Abstract
A new gene defect in Fleckvieh calves leads to a syndrome with partial phenotype overlap with bovine hereditary zinc deficiency. A mutation in a gene encoding phospholipase D4 (PLD4), an endosomal exonuclease, causes the disorder. In mice, PLD4 activity indirectly regulates the Toll-like receptor 9 (TLR9) pathway via degradation of microbial DNA. PLD4 absence thus results in visceral macrophage activation comparable to human macrophage activation syndrome. In this study, disease progression and the role of macrophages in affected calves were monitored clinically, clinicopathologically, and histologically over time. Breeding data identified 73 risk matings of heterozygous carriers resulting in 54 potentially PLD4-deficient calves born on farms. PLD4 status was examined via 5′-exonuclease assay, detecting 6 calves carrying the defect. These were purchased and monitored daily until final necropsy. The calves developed progressive skin lesions starting with small scaling areas terminating in severe crusting dermatitis, especially in areas with mechanical exposure. Histological and immunohistochemical analyses indicated that macrophages with cytoplasmic vacuolation increased considerably in skin sections obtained weekly during the disease course. Macrophage increase correlated with increased dermal lesion severity. Macrophage activation was confirmed by prominent phagocytic activity in the superficial dermis using electron microscopy. Dermal mRNA abundance of CCL2 and CCL3 measured by quantitative polymerase chain reaction verified macrophage activation. Further increase in mRNA of downstream molecule MyD88 and cytokine IL12b connected bovine PLD4 deficiency to increased TLR9 pathway activation. In contrast to human macrophage activation syndrome, the main feature of bovine PLD4 deficiency was local disease in organs with contact to microbial DNA (skin, intestine, lungs).
Keywords
Over more than 20 years, sporadic cases of Bavarian Fleckvieh calves with severe skin lesions strikingly similar to bovine hereditary zinc deficiency were observed. An accumulation of 7 cases incurable by zinc supplementation appearing within a short period of only 4 years aroused suspicion of a possibly new gene defect circulating in the cattle population. 12,16 A p.W215X-nonsense mutation in a phospholipase D4 encoding gene (PLD4) was identified as causal polymorphism via SNP genotyping and subsequent gene sequencing in these calves. 12 The clinical phenotype of the affected animals was characterized by ulcerative and crusting dermatitis, frequently accompanied by pneumonia, enteritis, and growth retardation. Because of the phenotypic similarity to bovine hereditary zinc deficiency, the disease was tentatively named zinc deficiency–like syndrome. 12,16
PLDs serve as important phospholipid signaling enzymes, which are important players involved in epidermal development and different leukocyte functions. 1,2,18 PLD4 is a phospholipase without classical PLD enzyme activity and widespread distribution with predominant expression in Iba1-positive microglia and splenic marginal zone macrophages. 29 PLD4 is associated with macrophage/microglia activation and survival and it is colocalized with early phagosomes. 8,23 In the Japanese human population, PLD4 is a susceptibility gene for the development of rheumatoid arthritis and systemic sclerosis, 22,28 autoimmune diseases involving CD4+ T cell responses, which also involve macrophages. The function of PLD4 and its role in cellular signaling pathways remained unknown for a long period of time. However, a recent discovery linked it to endosomal nucleic acid degradation, thus modulating the activation of the endosomal foreign DNA sensor toll-like receptor 9 (TLR9). PLD-deficient mice present a condition similar to human macrophage activation syndrome (MAS), which is characterized by fever, lymphadenopathy, and symptoms associated with a cytokine storm. 9
In the past, the initial PLD4-deficient calves were presented in a markedly reduced general condition and had to be euthanized for animal welfare reasons and unresponsiveness to therapeutic interventions few days after presentation. The age and history of the calves varied considerably and a major impact of different husbandry and management conditions on disease onset was therefore assumed. In histological sections, the skin displayed severe crusting with bacterial and fungal (Malassezia sp.) overgrowth, ulceration, marked epidermal alterations, and dermal inflammation. Distinguishing the syndrome from other dermatoses (like BHZD-associated dermatitis or nutrient deficiency) or identifying a pathogenic principle proved to be highly challenging. 16 This prospective study was initiated by the hypothesis that husbandry and management condition might influence and mask the disease course considerably. Furthermore, the scientific knowledge and literature at that time did not connect PLD4 to keratinocyte metabolism and cornification at all, but rather pointed toward a relation to macrophage function. Thus, we further hypothesized that the keratinization disorder is only indirectly connected to the PLD4 defect and that the underlying cause may be a different molecular mechanism, probably not related to keratinocytes at all. To test the hypotheses, we designed a prospective longitudinal study to include calves in their first weeks of life and to follow the course of disease clinically, clinicopathologically, and histologically from the onset of the syndrome until demise of the calves. With this approach, we aimed to highlight early and primary lesions, minimize the potential impact of environmental factors, investigate the role of dermal macrophages, and discover key features of this zinc deficiency–like syndrome that are connected to its pathogenesis. Based on recent discoveries of PLD4 function in mice, 9 a further aim of this study was to assess whether the TLR9 pathway is altered in affected calves.
