Abstract
Medullary bone is a calcium-rich, labile bone normally occurring in female birds with each egg-laying cycle. The stimulus for formation of medullary bone is, in part, the cyclic increase in serum estrogens produced by preovulatory ovarian follicles. Increased bone density due to formation of medullary bone, particularly in pneumatic bones, has been termed polyostotic hyperostosis, even if physiologic. This study investigated the formation of medullary bone in nonpneumatic (femur) and pneumatic (humerus) bones in sexually mature male budgerigars submitted for autopsy. Of the 21 sexually mature male budgerigars submitted for autopsy, 7 (33%) had medullary bone in 1 or more bones examined. All 7 male budgerigars with medullary bone had a testicular neoplasm, which was morphologically consistent with a testicular sustentacular cell tumor, seminoma, or interstitial cell tumor. Medullary bone was not present in the 14 cases with other diseases. Medullary bone formation in pneumatic and nonpneumatic bones can occur in male budgerigars with testicular neoplasms. Radiographic increases in medullary bone density, particularly in the humerus, could provide antemortem indication of testicular neoplasia in male budgerigars.
Keywords
Medullary bone (also known as laying bone and intramedullary bone) is a calcium-rich, labile bone that is both formed and removed during the avian egg-laying cycle, contributing to the mineralization of eggshells. 4 As such, medullary bone is a sex-specific tissue physiologically present in female birds about 10 days before ovulation until the mineralized egg passes through the oviduct (shell gland). 18 Depending on the relative size of the egg, 10%–20% of the total body calcium can be used to make each eggshell.7,8,10 With adequate dietary rations of calcium, approximately 25%–40% of the calcium in each eggshell is derived from the skeleton, including medullary bone.14,22 Like structural bone, medullary bone formation and removal is controlled by the actions of osteoblasts and osteoclasts, respectively. 25 Medullary bone is composed of irregular fibrils of type I collagen, similar to woven bone, although the increased proteoglycan content of medullary bone imparts basophilic staining with hematoxylin and eosin (HE) stain, resembling cartilage in some respects.2,6
Medullary bone is formed in medullary spaces along endosteal surfaces of structural bone in the female avian skeleton. 4 In general, medullary bone is deposited more frequently and in greater amount in nonpneumatic bones such as the femur and tibiotarsus, while pneumatic bones such as the humerus and bones of the skull are mostly spared.5,25 However, the skeletal locations in which medullary bone is formed during the egg-laying cycle vary between different species of bird, depending on natural or artificial selection pressures. 5 For example, domestic hens, which are largely sedentary birds that spend most weeks of a year reproductively cycling, often have medullary bone throughout the skeleton and even filling medullary spaces of pneumatic bones. 6
While the physiologic mechanisms controlling medullary bone are not completely defined, sex hormones and particularly estrogens likely play a key role.6,18 Medullary bone formation coincides with maturation of preovulatory ovarian follicles. Association between maturing ovarian follicles and formation of medullary bone is believed to involve the hypothalamic-pituitary-ovarian axis. Following binding of luteinizing hormone (LH) from the anterior pituitary gland, the theca externa layer of the ovum progressively synthesizes and releases estrogens that bind estrogen receptors expressed in the nuclei of some osteoblasts, leading to the deposition of type I collagen and other organic components of medullary bone. Postovulation, with reduction of serum estrogens, the stimulus for osteoblastic medullary bone formation is removed. As the egg reaches the shell gland and begins mineralization, decreased serum estrogens and hypocalcemia-induced parathyroid hormone secretion stimulate osteoclasts indirectly to remove medullary bone, releasing calcium into the bloodstream. 18 Experimental studies have emphasized the importance of estrogens20,26 and also suggested the necessity of testosterone in medullary bone formation. 3
In female birds, increased bone density due to formation of medullary bone, particularly in pneumatic bones, has been termed polyostotic hyperostosis, even if physiologic. 12 In disease, polyostotic hyperostosis occurs most commonly with female reproductive tract disorders, namely reproductive (ovarian or oviductal) adenocarcinoma, where endogenous estrogen production by neoplastic cells is thought to stimulate inappropriate medullary bone formation.12,21 In diagnostic cases, polyostotic hyperostosis is often presumed from radiographic changes in long bones. 12 The natural occurrence of medullary bone in male birds is poorly documented, particularly with postmortem examination and light microscopy. Increased radiographic medullary density throughout the skeleton has been observed in male budgerigars with testicular sustentacular cell (Sertoli cell) tumors. 13 One study reported finding medullary bone in the femurs of 2 male budgerigars with sustentacular cell tumors. 2 Testicular neoplasms, primarily sustentacular cell tumors, in male budgerigars can be associated with color change of the cere from the normal light blue to brown. Color change of the cere has been used as an external indicator of testicular neoplasia in this species.2,13
This study investigates the occurrence of medullary bone in nonpneumatic (femur) and pneumatic (humerus) bones in sexually mature male budgerigars submitted for autopsy between 2019 and 2021, including 1 archived case from 2017. The objectives of the study were to determine if medullary bone occurs in male budgerigars and to identify associated diseases. Based on few previous reports and reproductive physiology in female birds, it was hypothesized medullary bone formation in male budgerigars would occur with some testicular tumors.
