Abstract
Canine smooth muscle tumors (SMTs) commonly develop in the alimentary and female genital tracts and less frequently in soft tissue. The definition of histological criteria of malignancy is less detailed for SMTs in dogs than in humans. This study evaluated the clinicopathologic features of canine SMTs and compared the veterinary and human medical criteria of malignancy. A total of 105 canine SMTs were evaluated histologically and classified according to both veterinary and human criteria. The Ki67 labeling index was assessed in all SMTs. Estrogen receptor (ER) and progesterone receptor (PR) expression was evaluated for soft tissue SMTs. Follow-up data were available in 25 cases. SMTs were diagnosed in the female genital tract (42%), alimentary tract (22%), and soft tissue (20%). Soft tissue SMTs frequently arose in the perigenital area, pelvic cavity, and retroperitoneum. A subset of soft tissue SMTs expressed ER and/or PR, resembling the gynecologic type of soft tissue SMT in humans. SMTs were less frequently malignant when assessed with human criteria than with veterinary criteria, better reflecting their benign behavior, especially in the genital tract where human criteria tolerate a higher mitotic count for leiomyoma. Decreased differentiation was correlated with increased proliferation, necrosis, and reduced desmin expression. Mitotic count, Ki67 labeling index, and necrosis were correlated with metastases and tumor-related death. Further prognostic studies are warranted to confirm the better performance of the human criteria when assessing SMT malignancy, especially genital cases, to confirm their usefulness in ER/PR-expressing soft tissue SMTs, and to better define the most useful prognostic parameters for canine SMTs.
Keywords
Canine smooth muscle tumors (SMTs) arise more often in the female genital and alimentary tracts and less commonly in the lower urinary tract, soft tissue, and spleen. 1 In the alimentary tract, SMTs arise more frequently in the stomach, where they are mostly benign, and in the intestine, where they are more often malignant, while esophageal SMTs are less common. 1 Most information about canine alimentary SMTs precedes the first descriptions of canine gastrointestinal stromal tumors (GISTs). 1 This may have distorted the data in earlier reports as GISTs and SMTs often require differentiation by immunohistochemistry.
SMTs of the female genital tract are largely benign and often express estrogen receptor (ER) and progesterone receptor (PR). 1,13 SMTs of the lower urinary tract are less common but still represent the majority of urinary bladder mesenchymal tumors. 1 Splenic SMTs have been grouped among stromal tumors, but specific studies regarding their prognosis and behavior in dogs are lacking. 9
Canine cutaneous and subcutaneous SMTs are reported to have a good prognosis, 11 but information is mostly restricted to tumors derived from the arrector pili muscles or from vessel walls. 11,17 On the contrary, studies on deep-seated canine soft tissue SMTs are lacking. Furthermore, despite the knowledge available for human SMTs, 5,6 hormone receptor expression in canine soft tissue SMTs is largely unknown.
In dogs, the distinction between benign and malignant SMTs relies mainly on the morphological assessment of necrosis, infiltrative growth, and mitotic activity. However, current veterinary guidelines do not provide specific cutoff levels for these parameters and do not take into consideration the primary site of the tumor. 1 In contrast, specific guidelines are available for the morphological assessment of SMTs in humans. 12,18 In humans, morphologic criteria for a diagnosis of leiomyosarcoma, rather than leiomyoma, include mitotic count, nuclear atypia, and tumor cell necrosis. 12,18 The cutoffs for mitotic count vary according to tumor site: while any proliferative activity is considered an indication of malignancy in most sites, up to 9 mitoses in 10 high-power fields (HPFs) are tolerated in female genital tract SMTs, as these tumors are often considered to be benign. 12
Due to the paucity of up-to-date information regarding canine SMTs, the aims of this study were to: Describe the organ distribution and the clinicopathologic features of canine SMTs. Provide a detailed pathologic evaluation of canine soft tissue SMTs, including deep-seated tumors and expression of hormone receptors. Compare the performance of the morphological criteria of malignancy used in veterinary medicine (veterinary criteria) with those used in human medicine (human criteria) to separate benign and malignant SMTs in dogs.
Materials and Methods
Case Selection and Clinical Information
Cases from 2001 to 2017 of canine spindle cell neoplasms with a histological diagnosis (definitive or presumptive) of SMT, or for which a possible smooth muscle origin was hypothesized, were retrospectively collected from the archives of 2 different institutions. Cases morphologically consistent with a smooth muscle cell origin, 17 negative for CD117 (which excludes GISTs), and expressing α-smooth muscle actin (α-SMA) and/or desmin were included in this study as canine SMTs.
Data were collected regarding the breed, age, sex, and neutering status of each dog, as well as the site of development, and size of each neoplasm. The size was defined as the largest tumor diameter measured at the trimming station after fixation. Tumors of the female genital tract, alimentary tract, soft tissue, lower urinary tract, spleen, and miscellaneous SMTs were included in this study.
Diagnosis
Hematoxylin and eosin (H&E)-stained sections from each case were reevaluated at a multihead microscope by 2 pathologists (GA and VP). The following histological features were assessed according to their description in the human criteria:
12,18
Differentiation, classified as well-differentiated (similar to normal tissue), intermediate differentiation (histologic type can be determined), or poorly differentiated (undifferentiated tumors). Nuclear atypia, classified as absent, mild, moderate, or severe. Necrosis, classified as absent, <50% of the tumor, or ≥50% of the tumor (microscopically assessing all available sections). Nuclear shape, classified as oval, cigar-shaped, or slender (long, often hyperchromatic and occasionally twisted); classification was based on the nuclear morphology of the majority of neoplastic cells. Prominent vascularization, perivascular fibrosis, hyalinized stroma, trabecular pattern (neoplastic smooth muscle cells arranged in anastomosing trabeculae separated by extracellular matrix), myxoid differentiation (presence of myxoid matrix), mineralization, vesicular chromatin, nuclear palisading (nuclei lined up and alternating with anuclear zones), and multinucleation (all classified as present or absent).
The diagnosis of leiomyoma or leiomyosarcoma was based on both the veterinary criteria
1
and the human criteria.
12,18
Leiomyosarcoma was diagnosed, independent of the site of occurrence, when at least one of the following veterinary criteria indicative of malignancy was observed (cases with none of these features were diagnosed as leiomyoma): Mitotic count of at least 1 mitotic figure in 10 HPFs equivalent to the standard area of 2.37 mm2. Infiltration into adjacent tissues. Presence of necrosis.
The human criteria
12
indicative of malignancy for SMTs not located in the female genital tract included at least one of the following: Any mitotic figures within neoplastic cells. Nuclear atypia (defined as more than mild, clearly visible with a 10× objective lens, and including abnormal shape, karyomegaly, and prominent nucleoli). Rare, scattered, large hyperchromatic nuclei with intranuclear cytoplasmic inclusions were not considered as nuclear atypia but represented a degenerative change (so-called ancient change). Tumor cell necrosis. Infarct-type necrosis was not considered to be a criterion for malignancy and was differentiated from tumor cell necrosis by the presence of the following features: central location; abrupt transition with viable neoplastic cells; presence of either granulation tissue or hyalinized stroma between the necrotic and nonnecrotic areas; recent hemorrhage; mummified appearance showing outlines of the tumor cells; and both tumor and vessels appearing necrotic. In contrast, tumor cell necrosis had a scalloped outline and the neoplastic cells surrounding vessels were usually spared.