Material and Methods
This prospective case study was registered and performed in agreement with the Animal Welfare and Ethics Committee of the government of Upper Bavaria. Animal husbandry and care were carried out in accordance with national and institutional guidelines.
Animals and Husbandry
In order to identify calves carrying the causative p.W215X mutation, we screened the Fleckvieh breeding database for potential risk matings that happened by chance during normal routine breeding of Bavarian Fleckvieh farms. If both the sire and the maternal grandsire are heterozygous for the p.W215X mutation, the probability of descendant calves being homozygous for the defective allele is 12.5%. To determine PLD4 status of the descendant calves, nasal swab samples taken from the respiratory/olfactory mucosa in the first weeks of life were analyzed via 5′-exonuclease assay as previously described. 12
Six calves identified as homozygous for the PLD4 gene defect were included into the study and subsequently purchased and housed at the Clinic for Ruminants with Ambulatory and Herd Health Services, LMU Munich.
Examination and Sampling
The 6 calves underwent a clinical examination every day, with emphasis on behavior, posture, body temperature, integument, cardiovascular system, respiratory and digestive tract, and palpable lymph nodes. Skin areas including head, neck, trunk, and skin covering joints identified as predilection sites in a previous study 16 were clipped regularly and were given special consideration. Once a week, the clipped skin sites were photographed, jugular blood samples for laboratory analysis were taken, and a neck skin biopsy sample was taken under local anesthesia for histologic and cytokine mRNA examination. Severity of skin lesions was scored from 1 (mild) to 5 (severe) based on presence of scales, crusts, and ulcerations.
Hematologic and chemistry parameters and enzyme activities were analyzed as previously described, using validated in-house reference values. 17 Trace and bulk elements from plasma and serum were analyzed and serum protein gel electrophoresis was performed at Vet Med Labor GmbH/IDEXX Laboratories, Ludwigsburg, Germany.
Necropsy, Light and Electron Microscopy
For histology, neck skin samples taken weekly during the monitoring phase were routinely fixed, embedded in paraffin and plastic, cut, and stained. 14 Calves euthanized due to study endpoints (eg, severe weight loss, severe skin disease, fever without response to therapy, severe diseases of single organ systems, or severely reduced overall condition) were necropsied and tissue samples were processed likewise. For electron microscopy, skin samples were obtained from calves 1, 3, and 4 at necropsy. The skin samples were trimmed and fixed in 6.25% glutardialdehyde and routinely processed. 15
Immunohistochemistry (IHC) was performed using heat-induced epitope retrieval as pretreatment. For immunolabeling of macrophages in skin samples a primary rabbit antibody directed against ionized calcium-binding adapter molecule 1 (Iba1, CP290; Biocare Medical) was used. Bovine lymph node sections served as positive control. Skin samples of 4 age-matched (range 2–12 weeks of age) Fleckvieh calves from the same region without overt and histological skin lesions were used as normal control calves. A negative control was run by replacement of the primary antibody with rabbit IgG (ab172730; Abcam).
After incubation with primary antibody solutions, secondary biotinylated goat anti-rabbit antibody (BA-1000; Vector) incubation was followed by ABC-peroxidase (PK-6100; Vector) and diaminobenzidine-tetrahydrochloride (DAB, 4170; Biotrend) for conventional chromogenic IHC.
Examination of Selected Cytokine mRNA Profiles
The mRNA abundance of selected bovine cytokines/cellular factors were measured by reverse transcription-quantitative PCR (RT-qPCR) in calves 1, 2, and 4. The measured genes were 2 checkpoint molecules of the TLR9 pathway (myeloid differentiation factor-88 [MYD88] and tumor necrosis factor receptor-associated factor 6 [TRAF6]), one cytokine associated with the TLR9 pathway (interleukin 12b [IL12b]) and 2 chemokines associated with macrophage activation, chemokine ligand 2 (CCL2) and CCL3. GAPDH was used as reference (housekeeper) gene. Two samples from study entry and the last 2 samples before euthanasia were selected and analyzed in duplicate. RNA was isolated using the RNeasy Mini kit (Qiagen) according to the manufacturer’s instruction. For RT-qPCR the QuantiTect Probe RT-PCR kit (Qiagen) was used. Intron-spanning oligonucleotide primers described by Taraktsoglou and colleagues were complemented by newly designed intron-spanning or exon-exon-junction targeting primers or TaqMan probes in order to quantify specific mRNA levels of the target genes (see Supplemental Table S1). 27 Primer and probes were used at final concentrations of 800 nM and 400 nM, respectively. The thermal profile of the PCR was: 50 °C for 30 minutes, 95 °C for 15 minutes, and 42 cycles of 94 °C for 15 seconds, and 60 °C for 60 seconds.
Data were transformed using the 2-ΔΔCt method to plot x-fold increase. 26
Results
Descendant Calves Born From Risk Matings
Analysis of risk matings identified 73 potential pregnancies. In 24 of these, no calf was born. The remaining 49 pregnancies resulted in birth of 54 calves (including 5 twin births). Calves were quite widely geographically distributed throughout Bavaria, with a particular concentration to the southeast of Munich (Fig. 1). Molecular genetic examination identified 7/54 calves (12.5%) as homozygous for the defective allele that were consequently eligible for inclusion into the study. A total of 27 calves were heterozygous and 19 calves were homozygous for the wild-type allele. One calf was not examined.