Materials and Methods
Case Selection
Mature (age greater than 6 months) male budgerigars submitted for autopsy to the University of Tennessee, College of Veterinary Medicine between 2019 and 2021, as well as an archived case from 2017, were included in this study. The average age of the study population was 3.3 years (age range: 2–7 years). All budgerigars were kept in zoological collections or as companion pets. Budgerigars either died spontaneously, usually without clinical signs, or were euthanized at the discretion of the attending clinicians. Autopsies and tissue collection were performed by the University of Tennessee, College of Veterinary Medicine anatomic pathologists or anatomic pathology residents. Signalment, clinical findings, gross necropsy findings, and histologic findings were recorded for each budgerigar. Cere color was also recorded when it differed from the normal light blue of mature male budgerigars. An age-matched female budgerigar submitted for autopsy was used as a positive control.
Histopathology
All soft tissues were immersed in 10% neutral-buffered formalin for routine sample processing. Paraffin-embedded tissues were sectioned at 5 microns, mounted on glass slides, stained with HE, and examined by light microscopy. Prior to processing, humeri and femurs were additionally placed in a decalcifying solution (Formical-4, StatLab, 2090 Commerce Drive, McKinney, Texas) for approximately 24–30 hours for softening. Each bone was cut longitudinally, with each half submitted for processing when available. Additional tissues were examined for other disease processes. The humeri and femurs of the female budgerigar were processed as in the male budgerigars.
