Abstract
Increased acute mortality of farmed American alligators (Alligator mississippiensis) was observed in various pens from 2 different farms in Louisiana over 2 years (2019-2021). A total of 14 alligators from multiple events of increased mortality were subjected to postmortem investigations. Except for one alligator with acute neurologic signs, no premonitory signs were observed. All animals had pneumonia (14/14), coelomitis (14/14), and intravascular short Gram-negative bacilli (14/14). Myocarditis (13/14) was common. Yokenella regensburgei was isolated from all alligators tested (13/13). These data suggest the respiratory tract may be a primary target system and could be involved in transmission, either through exhaled bacteria or through swallowing of contaminated respiratory fluids with passage through the feces. Available sensitivity data for Y. regensburgei in this study indicates in vitro sensitivity to aminoglycosides, fluoroquinolones, chloramphenicol, and trimethoprim/sulphamethoxazole antibiotics. Yokenella regensburgei should be included in the differential diagnosis of septicemia and acute death in alligators.
Crocodilians (crocodiles, alligators, caimans, and gavials; 27 species) are widespread vertebrates primarily inhabiting tropical and equatorial regions. Many of the extant crocodilian species are endangered. 9 The American alligator (Alligator mississippiensis) population was at critically low levels during the 1960s; however, a 10-year ban on wild harvest contributed significantly to species’ recovery. Consequently, captive farming and ranching operations developed. In the United States, these are primarily located throughout Louisiana, Florida, Texas, and Georgia. 20
Captive rearing and health assessments have enabled advancement of medical knowledge on alligators. Bacterial disease is one of the most common causes of morbidity and mortality in farmed alligators. 21 Some of the most relevant bacteria are Mycoplasma alligatoris, Chlamydia spp., and Dermatophilus sp.19,21 Here we report the pathologic and bacteriologic analyses suggesting a role of Yokenella regensburgei infection in acute fatal septicemia in farmed American alligators.
Material and Methods
Epidemiologic and Farming Data
Increased acute mortality of farmed American alligators without premonitory signs (except one animal with acute neurologic signs) was observed in various pens from 2 different farms in Louisiana (United States) over a 2-year period (July 2019 and April 2021). Available historical data indicated mortality rates in these 2 farms prior to 2019 were approximately 2% to 3% for hatchlings and 1% for animals older than 3 months. Between 2019 and 2021, mortality rates in young 3- to 10-month-old alligators reached 5% to 10%, and appeared to be concentrated between March through July. Detailed mortality data per day were not available for 2019 and 2020; in March through July 2021, fatalities averaged 3 to 5 per day, and were particularly high in 2 of 25 barns in one of the farms and 5 of 13 barns in the other farm.
These 2 farms housed a total of 3000 to 4500 alligators year-round per barn, divided in 30 to 40 pens with approximately 110 animals per pen. The production system included breeding and/or egg collection from the wild, egg incubation, grow-out, and harvest. Individual pools sheltered approximately 110 individuals. The alligators were fed twice a day. Cleaning and disinfection operations were carried out 3 times per week. Pens were drained one-third per day for cleaning purposes; some were fully drained every 3 weeks. Hatchlings and younger alligators were kept within 31°C to 32°C), up to 4 months of age. Older animals were kept between 27°C to 29°C and primarily between 28.3°C to 28.9°C. Hygienic measures included the combination of chemical and ozone disinfection. There were no management modifications throughout the study period (2019–2021) except for a planned temperature drop (down to 27°C) in April 2021.
Historical confirmed causes linked to sporadic disease in these farms prior to 2019 were Mycoplasma spp. (presumably M. alligatoris), Aeromonas spp., Proteus spp., Escherichia coli, and Pseudomonas spp. An outbreak of chlamydiosis occurred in one of the farms in 2017.
Gross and Histopathologic Postmortem Analysis
A total of 14 alligators were subjected to postmortem investigations. Necropsies followed standard procedures. 12 Histologic sections were routinely prepared and stained with hematoxylin and eosin, and additional histochemical stains on selected samples included acid fast (Ziehl-Neelsen, Fite-Faraco), periodic acid-Schiff, Grocott-Gomori’s methenamine silver, Gram/Twort, and Pierce-van der Kamp modified Gimenez (PVK). Selected specimens of nasal cavity and sinuses were decalcified for 6 h by immersion in 12% (w/w) hydrochloric acid solution (Decal®, StatLab Medical Products, McKinney, U.S.).