The human criteria
12,18
provide the following specific diagnostic algorithm for SMTs located in the female genital tract: Concurrent nuclear atypia (more than mild, visible with a 10x objective lens) and tumor cell necrosis (as previously described, and independent of mitotic activity) indicates leiomyosarcoma. Presence of either nuclear atypia or tumor cell necrosis, combined with a mitotic count ≥10 in 10 HPFs (while not specified in the original studies,
12,18
in the present study a standard area of 2.37 mm2 was evaluated) indicates leiomyosarcoma. A mitotic count ≤9 in 10 HPFs (2.37 mm2) when both nuclear atypia and necrosis are absent indicates leiomyoma. A diagnosis of SMT with unknown malignant potential (SMT-UMP) is recommended in all other genital cases.
Histochemical Evaluation
Masson’s trichrome stain was performed with a commercially available kit (Code: 04-010802, Bio-Optica) in all cases to assess the amount and distribution of collagen within the canine SMTs. The amount of collagen was scored as absent, scant, moderate, or abundant. The collagen distribution pattern was classified as interfascicular (when separating bundles of neoplastic cells), interstitial (when surrounding single neoplastic cells), or mixed (a combination of the previous two).
Immunohistochemistry
Immunohistochemistry for α-SMA, desmin, and CD117 was performed on tissue microarrays using a previously validated protocol. 16 Any further staining (histochemical and immunohistochemical) for Ki67 and hormonal receptors was performed on full sections of selected cases. Immunohistochemistry for α-SMA, desmin, CD117, and Ki67 was performed in all cases while ER and PR expression was assessed only in soft tissue SMTs.
Three-micrometer-thick sections were dewaxed and rehydrated. Endogenous peroxidase was blocked by immersion in 3% H2O2 in methanol for 30 minutes. Source, dilution, and retrieval protocols for each antibody are reported in Supplementary Table S1.
The reaction was amplified by the avidin-biotin method (Vectastain Elite ABC-HRP kit, Vector) and visualized with 0.04% 3,3′-diaminobenzidine (Code: 10-0048, Histoline) for 4 minutes. Sections were counterstained with hematoxylin, rinsed in tap water, and dehydrated, before a coverslip was added. The following positive controls were used: sections of canine small intestine for α-SMA, desmin, and Ki67 staining; sections of a canine GIST for CD117 staining; and sections of canine uterus for ER and PR expression. Negative controls comprised slides incubated with omission of the primary antibody and normal tissues known to be nonreactive for the specific antibody. Ki67 expression was evaluated as the labeling index and defined as the percentage of Ki67-positive cells. Ki67-positive cells were counted in 10 HPFs (400×) counting at least 1000 cells for each case, using the manual count tool of the ImageJ 1.48 analysis software.
Follow-Up
Collection of follow-up data was attempted for all cases by phone calls with referring veterinarians. Follow-up data included tumor recurrence, metastasis, and tumor-related death.
Statistical Analysis
Correlations between histologic variables were obtained with the Spearman test. A P value ≤.05 was considered significant. The normality of data distribution was assessed according to the D’Agostino and Pearson omnibus test. Statistical analysis was performed using GraphPad Prism 8.3 (GraphPad Software, Inc).
