Abstract
Mammary gland neoplasms in macropods are uncommonly reported, and the morphological and immunohistochemical characteristics are incompletely described. The goal of this study was to describe the morphologic features of macropod mammary neoplasms and to determine the molecular subtypes of mammary carcinomas using a panel of antibodies against estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (Her-2), p63, smooth muscle actin (SMA), and epidermal growth factor receptor (EGFR). Biopsy and necropsy specimens were examined from 21 macropods with mammary tumors submitted to Northwest ZooPath from 1996 to 2019. In accordance with the histologic classification of canine mammary tumors proposed by Goldschmidt and colleagues, tubulopapillary (2), tubular (10), and comedo-carcinomas (2), adenoma (1), lobular hyperplasia (3), fibroadenomatous hyperplasia (1), and mastitis (2) were diagnosed. Red kangaroos (Osphranter rufus) were most commonly diagnosed with mammary carcinomas (79% of all carcinomas). Seven carcinomas had lymphovascular invasion and 2 also had pulmonary metastases. Six of these 7 carcinomas were classified as grade 3. Immunohistochemistry (IHC) for all antibodies was performed on 9/14 carcinomas, and partial IHC was performed for 3 cases. All 12 carcinomas were immunoreactive for PR, 5 for ER, 9 for EGFR, and none for Her-2. Five of the 9 mammary carcinomas with complete IHC data were classified as luminal A subtype, and 4 were normal-like subtype. Accurate classification of mammary tumors in macropods based on morphology, immunohistological characteristics, and molecular subtype may be helpful in guiding clinical management, prognosis, and potential therapeutic targets.
The family of Macropodidae, hereafter referred to as macropods, includes kangaroos, tree-kangaroos, and wallabies, as well as wallaroos, quokkas, pademelons, and potoroos. The mammary glands of macropods differ from those of domestic mammals as they do not require a preceding pregnancy to establish lactation and all 4 glands may differentiate independently. 7 Different glands can even produce different types of milk at the same time to simultaneously feed an in-pouch infant as well as a joey that has already left the pouch. 13 The mammary glands of juvenile female macropods consist of few ducts within abundant collagenous stroma and bundles of skeletal muscle with no alveoli. 7 Primary and secondary ducts develop at sexual maturity, and rudimentary alveoli lined by a single layer of cuboidal epithelium are present. 7 Throughout estrus and pregnancy, the mammary glands of macropods remain similar, as opposed to domestic mammals where there is marked glandular development. 13 Lobular architecture begins to emerge during pregnancy; however, mammary glands remain predominantly composed of connective tissue with few alveoli.6,7 These alveoli are round, have small lumina, and secretion may be found near the end of pregnancy. 7 Normal lactation will ensue if a sucking stimulus is applied to the gland at the correct stage of the cycle. 29 By the first week of lactation, there are increased numbers of alveoli with minimal ducting. 6 Ducts and alveoli continue to develop and gradually replace previously prominent connective tissue over the following 4 weeks of lactation, forming secondary and tertiary ducts and increasingly large and defined alveoli.6,7 At week 5 of lactation and beyond, the mammary glands of macropods appear developmentally similar to lactating mammary glands of domestic mammals, notably there is replacement of the abundant connective tissue by ducts and secretory alveoli. 6
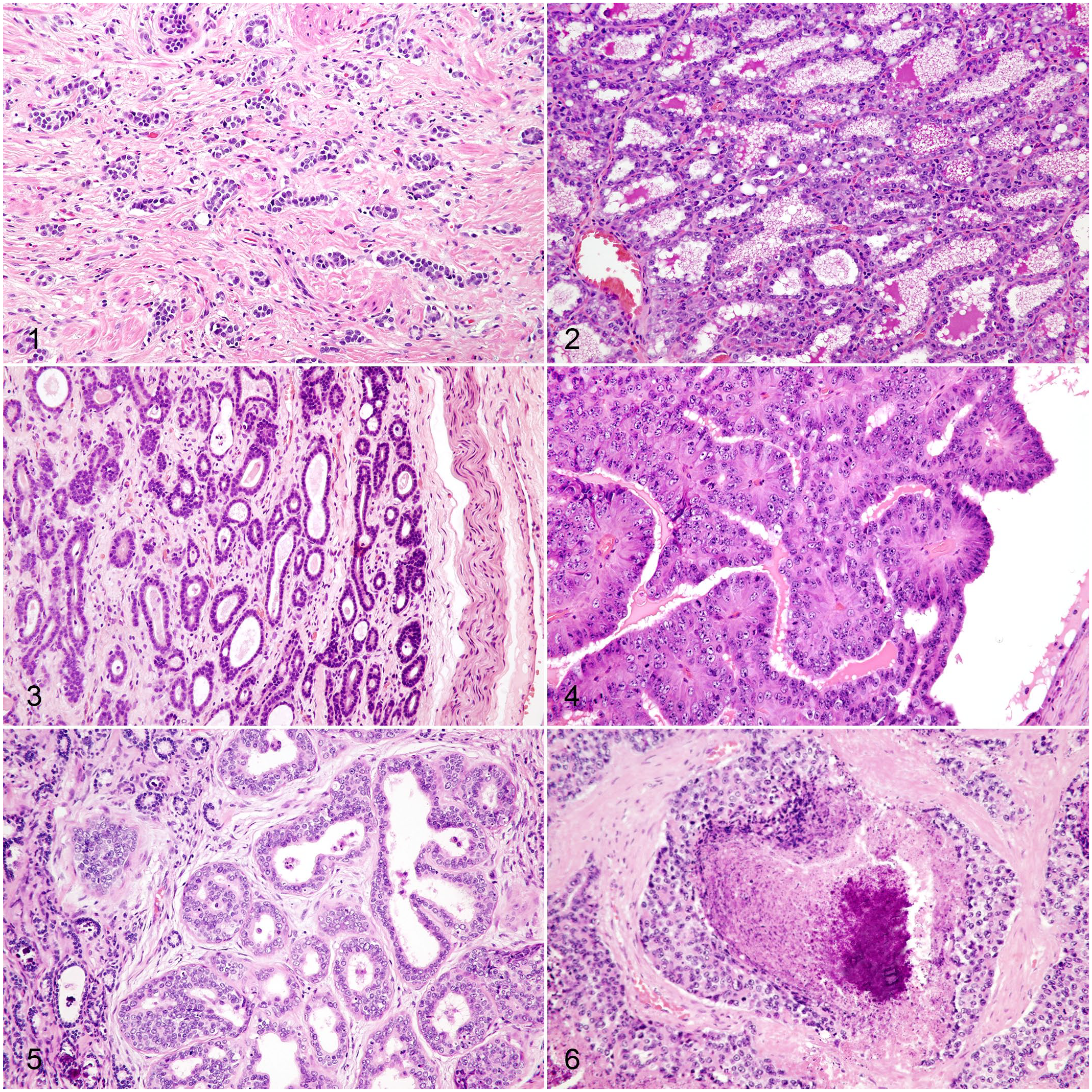
Histologically, the nonlactating mammary gland of macropods appears relatively amorphous and distinct lobules are difficult to recognize as the normal architecture is obscured by abundant amounts of collagenous stroma. 13 Tubules and acini are small, with indistinct lumina, lined by a single layer of cuboidal epithelial cells,6,17 and are widely separated by moderately cellular connective tissue that contains blood vessels and striated muscle (Fig. 1). Glandular epithelial cells have minimal eosinophilic cytoplasm, round to ovoid nuclei, euchromatic chromatin, and occasionally a single small nucleolus. There is absence of distinct lobules composed of alveoli with prominent open lumina, attenuated epithelium with foamy cytoplasm, and complex ductal structures.6,7,17