Microbiologic and Molecular Analyses
For isolation of bacteria, fresh tissue samples were streaked onto 5% sheep blood agar, tergitol-7 agar and phenylethyl alcohol agar plates followed by aerobic incubation at 37°C for 48 h. Isolated bacterial colonies were identified to genus and species level based on biochemical tests, matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF), and 16sRNA sequencing, if required. Antibiotic susceptibility testing was performed using minimum inhibitory concentration described by Clinical and Laboratory Standards Institute (CLSI). 3 Fresh tissue samples were processed and tested for Mycoplasma spp. by conventional PCR 15 and for Chlamydia spp. 22 (targeting the 16S rRNA gene) and West Nile virus 11 by real-time polymerase chain reaction (RT-PCR) following standard protocols.
Results
Biologic and epidemiologic data are recorded in Supplemental Table S1. Sex distribution was 1 female, 10 males, and 3 not recorded. Age distribution was 8-month-old (n = 6) and 1-year-old (n = 3); 5 alligators were considered juvenile without further specification. All but one case died acutely without identifiable premonitory clinical signs of disease. One of the cases presented with acute neurologic signs characterized by altered locomotion and unresponsiveness. Detailed pathologic data are recorded in Supplemental Table S1. The main gross pathologic findings were pleuritis, (including pleural effusion) (11/11), pneumonia (11/11) (Fig. 1); coelomitis, including coelomic (peritoneal) effusion (7/11) (Fig. 2); pericardial effusion (pericarditis) (7/11); rhinitis (4/11) (Fig. 3); enteritis or enterocolitis (2/11); hepatitis (1/11); subcutaneous edema (1/11); hypopyon (1/11) (Fig. 4); meningitis (1/11) (Fig. 4); and subcutaneous and muscle hemorrhage (1/11).
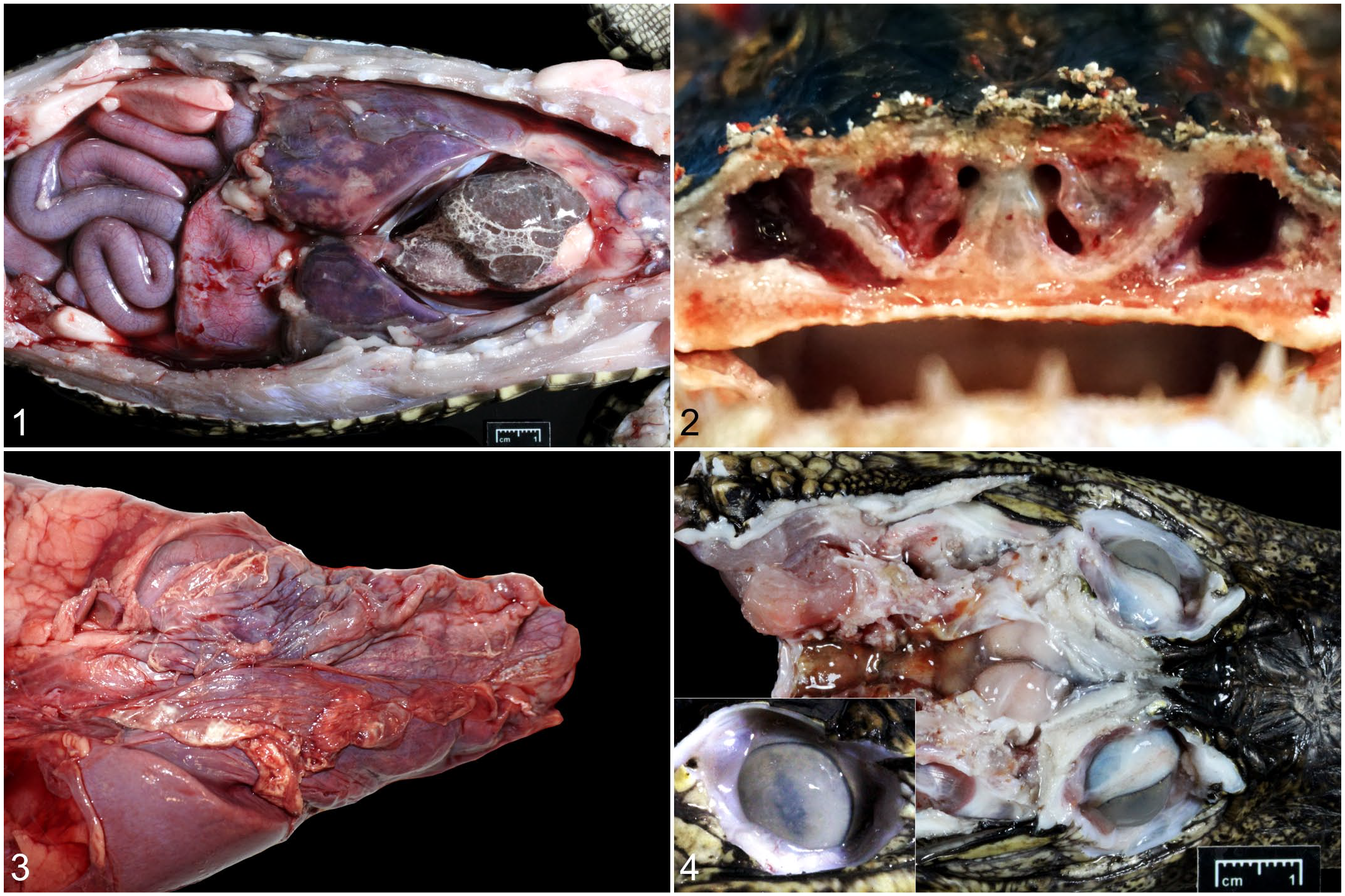
Yokenella regensburgei infection, farmed American alligators.
Microscopically, all 14 cases had pneumonia with pleuritis and pleural exudate (14/14; [affected/evaluated]). Patterns of lung lesions included fibrinoheterophilic pleuropneumonia (6/14), bronchointerstitial pneumonia (3/14), not further specified (3/14), heterophilic bronchopneumonia (1/14), and embolic pneumonia (1/14) (Figs. 5 and 6). Intravascular Gram-negative coccobacillary to bacillary bacteria (Fig. 6) with or without thromboembolism (14/14) were also consistently observed. All cases had coelomitis (14/14) and myocarditis was observed in 13 animals (Fig. 7). The remaining histologic findings were rhinitis (2/2), uveitis (1/1), enterocolitis (6/12), hepatitis (7/14), myonecrosis (4/8), steatitis (4/8), oropharyngitis (1/2), oophoritis (1/2), nephritis (6/13), meningitis (4/11) (Fig. 8), tracheitis (2/8), and splenitis (2/7).