Furthermore, 3 statistical models were used to determine the association of each variable with the diagnosis (with and without confounding variables) and to identify a multivariable predictive model for the diagnosis.
Univariate logistic regression models were fitted for each variable to test its association with the diagnosis (according to both the veterinary and human criteria). The Wald’s test and the likelihood ratio test were used to assess statistical significance. Each model was evaluated by adjusting the variables for confounding effects (age, tissue, and sex) and with no adjustment. P values were adjusted for multiple testing using the BH procedure. Results were sorted according to the residual deviance. A smaller residual deviance means that the variable better predicts the diagnosis.
A multivariable model was obtained by elastic net regression. The penalization parameter was evaluated using the cross-validation procedure of the cv.glment function from the glmnet library in R (accessed June 2014). A Leave-One-Out (LOO) cross-validation was used to test the prediction accuracy and the AUC (area under the curve) of ROC (receiver operating characteristic curve) was thus calculated. The importance of each variable in the model was estimated as the average of the coefficients obtained in each LOO iteration and standard deviations were also reported. For the diagnosis according to the human criteria, the model was computed 3 times, considering only one pair of outcomes at a time. All analyses were performed in R 3.6.3. Missing values were imputed with the mice algorithm v3.5.0 in R. 19
Microscopic images depicting the histological features assessed are available as supplemental material.
Results
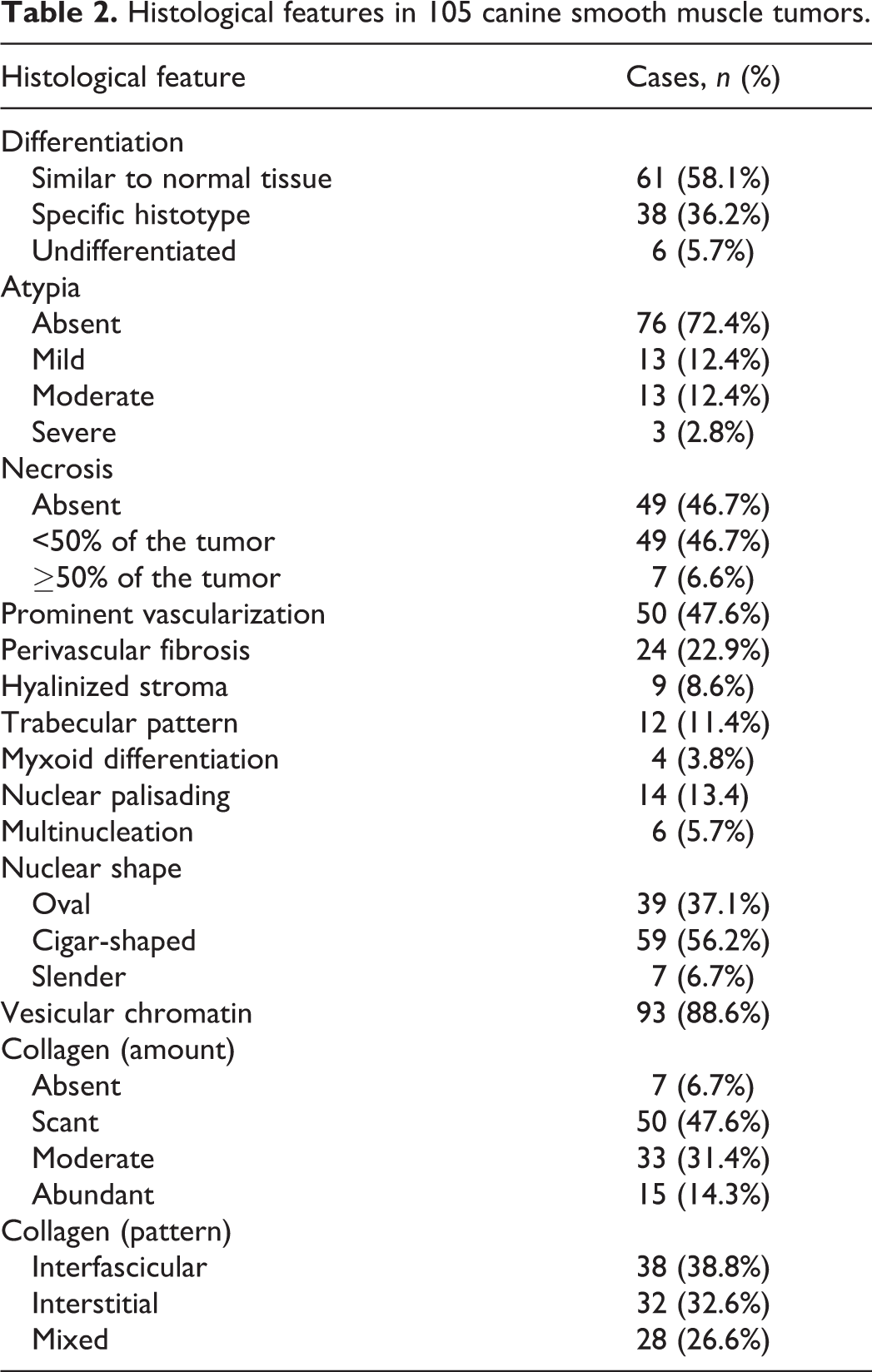
Selection criteria were met for 105 canine SMTs from 104 dogs. A total of 71/104 dogs were female (69%, 14 of which were spayed) and 32/104 dogs were male (31%, 9 of which were castrated). In one case, the sex was unknown. Table 1 lists the sex distribution parsed by the primary site of the neoplasm. Most dogs were crossbreed, 41/104, followed by retrievers, 12/104, and boxers, 6/104. Sixteen other breeds were represented, with 1 to 3 cases each. The median age of the total cohort was 11 years (range 3–17), the median age of dogs with splenic SMTs was 11.2 years, and the median age was 11 years in all other groups. The age range was 3 to 15 years for dogs with tumors of the genital tract, 4 to 17 years for dogs with alimentary tumors, 4 to 15 years for dogs with soft tissue tumors, 6 to 13 years for dogs with lower urinary tract tumors, and 10 to 12 years for dogs with splenic tumors. No statistically significant differences were found in the age of dogs based on sex or tumor site. SMTs of the female genital tract developed in younger dogs, with a peak incidence at 8 to 9 years of age (Fig. 1). Forty-four tumors were in the female genital tract, 23 in the alimentary system, 21 in soft tissue, 11 in the lower urinary tract, 4 in the spleen, and 2 in other sites (Fig. 2). Tumor size ranged between 0.5 cm and 15 cm (median = 3.2 cm; mean = 4.6 cm).
Sex distribution of 104 canine smooth muscle tumors stratified by location.
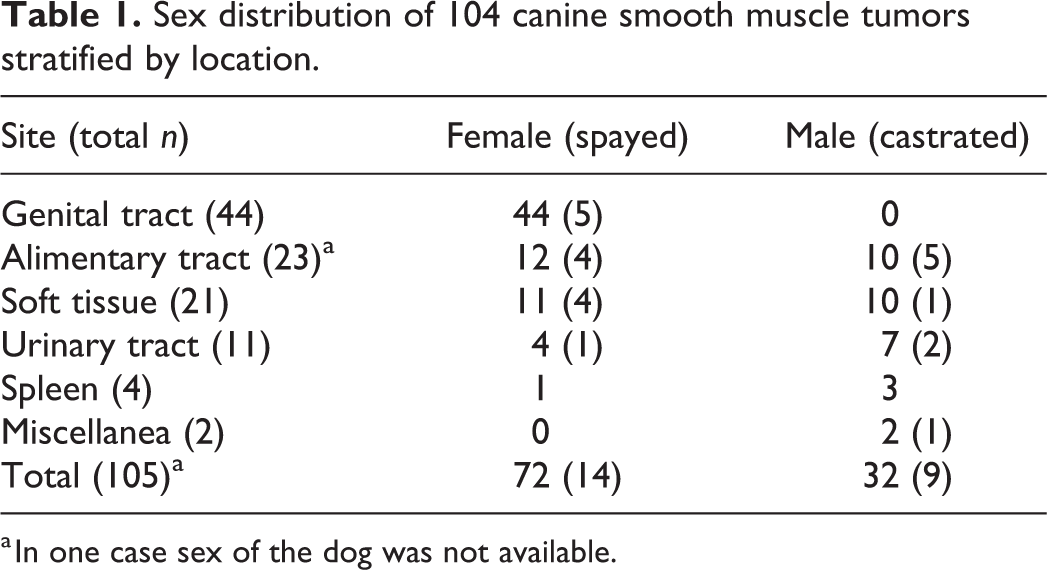
a In one case sex of the dog was not available.
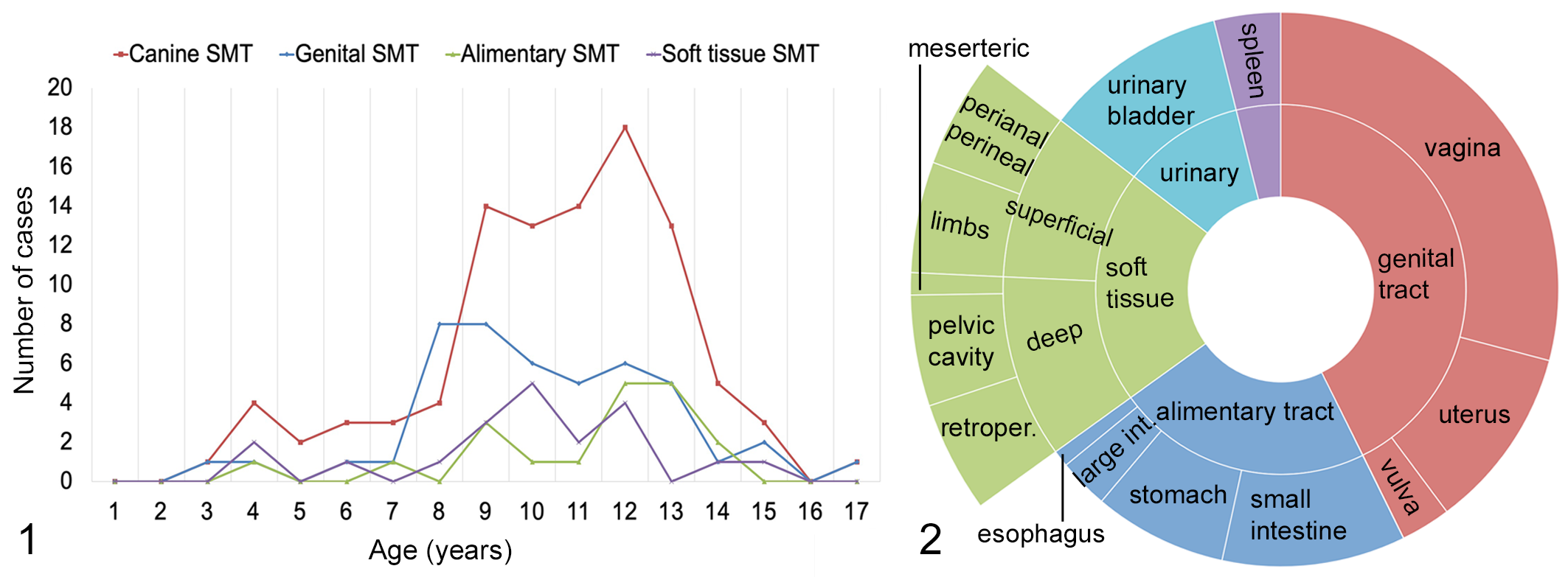
Age distribution of dogs with smooth muscle tumors (SMTs). Comparison of all cases (red line) with the genital, alimentary, and soft tissue SMT groups.