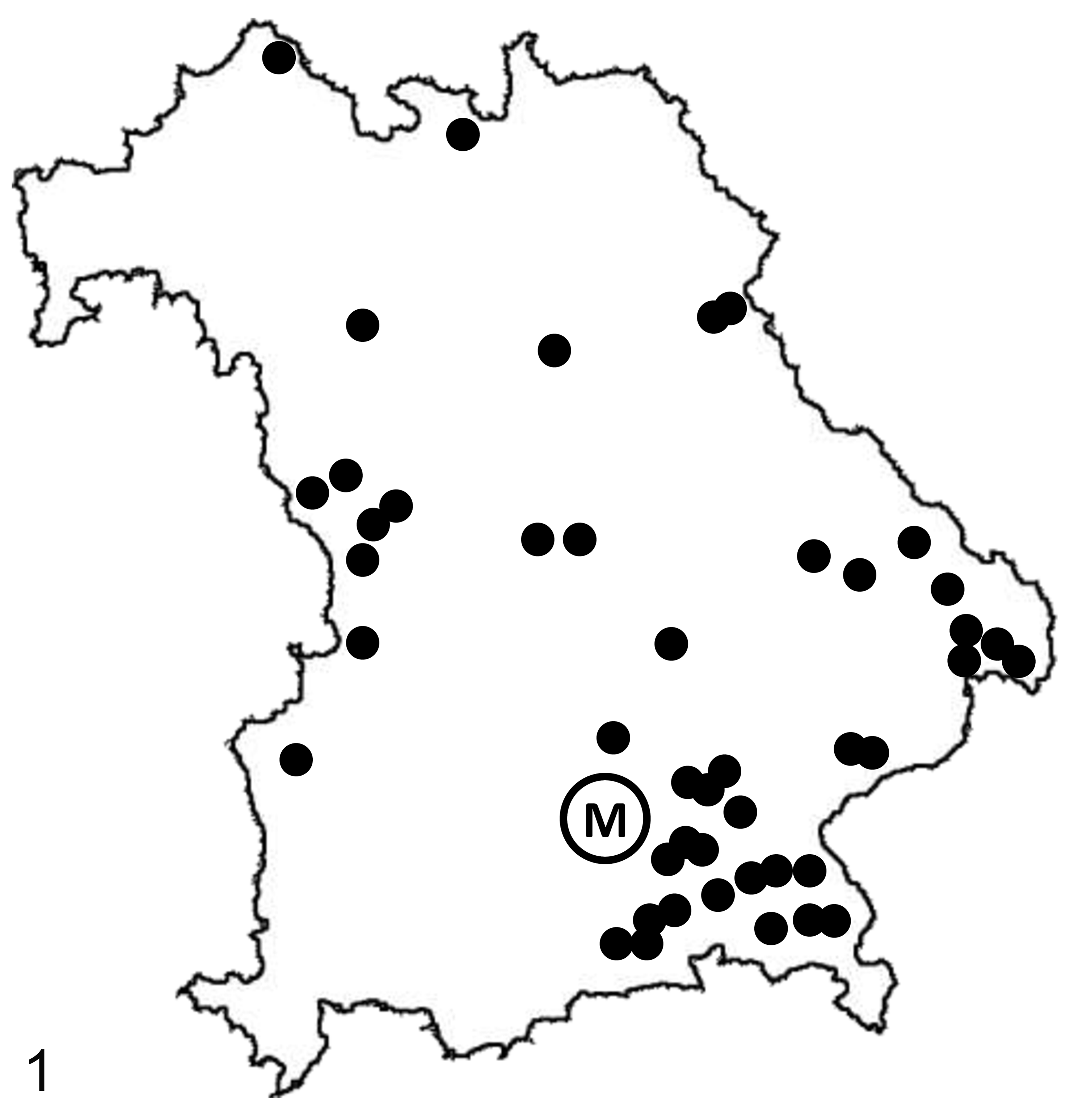
Distribution of pregnancies resulting from risk matings of sires heterozygous for the phospholipase D4 gene defect all over the state of Bavaria. M, Munich.
Clinical Findings
Five of the 7 homozygous Fleckvieh calves were males and 2 were females. Two of the males were twins. The ear biopsy of one female calf was BVDV-positive and the calf was therefore excluded from the study. The remaining 6 calves were purchased and included in the study.
Both twins (cases 5 and 6) and one other calf (case 3) displayed progressive disease development and a markedly reduced general condition at the time of purchase and had to be euthanized a few days later due to study endpoint criteria. The remaining 3 calves were monitored for a period of 49, 68, and 181 days, respectively (Table 1). Cases 1 and 4 developed quite severe scaling and crusting skin lesions when 8 weeks old, with progressive deterioration in the following weeks (Figs. 2 –10). The twin calves (cases 5 and 6) displayed similar lesion severity at a similar age (Fig. 2). Calf 3 displayed similar lesion severity much earlier, in the 4th week of life. Calf 2 developed waxing and waning oral mucosal erosions/ulcerations starting in the 8th week of life, and developed moderate skin lesions later, in the 28th week of life. Skin lesions were first detected above skin sites with mechanical exposure (such as knee folds and joint areas) in calves 1 and 2 and on the head in calf 4. Pyrexia lasting for several days was not observed. After reaching study endpoints, calves 1, 2, and 4 were euthanized and subsequently necropsied at the age of 85, 210, and 73 days, respectively (Table 1).