While macropods are commonly kept in zoological collections in North America, there are few reports of neoplastic diseases in any macropods,2,27,31–33 including a limited number of mammary carcinomas.2,33 Despite the few published reports, mammary gland neoplasms are 1 of the most commonly diagnosed neoplasms in macropods submitted to Northwest ZooPath from 1996 to 2019. Since mammary tumors are among the most common neoplasms in dogs and cats, much progress has been made to accurately diagnose and prognosticate their mammary tumors based on morphological and immunohistochemical characteristics.11,28,37 Furthermore, breast cancer represents 1 of the most common neoplastic diseases in humans, and some feline and canine mammary tumors have been proposed as models for human breast tumors.12,23,35
To date, there is no standardized histologic classification of mammary tumors in macropods. The establishment of a standardized classification system is important for 2 primary reasons. First, it allows for the comparison between studies and across species. Second, a consistently accurate diagnosis of a specific mammary tumor can be helpful in guiding clinical management. Prognosis based on histologic types of macropod mammary neoplasms has yet to be demonstrated, but the standardization of the morphologic diagnoses will aid in this endeavor. To facilitate further research in macropod mammary tumors, this study investigated our ability to apply the canine mammary tumor classification system proposed by Goldschmidt and colleagues to macropods. 11 Furthermore, the immunohistochemical characteristics of macropod mammary tumors have yet to be studied. The expression or the lack of various combinations of receptors and other antigens are the foundation for the different molecular subtypes of human breast cancer. These subtypes represent the current standard for prognostication and treatment of human breast cancer.8,26 Efforts have been made to utilize similar classification systems in dogs. An additional goal of our study was to apply a panel of antibodies targeting estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (Her-2), p63, smooth muscle actin (SMA), and epidermal growth factor receptor (EGFR) to investigate the translatability of the immunophenotypical classification of canine mammary carcinomas or human breast carcinomas to macropod mammary carcinomas.
Materials and Methods
Case Selection
A retrospective search was performed of all macropod cases submitted to Northwest ZooPath between 1996 and 2019 and identified 1036 macropod cases including 13 eastern gray kangaroos (Macropus giganteus), 373 red kangaroos (Osphranter rufus), 18 western gray kangaroos (Macropus fuliginosus), 18 nonspeciated kangaroos, 52 Matschie’s tree-kangaroo (Dendrolagus matschiei), 38 nonspeciated tree-kangaroos (Dendrolagus sp.), 1 long-nosed rat kangaroo (Potorous tridactylus), 9 brush-tailed bettongs (Bettongia penicillata), 8 agile wallabies (Macropus agilis), 7 black-striped wallabies (Macropus dorsalis), 81 Parma wallabies (Macropus parma), 147 red-necked wallabies (Macropus rufigriseus), 10 swamp wallabies (Wallabia bicolor), 76 Tammar wallabies (Macropus eugenii), 68 yellow-footed rock wallabies (Petrogale xanthopus), 8 nonspeciated rock wallabies (Petrogale sp.) and 109 nonspeciated wallabies. Mammary tumors had been diagnosed in 13 red kangaroos (O. rufus), 1 Matschie’s tree-kangaroo (D. matschiei), 1 yellow-footed rock wallaby (P. xanthopus), 1 red-necked wallaby (M. rufogriseus), and 1 Parma wallaby (M. parma). In addition, a search of the archives of the Veterinary Diagnostic Laboratory (VDL) at Michigan State University identified mammary tumors in 3 Matschie’s tree-kangaroos (D. matschiei) and 1 Parma wallaby (M. parma) between 2016 and 2019. These cases were also included in the study. Patient data included age and sex. All neoplasms had been submitted from zoos, as either surgical biopsies or had been discovered at necropsy.
Histopathology
All mammary tumors had been fixed in 10% neutral-buffered formalin and sections had been routinely processed into paraffin blocks. Paraffin-embedded tissues were sectioned at 3 μm, mounted on frosted glass slides, stained with hematoxylin and eosin (HE), and examined by light microscopy.
Mammary tumors were classified morphologically in accordance with the classification system for canine mammary tumors proposed by Goldschmidt et al. 11 For all carcinomas, a histologic grade was determined in accordance with the grading system established for canine mammary carcinomas by Peña et al. 19 According to this system, points were assigned based on tubule formation, nuclear pleomorphism, and mitotic count. The sum of the points assigned for these criteria determined the histologic grade (Table 1).