Histologic and Immunohistochemical Features
Histologic features are listed in Table 2 and illustrated in Figures 3 to 17. All cases expressed α-SMA diffusely and 73/105 cases (70%) expressed desmin either diffusely or multifocally (Figs. 18–21). Estrogen and/or progesterone receptors were expressed in 7/21 soft tissue SMTs (Figs. 22, 23).
Histological features in 105 canine smooth muscle tumors.
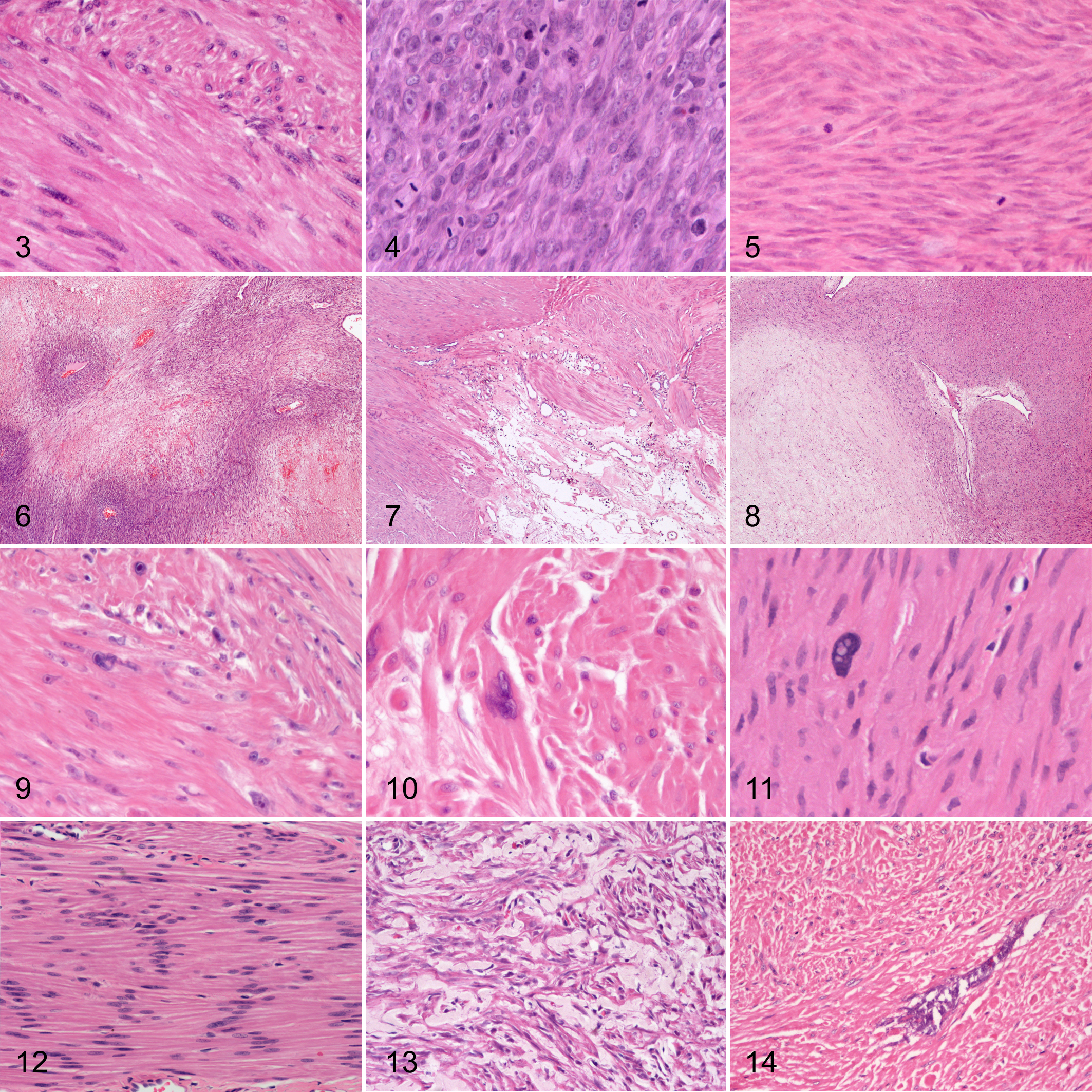
Smooth muscle tumors (SMTs), dog. Hematoxylin and eosin.
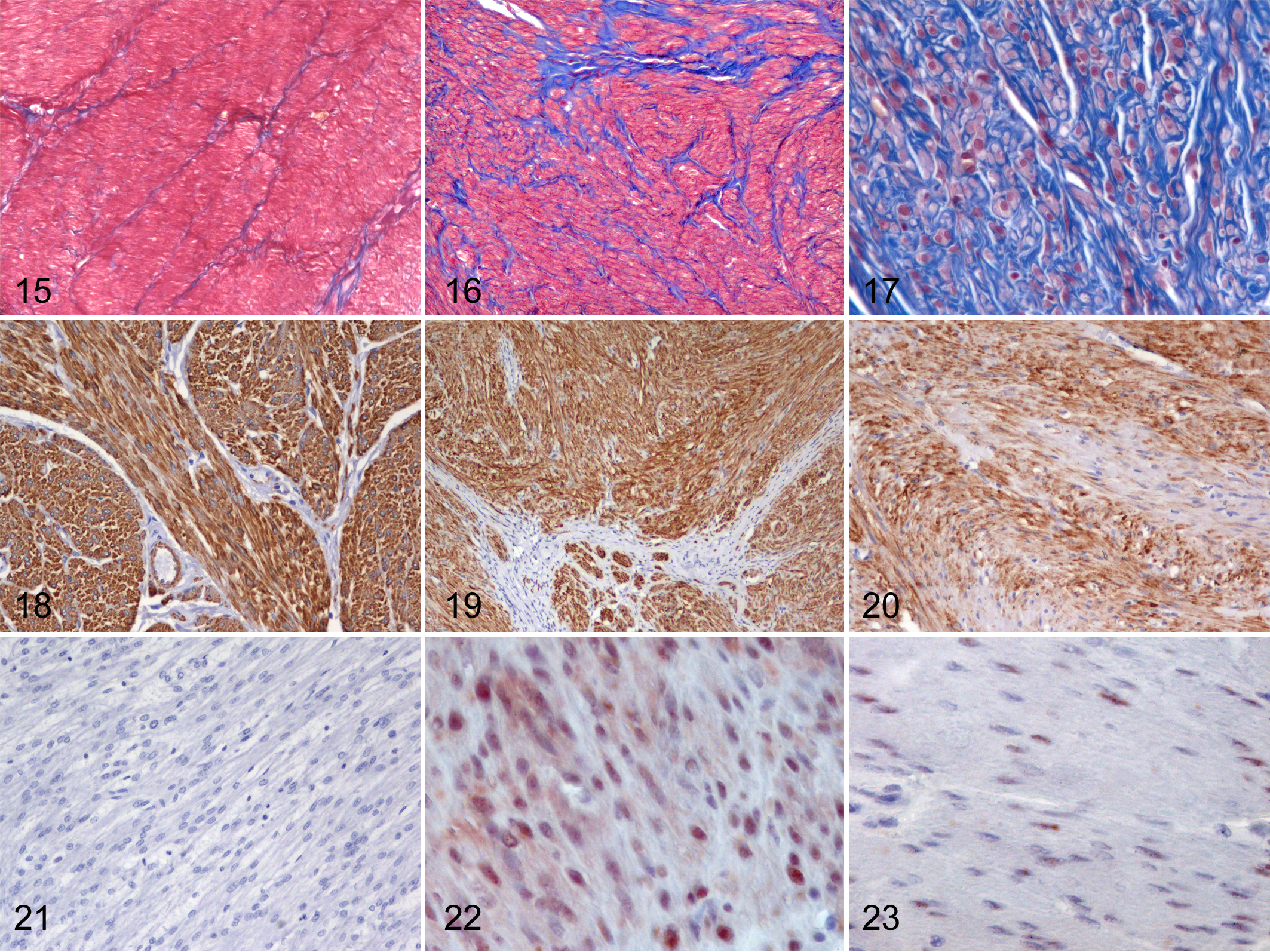
Smooth muscle tumors (SMTs), dog. Masson’s trichrome.