Basic information on phospholipase D4–deficient Fleckvieh calves. The data show the calf age (days) for study events, and body and organ weights at death.
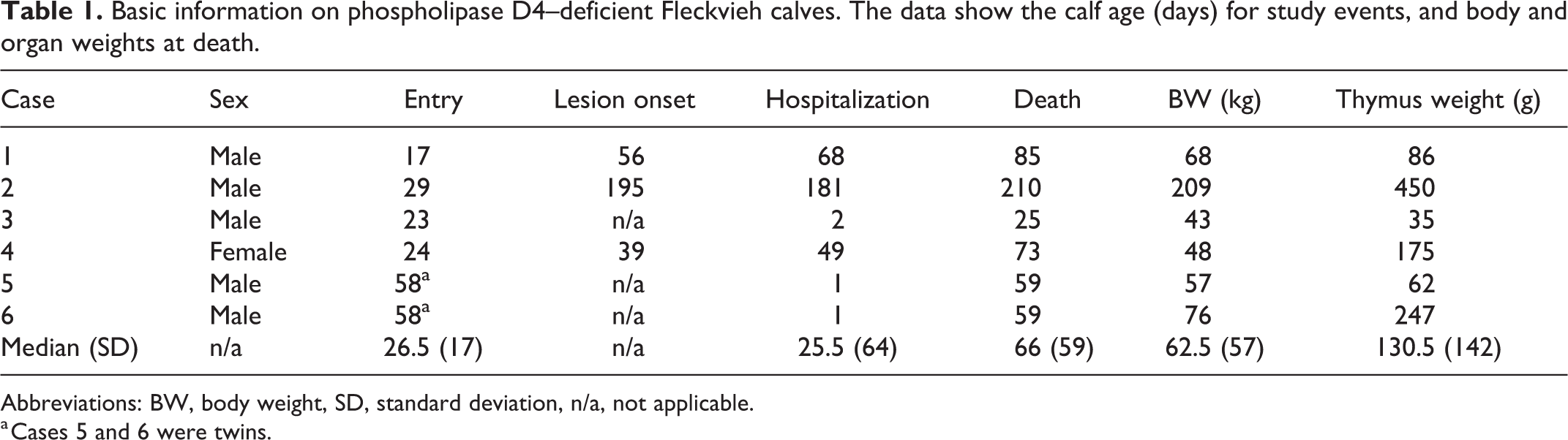
Abbreviations: BW, body weight, SD, standard deviation, n/a, not applicable.
a Cases 5 and 6 were twins.
Phospholipase D4 deficiency, skin, calves.
Laboratory Parameters
In the weekly blood samples, calves 1, 2, 4, and 5 had a mild normochromic microcytic anemia (hematocrit <0.30; hemoglobin <6.2 mmol/L; mean cellular volume <40 fL) over the whole observation period. Calves 3 and 6 did not have anemia. Thrombocyte and leukocyte counts were mostly normal, with single values above or below the reference ranges. Serum iron concentrations were low in calves 1, 3, and 5. Plasma copper concentration was within the reference range in all calves, except for calf 2, which was slightly below the reference range. Serum selenium concentrations were low in all calves (25.4–47.3 µg/L). Serum zinc concentrations were low in calves 3, 5, and 6 (294–646 µg/L) and marginal or within reference range in the remaining calves. Serum albumin concentration was marginally below the reference range in calves 1 and 5 (mean 29 g/L and 28 g/L, respectively; range: 30–40 g/L) and within reference range in the other calves. Globulin concentration was within reference limits in all calves. The remaining serum parameters and enzyme activities displayed no specific pattern over the observation period and were within reference ranges, with single values lying outside the upper or lower limits (see Supplemental Table S2).