Histologic grading of mammary carcinomas according to Peña et al. 19

Abbreviation: HPF, high-power field.
Immunohistochemistry
Based on availability of sufficient tissue in paraffin blocks, serial sections from 12 mammary carcinomas were immunohistochemically labeled with antibodies against p63, SMA, ER, PR, Her-2, and EGFR as previously published.15,34,35 Deparaffinization, antigen retrieval, immunohistochemical labeling, and counterstaining were performed on the BOND-MAX automated staining system (Leica BioSystems, Buffalo Grove, Illinois) using a diaminobenzidine (DAB) detection system (Leica BioSystems) for p63, EGFR, SMA or on the Discovery Ultra automated staining system (Ventana Medical Systems, Tucson, Arizona) using an alkaline phosphatase detection system (Ventana Medical Systems) for ER, PR, and Her-2. Information on the primary antibodies is listed in Table 2. Canine mammary gland, canine uterus, and canine basal cell carcinoma tissues, normal wallaby mammary gland tissue, normal red kangaroo mammary gland (Supplemental Figs. 8, 10, 12), uterus (Supplemental Figs. 1, 3), skin (Supplemental Figs. 7, 9), smooth muscle (Supplemental Fig. 11), hyperplastic mammary gland (Supplemental Figs. 2, 4), mammary carcinoma (Supplemental Fig. 5), and squamous cell carcinoma (Supplemental Fig. 6) from red kangaroos were used as the positive control tissues. For negative controls, the primary antibodies were replaced with homologous nonimmune sera.
Primary antibodies used for IHC.
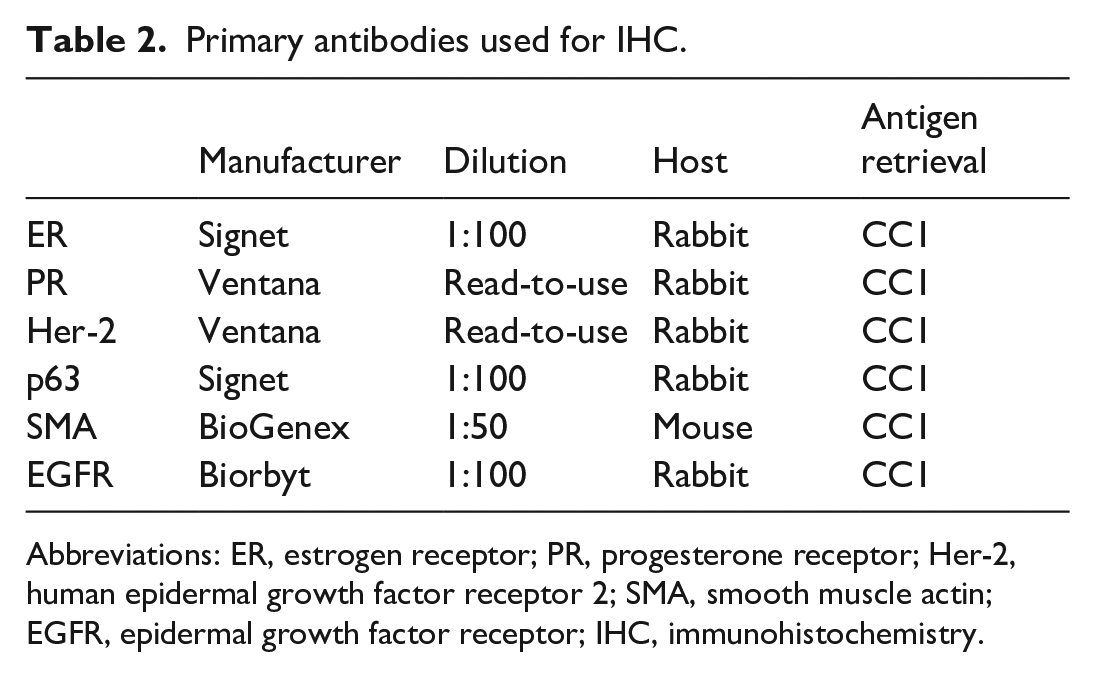
Abbreviations: ER, estrogen receptor; PR, progesterone receptor; Her-2, human epidermal growth factor receptor 2; SMA, smooth muscle actin; EGFR, epidermal growth factor receptor; IHC, immunohistochemistry.