Several statistically significant correlations were identified with the Spearman test (P and R values are listed in Supplemental Table S2). Reduced differentiation was correlated with increased tumor size, higher proliferative activity (both mitotic count and Ki67 labeling index), increased amount of tumor cell necrosis, presence of multinucleation, and lack of desmin expression (Figs. 24 –27). Greater amount of tumor cell necrosis correlated with increased size, higher mitotic count, greater collagen amount, and interstitial or mixed collagen pattern. Higher mitotic count correlated with higher Ki67 labeling index, lack of desmin expression, and presence of multinucleation. Presence of nuclear atypia correlated with increased proliferative activity (both mitotic count and Ki67 labeling index) and presence of multinucleation. The finding of hyalinized areas correlated with trabecular pattern, multinucleated cells, and myxoid areas. The amount of collagen was correlated with its distribution pattern, being more abundant in cases with interstitial and mixed distribution.
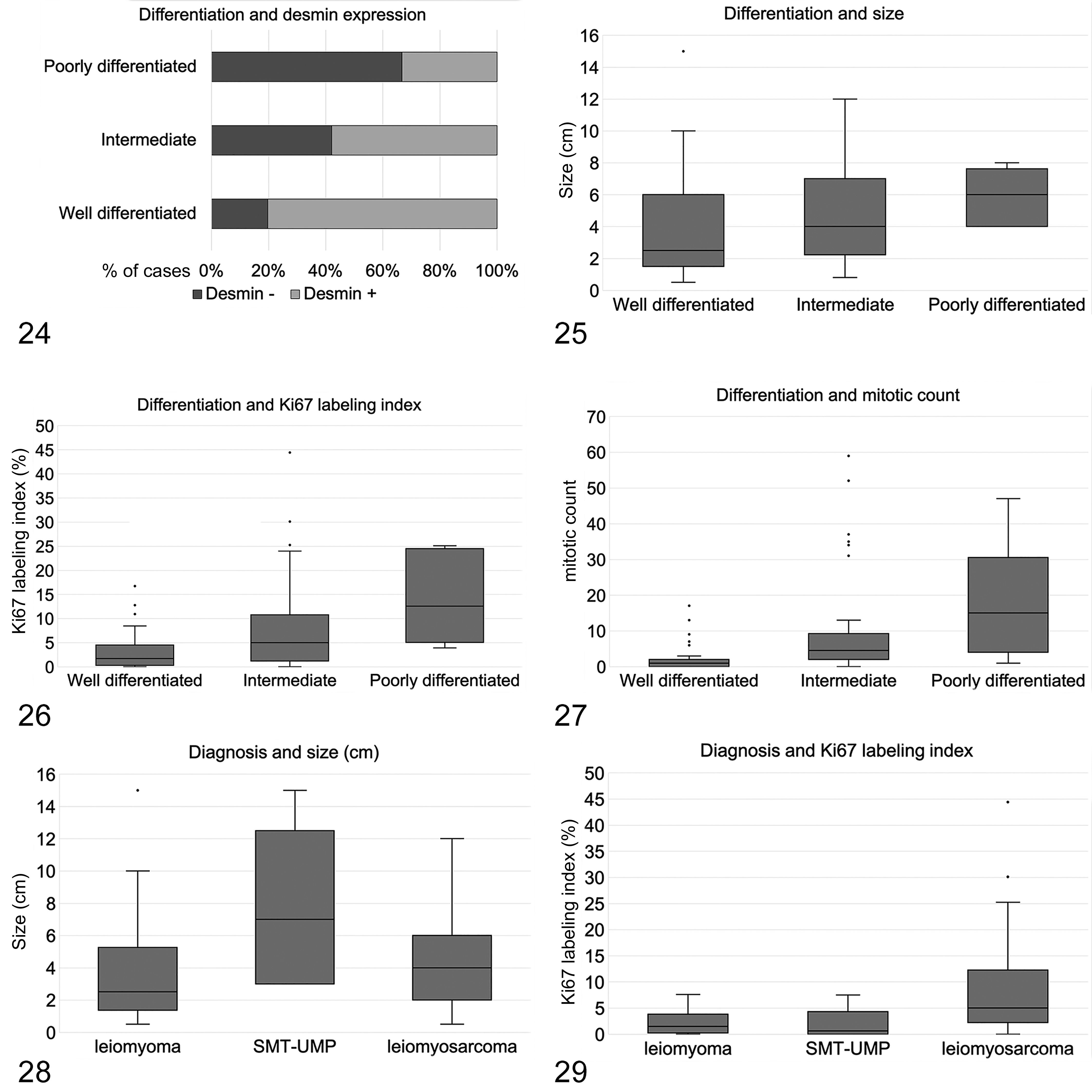
The correlation between histological differentiation of canine smooth muscle tumors (SMTs) with desmin expression (Fig. 24), tumor size (Fig. 25), Ki67 labeling index (Fig. 26), and mitotic count (Fig. 27). The box plots show median and quartiles, whiskers show minimum and maximum, and dots show outliers.
