Abstract
Proventricular dilatation disease is a lethal disease of psittacine birds. In this study, we characterized the local cellular immune response in the brain, proventriculus, and small intestine of 27 cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2 (PaBV-2). Perivascular cuffs in the brain were composed of CD3+ T-lymphocytes and Iba1+ macrophages/microglia in most cockatiels (n = 26). In the ganglia of the proventriculus, CD3+ T-lymphocytes (n = 17) and Iba1+ macrophages (n = 13) prevailed. The ganglia of the small intestine had a more homogeneous distribution of these leukocytes, including PAX5+ B-lymphocytes (n = 9), CD3+ T-lymphocytes (n = 8), and Iba1+ macrophages (n = 8). Our results indicate that perivascular cuffs in the brain and the inflammatory infiltrate in the proventriculus of PaBV-2-infected cockatiels is predominately composed of T-lymphocytes, while the inflammatory infiltrates in the ganglia of the small intestine are characterized by a mixed infiltrate composed of T-lymphocytes, B-lymphocytes, and macrophages.
Keywords
Parrot bornaviruses are the causative agents of proventricular dilatation disease (PDD), a lethal disease of psittacine birds.2,19 Macroscopically, the main lesion observed in PDD is dilatation of the proventriculus. Histologically, the hallmark lesion is pronounced mononuclear inflammation that affects the central nervous system (CNS) and also ganglia and nerves of the peripheral nervous system (PNS), mainly in the gastrointestinal tract, heart, and adrenal glands.14,20
Several studies have documented the inflammatory infiltrates in the CNS and PNS of natural and experimental PDD cases,7,8,11,14,20 but the immunophenotype of the local inflammatory response in PDD remains unknown. This study characterized the local inflammatory response in the central and enteric nervous system of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus-2 (PaBV-2).
Cockatiels (n = 27) were experimentally infected (CK1-27) with PaBV-2 as previously described. 7 Seven mock-inoculated cockatiels served as negative controls (CK28-34) at 20 days postinfection (dpi), 25, 30, 35, 40, 60, and 114 dpi. Cockatiels were identified as CK1 to CK34.
Formalin-fixed and paraffin-embedded samples of cerebrum at the level of thalamus and midbrain, proventriculus and small intestine were subjected to immunohistochemistry (IHC) using the following primary antibodies: CD3 (T-cells) (rabbit polyclonal anti-CD3; 1:1500 dilution; Dako, California, USA), paired box protein (PAX)5 (B-cell) (mouse monoclonal anti-PAX5; BC/24; 1:400 dilution; Biocare Medical, California, USA), and ionized calcium-binding adaptor molecule 1 (Iba1; macrophage/microglia) (mouse monoclonal anti-Iba1; NCNP24; 1:500 dilution; FUJIFILM Wako Pure Chemical, Chou-Ku, Osaka, Japan). Briefly, serial sections were cut at 3-μm-thick and onto charged slides. Antigen retrieval (citric acid; 1mM; pH = 6; temperature = 120°C) was followed by endogenous peroxidase blocking and nonspecific binding blocking with normal serum of the same species where antibodies were raised. Antibodies were incubated overnight (18h, 4°C). Amplification and visualization were achieved by the Novolink HRP polymer detection system kit (Leica Biosystems, Newcastle, UK) and counterstaining with Harris’ hematoxylin. Normal cloacal bursa from a blue-fronted Amazon parrot (Amazona aestiva) was used as positive control for validation of all antibodies (Supplemental Material). Antibodies were replaced by nonimmune homologous serum for negative controls.
The number of immunolabeled cells was semiquantitatively scored for each antibody. The number of positive cells in the ganglia present in the section of proventriculus and small intestine and the number of positive cells in 10 perivascular areas of the brain were scored as follows: − (absent), + (1-10 positive cells, minimal); ++ (11-50 positive cells, mild); +++ (51-100 positive cells, moderate); ++++ (100-150 positive cells, moderate to marked), and +++++ (>150 positive cells, marked).
The proportion of positive immune cells in different tissues and different markers were evaluated by McNemar’s test for paired observations. The difference in proportion of positive immune cells in early (up to 44 days) and late (after 66 days) stages of infection were compared by chi-square test. The difference in frequencies of positive scores was evaluated by Friedman test followed by the Conover’s post hoc test. The significance level of 0.05 was considered for all analyses. Statistical evaluations were performed using R v3.6.0. 15
IHC results are recorded in Table 1 and Figs. 1–10. Consistent immunolabeling was detected for CD3, PAX5, and Iba1 in all organs/tissue sections examined. The 7 mock-inoculated cockatiels exhibited strong immunolabeling for all markers in their expected microanatomical locations. The following alterations in immunophenotypic profiles of local inflammatory responses are in comparison to control cockatiels.