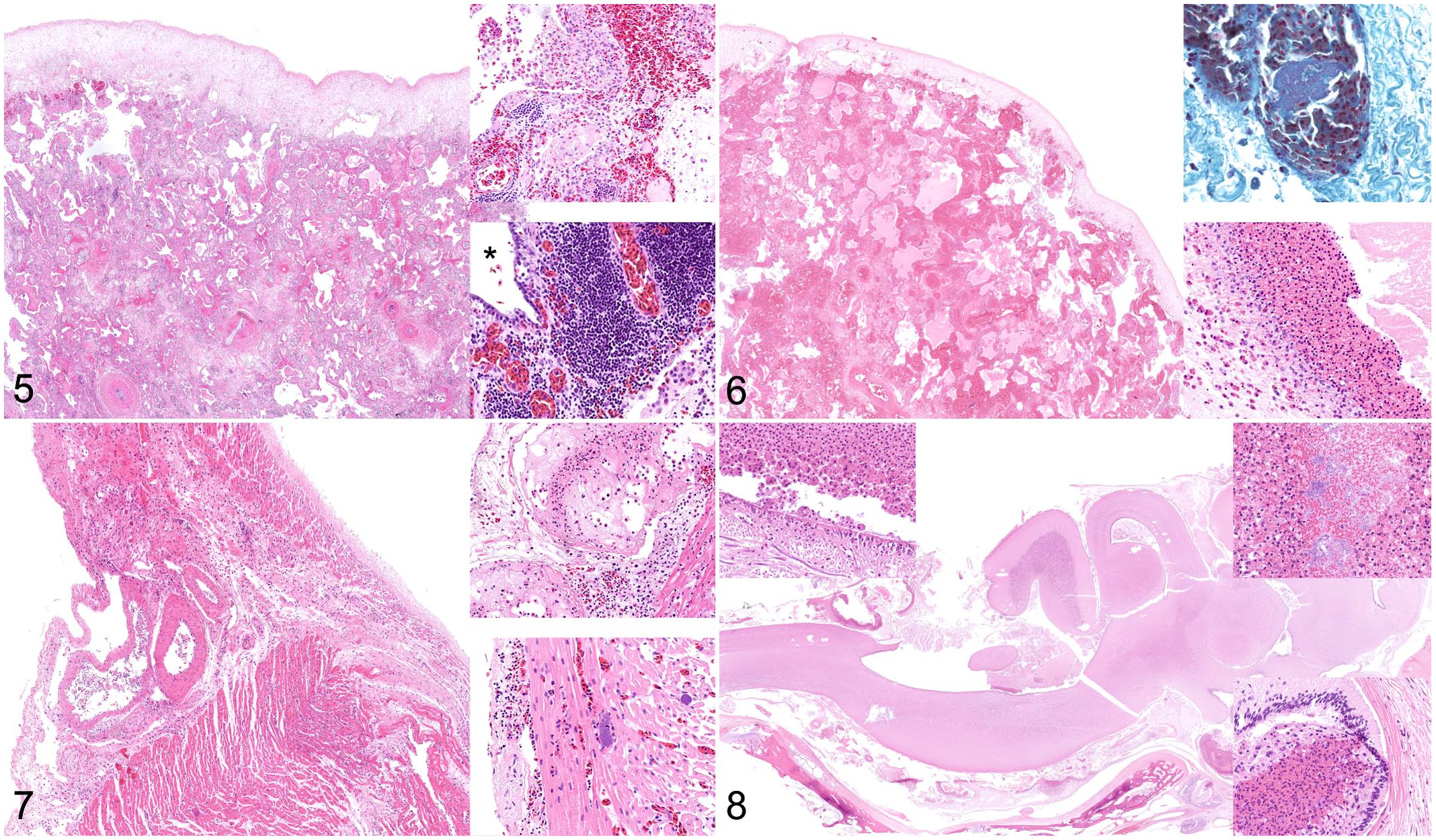
Yokenella regensburgei infection, farmed American alligators.
Bacterial culture and analyses identified Y. regensburgei in 13 of 13 (100%) cases tested. The bacterium was isolated from pleural exudate (6/6 [isolates/tested]), pericardial exudate (5/5), liver (2/2), whole blood (1/1), and lung (3/4) (Supplemental Table S1). One animal had no bacterial culture performed. Concomitant bacterial isolates (all from lung) were: Aeromonas sp. (n = 5), Proteus sp. (n = 2), Escherichia coli (n = 1), Enterococcus sp. (n = 1), Clostridium sp. (n = 1), and mixed bacterial culture (n = 6). All Y. regensburgei isolates were consistently susceptible to aminoglycosides (amikacin and gentamycin), fluoroquinolones (enrofloxacin, marbofloxacin, orbifloxacin, and pradofloxacin), chloramphenicol, and trimethoprim/sulphamethoxazole but they were variably resistant to β-lactams (ampicillin, amoxicillin/clavulanic acid, piperacillin/tazobactam), cephalosporins (cefazolin, cefovecin, cefpodoxime, ceftazidime, and cephalexin), and tetracyclines (doxycycline and tetracycline) (Supplemental Table S2). Mycoplasma spp. nucleic acid was detected in the lung of 6/12 cases, whereas 6/12 were negative. Molecular analyses for West Nile virus (7/7; brain) and Chlamydia spp. (2/2; brain) were negative.