Diagnoses According to the Veterinary and Human Criteria for SMT Malignancy
According to the veterinary criteria, 22/105 cases were diagnosed as leiomyoma and 83/105 were diagnosed as leiomyosarcoma. According to the human criteria, 42/105 cases were identified as leiomyoma, 11/105 cases as SMT-UMP, and 52/105 cases as leiomyosarcoma. There was disagreement in the veterinary and human definition of malignancy in 31 SMT cases, which were all classified as malignant according to the veterinary criteria but were diagnosed as leiomyoma (20 cases) or SMT-UMP (11 cases) using the human criteria. Twenty-six of the 31 discordant cases were in the female genital tract, 3/31 were in the alimentary tract, 1/31 was in soft tissue, and 1/31 was in the kidney.
Reclassification according to the human criteria was based on the following:
Ten cases were diagnosed as leiomyosarcoma based on the veterinary criteria because of the presence of necrosis. These cases were reclassified as leiomyoma based on the human criteria because the necrosis was of the infarct-type and concurrent atypia and mitotic activity were absent. Five of these cases were genital and 5 were extragenital.
Ten cases were diagnosed as leiomyosarcoma based on the veterinary criteria because of the presence of mitotic activity. These cases were reclassified as leiomyoma based on the human criteria because the mitotic count was below 10 and the tumors arose in the female genital tract.
Ten cases were diagnosed as leiomyosarcoma based on the veterinary criteria because of the concurrent presence of mitotic activity and tumor cell necrosis. These cases were reclassified as SMT-UMP according to the human criteria because the mitotic count was below 10 and the tumors developed in the female genital tract.
One case was diagnosed as leiomyosarcoma based on the veterinary criteria because of the presence of mitotic activity. This case was reclassified as SMT-UMP based on the human criteria because, despite a mitotic count of 13, atypia and tumor cell necrosis were absent, and the tumor was in the female genital tract.
Clinicopathologic Features of Canine SMTs in Distinct Anatomical Sites
There were 44 genital tract SMTs, of which 30 were vaginal, 11 uterine, and 3 vulvar. The veterinary criteria identified 12 leiomyomas and 32 leiomyosarcomas, while the human criteria identified 27 leiomyomas, 11 SMTs-UMP, and 6 leiomyosarcomas.
There were 23 alimentary SMTs, of which 1 was in the esophagus, 8 were in the stomach, 11 were in the small intestine, and 3 were in the large intestine. The veterinary criteria identified 4 leiomyomas and 19 leiomyosarcomas, while the human criteria identified 7 leiomyomas and 16 leiomyosarcomas.
There were 21 SMTs located in the soft tissue, of which 10 were superficial (subcutaneous) and 11 were deep-seated (below the subcutis or intracavitary). Superficial cases were located on the limbs (5 cases), perineal region (3 cases), or perianal region (2 cases). Deep-seated cases were retroperitoneal (5 cases), intrapelvic (4 cases), mesenteric (1 case), or within the muscles of the pelvic diaphragm (1 case). The veterinary criteria identified 2 leiomyomas and 19 leiomyosarcomas, while the human criteria identified 3 leiomyomas and 18 leiomyosarcomas. Of the 21 soft tissue SMTs, 7 expressed hormone receptors (6 leiomyosarcomas and 1 leiomyoma, according with both veterinary and human criteria); 5 of these were ER-positive and PR-negative, 1 was PR-positive and ER-negative, and 1 was ER- and PR-positive (Figs. 22, 23). Three of the hormone receptors-expressing soft tissue SMTs were deep-seated (2 retroperitoneal and 1 intrapelvic) and 4 were subcutaneous (all in the perianal or perineal region).
There were 11 SMTs of the urinary bladder. The veterinary and human criteria were concordant in these cases and both sets of criteria identified 4 leiomyomas and 7 leiomyosarcomas. There were 4 splenic SMTs, all diagnosed as leiomyosarcomas with both the veterinary and human criteria. Other sites (miscellaneous) included 1 kidney and 1 gallbladder SMT, and these were diagnosed as leiomyoma and leiomyosarcoma, respectively, with both the veterinary and human criteria.
Clinical Follow-Up
Clinical follow-up data were available for 25 cases. Overall, 4/25 dogs had evidence of local recurrence or metastasis. Specifically, 1 dog with soft tissue (pelvic diaphragm) SMT developed local recurrence; 2 dogs (one with perineal SMT and one with small intestinal SMT) developed suspected metastases; and 1 dog with genital SMT developed both local recurrence and suspected metastases. Metastases were confirmed in the liver by histopathology in 1 dog and by diagnostic imaging (suspected metastases) in the brain of 1 dog and in the liver of another dog. Six dogs died due to tumor-related causes: 4 due to relapse (recurrence, metastasis/or suspected metastasis, or both) and 2 were euthanized during surgery for resection of an alimentary tract SMT. The status of surgical margins was unknown in 1 of the 2 cases of recurrence (located in the soft tissue) and infiltrated in the second case (located in the female genital tract).
The presence of metastases or suspected metastases correlated with mitotic count, tumor cell necrosis, Ki67 labeling index, and tumor-related death. Tumor-related deaths were correlated with the size, atypia, mitotic count, tumor cell necrosis, presence of nuclear atypia, Ki67 labeling index, differentiation, and lack of desmin expression. Recurrence was associated with perivascular fibrosis, palisading, multinucleated cells, and tumor-related death.
Of the 25 cases with available follow-up:
Four were diagnosed as leiomyoma by both the human criteria and the veterinary criteria; none of these developed relapses or died of tumor-related causes during the follow-up period.
Six were diagnosed as leiomyosarcoma with the veterinary criteria and as leiomyoma or SMT-UMP with the human criteria; none of these developed relapses or died of tumor-related causes during the follow-up period.
Fifteen were diagnosed as leiomyosarcoma with both the veterinary and human criteria; all the cases which developed relapses/suspected relapses or died of tumor-related causes belong to this group.
There was no statically significant correlation between diagnosis and relapse or death from tumor-related causes.
Association Between the Clinical and Pathological Variables and Diagnoses
After adjusting for confounding effects, univariate analysis showed that 4 variables were significantly associated with diagnosis according to the veterinary criteria: mitotic count, necrosis, Ki67 labeling index, and differentiation (Supplemental Table S3). The variables that were significantly associated with diagnosis according to the human criteria were mitotic count, tumor cell necrosis, Ki67 labeling index, differentiation, nuclear atypia, and presence of multinucleated cells (Supplemental Table S4).
The elastic net model (multivariable model) identified a set of variables whose combination was associated, with good accuracy, with the diagnosis according to the veterinary criteria (AUC of ROC = 0.78 when considering 4 categories for sex; AUC of ROC = 0.811 when considering 2 categories for sex; Supplemental Table S5). Specifically, the variables that showed a partial association with a diagnosis of leiomyosarcoma were a higher amount of necrosis, increased mitotic count, presence of nuclear atypia, increased Ki67 labeling index, soft tissue origin, sex (female intact, male intact and male castrated; or male when considering 2 categories for sex), mixed collagen pattern, and poor differentiation. The variables associated with a diagnosis of leiomyoma were presence of perivascular fibrosis, desmin expression, vesicular chromatin, lower urinary tract origin, and increased collagen amount. Results obtained considering either 4 or 2 categories for sex were consistent with those listed above, but 2 other variables showed association with the diagnosis of leiomyosarcoma when considering 2 categories for sex: the presence of hyalinized stroma and slender nuclei.