Necropsy, Light and Electron Microscopy
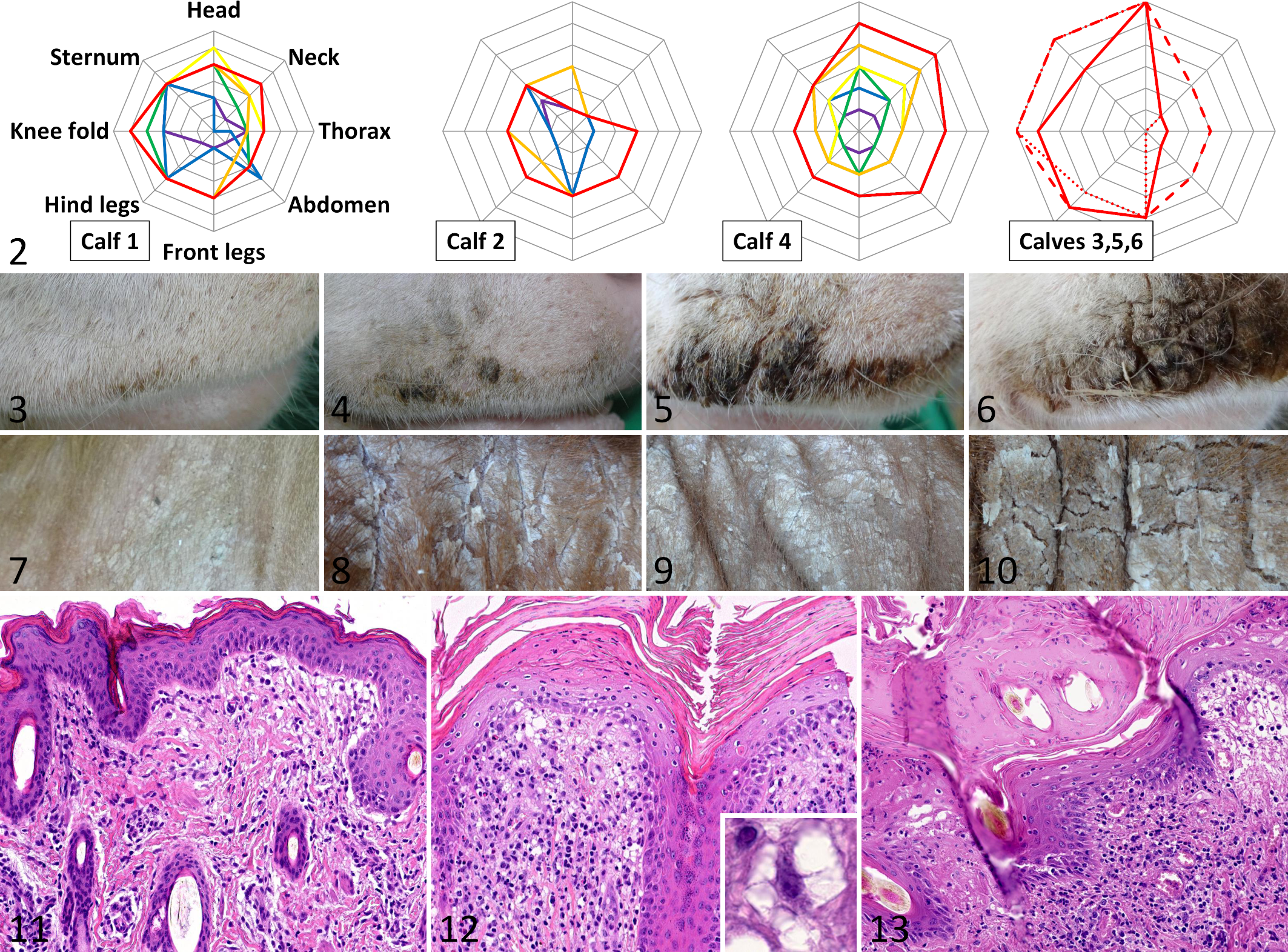
The histological progression of disordered keratinization reflected the detected clinical progression of increasing scabs and crusts. With the onset of clinical signs (scabs and crusts) a few weeks prior to necropsy, the histological lesions developed from early mild orthokeratotic hyperkeratosis and epidermal hyperplasia to diffuse severe orthokeratotic and parakeratotic hyperkeratosis with serum lakes, epidermal hyperplasia or erosion and ulceration. In parallel, the dermal inflammation, consisting of macrophages, lymphocytes, and few plasma cells, also increased in severity (Figs. 11 –13).
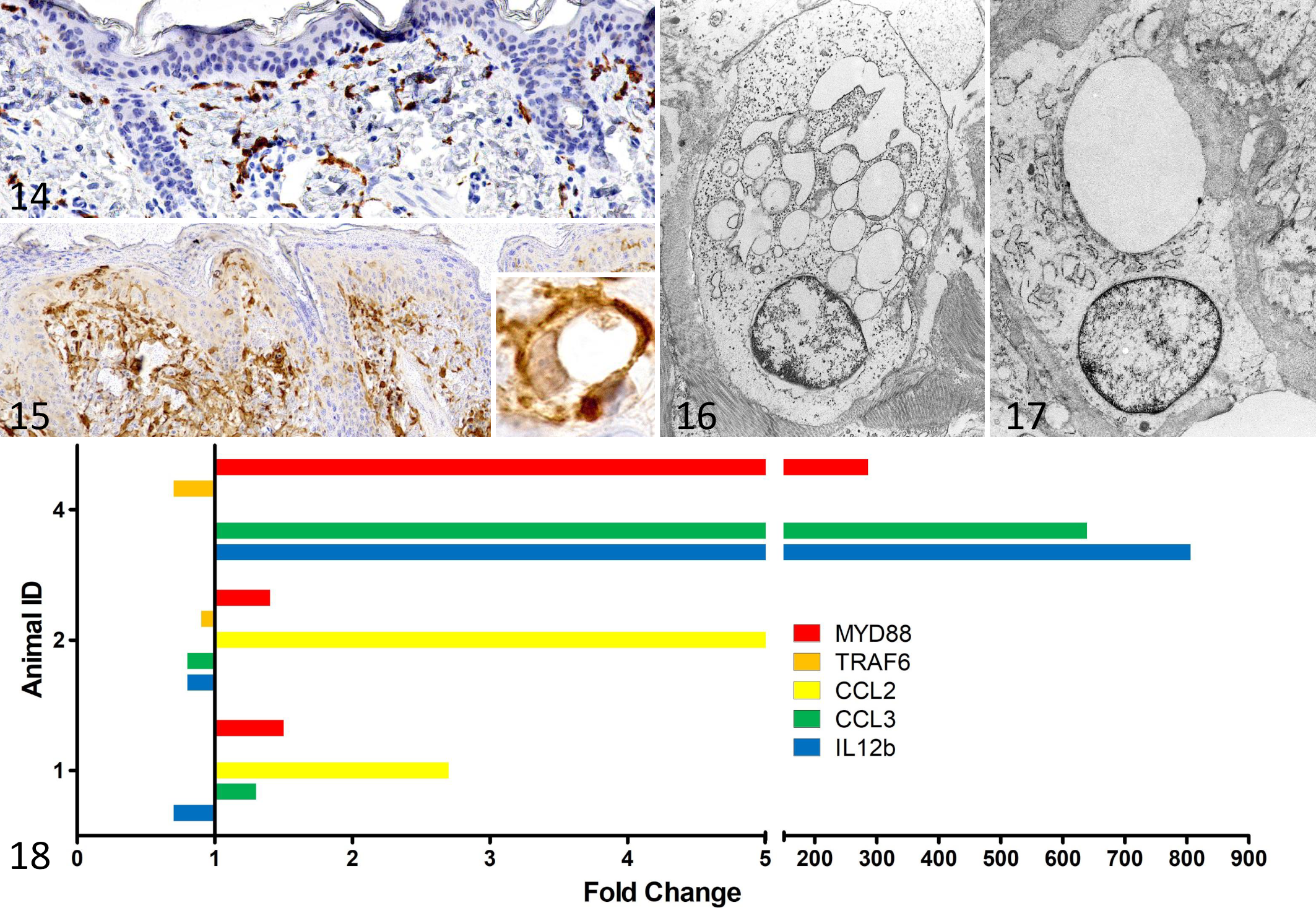
A distinct feature of the dermal lesions was accumulation of large vacuolated histiocytoid cells that were situated either dermal perivascular or along the dermo-epidermal interface. These cells had multiple clear, macrovesicular cytoplasmic vacuoles with thin bridges in between (Fig. 12 inset). The cells were positive with Iba1-immunohistochemistry (confirming histiocytic origin). In all 3 animals in which disease course was studied, dermal macrophages were already present in moderate to large numbers in the dermis at an early age, when compared with normal control calves (Fig. 14). With increasing disease severity, the number of vacuolated macrophages in the dermis increased (Fig. 15). Infiltration of dermal macrophages preceded clinical visible skin lesions and histologically detectable epidermal lesions like crusting and hyperkeratosis. In late disease, there were extensive crusts, epidermal intracellular and intercellular edema, erosions and ulcerations, and severe dermal inflammation.
Phospholipase D4 deficiency, skin, calves.
Electron microscopy revealed vacuolated macrophages with multiple lysosomes and formation of pseudopodia (activation) and neutrophils along the dermo-epidermal interface. Several cytoplasmic vacuoles with clear or flocculent content corresponded to the vacuolated appearance of these cells in light microscopy. Macrophages and neutrophils frequently displayed engulfing of extracellular material (phagocytotic activity, Figs. 16, 17).