Immunohistochemistry scores for the frequency of CD3, PAX5, and Iba1 positive cells in the brain, proventriculus, and small intestine of 34 cockatiels (CK) experimentally inoculated with parrot bornavirus 2 (infected) or placebo (*, control).
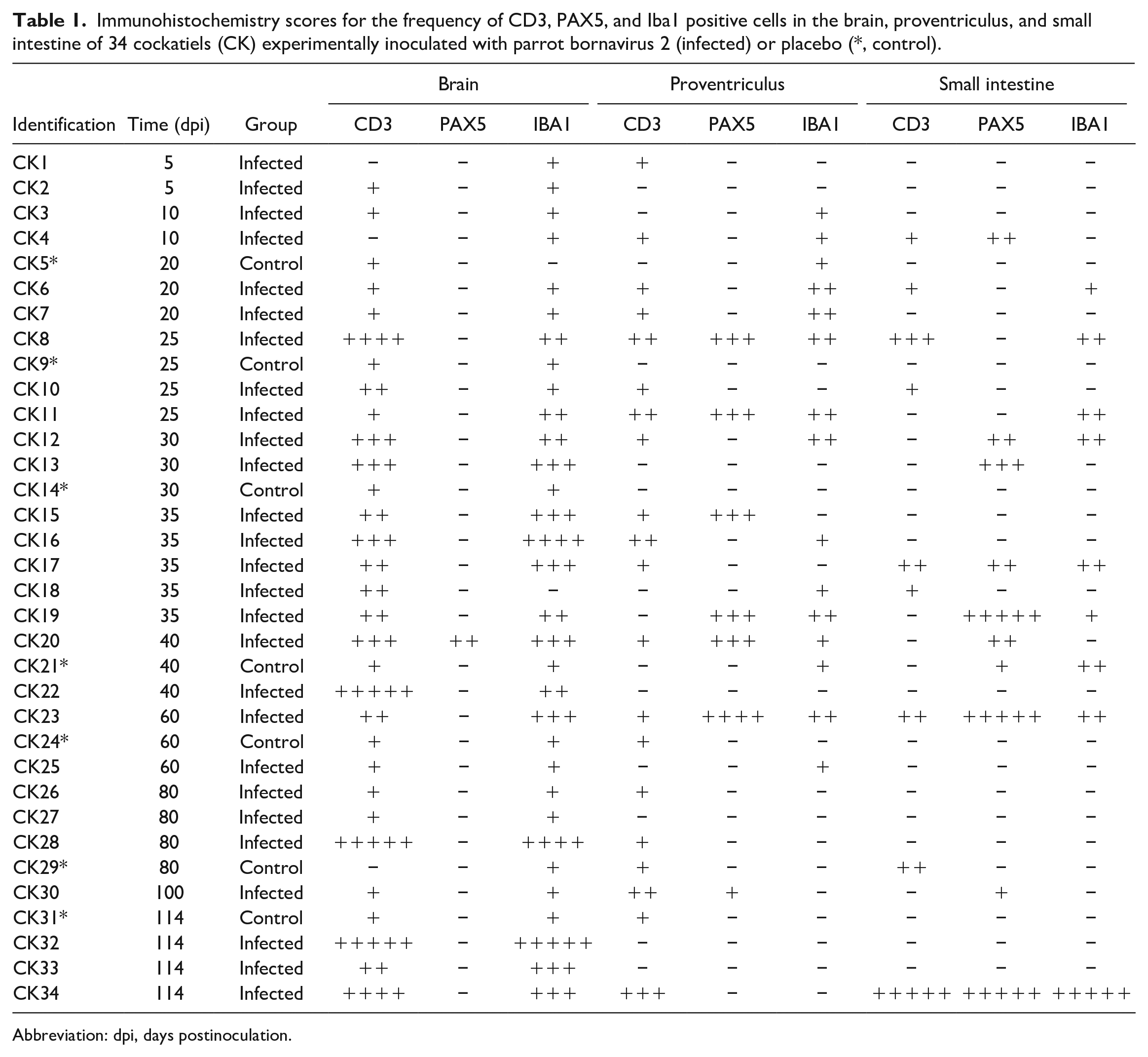
Abbreviation: dpi, days postinoculation.