Results
Case Selection and Postmortem Findings
Twenty-one male budgerigars were examined for the presence of medullary bone from 2017 to 2021. Case information and primary postmortem findings for each budgerigar are summarized in Supplemental Table S1. The most common postmortem finding in this population, either alone or concurrent with another condition, was a testicular neoplasm that expanded 1 or both testes. Five budgerigars with testicular neoplasms had color change of the cere, which included regional to diffuse brown, blue-brown, red-brown, or dark-blue discoloration. All male budgerigars without testicular neoplasms had normal light blue ceres. The average age of male budgerigars with and without testicular neoplasia was 4 years (age range: 3–7 years) and 2.9 years (age range: 2–4 years), respectively. The age-matched, postive control female budgerigar was euthanized for a large subcutaneous mass that was unrelated to the reproductive system or hypothalamic-pituitary-gondal axis. The ovary contained a 5-mm, yellow follicle (active ovary).
Gross Findings—Medullary Bone
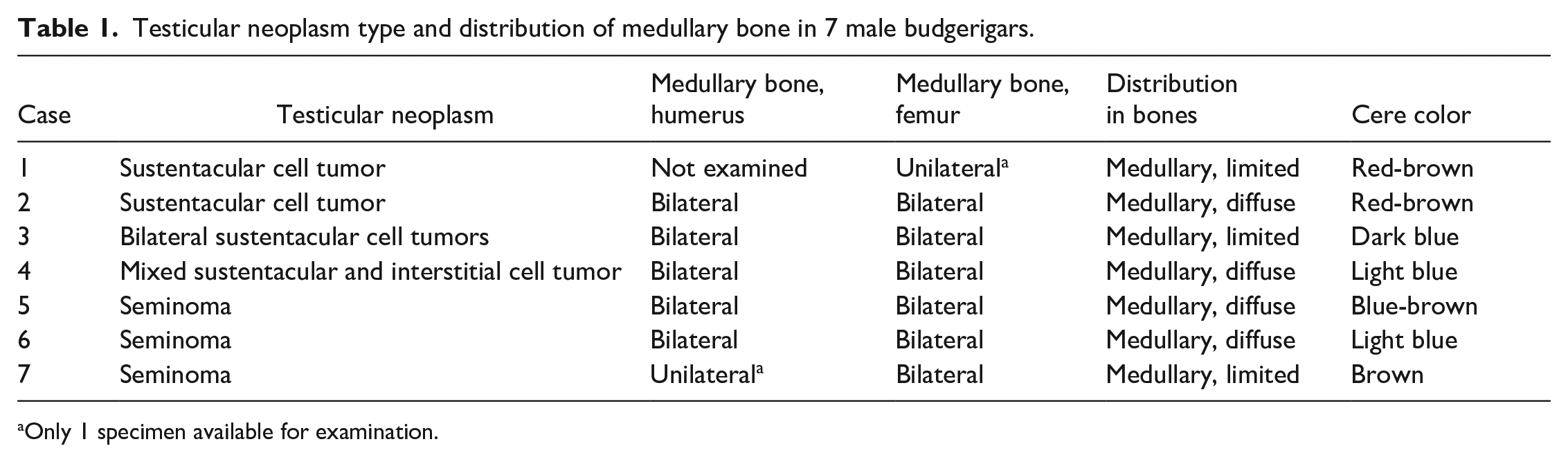
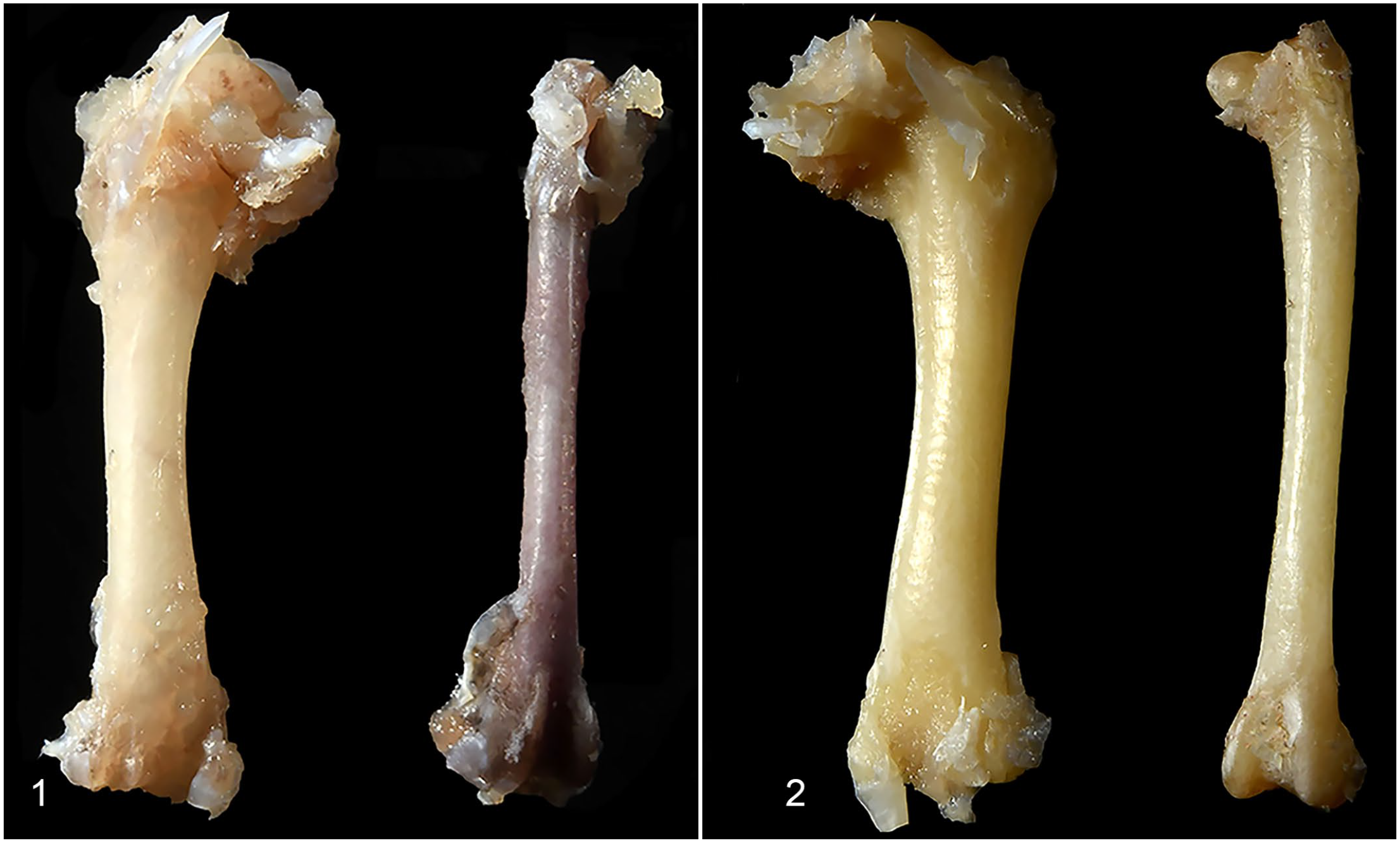
Medullary bone could be recognized grossly when diffusely deposited (Table 1; cases 2, 4, 5, and 6). In the femur, the normal red-gray imparted by hematopoietic tissue within marrow spaces (Fig. 1) became discolored tan to pale-tan (Fig. 2). The humerus (a pneumatic bone) sank in formalin, as normal pneumatic spaces (Fig. 3) were replaced by solid, yellow-tan, firm material (Fig. 4).