At necropsy, in addition to skin lesions, 4 of the calves were underweight when compared to other Fleckvieh calves. 24 Each of the 6 calves displayed inflammation of the respiratory and digestive tracts. Suppurative rhinitis was detected in 3/3 calves, and there was suppurative (2/5) and lymphoplasmacytic (3/5) tracheitis. Suppurative bronchopneumonia was evident in 5/6 calves. Small intestinal lesions consisted of villous blunting or fusion and increased number of lymphocytes and plasma cells. Widespread hemorrhages were not observed. Lung cultures did not reveal specific bacterial pathogens in any of the 5 calves tested. Four of 6 intestinal samples showed growth of Escherichia coli and Salmonella enrichment was negative in 5/5 calves. Three of 6 calves had intestinal coronavirus virions detected by electron microscopy. Mean thymus weight was 176 g, and mean thymus–body weight ratio was 0.2%. The thymus weights were 9.8-fold higher than the average weight of hypoplastic thymuses of calves with bovine hereditary zinc deficiency. 5
Dermal Cytokine mRNA Profiles Over the Course of the Disease
The mRNA abundance of macrophage-associated chemokines CCL2 and CCL3 increased over the study period in 2/2 and 2/3 calves studied, respectively (Fig. 18). In calf 4, CCL2 expression was not calculated because the Ct values were 42. CCL3 expression was decreased in calf 2. Of the 3 factors associated with the TLR9 pathway, MYD88 expression increased in 3/3 calves. TRAF6 and IL12b expression decreased over the study period in 2/3 calves, but IL12b concentration increased in calf 4.
Discussion
After the initial characterization of zinc deficiency–like syndrome based on spontaneous cases examined in different stages of the disease, 12,16 we investigated the pathogenesis of the gene defect using a prospective study design to include calves before clinical signs were present. The findings of the present study provide evidence that exuberant macrophage activation is a key feature of the dermal disease course and likely plays a central role in the pathogenesis of skin lesions of PLD4-deficient calves.