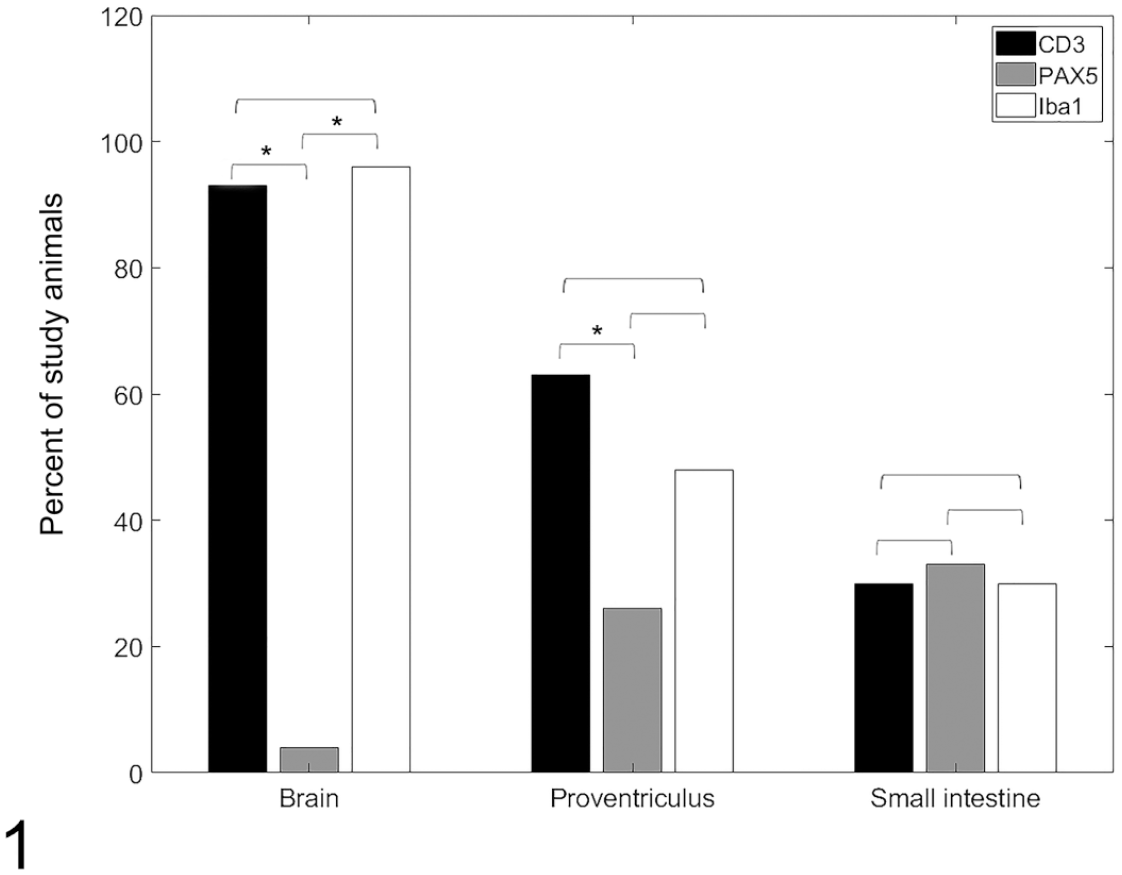
Comparison of the percentage of cockatiels experimentally infected with parrot bornavirus 2 that presented cells of each immunophenotype in the inflammatory infiltrates.