Discussion
These results support a pathogenic role of Yokenella regensburgei in farmed American alligators. The pathologic findings associated with Y. regensburgei infection in these alligators aligns with typical observations in pneumonic and septicemic Gram-negative bacterial diseases in crocodilians.11,21 Grossly, the respiratory system was the most consistently affected, characterized by pleural effusion and pleuritis with various patterns of pneumonia. Coelomic (peritoneal) and pericardial effusions associated with coelomitis and pericarditis were also highly prevalent. Other findings, namely, rhinitis, enteritis, or enterocolitis, subcutaneous edema, and subcutaneous and muscle hemorrhages were common. Multiorgan petechiae, hypopyon, hepatitis, and meningitis were less frequent. Histologic examination confirmed pleuritis and pneumonia in all cases; fibrinoheterophilic pleuropneumonia was most frequent, but few cases presented bronchointerstitial pneumonia. Coelomitis (affecting serosae of multiple organs) was confirmed microscopically in all cases. Myocarditis and multiorgan intravascular emboli of mostly extracellular but also intrahistiocytic gram-negative short-bacillary bacteria were common. Other histologic features were seen at lesser incidence or were recorded in fewer animals.
Yokenella regensburgei was first identified as National Institutes of Health biogroup 9 in Japan. Later, the bacterium was named Koserella trabulsii and classified as Enteric Group 45 by the Centers for Disease Control and Prevention. It was first isolated in Regnesburg, Germany, from the intestine of firebugs (Pyrrhocoris apterus). 14 Recently, Y. regensburgei gained scientific attention for its association with high mortality in pheasantshell mussel (Actinonaias pectorosa) populations in the Clinch River in Virginia and Tennessee, 16 and ebonyshell mussels (Fusconaia ebena) in the Tennessee River, Alabama. 27 In humans, Y. regensburgei is considered a rare opportunistic pathogen with clinical significance, primarily in immunocompromised patients. 17 However, a record in a presumably immunocompetent human patient exists. 24 This bacterium has been isolated from a variety of human clinical specimens including synovial exudate, blood, cellulitis, urine, feces, and sputum, 29 and has been associated with soft-tissue infection with bacteremia, 17 septic arthritis and transient subclinical bacteremia, 1 venous ulcer, 6 osteomyelitis, 24 urinary tract infection, 26 respiratory tract infection, 7 digestive tract infection, 13 brain abscess, 25 and necrotizing fasciitis. 29 Alcoholic steatohepatitis and cirrhosis, infection with human immunodeficiency virus, immunosuppressive therapy, long-term renal disease, and diabetes mellitus have been listed as predisposing factors for Y. regensburgei infections in humans. 29 A possible association between Y. regensburgei infection and the use of untreated and stored water in tanks has been suggested.9,17 Yokenella regensburgei has also been isolated in soil, insects, and wild grass snake (Natrix natrix), where it was of no pathologic significance.14,23
The source of infection in these alligators is unclear, yet the soil, water, food, fomites, and introduced animals are given reasonable consideration. These animals had no evident predisposing factors such as fractures or skin wounds. Environmental variables could have played roles in this scenario. 14 Despite a planned temperature drop (down to 27°C) in April 2021, temperature disturbance was not identified as significant factor in these mortalities. Inappropriate temperature, lighting, and humidity conditions, among others, are key stressors leading to immunological susceptibility and disease occurrence in reptiles including alligators. 5
The mechanism and route of transmission among these alligators is not known. We surmise that the respiratory tract may be a primary target system and could be involved in transmission, either through exhaled bacteria or through swallowing of contaminated respiratory fluids with passage through the feces. The fact that cases have been recorded over 2 years suggests the bacterium could possibly survive in the environment where these alligators are raised or the existence of persistent subclinical respiratory infections; however, both remain to be proven. Also, bacterial cultures of water column from pens with infected alligators performed after the study period (after April 2021) have not yielded Y. regensburgei (data not shown). The bacterium has been isolated only from animals between 3 to 12 months of age and batches of alligators are renewed on a yearly basis (data not shown).