Recently, the role of PLD4 has been identified as an endosomal exonuclease directly connected to the TLR9 pathway. TLR9 senses viral and bacterial DNA and leads to activation of macrophages via induction of proinflammatory cytokines and type I interferons. 4,31 PLD4 and PLD3 degrade foreign oligonucleotides, which are sensed by TLR9 and thus decrease TLR9 activation. In other words, PLD4 and PLD3 activity exerts an inhibitory effect on TLR9, which is supported by the finding that PLD4-null mice had a similar phenotype as mice with overstimulated TLR9. 4 These experimental studies link TLR9 activation in mice to macrophage-activation syndrome in humans. 4,9
Macrophages mount first-line inflammatory responses to microbial pathogens in many organs. In the investigated calves, the disorder thus resulted in inflammation of organs which are in contact to the outer environment (skin, respiratory tract, digestive tract).
TLRs detect pathogen specific molecular patterns (PAMP). TLR9 is situated intracellularly in endosomes or lysosomes preferentially in macrophages or dendritic cells. 23,29,31 Its pathway starts with activation through microbial oligonucleotides containing specific unmethylated CpG motifs. MyD88, an adaptor molecule located on the cytosolic side, is essential for TLR9 downstream signaling. Recruitment of further proteins, including TRAF6, leads to a signal cascade which eventually activates NF-κB and activator protein 1 (AP-1). These are transcription factors that affect expression of several genes related to immunity. 13
With increasing disease severity, the number of macrophages in the calf skin increased in hematoxylin–eosin-stained sections, which was confirmed by immunohistochemistry and electron microscopy. Also, cytoplasmic vacuolation and frequently detected phagocytotic activity detected by light and electron microscopy indicate macrophage activation. Thus, we investigated mRNA of chemokines associated with macrophage activation (CCL2, CCL3) and factors of the TLR9 signaling pathway (myD88, TRAF6, IL12b). Although only few calves were examined and the magnitude of increase were considerably different, the partial increase of expression of these genes further hints toward excessive macrophage activation. CCL2 and CCL3, involved in recruitment and activation of macrophages, were increased with progressing disease in 2 calves. CCL2 was also increased in PLD3−/−/PLD4−/− mice, which showed increased macrophage activation. 9 In calf 4, IL12b mRNA was increased, and this is one of the proinflammatory factors released by activated macrophages following TLR9 receptor activation. This reflects the situation in PLD4−/− mice, where excessive TLR9 activation leads to increased IL12b production. 9 Further investigation of MyD88, a main TLR9 pathway contributor processing TLR9 downstream signals, 31 displayed an increase as well.
Interestingly, the mRNA of TRAF6 and IL12b, a downstream molecule of MyD88, was decreased in 2 examined calves. This may be due to a negative feedback loop owing to consistent NF-κB activation. Although we were not able to test this, mouse experiments studying the NF-κB regulation axis support this hypothesis. In mice, NF-κB activation leads to ovarian tumor deubiquitinase 1 (OTUD1) induction, which negatively regulates TRAF3 and TRAF6. 30 Studies of cancer cells reveal that micro-RNAs also negatively regulate TRAF6. 11 The reasons for the high differences in mRNA abundance between calf 4 and calves 1 and 2 can only be speculated. Maybe the observed magnitude might reflect the variability of the whole syndrome, which includes considerable differences in disease onset and disease severity at different ages. Alternatively, environmental (microbial) factors might contribute to the differences in macrophage activation as discussed below.
Macrophage activation syndrome in humans is characterized by fever and lymphadenopathy, and further consequences of a cytokine storm such as hemorrhages, cytopenia, and hepatosplenomegaly. 6 The phenotype of PLD4−/− mice has been linked to a mild form of human macrophage activation sydrome, 9 which can also be provoked by repeated TLR9 activation in mice on a normal genetic background, without using exogenous antigen. 4 In contrast, the phenotype of PLD4-deficient calves did not include persistent high pyrexia, hemorrhages, platelet abnormalities, or cytopenia. Lymphadenopathy and increased dermal factors associated with macrophage activation were present in calves; however, the lymphadenopathy might also have been caused by the inflammation in the dermis and other organs. We did not measure systemic cytokines, which is a limitation of this study. A fulminant increase of systemic cytokines seems unlikely, however, because calves were not apathetic or constantly pyrexic. Furthermore, serum albumin as negative acute phase protein was not considerably decreased. Thus, it seems as if the syndrome in calves does not result in a generalized macrophage activation. Instead, PLD4-deficient calves display local exuberant macrophage activation without severe systemic involvement, thus clearly distinguishing the calf phenotype from human macrophage activation syndrome and the phenotype of PLD4−/− mice. To our knowledge, a direct connection between PLD4 and human macrophage activation syndrome has not been established so far. A direct comparison between calves and humans does not seem appropriate at the moment. Lesions associated with macrophage activation were not seen in spleen and brain, where PLD4 is expressed in mice, 29 so the reason for the differences between the murine and bovine phenotype remains unclear. However, we know that there are considerable differences between immunological functions in different species. This could provide an explanation for the observed variance, without knowing the specific differences between pathways.