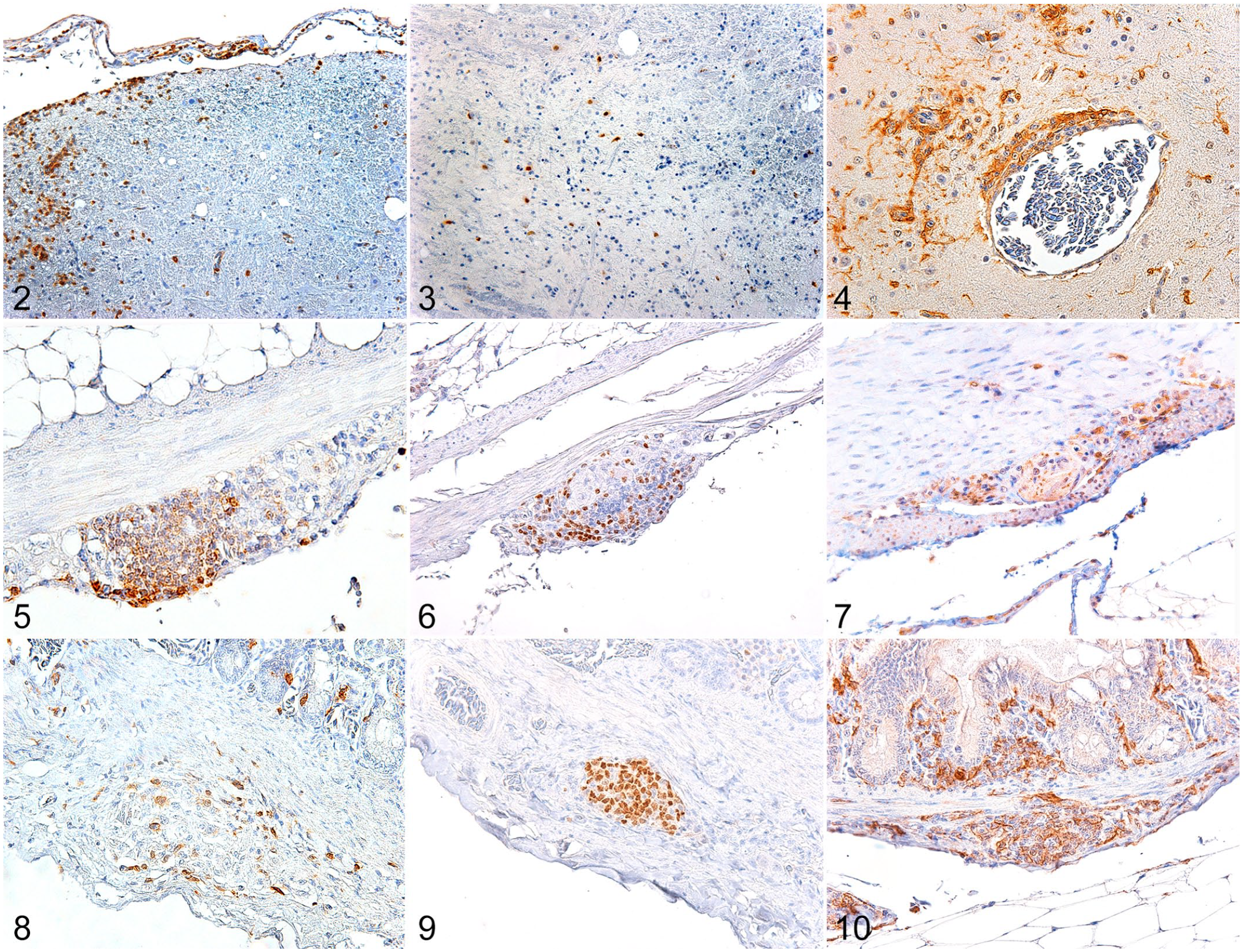
Experimental parrot bornavirus-2 infection, cockatiels. Immunohistochemistry for CD3 (T cells), PAX5 (B cells), and Iba1 (macrophages/microglia).
In the brain, inflammatory perivascular cuffs were predominantly composed of CD3+ T-lymphocytes (Fig. 2) and Iba1+ macrophages/microglia (Fig. 4). The immunolabeling for these 2 cell populations were similar (P > .05). Two cockatiels had no detectable CD3+ T-lymphocytes (CK1 and CK4) and one cockatiel had no Iba1 immunolabeling (CK18). CD3+ T-lymphocytes and Iba1+ macrophages/microglia were largely absent or were present in small numbers (mild) in the initial timepoints until 20 dpi. Scores for timepoints between 25 to 114 dpi varied greatly, ranging from absent to marked numbers of CD3+ T-lymphocytes and/or Iba1+ macrophages/microglia. CD3+ lymphocytes were also present in the leptomeninges. Iba1 consistently labeled aggregated and/or isolated macrophages/microglia. Rare Iba1+ cells were present in the leptomeninges. PAX5 immunolabeling was significantly lower (P = .002) that that of CD3 and IBA1 (Fig. 1). Only one cockatiel presented with small numbers (mild) of PAX5+ B-lymphocytes (Fig. 3) in the perivascular infiltrates.