Testicular neoplasm type and distribution of medullary bone in 7 male budgerigars.
Only 1 specimen available for examination.
Humerus and femur, male budgerigars (fixed specimens).
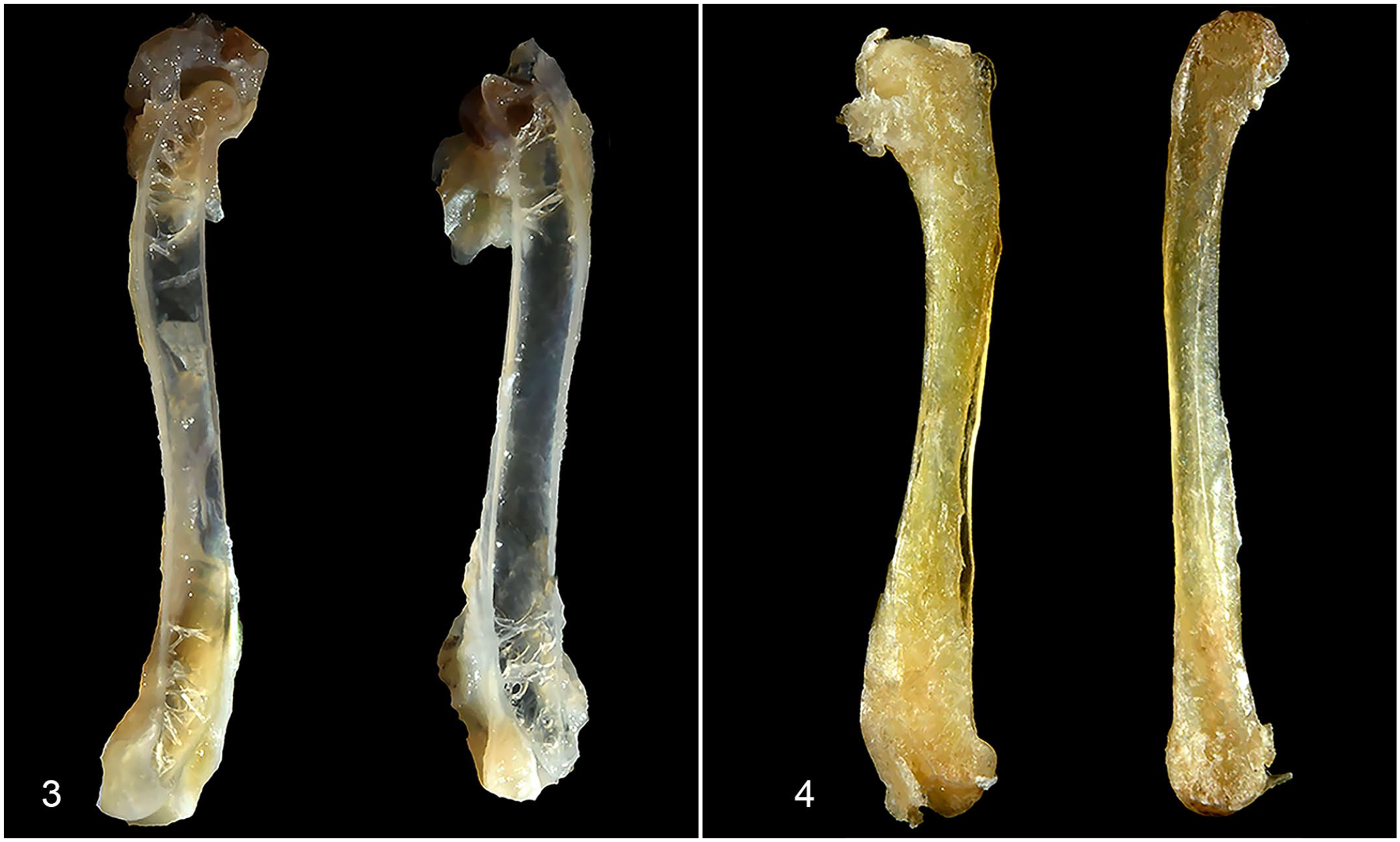
Humerus, male budgerigars (bisected fixed specimens).