Concomitant bacterial isolates in these cases included Aeromonas sp., Proteus sp., Escherichia coli, Enterococcus sp., and Clostridium sp. From these, Aeromonas is a known cause of disease in crocodilians, 18 but it has also been isolated from healthy individuals. 8 Proteus sp., Escherichia coli, Enterococcus sp., and Clostridium sp. have been isolated from antemortem and postmortem tissue samples in alligators with clinical disease and lesions; however, their pathogenic relevance is uncertain.19,21 Their low prevalence suggests these bacteria did not play a main role in these cases.
Interestingly, Mycoplasma spp. was detected in lung tissue of 6/12 alligators using a PCR assay that amplifies DNA from many different species of mycoplasmas. Some samples yielded multiple bands indicating the presence of more than one mycoplasma. A few yielded what appeared to be single bands, but on Sanger sequencing, multiple peaks were detected throughout the sequence which prevented further analysis. Although no further Mycoplasma speciation was achieved in these cases, M. alligatoris and M. crocodyli are common significant pathogens in crocodilians. Specifically, M. alligatoris is associated with pneumonia, pericarditis, and polyarthritis in natural infections in farmed alligators and experimentally infected alligators.2,3 Histologically, experimentally infected alligators developed pulmonary edema, interstitial pneumonia, pericarditis, myocarditis, meningitis, and synovitis. 2
In this study, despite considerable pathologic similarities with those reported in naturally occurring and experimental mycoplasmosis in alligators,2,4 the following reasons support a main pathogenic role for Y. regensburgei: (1) Y. regensburgei was isolated in all the animals tested; (2) Mycoplasma spp. was identified by PCR (highly sensitive and specific technique) only in 6/12 alligators; (3) intravascular, extracellular, and intrahistiocytic, Gram-negative bacilli (compatible with Y. regensburgei but not with Mycoplasma sp) were histologically visible in 14/14 cases; and (4) polyarthritis was not a pathologic feature in the present alligators. It is possible that Y. regensburgei and Mycoplasma sp. interplayed in those animals with demonstrable coinfection. Prospective experimental studies may be of value to address possible coinfection dynamics of Y. regensburgei and Mycoplasma sp. in alligators. Molecular analyses for West Nile virus and Chlamydia spp. were negative.
Available sensitivity data for Y. regensburgei in this study (Supplemental Table S2) indicates in vitro sensitivity to aminoglycosides, fluoroquinolones, chloramphenicol, and trimethoprim/sulphamethoxazole antibiotics. However, case-specific testing of antibiotic susceptibility is highly recommended to choose the appropriate antibiotic for a particular case, as members of Enterobacteriaceae family have unpredictable susceptibility patterns. Limited data on antibiotic resistance for Y. regensburgei in the literature revealed the presence of ampC genes and expression of potent betalactamases. 28 In this study, Y. regensburgei isolates were variably resistant to β-lactam/penicillin combination, cephalosporin, and tetracycline, which might suggest that multiple clones of Y. regensburgei were present in the farm population. However, this needs to be confirmed by molecular methods like whole genome sequencing and pulsed field gel electrophoresis.
At the time of writing, we have pathologic and bacterial evidence of Y. regensburgei infection in alligator farming operations in Texas (unpublished observations). Future analyses will address potential relevant host, pathogen and environmental factors, sources and routes of transmission, potential for spread to wildlife (because these farms release some animals according to laws in place), and monitoring.
In summary, Y. regensburgei infection was associated with acute mortality in farmed American alligators. The pathologic findings in this cohort of alligators were typical for pneumonia and septicemia due to Gram-negative bacteria. Concomitant pathogens were Mycoplasma spp., Aeromonas sp., Proteus sp., Escherichia coli, Enterococcus sp., and Clostridium sp. Yokenella regensburgei should be included in the differential diagnosis of septicemia and acute death in alligators. These results may be of value to diagnosticians and personnel involved in alligator farming.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858211069165 – Supplemental material for Yokenella regensburgei, a novel pathogen in farmed American alligators
Supplemental material, sj-pdf-1-vet-10.1177_03009858211069165 for Yokenella regensburgei, a novel pathogen in farmed American alligators by Gayathriy Balamayooran, Clark Cooper, Narayan C. Paul, Pamela J. Ferro, Laura Rice, Gabriel Gomez and Josué Díaz-Delgado in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank the histology technicians at TVMDL for their excellent work and commitment.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