Interpretation of Immunohistochemical Labeling
Cytoplasmic labeling within neoplastic cells was evaluated as positive for SMA, perimembranous labeling was evaluated as positive for EGFR, and nuclear labeling was evaluated as positive for p63. The Allred scoring system was used to evaluate ER and PR immunolabeling1,20 (Table 3). The Allred score is the summation of the proportion score (PS) and intensity score (IS). Proportion score accounts for the percentage of immunoreactive cells, while IS accounts for the intensity of the reaction product. The maximum total value (Allred score) is 8. An Allred score of 0–2 was considered negative, and a score of 3–8 was considered positive. Only nuclear labeling of neoplastic cells was counted in accordance with the Allred scoring system. The guidelines of the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) were applied to evaluate Her-2 (Table 4) expression. 36 IHC scores of 0 to 1+ were interpreted as negative, and Immunohistochemistry (IHC) scores of 2+ and 3+ were interpreted as positive. 20
Estrogen and progesterone receptor evaluation according to Allred et al. 1
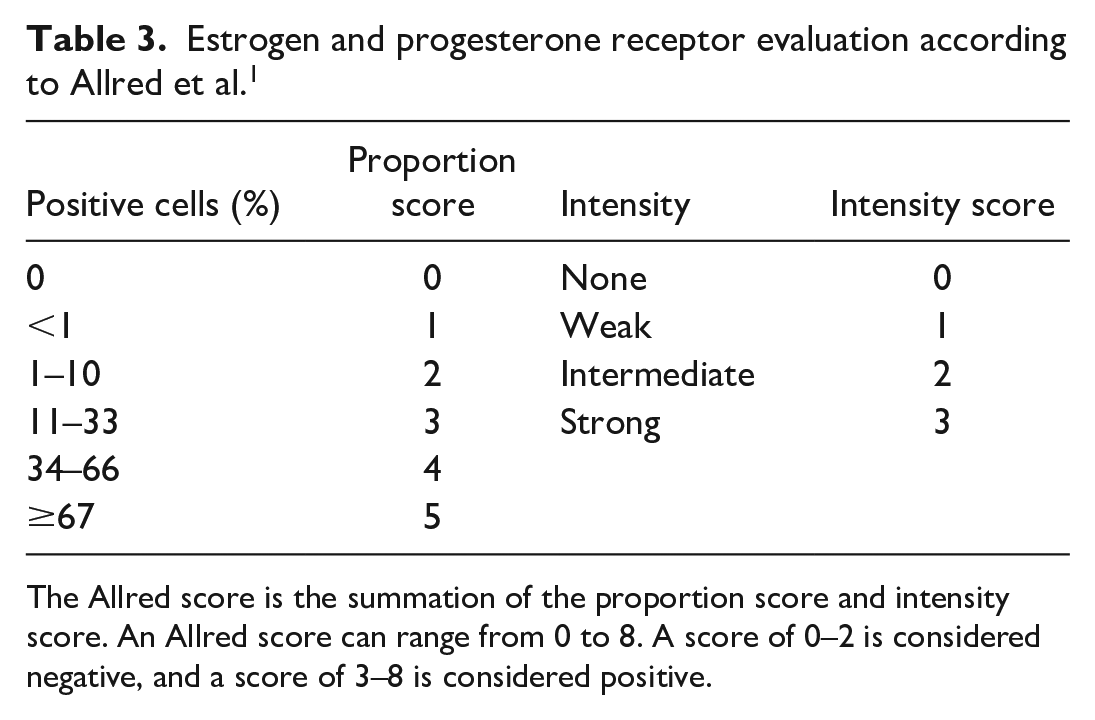
The Allred score is the summation of the proportion score and intensity score. An Allred score can range from 0 to 8. A score of 0–2 is considered negative, and a score of 3–8 is considered positive.
Her-2 evaluation according to Wolff et al. 36

IHC scores of 0 to 1+ were interpreted as negative, and IHC scores of 2+ and 3+ were interpreted as positive. 20
Abbreviation: Her-2, human epidermal growth factor receptor 2; IHC, immunohistochemistry.
Molecular Classification
Molecular subtypes, including luminal A, luminal B, Her-2 overexpressing, basal-like, and normal-like, were determined in accordance with previous published studies in dogs9,28 (Table 5).
Molecular subtypes in mammary carcinomas according to Gama et al., 2008 and Sassi et al., 2010.
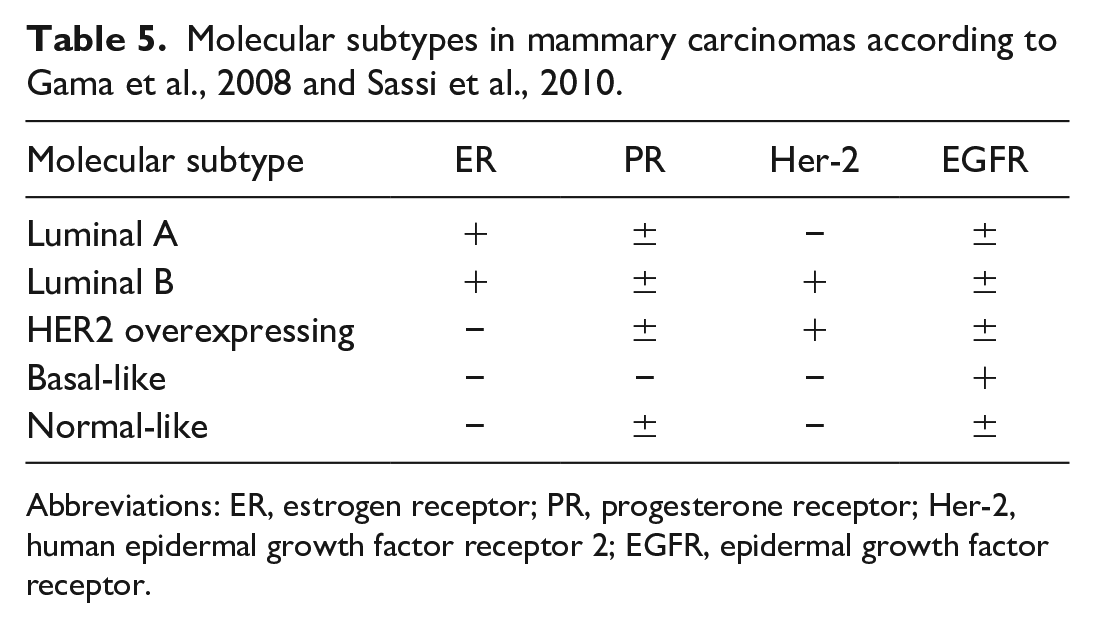
Abbreviations: ER, estrogen receptor; PR, progesterone receptor; Her-2, human epidermal growth factor receptor 2; EGFR, epidermal growth factor receptor.