Histopathology—Medullary Bone
Humeri and/or femurs of 7 of 21 budgerigars had medullary bone, which with HE staining, appeared as irregular spicules and lamellae of nonbirefringent, basophilic matrix containing scattered osteocytes that covered endosteal surfaces of trabecular and cortical bone (Figs. 5, 6). In the positive control female budgerigar, medullary bone was microscopically identical to that in humeri and femurs of the male budgerigars. However, medullary bone in the female budgerigar often had scalloped margins filled by osteoclasts with foamy cytoplasm, suggesting active resorption (Figs. 7, 8). Medullary bone was not present in the other 14 male budgerigars. Figs. 9–12 show the contrast between normal humeri and femurs and those bones with medullary bone deposition in adult males.
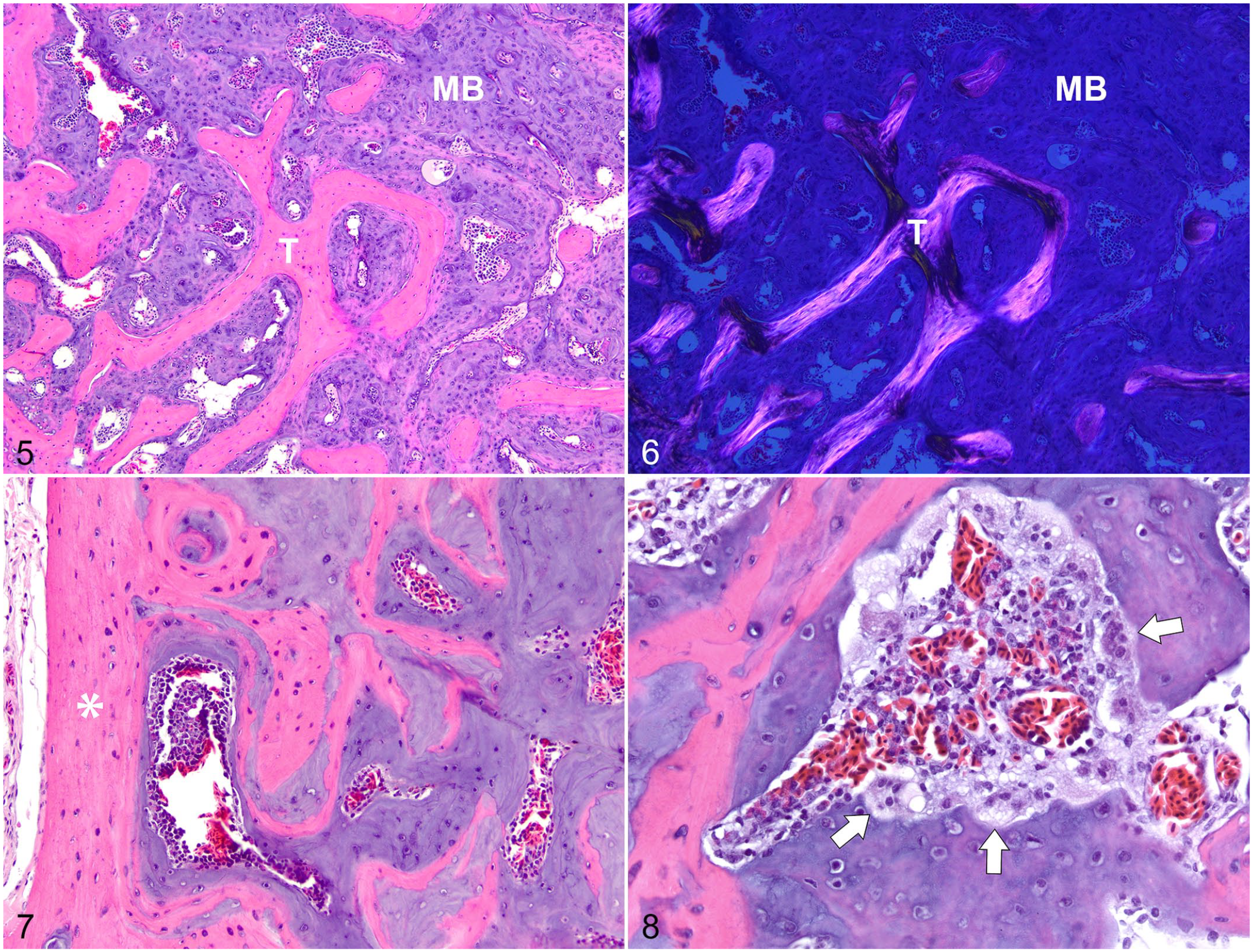
Medullary bone (MB), humerus, male budgerigar, case 6.