In our preceding study, we hypothesized about PLD4 being involved in keratinocyte differentiation as the molecular basis for the keratinization disorder. 16 However, skin lesions were not reported in PLD4−/− mice or in mice with overstimulated TLR9. 4,9 Furthermore, keratinocyte development is completed prenatally and calves were born without skin lesions, which does not support a congenital keratinization disorder. In dogs, feeding inadequate diets (generic dog food dermatosis) and metabolic disease (superficial necrolytic dermatitis) are associated with comparable lesion quality, severity, and distribution. 10 In particular, superficial epidermal degeneration and necrosis are lesions related to metabolic disease in dogs 10 and are not specified for bovine hereditary zinc deficiency. 19 Therefore, it is tempting to speculate that the keratinization disorder might be caused by an epidermal or dermal malnutrition due to unknown nutritional deficiencies, which may be multiple and overlapping. On the other hand, the chronic dermal inflammation itself might be able to disturb epidermal growth and integrity. Keratinocyte differentiation and proliferation might be negatively influenced by the abundance of dermal inflammatory mediators as it is in human psoriasis. 21
At the beginning of this study, we hypothesized that environmental factors (ie, different husbandry conditions) are a major contributing factor of disease evolution, because disease onset and course differed considerably between affected calves. 16 We aimed to get access to calves early in the disease; however, we were able to observe disease evolution in only 3 calves. This limits the testing of this hypothesis, because animal numbers were too low and calves were not bought at the very same age. However, data from the mouse model 9 and the exuberant macrophage activation in calves can support this hypothesis as environmental pathogens result in different but constantly increasing amounts of stimulatory endosomal bacterial and viral DNA. Calves with slower disease course might have had more favorable environmental microbial conditions than the other ones, maybe leading to the differences in disease severity and onset as also observed in calf 2 in the present study.
In this light, the term zinc deficiency–like syndrome is only appropriate to the clinical phenotype. However, it is a misnomer with regard to molecular basis of the disease, because there is no connection between the PLD4 exonuclease and the zinc pathway. Mutations in zinc-pathway-associated genes were not found in calves examined before. 12 Also, this explains why zinc supplementation had only palliative and no ameliorating effects in 9 calves. 16 Nevertheless, zinc supplementation might be still beneficial because zinc not only has a positive effect on immune system and leukocytes, 7,25 but also on cellular barrier functions, 3 in this way it might decrease the amount of foreign DNA which enters beyond physiological epithelial barriers.
The laboratory finding of mild anemia, which was also present in previous cases, 16 is probably the result of a chronic proinflammatory state and can therefore be interpreted as anemia of chronic disease. The low serum selenium concentrations are not necessarily related to this syndrome as Bavaria is a selenium deficient region. 20
This study has several limitations. The prospective study design did not allow collection of a large number of cases due to the low frequency of the gene defect in the population. Thus, the interpretations of this study are limited by systemic bias due to the small sample size. Although we tried to recruit calves as fast as possible, the age at study entry still varied leading to only 3/6 calves being monitored over an extended period of time.
In conclusion, this “genodermatosis” highlights the importance of interdisciplinary cooperation which led to the identification of individuals with similar disease and detection of an underlying genetic cause. Although the phenotype of affected calves showed similarity to bovine hereditary zinc deficiency, the underlying molecular mechanism is completely different. This knowledge helps in clinical decision making to prevent futile treatment plans, which are unable to ameliorate the suffering of affected calves.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211062629 - Macrophages in dermal disease progression of phospholipase D4–deficient Fleckvieh calves
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211062629 for Macrophages in dermal disease progression of phospholipase D4–deficient Fleckvieh calves by Martin C. Langenmayer, Simone Jung, Robert Fux, Christina Wittlinger, Theresa Tschoner, Monir Majzoub-Altweck, Gabriela Knubben-Schweizer, Ruedi Fries, Walter Hermanns and Florian M. Trefz in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-xlsx-1-vet-10.1177_03009858211062629 - Macrophages in dermal disease progression of phospholipase D4–deficient Fleckvieh calves
Supplemental Material, sj-xlsx-1-vet-10.1177_03009858211062629 for Macrophages in dermal disease progression of phospholipase D4–deficient Fleckvieh calves by Martin C. Langenmayer, Simone Jung, Robert Fux, Christina Wittlinger, Theresa Tschoner, Monir Majzoub-Altweck, Gabriela Knubben-Schweizer, Ruedi Fries, Walter Hermanns and Florian M. Trefz in Veterinary Pathology
Footnotes
Acknowledgements
We are indebted to Dr N. S. Gollnick for project conceptualization and critical proofreading of the manuscript. We acknowledge the kind support provided by Dr A. Müller (Vet med Labor GmbH/IDEXX Laboratories) with the analyses of trace and bulk elements from plasma and serum. We acknowledge the assistance of B. Luntz in identification of risk matings. We thank the breed wardens for the nasal swab sampling and the animal caretakers and the veterinary and laboratory personnel at the Clinic for Ruminants and the Institute of Veterinary Pathology for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Münchener Universitätsgesellschaft (31592, MUG).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