Results
Epidemiological Findings
A total of 1040 macropod cases were reviewed for this study. These cases included 423 kangaroos, 90 tree-kangaroos, 10 rat-kangaroos, and 517 wallabies. We identified 76 macropods with neoplastic lesions, of which 15 (1.4% of all cases and 19.7% of all neoplasms) were of mammary gland origin. Mammary gland neoplasms and squamous cell carcinomas (15, 1.4%) were most common, followed by endocrine neoplasms (12, 1.2%) including 9 pheochromocytomas. Other neoplasms included cases of lipoma (4), squamous papilloma (3), granulosa cell tumor (3), transitional cell carcinoma (3), thyroid adenoma (2), sarcoma (2), and individual cases of myelolipoma, lymphoma, leiomyoma, leiomyosarcoma, hepatocellular carcinoma, pulmonary adenocarcinoma, ovarian papillary cystadenoma, gastric adenocarcinoma, colonic adenocarcinoma, and carcinoma of unknown origin (8). Red kangaroos were the most common species in this study (373) and the species most commonly affected by mammary lesions (13/21 mammary lesions, 11/14 mammary carcinomas) representing a prevalence of 3.5% for mammary lesions and 2.9% for mammary carcinomas, respectively. The remaining 3 mammary carcinomas were identified in 2 different wallaby species and a Matschie’s tree-kangaroo (see Table 6). The mean age of red kangaroos with mammary carcinomas was 14 years (ranging from 12 to 19 years; median of 13 years), while the Matschie’s tree-kangaroo was 21 years old and the red-necked and Parma wallabies were 4 and 12 years old, respectively. Pulmonary metastases were found in 2 of the 14 carcinomas, both of which were in red kangaroos. Six carcinomas presented grossly as a single mass that ranged from 2 to 6 cm in diameter, 2 presented as multiple masses, and 1 presented as a firm mammary gland. All macropods with mammary lesions included in this study were females. Twelve of the mammary gland lesions were collected as an excisional biopsy during surgery, and the other 9 were collected at necropsy.
Histologic subtypes and histologic grade of macropod mammary carcinomas.
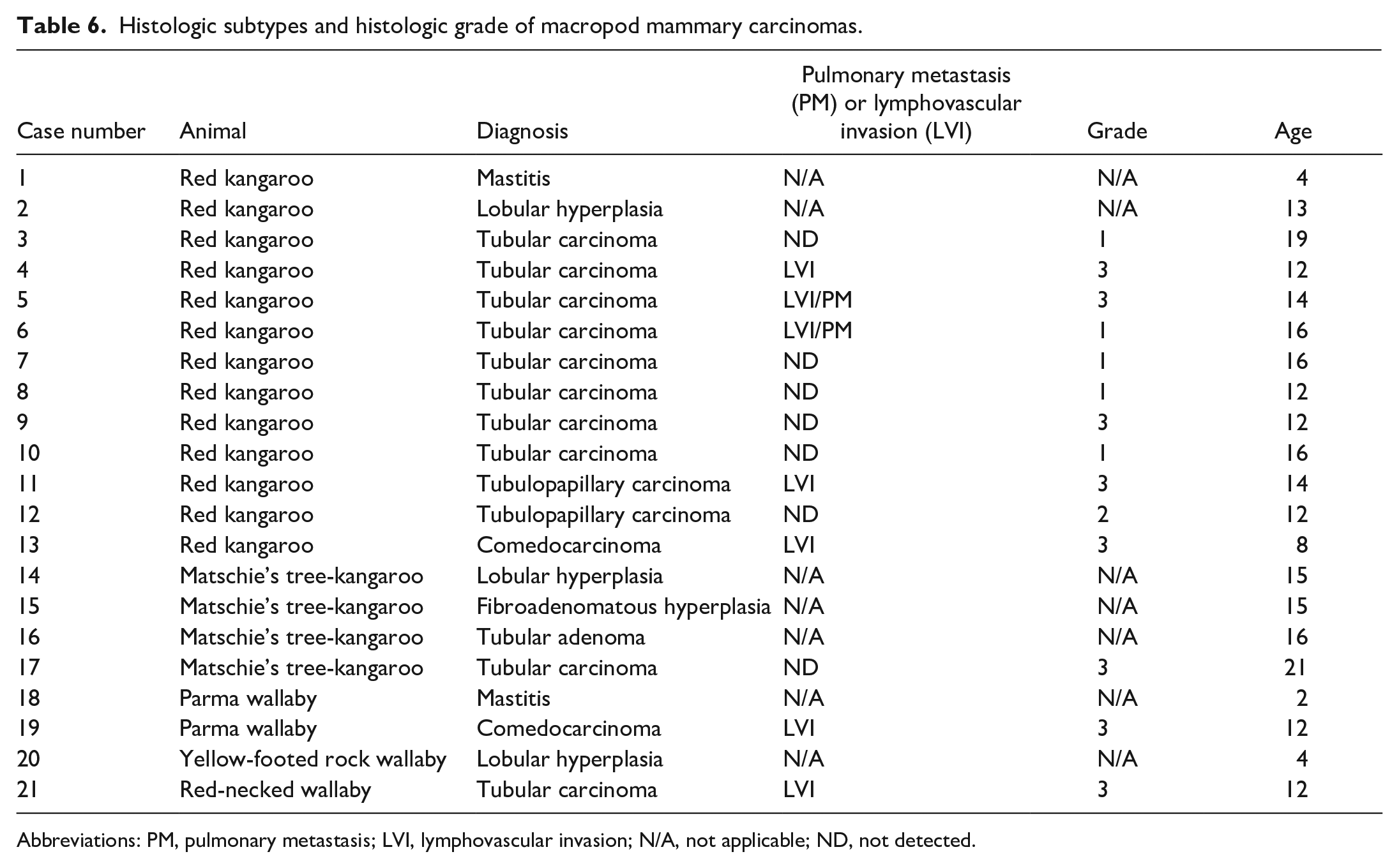
Abbreviations: PM, pulmonary metastasis; LVI, lymphovascular invasion; N/A, not applicable; ND, not detected.
Histologic Findings
Three mammary lesions were diagnosed as lobular hyperplasia (cases: 2, 14, and 20), and 1 was diagnosed as fibroadenomatous hyperplasia (case 15). Hyperplastic glands were characterized by the proliferation of well-differentiated acinar and tubular structures that did not compress or invade the adjacent tissue. Acini and tubules were lined by a single layer of well-differentiated cuboidal to columnar epithelial cells. Epithelial cells exhibited minimal anisocytosis and anisokaryosis and maintained nuclear polarity. Individual glands were separated by thin streams of fibrovascular stroma (Fig. 2). Fibroadenomatous hyperplasia was characterized by concurrent proliferation of fibroblasts within an abundant collagen matrix.