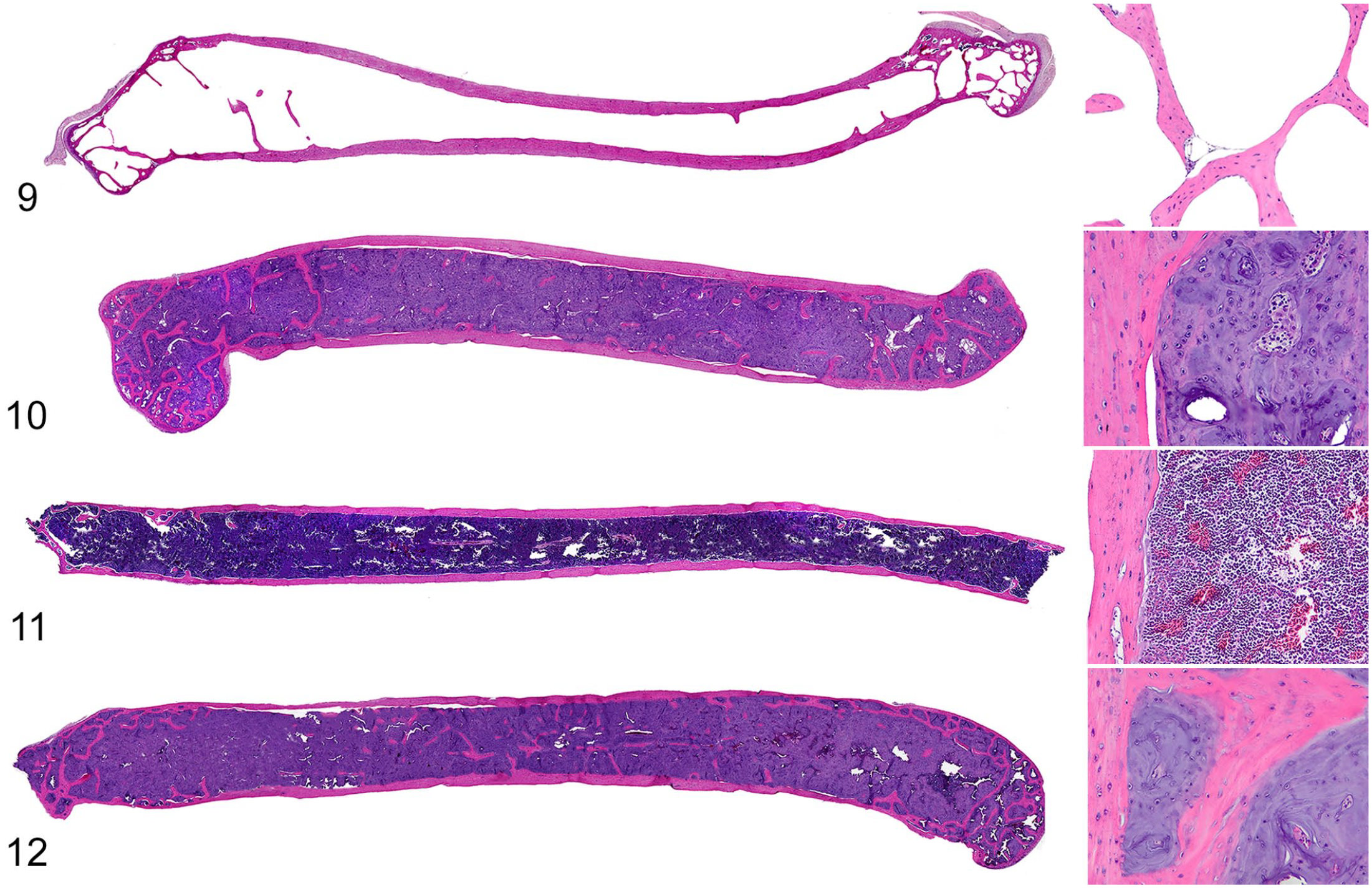
Humerus and femur, male budgerigars.