When considering the diagnosis according to the human criteria, the prediction accuracy of the elastic-net model ranged from 0.688 (when discriminating between leiomyoma and SMT-UMP, and considering 2 categories for sex) to 0.974 (when discriminating between SMT-UMP and leiomyosarcoma, and considering 2 categories for sex), indicating good performance in all comparisons (Supplemental Tables S6, S7, and S8).
When comparing leiomyoma and leiomyosarcoma, the variables associated with a diagnosis of leiomyosarcoma were presence of nuclear atypia, increased mitotic count, poor differentiation, soft tissue or splenic origin, increased Ki67 labeling index, sex (male and male castrated, or male, when considering 2 categories for sex), mixed collagen pattern, increased amount of tumor cell necrosis, oval nucleus, and perivascular fibrosis. The variables associated with a diagnosis of leiomyoma were genital origin, sex (female intact), slender nucleus, absence of collagen, and myxoid differentiation. Results obtained considering 4 or 2 categories of sex were consistent with those above, and when considering 2 categories for sex, we also identified interfascicular collagen pattern to have a weak association with a diagnosis of leiomyoma.
When comparing leiomyoma and SMT-UMP, the variables associated with a diagnosis of SMT-UMP were increased amount of tumor cell necrosis, increased mitotic count, female genital tract origin, presence of trabecular pattern, loss of differentiation, presence of nuclear palisading, and oval nucleus. The variables associated with the diagnosis of leiomyoma were cigar-shaped nucleus and alimentary tract origin. Results obtained considering 4 or 2 categories of sex were consistent with those above, and when considering 2 categories of sex, oval nucleus, mixed collagen pattern, and collagen amount were also associated with a diagnosis of SMT-UMP, while hyalinized stroma was associated with a diagnosis of leiomyoma.
Finally, when comparing SMT-UMP and leiomyosarcoma, the variables associated with a diagnosis of SMT-UMP were female genital tract origin, presence of nuclear palisading, presence of trabecular pattern, slender and oval nucleus, interfascicular collagen pattern, sex (female intact), increased amount of collagen, tumor cell necrosis, and age. The variables associated with a diagnosis of leiomyosarcoma were reduced desmin expression, increased mitotic count, increased nuclear atypia, increased Ki67 labeling index, presence of perivascular fibrosis, soft tissue origin, splenic and lower urinary tract origin, reduced differentiation, vesicular chromatin, sex (male intact or male when considering 2 categories for sex), hyalinized stroma, cigar-shaped nucleus, presence of multinucleated cells, and mixed collagen pattern. The results obtained considering 4 or 2 categories of sex were consistent.
Raw data for each of the cases included are reported in Supplemental Table S9. Examples of histological features not reported in Figures 3 to 14 are reported in Supplemental Figures S10.
Discussion
One of the main difficulties in the diagnosis and prognosis of SMTs in veterinary medicine is the lack of specific guidelines to differentiate leiomyomas from leiomyosarcomas by histopathology. Based on this premise, we applied and compared the histopathological features used in veterinary and human medicine to a series of 105 canine SMTs to assess the best morphological features that distinguish between benign and malignant tumors. 1,12,14,18 The statistically significant correlation between differentiation, proliferative activity, and tumor cell necrosis was expected, because these parameters are often included in grading systems as indicators of malignancy. 2,14
Interestingly, when nuclear atypia was defined as more than mild and visible at 10×, it correlated with differentiation, mitotic count, Ki67 labeling index, and multinucleation. This result was unexpected as the assessment of atypia has been reported to be subjective. Our findings suggest that the guidelines used in human medicine 12 allow for a more reliable identification of nuclear atypia and avoid overestimation of this feature. Importantly, nuclear atypia is a relevant feature of malignancy in human SMTs, 12 and the application of the same criterion to canine SMTs represents a promising method to facilitate the distinction between benign and malignant SMTs.
Tumor cell necrosis was found to correlate with differentiation and proliferative activity, as well as with the size of the tumor and amount of collagen. These correlations suggested that angiogenesis may not always be efficient in SMTs and that hypoxia may develop, leading to necrosis in larger tumors.
Another interesting finding was the correlation of mitotic count with reduced differentiation and with a lack of desmin expression, suggesting that the loss of differentiated smooth muscle cell markers may parallel an increase in proliferative potential. Finally, the amount of collagen identified in canine SMTs was variable and correlated with the pattern of collagen distribution, being more abundant in cases with interstitial and mixed collagen. The amount of collagen also correlated with the amount of tumor cell necrosis. These findings, and the lack of correlation with desmin expression or degree of differentiation, suggest that the presence of collagen, even when surrounding individualized neoplastic cells, does not imply a reduced differentiation of the tumor, but may more likely represent a reaction to hypoxia.
Common histologic features of canine SMTs included nuclear palisading and trabecular pattern, and both were more frequent in genital SMTs than in SMTs of other sites. These features may represent features typical of genital SMTs and should be kept in mind especially to avoid a misdiagnosis of peripheral nerve sheath tumors based on the presence of nuclear palisading.
The veterinary and human criteria systems to discriminate between benign and malignant SMTs disagreed in 30% of the canine SMT cases. All of these were diagnosed as leiomyosarcomas according to the veterinary criteria but were reclassified as leiomyomas or SMTs-UMP when following the human criteria. Most of these discordant cases were in the female genital tract where the veterinary criteria identified 73% of genital SMT cases as leiomyosarcoma, while the human criteria identified only 14% of these cases as malignant. This result was expected considering that there are human criteria specific for this location. Furthermore, the human criteria tolerate a greater mitotic count for a diagnosis of genital leiomyoma, and that was the main reason for reclassification in this study. 12,18 The human criteria seemed to better reflect the benign behavior of the majority of genital SMTs in dogs, 1 while the veterinary criteria seem to overestimate the diagnosis of leiomyosarcoma in this site. Unfortunately, in our case series, follow-up data were available only for a minority of dogs with SMTs. However, in that subset of patients, all the cases associated with local recurrence and/or distant metastasis or tumor-related death were classified as leiomyosarcoma with both the human and veterinary criteria. Thus, further prospective prognostic studies on canine SMTs of the female genital tract are needed to confirm that the human criteria predict their biological behavior better than the veterinary criteria.
The second most frequent reason for reclassification was the morphological type of necrosis as the human criteria specifically excludes infarct-type necrosis from the morphological criteria of malignancy. 12 This distinction between infarct-type and tumor cell necrosis allowed the reclassification of only 5 extra-genital SMTs that were diagnosed as leiomyosarcoma based on necrosis only, when using the veterinary criteria. The exclusion of infarct-type necrosis is based on the fact that leiomyomas can reach a large size (up to 15 cm in this case series), leading to a hypoxic microenvironment causing central necrosis despite the benign nature of the tumor. Nevertheless, early small foci of infarct-type necrosis might be difficult to differentiate from tumor-cell necrosis. Sections should be carefully examined to identify specific morphological features to facilitate this distinction.