One case was diagnosed as a tubular adenoma (case 16). The adenoma was a well-demarcated mass that compressed the surrounding tissue and was composed of irregular tubules supported by bands of collagenous stroma (Fig. 3). These tubules were lined by 1 to occasionally 2 layers of cuboidal epithelial cells. Lining epithelial cells exhibited mild anisocytosis and anisokaryosis and maintained nuclear polarity. There were 3 mitoses per 10 contiguous high-power fields (HPF, 400×, 2.37 mm2).
Fourteen mammary tumors were diagnosed as carcinomas. All mammary carcinomas were poorly demarcated, infiltrative masses, and 7 of the carcinomas had lymphovascular invasion (7 cases: 4, 5, 6, 11, 13, 19, and 21). These carcinomas had tubulopapillary (Fig. 4; 2 cases: 11 and 12), tubular (Fig. 5; 10 cases: 3, 4, 5, 6, 7, 8, 9, 10, 17, and 21), or solid/comedo-like (Fig. 6; 2 cases: 13 and 19) patterns. Tubulopapillary carcinomas were composed predominantly of tubules and papillae-like structures, although markedly ectatic tubular lumina did not have papillary infoldings. Neoplastic cell-lined papillae extended into tubular lumina and were supported by a fine fibrovascular stroma. Neoplastic cells were piled along the basement membranes, and had distinct cell boundaries, moderate amounts of eosinophilic cytoplasm, and variable loss of nuclear polarity. These cells were cuboidal to polygonal and exhibited moderate anisokaryosis. Nuclei were round to ovoid, generally basaloid, and chromatin density was highly variable, with either single prominent or multiple indistinct nucleoli. An average of 34 mitoses (ranging from 30 to 38) were observed in 10 contiguous HPF (2.37 mm2). One case (case 11) had evidence of lymphovascular invasion.
Tubular carcinomas were the most commonly observed subtype and composed predominantly of neoplastic cells arranged in tubules. Tubules were lined by 1–2 layers of neoplastic cells in well-differentiated areas, while in poorly differentiated areas, neoplastic tubules were up to 10 cells thick with loss of nuclear polarity. The neoplastic cell morphology was variable, ranging from cuboidal to columnar to polygonal. Cell borders were variably distinct. Neoplastic cells typically had small to moderate amounts of eosinophilic cytoplasm. Nuclei were round to ovoid, occasionally basaloid, and ranged from hyperchromatic to normochromatic to hypochromatic among the cases. Nuclei typically had single, prominent nucleoli. Mitotic activity was variable among cases with an average of 30.2 mitoses (ranging from 10 to 53) in 10 contiguous HPF (2.37 mm2). There were variable amounts of collagenous intertubular stroma that was infiltrated by varying numbers of lymphocytes, plasma cells, and histiocytes. Tumor invasion into the surrounding tissue invoked a desmoplastic response and 4 cases had lymphovascular invasion (cases: 4, 5, 6, 21). The combination of tissue invasion (with or without lymphovascular invasion), cellular pleomorphism, and mitotic activity readily differentiated tubular carcinomas from lobular hyperplasia and tubular adenoma.
Comedocarcinomas presented as solid carcinomas with central areas of necrosis. Necrotic centers were composed of abundant amounts of amorphous eosinophilic material mixed with cellular debris and degenerate neutrophils. Few necrotic centers were mineralized. The neoplastic cells that surrounded the necrotic centers were arranged in solid sheets supported by variably thick bands of fibrovascular stroma. Neoplastic cells had moderate amounts of eosinophilic cytoplasm. Nuclei were round to ovoid and contained single or multiple prominent nucleoli. Nests of neoplastic cells were commonly surrounded by hyalinized stroma characteristic of desmoplasia. Cellular and nuclear morphology was as described for the tubular carcinomas. An average of 17 mitotic figures (ranging from 15 to 19) was observed in 10 contiguous HPF (2.37 mm2). Both cases had evidence of lymphovascular invasion.
None of the examined neoplasms exhibited osseous or cartilaginous metaplasia or proliferation of myoepithelial cells; therefore, mixed or complex mammary tumors were not observed. Cases 1 and 18 were diagnosed as mastitis and characterized by acini containing large amounts of eosinophilic fluid admixed with neutrophils and lymphocytes. The supporting stroma was infiltrated by moderate numbers of scattered lymphocytes and fewer plasma cells.
Based on the canine mammary carcinoma grading system established by Peña et al. 19 (Table 1), 8 carcinomas were classified as grade 3 mammary carcinomas (cases: 4, 5, 9, 11, 13, 17, 19, and 21), 1 as grade 2 (case 12), and 5 as grade 1 (cases: 3, 6, 7, 8, and 10). Six of the 8 grade 3 carcinomas (cases: 4, 5, 11, 13, 19, and 21) exhibited lymphovascular invasion, and 1 exhibited pulmonary metastasis (case 5). One red kangaroo with a grade 1 tubular carcinoma exhibited lymphatic invasion and pulmonary metastasis (case 6). Five of the 7 animals with mammary carcinomas that exhibited lymphatic invasion or pulmonary metastasis (cases: 4, 5, 6, 11, and 13) were red kangaroos.
All 5 grade 1 carcinomas were tubular carcinomas. The 1 grade 2 carcinoma was a tubulopapillary carcinoma. Grade 3 carcinomas comprised 3 histologic subtypes, 5 tubular, 1 tubulopapillary, and 2 comedocarcinoma(s). Both comedocarcinomas had evidence of lymphovascular invasion.