The distribution of medullary bone in male budgerigars is summarized in Table 1. In all cases with both bones available, medullary bone was bilateral and symmetrical. Medullary bone filled medullary spaces through the epiphyses, metaphyses, and diaphyses (diffuse distribution) in 4/7 budgerigars (Figs. 10 and 12). The other 3/7 budgerigars had patchy, multifocal deposition of medullary bone (limited distribution), usually forming scattered, irregular spicules along preexisting trabecular bone. Active osteoblasts and osteoclasts were usually inapparent, except in femurs of 1 budgerigar (case 3) in association with metastatic tumor cells. In the humeri, spaces between medullary bone contained hematopoietic tissue.
Associated Pathologic Findings
All 7 budgerigars with medullary bone had testicular tumors, which included sustentacular cell tumor (4/7), seminoma (3/7), and interstitial cell tumor (1/7). Testicular tumor diagnosis was based on histomorphology alone. Briefly, sustentacular cell tumors were poorly demarcated neoplasms composed of polygonal cells arranged in cords and irregular tubules, separated by frequent streams of tortuous fibrous connective tissue. Seminomas were poorly demarcated neoplasms with round cells filling tubules, with increased pleomorphism and frequent mitoses. The interstitial cell tumor was a poorly demarcated neoplasm composed of polygonal cells with brightly eosinophilic cytoplasm arranged in nests, separated by thin, fibrovascular trabeculae. One budgerigar had bilateral sustentacular cell tumors (case 3), and another had a mixed sustentacular and interstitial cell tumor (case 4). In 2/4 cases with sustentacular cell tumors, malignancy was indicated by metastasis to the kidney (case 4) or marrow of both femurs (case 3). One budgerigar with a testicular neoplasm had concurrent epididymal cysts (case 3). No other reproductive disorders were present in the male budgerigars.
Discussion
Medullary bone is a unique tissue of the female avian skeleton that supplies calcium to developing eggshells. The natural occurrence of medullary bone in male birds is poorly characterized. In this study, pneumatic (humeri) and nonpneumatic (femurs) bones from 21 male budgerigars submitted for autopsy were microscopically examined for the presence of medullary bone. Of those, 7 had medullary bone in medullary spaces of the humeri and/or femurs. All male budgerigars with medullary bone in this study had a testicular tumor consistent with sustentacular cell tumor, seminoma, or interstitial cell tumor. Diagnoses in the remaining 14 male budgerigars were not associated with medullary bone formation.
Microscopically, medullary bone is a nonbirefringent, basophilic matrix with embedded osteocytes that covers endosteal surfaces of structural bone and fills medullary spaces. The composition of medullary bone differs from that of structural bone. The increased proteoglycan content of medullary bone imparts the basophilic staining pattern and isotropic (nonbirefringent) properties observed with light microscopy. The absence of birefringence with polarized light is helpful in distinguishing medullary from structural bone (Figs. 5, 6). Deposition of medullary bone in male budgerigars of this study was usually bilateral and symmetric, filling medullary spaces of the humeri and femurs similarly. The extent of deposition varied from diffuse filling of medullary spaces to multifocal, patchy areas along preexisting trabecular bone. Although beyond the focus of this study, differences in distribution likely reflect relative serum concentration of reproductive hormones perhaps related to cyclic production of reproductive hormones by neoplastic cells in the testes. Diffuse deposition of medullary bone, particularly in the humerus, likely could be recognized radiographically. The microscopic appearance and location of medullary bone in the male budgerigars of this study did not differ from that of the positive control female budgerigar. The presence and degree of medullary bone mineralization could not be evaluated in the decalcified samples used in this study.
Budgerigars have a higher incidence of testicular neoplasia relative to other avian species.15,17 In this study, sustentacular cell tumor and seminoma predominated, and interstitial cell tumor was less frequent. Malignant sustentacular cell tumors were indicated by metastasis to the kidney or bone (cases 4 and 3, respectively).