The inflammatory infiltrate in the proventriculus was a mixed population of T- and B-lymphocytes and as well as macrophages. Immunolabeling for CD3+ T-lymphocytes (Fig. 5) was detected in the proventriculus of 17/27 cockatiels (63%) and was more frequent than for PAX5+ B-lymphocytes (P = .009, Fig. 6), which was detected in 7/27 cockatiels (26%) and ranged from minimal to moderate. However, the frequency of immunolabeling for CD3 was similar (P > .05) to that for Iba1+ macrophages (Fig. 7), which was detected in 13 cockatiels (48%). T-lymphocytes, macrophages, and B-lymphocytes infiltrated the ganglia and nerves of the proventriculus (Figs. 5–7). Of the cellular infiltrates in the ganglia, more than 80% were CD3+ T-lymphocytes, while 40% to 80% were PAX5+ B-lymphocytes in 6/7 cockatiels. CD3+ T-lymphocytes and PAX5+ B-lymphocytes were present in the lymphoid aggregates of the mucosa and submucosa of the proventriculus in infected and control cockatiels. While CD3+ T-lymphocytes were consistent for most timepoints, PAX5+ B-lymphocytes were reduced or absent in initial (until 20 dpi) and late (after 60 dpi) timepoints. Iba1+ macrophages were reduced in late timepoints.
The inflammatory infiltrate of the small intestine had a homogenous distribution (P > .05) of T-lymphocytes, B-lymphocytes, and macrophages, throughout all timepoints. CD3+ T-lymphocytes (Fig. 8) and Iba1+ macrophages (Fig. 10) comprised the mononuclear infiltrates effacing the ganglia of the small intestine in 8 cockatiels (30%), while PAX5+ B-lymphocytes (Fig. 9) were observed in the intestinal ganglia of 9 cockatiels (33%). PAX5 immunolabeling received a marked score more frequently (3 cases) than CD3 and Iba1 markers (1 case each).
Immunolabeling for CD3 was more frequent in the brain when compared to the proventriculus or small intestine (P = .04), and more frequent in the proventriculus when compared to the small intestine (P = .0002). On the contrary, PAX5 was more frequent in the proventriculus and small intestine when compared to the brain (P = .04) but did not differ between proventriculus and small intestine (P > .05). No differences were seen for Iba1 in any of the tissues (P > .05). Regarding the grading scores (Fig. 2), most of CD3 and Iba1 immunolabeling (30%-40%) in the brain was mild (+), while 96% of the cases were negative for PAX5.
In this study, the perivascular inflammatory infiltrates in the brain of cockatiels experimentally infected with PaBV-2 were comprised mostly of CD3+ lymphocytes while glial nodules and resident microglia were immunolabeled for Iba1. Interestingly, PAX5+ lymphocytes were detected in the perivascular infiltrates of only one cockatiel at 40 dpi. This finding is in accordance with studies with Lewis rats experimentally infected with BoDV-1, where infiltrates were composed mostly of CD4+ and CD8+ T-cells and macrophages while B-cells and plasma cells only occurred at later timepoints and in lower numbers.5,9 This predominance of CD3+ lymphocytes in the perivascular spaces, accompanied by fewer macrophages and B-cells, is corroborated by several studies in rodents,5,17 naturally BoDV-1-infected horses, 3 and humans naturally infected with variegated squirrel bornavirus 1. 18 The onset of clinical neurological signs has been linked to the occurrence of CD3+ T-cells in both mammals and birds,4,9,12 which suggests a T-cell-mediated mechanism with subsequent cytotoxic effects may be a key factor in bornaviral pathogenesis. 10
In one case report of PDD in a blue-winged macaw (Primolius maracana) 13 CD3+ cells but no CD79+ cells were identified in the perivascular inflammatory infiltrates in the brain. In our experience, commercial primary anti-CD79 antibodies show inconsistent or no cross-reactivity with lymphoid cellular populations in avian tissues. By contrast, PAX5 exhibits reliable cross-reactivity as observed in a recent study. 6 In another study, 1 CD3+ cells were detected in the brain of cockatiels experimentally infected with PaBV-4 but not in the brain of cockatiels experimentally infected with PaBV-2, while the IHC technique was not successful in detecting B-cells or macrophages in the CNS of any of the groups. However, it is not clear how many cockatiels presented this pattern in the study. The latter study evaluated B-cells immunohistochemically using primary antibodies against IgY, CD20, CD45, IgG, MUM1, and CD79α, while macrophages were evaluated using primary antibodies against myeloid histiocyte antigen clone MAC378, human lysosome, and KUL01. 1
Lymphoplasmacytic inflammatory infiltrates have been consistently described in the ganglia and peripheral nerves of a wide variety of organs of natural and experimentally PaBV-infected birds, especially those in the gastrointestinal tract.12,19,20 This study demonstrated that CD3+ T-lymphocytes are indeed the predominant inflammatory cell type in the ganglia of the proventriculus, followed by macrophages and fewer B-lymphocytes. On the other hand, inflammatory infiltrates were less frequent in the ganglia of the small intestine when compared to the proventriculus; with more equal numbers of T-lymphocytes, B-lymphocytes, and macrophages.