Immunohistochemical Findings
There was sufficient tissue to perform IHC on 9/14 carcinomas (Table 7; cases: 3, 4, 6, 7, 10, 11, 12, 17, 21), while for 3 cases, there was only sufficient tissue for a partial analysis (cases: 8, 9, 13). All 12 carcinomas were positive for PR. Five of the 9 carcinomas were also immunoreactive for ER (cases: 3, 4, 7, 10, and 17). Four cases (cases: 3, 6, 10, and 21) had a Her-2 IHC score of 1+, and all remaining cases had a score of 0. According to the evaluation criteria, all 9 carcinomas were therefore negative for Her-2. None of the examined carcinomas were immunoreactive for p63 or SMA. Nine carcinomas were immunoreactive for EGFR (cases: 3, 6, 7, 8, 9, 10, 12, 17, and 21). Based on these immunohistochemical profiles, 5/9 mammary carcinomas with complete IHC data were classified as molecular subtype luminal A (cases: 3, 4, 7, 10, and 17) and 4/9 as molecular subtype normal-like (cases: 6, 11, 12, and 21).
Molecular subtypes of macropod mammary carcinomas.
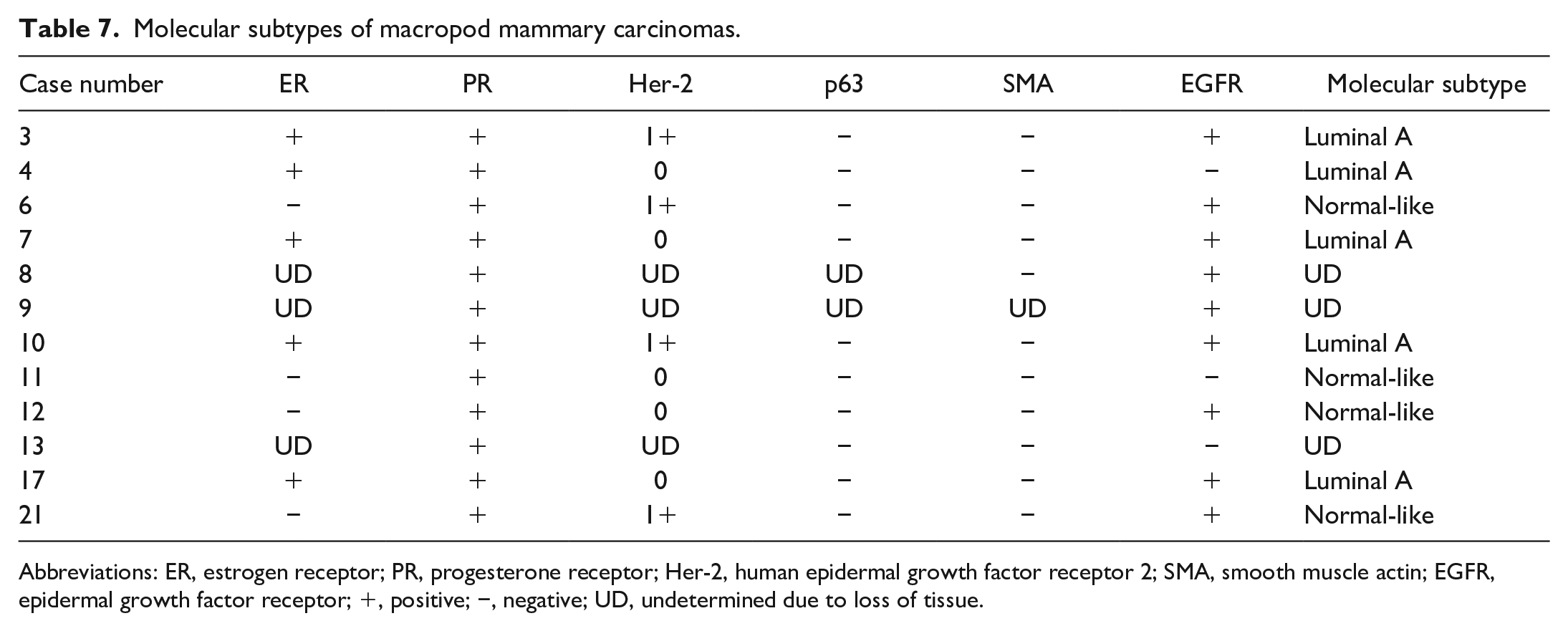
Abbreviations: ER, estrogen receptor; PR, progesterone receptor; Her-2, human epidermal growth factor receptor 2; SMA, smooth muscle actin; EGFR, epidermal growth factor receptor; +, positive; −, negative; UD, undetermined due to loss of tissue.
Discussion
Based on our data set and previous publications, neoplasms appear to be uncommon among macropods in zoological collections. Only 76 (7.3%) neoplasms were diagnosed from 1040 submissions. Our data set spans a 23-year period and reflects a wide range of geographic locations and animals at different ages and of different sex thereby strengthening the argument that macropods are at lower risk for developing neoplasms compared with other mammals. The low prevalence of neoplasms in macropods is in sharp contrast to companion animals, such as domestic dogs, where 1 study found that 58.7% of surgical biopsies represented neoplasms. 25
Interestingly, 61.9% of all mammary lesions and 78.6% of all mammary carcinomas occurred in red kangaroos, representing 35.9% of the macropods in this study. The results of our study are similar to a previously published survey of neoplasms in red kangaroos, 33 that reported mammary carcinomas and squamous cell carcinomas as the 2 most prevalent neoplasms. Prior to our study, only a single case of a mammary carcinoma was reported in a macropod other than a red kangaroo, a Parma wallaby. 2 While our study population only included 81 Parma wallabies, 2 of the 21 mammary lesions were also observed in this species, including 1 of the highly aggressive comedocarcinomas. Furthermore, 4 mammary lesions, including 2 mammary carcinomas, were presented in Matschie’s tree-kangaroos, a species that comprised only 55 of the 1040 cases. As 3 of these cases originated from the archives of the VDL, the slightly higher prevalence in this species may indicate a selection bias.
Overrepresentation of mammary carcinoma in 1 species within a family is not unprecedented. A well-known parallel can be found in the family of Muridae, where mammary carcinomas are more prevalent in mice (Mus musculus) compared with rats (Rattus rattus). 22 In the Macropodidae-Muridae parallel, benign mammary neoplasms (i.e. fibroadenomas) are more prevalent in rats compared with mice, and benign mammary tumors are more common in macropods other than red kangaroos. The reason for this species variation between members of the same family remains to be elucidated.