The role of sex hormones, particularly estrogens, in structural bone formation and maintenance is well studied in humans. 1 As previously discussed, a similar association has been suggested for medullary bone formation in birds. This study suggests an association between different types of testicular neoplasms and the formation of medullary bone. Although the relationship is unclear, increased serum estrogens may contribute to the pathogenesis. In other bird species, the synergistic action of estrogens and testosterone is suggested to be necessary for medullary bone formation in male birds. 3 As in mammals, sustentacular and interstitial cells in the avian testes produce estrogens and testosterone, respectively. As the sole interstitial cell tumor was concurrent with a sustentacular cell tumor (case 4), estrogen production by neoplastic sustentacular cells may have led to medullary bone formation alone. Three male budgerigars with medullary bone had seminomas, but germ cells are not expected to produce hormones. In humans, hyperestrogenemia has been reported in a case of testicular seminoma with the ability to synthesize the enzyme aromatase (estrogen synthetase). 9 Paraneoplastic feminization has been reported in a dog with a testicular seminoma. 11 In the human and canine cases, removal of the testicular seminomas led to normalization of serum estrogen and resolution of clinical signs.
Paraneoplastic feminization is the acquisition of feminine characteristics as an indirect consequence of a neoplasm, usually due to production of chemical signaling molecules such as hormones. In domestic animals, the most common paraneoplastic feminization syndrome occurs in male dogs with sustentacular cell tumors.16,23 In part, increased estrogen production by neoplastic cells leads to feminine behaviors and gynecomastia, as well as other paraneoplastic effects (alopecia, prostatic squamous metaplasia, and myelosuppression).19,23,24 Medullary bone formation is 1 component of a proposed paraneoplastic feminization syndrome in male budgerigars, which has previously been reported to include cere color change, widening of the pelvic inlet, and protrusion of the cloaca. 13 The latter 2 were not observed in this study. Cere color change, from the normal light blue to brown, blue-brown, or red-brown, occurred in 4/7 male budgerigars with testicular neoplasms and medullary bone in this study, and an additional case had a dark-blue cere. While cere color change can be an external indicator of testicular neoplasia in male budgerigars, this is insensitive. Two male budgerigars with testicular neoplasms in this study did not have color change of the cere, yet these 2 budgerigars had diffuse deposition of medullary bone in the humeri and femurs. The relationship between cere color, medullary bone formation, and testicular neoplasms is unknown. It should be noted that the normal color of the female cere in budgerigars is bright pink, rather than brown or a brown variation. Given the similar gross appearance, location in the caudal coelom, and high frequency15,17 in budgerigars, antemortem differentiation of testicular and renal neoplasms can be difficult. Recognition of these paraneoplastic changes may be helpful.
Polyostotic hyperostosis has been used to denote both physiologic and pathologic formation of medullary bone.2,12,21 Distinguishing physiologic from pathologic formation of medullary bone in female birds is difficult. Polyostotic hyperostosis in female birds is indicated when medullary bone is formed in areas it is usually not present (often pneumatic bone) or when medullary bone formation is asynchronous with the egg-laying cycle. The diagnosis of polyostotic hyperostosis in female birds requires knowledge of both the reproductive cycle in each individual bird and the normal extent of medullary bone deposition in that particular species. To our knowledge, the occurrence of medullary bone in male birds is always pathologic.
In conclusion, medullary bone formation in pneumatic and nonpneumatic bones can occur naturally in male budgerigars and has a similar microscopic appearance to that of female birds. All male budgerigars with medullary bone in this study had testicular neoplasms. The mechanisms linking testicular neoplasia and medullary bone formation are not entirely determined, although a key role of sex hormones, particularly estrogens, is suspected. Additional studies measuring serum estrogens and testosterone across multiple time points in male budgerigars are necessary. Medullary bone is an important example of rapid bone remodeling and sex hormone-responsive tissue, possibly providing a relevant model of human skeletal disease, such as postmenopausal osteoporosis. In addition, increased radiographic bone density, particularly in pneumatic bones, such as the humerus, could provide antemortem indication of testicular neoplasia in male budgerigars.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858211069126 – Supplemental material for Medullary bone in male budgerigars (Melopsittacus undulatus) with testicular neoplasms
Supplemental material, sj-pdf-1-vet-10.1177_03009858211069126 for Medullary bone in male budgerigars (Melopsittacus undulatus) with testicular neoplasms by Nathan K. Hoggard and Linden E. Craig in Veterinary Pathology
Footnotes
Acknowledgements
We thank the managers, necropsy technicians, and histotechnicans in the Department of Biomedical and Diagnostic Sciences at the University of Tennessee, College of Veterinary Medicine. We would also like to thank the anatomic pathologists and anatomic pathology residents at the University of Tennessee, Collage of Veterinary medicine who provided indirect assistance in shaping this article.
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by research funds provided by the University of Tennessee, College of Veterinary Medicine.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
