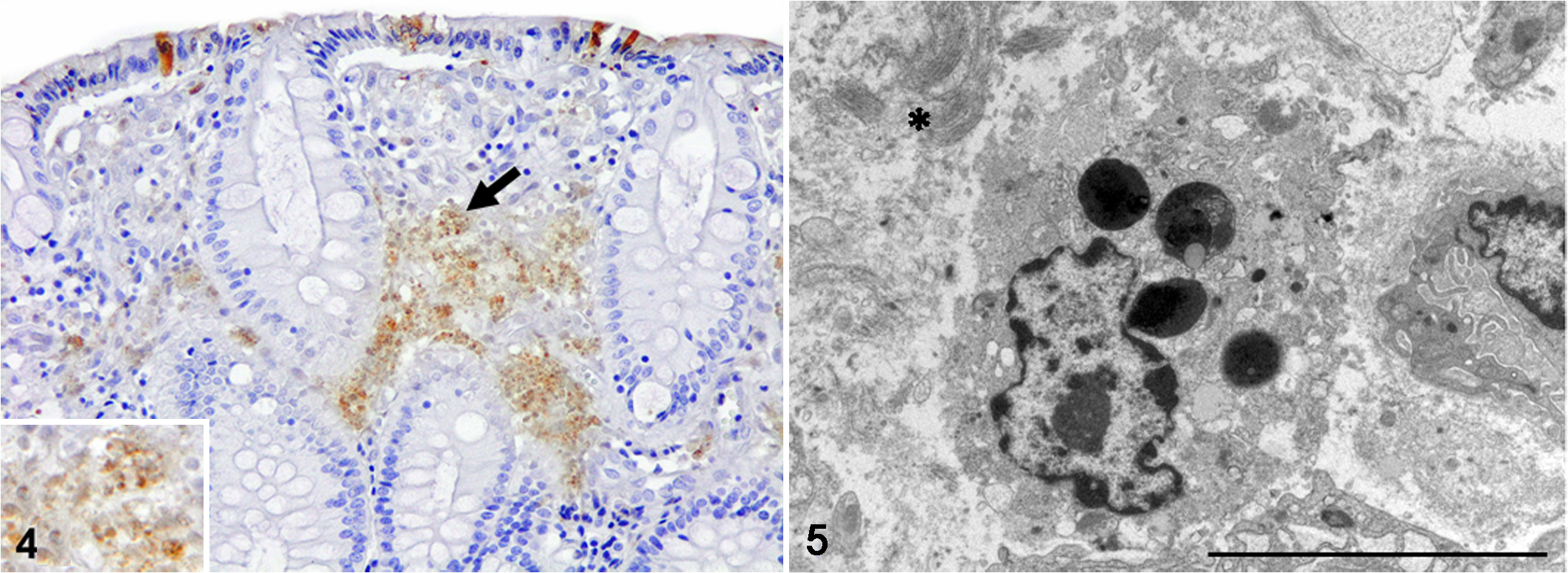
Melanosis coli, colon, pig. Pigment-laden macrophages, free pigment (black arrow) and some colonic epithelial cells positively stained for cleaved caspase-3. Inset: detail of the pigment. Immunohistochemistry.
Analysis of drinking water samples showed that sulfate levels from all finishing units were much higher than the maximum concentration allowed (250 mg/l). Water supply came from groundwater wells and the sulfate concentrations were 4920 mg/l, 3390 mg/l, 4710 mg/l, and 3700 mg/l for fattening units 1 to 4, respectively (ie, from 13.6- to 19.7-fold increase over the allowable level). In contrast, sulfate concentrations from nonaffected units, from the same geographic area and sampled after knowing the results of case herds, were within the normal range. Affected pigs from the different fattening units differed in their diets; however, all these diets were being used in other units without reported cases of melanosis coli. Health programs were up to date and no clinical signs of any particular disease were observed prior to slaughter.
Taken together, we describe a series of cases of melanosis coli in pigs affecting different finishing units with a high content of sulfates in drinking water.
All submitted cases were characterized by diffuse pigmentation of the colonic and rectal mucosa, although the tone of the discoloration was variable between pigs. This contrasts with the snake-skin appearance in human cases, 3,7,8,10,11,16 possibly due to a different arrangement of the intestinal glands in humans and pigs. Moreover, melanosis coli is different from so-called intestinal lipofuscinosis of dogs, characterized by a tan-brown pigmentation of the muscular layer of the intestine, which is visible through the serosa. 18
In the current study of porcine melanosis coli, histochemical, immunohistochemical, and ultrastructural studies identified that the pigment found free and in the cytoplasm of macrophages was lipofuscin and it colocalized with immunolabeling for the apoptosis marker cleaved caspase-3. Lipofuscin is a type of ceroid pigment which has been linked to the breakdown and phagocytosis of apoptotic colonic epithelial cells in melanosis coli of humans and animals. 3,5,6,11,19,20 This process has been well described after anthranoid laxatives consumption, where these substances appear to irreversibly damage the organelles of the colonic surface epithelium triggering their apoptosis. In fact, frequencies of apoptotic cells were significantly greater in melanosis coli subjects than in controls. 3,6,19 Wilberts et al 21 described an increase in protein nitration and lipid peroxidation in pigs with melanosis coli. In addition, the levels of vitamin E were low in some of the animals; however, no correlation was found between the footprints of oxidative stress and hypovitaminosis E.
In our study, we found high levels of sulfates in the drinking water of all affected finishing units and cleaved caspase-3+ pigment in the lamina propria of pigs with melanosis coli. Thus, we hypothesize that the increased concentration of sulfates in the lumen of the colon could be reduced to hydrogen sulfide by sulfate-reducing bacteria, such as Proteobacteria, Actinobacteria, and Firmicutes, that are part of the commensal bowel microbiota. 14 This metabolic end-product is cytotoxic, crossing the cell membrane due to its lipophilic properties and potentially inducing the death of colonic epithelial cells, as has been reported for Jurkat cells and mononuclear leukocytes, where a significant increase in the activity of the apoptotic markers cleaved caspase-9 and -3 were detected after treatment with sodium hydrosulfide. 4,15 Uptake and degradation of these apoptotic cells may finally result in the pigment lipofuscin. In humans, Ennaifer et al 7 reported 2 cases of melanosis coli in patients with chronic constipation and consumption of osmotic laxatives, among which are sulfates; however, no additional studies of the pathogenesis were conducted. One might wonder why the pigs did not suffer from diarrhea due to the high sulfate content and their laxative effect, at least in humans; however, exposure of pigs to water containing high levels of sulfates appears to have no significant health and management consequences. 1
To conclude, we report a case series of melanosis coli in fattening pigs coinciding with elevated levels of sulfates in drinking water.
Footnotes
Acknowledgements
We gratefully acknowledge Gema Muñoz and Alberto Alcántara for their technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Jaime Gómez-Laguna is supported by a “Ramón y Cajal” contract of the Spanish Ministry of Economy and Competitiveness (RyC-2014-16735).