To study mammary tumors in macropods, correct distinction between normal nonlactating mammary gland and its neoplastic counterparts, and consistent morphologic classification of these tumors are paramount. As previously described, the nonlactating mammary gland of macropods is morphologically distinct from domestic mammalian species. The large amounts of well-organized collagenous stroma that widely separates regressing acini and ducts should not be mistaken for a sclerosing carcinoma. Desmoplasia caused by mammary carcinomas in macropods is characterized by hyalinized stroma with minimal cellular infiltrates and associated with proliferations of pleomorphic glandular epithelial cells.
The current lack of a classification system necessitated the use of comparative studies to establish morphological type and grade for the various entities. Despite the distinct architecture of the macropod mammary gland, we applied the canine mammary tumor classification system to morphologically classify macropod mammary tumors. 11 By utilizing the canine mammary classification for macropod tumors, we were also able to apply the current grading system for mammary carcinomas to macropod mammary carcinomas. 19 While the canine mammary tumor classification system and the grading system have prognostic importance and predict local recurrence, metastasis, and survival, 24 the prognostic value of applying the canine classification and grading to macropod mammary carcinomas remains unknown. However, some parallels between macropod and canine mammary carcinomas were observed. First, pulmonary metastasis and/or lymphovascular invasion was observed in the majority (6/8) of grade 3 carcinomas, while only 1 (1/5) grade 1 mammary carcinoma had lymphovascular invasion and pulmonary metastasis. The metastatic rate for grade 3 macropod mammary carcinomas was 75%, similar to the metastatic rate of 87% reported for canine grade 3 mammary carcinomas. 24 Second, both macropod mammary comedocarcinomas were grade 3 carcinomas with lymphovascular invasion. This entity has been shown to have aggressive biological behavior and to metastasize in 82% of dogs. 24 These limited data support that the canine mammary classification and grading system may have a similar prognostic importance in macropods as in dogs.
Estrogen receptor expression in human breast cancer is related to disease-free survival and overall survival. Furthermore, women with ER-expressing breast cancer have improved response rates with antiestrogen therapy, such as tamoxifen.3,10 Progesterone receptor expression was also correlated with a favorable prognosis. 21 However, overexpression of Her-2 is associated with decreased overall survival and relapse. 30 These early studies using ligand-binding assays examined ER, PR, or Her-2 alone. With the advent of an IHC-based classification system based on evaluating the combination of ER, PR, and Her-2 expression, 14 prior classifications based on ER and PR or Her-2 alone were found to be inferior for prognostic and therapeutic information. 18 Early veterinary literature similarly examined the prognostic importance of ER or PR alone; 16 however, recent publications have followed the human classification system.9,28 Two studies combined the analysis of ER, PR, and Her-2 with the expression patterns of other antigens such as CK5/6, EGFR, CK14, p63, and P-cadherin to develop an immunohistochemical classification system for canine mammary carcinomas that closely mimics its human counterpart.9,28
This study demonstrated the translatability of the canine mammary carcinoma immunohistochemical classification system to macropod mammary carcinomas, based on a panel of antibodies targeting ER, PR, Her-2, p63, SMA, and EGFR. One limitation of this study is the lack of extensive validation of the applied antibodies in macropod tissues. Regardless, immunohistochemical labeling was interpretated in combination with microscopic findings. The immunoreactivity of the different antibodies in normal mammary tissues and tumors of wallabies and red kangaroos was morphologically similar to the expression patterns observed in canine mammary tissues and tumors, thereby, supporting cross-reactivity of these antibodies (Supplemental Figs. S1–S12). This immunohistochemical classification of mammary carcinomas is important because it allows for the additional classification by molecular subtype that has been directly linked to outcome and therapeutic response in human breast cancer.4,5 Similar prognostic significance has been shown for some molecular subtypes, especially an aggressive behavior for the basal-like subtype, in dogs. 9 Given that all carcinomas included in this data set were ER and/or PR positive and Her-2 negative, they were subtyped as luminal A or normal-like subtype, a classification that would provide little additional prognostic information. Whether luminal A carcinomas in macropods are also candidates for antiestrogen therapies has to be investigated in future studies.
In conclusion, we were able to demonstrate the translatability of the morphologic, immunohistologic, and molecular classification system of canine mammary tumors to mammary tumors of macropods. The application of these classification systems to macropods may lead to more consistent classification of mammary tumors in macropods and potentially to the identification of important prognostic factors and tumor type-specific susceptibility to therapy.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858211069164 – Supplemental material for Morphological and immunohistochemical characterization and molecular classification of spontaneous mammary gland tumors in macropods
Supplemental material, sj-pdf-1-vet-10.1177_03009858211069164 for Morphological and immunohistochemical characterization and molecular classification of spontaneous mammary gland tumors in macropods by Tu Chun Hsu, Michael M. Garner and Matti Kiupel in Veterinary Pathology
Supplemental Material
sj-tiff-2-vet-10.1177_03009858211069164 – Supplemental material for Morphological and immunohistochemical characterization and molecular classification of spontaneous mammary gland tumors in macropods
Supplemental material, sj-tiff-2-vet-10.1177_03009858211069164 for Morphological and immunohistochemical characterization and molecular classification of spontaneous mammary gland tumors in macropods by Tu Chun Hsu, Michael M. Garner and Matti Kiupel in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank the Michigan State University Veterinary Diagnostic Laboratory histology section for performing the immunohistochemistry for this study. The authors are also grateful to Cathy Minogue of Northwest ZooPath for data retrieval, and Leroy Brown of Histology Consulting Service for original slide preparation. Dr Tu Chun “Steven” Hsu is a Molecular Pathology Fellow in the National Institutes of Health (NIH) Comparative Biomedical Scientist Training Program supported by the National Cancer Institute in partnership with Michigan State University.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