Furthermore, the human criteria include a third category of SMT-UMP in the female genital tract: this category had a Ki67 labeling index lower than leiomyosarcomas (similar to leiomyomas) but a size larger than leiomyomas. Thus, SMTs-UMP may also have intermediate features between benign and malignant SMTs in dogs. Interestingly, all the cases with available follow-up data that were reclassified from leiomyosarcoma to a more benign category (leiomyoma or SMT-UMP) had benign tumor behavior. Since data regarding the behavior of neoplasms with intermediate histologic features of malignancy are still scarce in human medicine 18 and have never been investigated in dogs, this topic warrants further evaluation.
The most frequent sites of SMT development in our caseload were the female genital and gastrointestinal tracts, paralleling previous reports. 1,8,13 The third most-represented site was soft tissue, which was unexpected considering the paucity of reports on soft tissue SMTs in dogs. 11 The spleen was less represented than expected, but the number of splenic cases in this study may underestimate the true incidence of splenic SMTs since most SMTs in this study were collected from referral practices while splenectomy is often performed in general practice.
The distribution of SMTs within the different organ systems was expected. We found vaginal tumors to be the most frequent, as previously reported in the literature. 1 In the gastrointestinal tract, the small intestine was the most common site, followed by the stomach, while cases in the large intestine and esophagus were rare. These data partially confirm the reported low frequency of SMTs in the large intestine and esophagus, 3,4,7 but differ with the reported frequency of SMTs in the stomach compared with the small intestine. 3,4
In the human literature, soft tissue SMTs are divided into 2 major groups: superficial and deep-seated. However, deep-seated SMTs have not been previously identified in veterinary medicine. Interestingly, in this study, half of the superficial cases were in the perineal/perianal region, and half of the deep-seated cases were in the pelvic cavity or within the tissues of the pelvic diaphragm. Thus, 10/21 cases (15/21 if the retroperitoneal SMTs are included) arose in the soft tissues adjacent to the genital system. While the retroperitoneal location of leiomyosarcoma has been occasionally reported in the dog, 10 the occurrence of SMTs in the pelvic cavity and perigenital soft tissue of dogs is novel and parallels reports in human medicine. 5 Human deep-seated leiomyomas arise most frequently in the pelvic cavity and retroperitoneum, and are believed to arise from hormonally sensitive, resident smooth muscle cells. 5,6,15 These tumors express the ER and PR, and are referred to as leiomyoma of the gynecologic type. 5,6,12 In our case series, 7 soft tissue SMTs expressed one or both hormone receptors and were all located in perigenital soft tissues. There is a discrepancy between our findings and the human literature, since 6/7 of the perigenital SMT cases in this study were diagnosed as leiomyosarcoma, not leiomyoma, by both the veterinary and human criteria. However, if we classified the perigenital soft tissue SMTs using the human genital SMT criteria, 12 only 2 cases were diagnosed as canine leiomyosarcoma, and one of these had distant metastases. The application of the human criteria used for genital SMTs to extra-genital, hormone receptor-positive cases seems reasonable, but as for genital SMTs, prospective studies are recommended to justify and validate these diagnostic criteria.
In this study, the veterinary and human criteria led to the same diagnoses for urinary bladder and splenic SMTs. The majority of SMTs in the urinary bladder were diagnosed as leiomyosarcoma with both the veterinary and human criteria. This result contrasts with previous data that report benign SMTs of the urinary bladder to be more frequent. 1 Nevertheless, the lack of prognostic studies on SMTs in this location makes hypotheses on this matter speculative.
In the cases for which follow-up data were available, proliferative activity and tumor cell necrosis strongly correlated with metastasis and tumor-related death. Tumor cell necrosis also correlated with decreased differentiation and lack of desmin expression. Even though these parameters were part of the diagnostic algorithm, the diagnosis of leiomyoma or leiomyosarcoma, based on the veterinary or human criteria, did not correlate with clinical variables. This may be a consequence of the small number of events available in this case series. Therefore, further prognostic studies are necessary to confirm the prognostic value of those parameters. Prediction of local recurrence is also a topic for future studies and should include assessment of the status of surgical margins and infiltrative growth. In the present case series, only 2 cases of recurrence were recorded. Of these, only one had a known status of the surgical margins. Furthermore, only one of the cases in this study had clear evidence of infiltrative growth, which is included in the veterinary criteria. These limitations are likely the consequence of the retrospective nature of this study, and conclusions on this matter cannot be drawn based on the present data.
The multivariable analysis identified the following variables associated with the diagnosis of leiomyosarcoma independent of the criteria used: soft tissue location, male sex, Ki67 labeling index, reduced differentiation, and mixed collagen pattern. These parameters may provide further support to the diagnosis of malignancy, and therefore warrant further consideration as to whether they should be added to the current diagnostic criteria. The veterinary criteria currently include, tissue infiltration, mitotic count, and tumor cell necrosis. The human criteria include nuclear atypia, in addition to mitotic count and tumor cell necrosis. Intact female sex was associated with malignancy when we applied the veterinary criteria, while it was associated with leiomyoma and SMT-UMP when we used the human criteria. This discrepancy is likely a consequence of the reclassification of many of the SMTs located in the female genital tract using the human criteria.
Further variables associated with the diagnosis of leiomyoma and SMT-UMP when applying the human criteria included: female genital tract, presence of slender nuclei, nuclear palisading, myxoid change, and trabecular pattern. These data suggest that these features, although uncommon, may be particular to SMTs of genital origin.
In conclusion, this study describes and compares the clinicopathological features of canine SMTs in different organ systems and describes for the first time soft tissue SMTs of gynecologic type in dogs. Our results also expand the knowledge of SMTs of soft tissues, by describing deep-seated SMTs, their preferential perigenital location, and the ER and/or PR expression in a subset of these tumors. These data suggest the usefulness of the human criteria to differentiate benign from malignant SMTs of the female genital tract in dogs because the human criteria better predicted the biological behavior of the tumors. 1 Furthermore, the application of guidelines from the human criteria to assess nuclear atypia and tumor cell necrosis 1 seem to help prevent overdiagnosis of malignant SMTs. Further prognostic studies are warranted to confirm the better performance of the human criteria in genital SMTs, where the diagnosis of malignancy seems to be overestimated by the veterinary criteria and where a benign behavior is generally expected. Further work is also needed to assess the usefulness of the human criteria in hormone receptor-expressing SMTs of soft tissue in dogs and to define the prognostic parameters for canine SMTs in general.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211066862 - Canine smooth muscle tumors: A clinicopathological study
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211066862 for Canine smooth muscle tumors: A clinicopathological study by Giancarlo Avallone, Valeria Pellegrino, Luisa Vera Muscatello, Paola Roccabianca, Gastone Castellani, Claudia Sala, Marco Tecilla, Paola Valenti and Giuseppe Sarli in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-xlsx-1-vet-10.1177_03009858211066862 - Canine smooth muscle tumors: A clinicopathological study
Supplemental Material, sj-xlsx-1-vet-10.1177_03009858211066862 for Canine smooth muscle tumors: A clinicopathological study by Giancarlo Avallone, Valeria Pellegrino, Luisa Vera Muscatello, Paola Roccabianca, Gastone Castellani, Claudia Sala, Marco Tecilla, Paola Valenti and Giuseppe Sarli in Veterinary Pathology
Footnotes
Acknowledgement
We thank Leah Cannon, PhD, for editing a draft of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