Overall, there were few inflammatory cells until 20 dpi in all cockatiels, and no immunolabeling was detected in the small intestine until 10 dpi. Beginning at 25 dpi, immunolabeling became more prominent, and it was expressed by more cells in all 3 tissues examined, reaching in some cases, moderate to marked scores. These results corroborated the histopathologic and PaBV-2 IHC results published by our group. 7
It is suggested that the macrophages within the ganglia and brain in PDD are recruited by CD4+ T-cell responses, and it has been speculated that these inflammatory responses lead to antibody-mediated phagocytosis of axons. 16 Demyelination, however, is not a primary finding in PDD and mammalian bornaviral infections. 10 In this study, Iba1+ macrophages were detected as part of the inflammatory infiltrates in the ganglia of the proventriculus and intestine, and Iba1+ macrophages/microglia were detected in the brain of the cockatiels; however, demyelination was not observed.
In conclusion, the local inflammatory response in the brain of the PaBV-2 experimentally infected cockatiels is largely represented by CD3+ T-lymphocytes and Iba1+ macrophages/microglia. In the gastrointestinal tract, CD3+ T-lymphocytes, PAX5+ B-lymphocytes and Iba1+ macrophages occur in variable proportions over time and between infected birds. These results shed light on the immunopathology of PaBV-2 infection, suggesting that a CD3+ T-cell-mediated mechanism, similar to VSBV-1 and BoDV-1, might mediate the tissue injury in PDD.
Supplemental Material
sj-docx-2-vet-10.1177_03009858211069166 – Supplemental material for Immunophenotype of the inflammatory response in the central and enteric nervous systems of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2
Supplemental material, sj-docx-2-vet-10.1177_03009858211069166 for Immunophenotype of the inflammatory response in the central and enteric nervous systems of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2 by Jeann Leal de Araújo, Raquel R. Rech, Aline Rodrigues-Hoffmann, Paula R. Giaretta, Cinthya Cirqueira, Raphael Rocha Wenceslau, Ian Tizard and Josué Diaz-Delgado in Veterinary Pathology
Supplemental Material
sj-docx-3-vet-10.1177_03009858211069166 – Supplemental material for Immunophenotype of the inflammatory response in the central and enteric nervous systems of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2
Supplemental material, sj-docx-3-vet-10.1177_03009858211069166 for Immunophenotype of the inflammatory response in the central and enteric nervous systems of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2 by Jeann Leal de Araújo, Raquel R. Rech, Aline Rodrigues-Hoffmann, Paula R. Giaretta, Cinthya Cirqueira, Raphael Rocha Wenceslau, Ian Tizard and Josué Diaz-Delgado in Veterinary Pathology
Supplemental Material
sj-pdf-1-vet-10.1177_03009858211069166 – Supplemental material for Immunophenotype of the inflammatory response in the central and enteric nervous systems of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2
Supplemental material, sj-pdf-1-vet-10.1177_03009858211069166 for Immunophenotype of the inflammatory response in the central and enteric nervous systems of cockatiels (Nymphicus hollandicus) experimentally infected with parrot bornavirus 2 by Jeann Leal de Araújo, Raquel R. Rech, Aline Rodrigues-Hoffmann, Paula R. Giaretta, Cinthya Cirqueira, Raphael Rocha Wenceslau, Ian Tizard and Josué Diaz-Delgado in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank the Schubot Avian Health Center and Department of Veterinary Pathobiology at Texas A&M University for the financial support and the Adolf Lutz Institute for the technical cooperation. They also thank the CAPES—Science without borders program for supporting this study.
Supplemental material for this article is available online.
Author’s Note
Aline Rodrigues-Hoffmann is now affiliated to University of Florida, Gainesville, FL.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Schubot Avian Health Center and Department of Veterinary Pathobiology at Texas A&M University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
