Abstract
A case of epidermolysis bullosa in a calf descendent from a Gir bull and a Gir crossbreed cow is reported. The calf presented with exungulation of all hooves, widespread erosions and crusts on the skin, and ulcers in the oral cavity. Histologically, the skin showed subepidermal separation with clefts occasionally filled with eosinophilic clear fluid, cellular debris, or neutrophils. Ultrastructurally, there was epidermal–dermal separation at the level of the lamina lucida, with the lamina densa attached to the papillary dermis. The hemidesmosomes were poorly defined and small. The clinical, histological, and ultrastructural findings are characteristic of junctional epidermolysis bullosa.
Epidermolysis bullosa (EB) is a hereditary mechanobullous skin disease of animals and human beings, characterized by marked skin and mucous membrane fragility, which triggers the formation of blisters and ulcers in response to minor trauma. The lesions occur due to insufficient connections at the dermis–epidermis junction, due to defects at the basement membrane zone.4,6 In addition to the formation of blisters, there is a loss of nails, claws, or hooves, and detachment of the epithelium of the oral mucosa.7,8 The different EB types in domestic animals follow human classification, based on the ultrastructural lesion at the skin basement membrane zone. There are 3 types of EB: epidermolysis bullosa simplex (EBS), characterized by cytolysis of basal keratinocytes producing intraepidermal clefts; junctional epidermolysis bullosa (JEB), in which the separation occurs in the lamina lucida; and dystrophic epidermolysis bullosa (DEB) with cleavage within or below the lamina densa. 3 Most types of EB are caused by autosomal recessive genes and appear with low frequency. It is estimated that, in human beings, the disease affects 1 in 17,000 live births in the entire world population. 8 In animals, the frequency of EB is not estimated, but there are few reports of the disease. The aim of the current study was to report the clinical, histological, and ultrastructural aspects of a case of EB in a 2-month-old calf, examined in April 2010, at the Veterinary Hospital of the Federal University of Campina Grande (HV/UFCG), Paraíba, Brazil. The calf belonged to a herd of 18 animals. It was descendent from a Gir bull and a Gir crossbreed cow. According to the farmer, a male calf (sired by the same bull) with similar lesions died previously on the farm.
The calf had exungulation of all hooves, widespread erosions, and crusts on the skin (Fig. 1A, 1B) and ulcers in the oral cavity (Fig. 1C). The epidermis loosened easily when a little pressure was exerted on the intact skin (positive Nikolsky sign 8 ). Due to the severity of clinical signs and unfavorable prognosis, the animal was euthanized and necropsied. Varying degrees of alopecia, erosions, and crusts were observed on the skin of the metacarpal and metatarsal regions, elbows, knees, ventral thorax, and abdomen and gluteal region. There were also multifocal ulcers on the gums, hard and soft palates, mucosa of the lips, cheek mucosa, and dorsum of the tongue. The other organs showed no significant lesions.
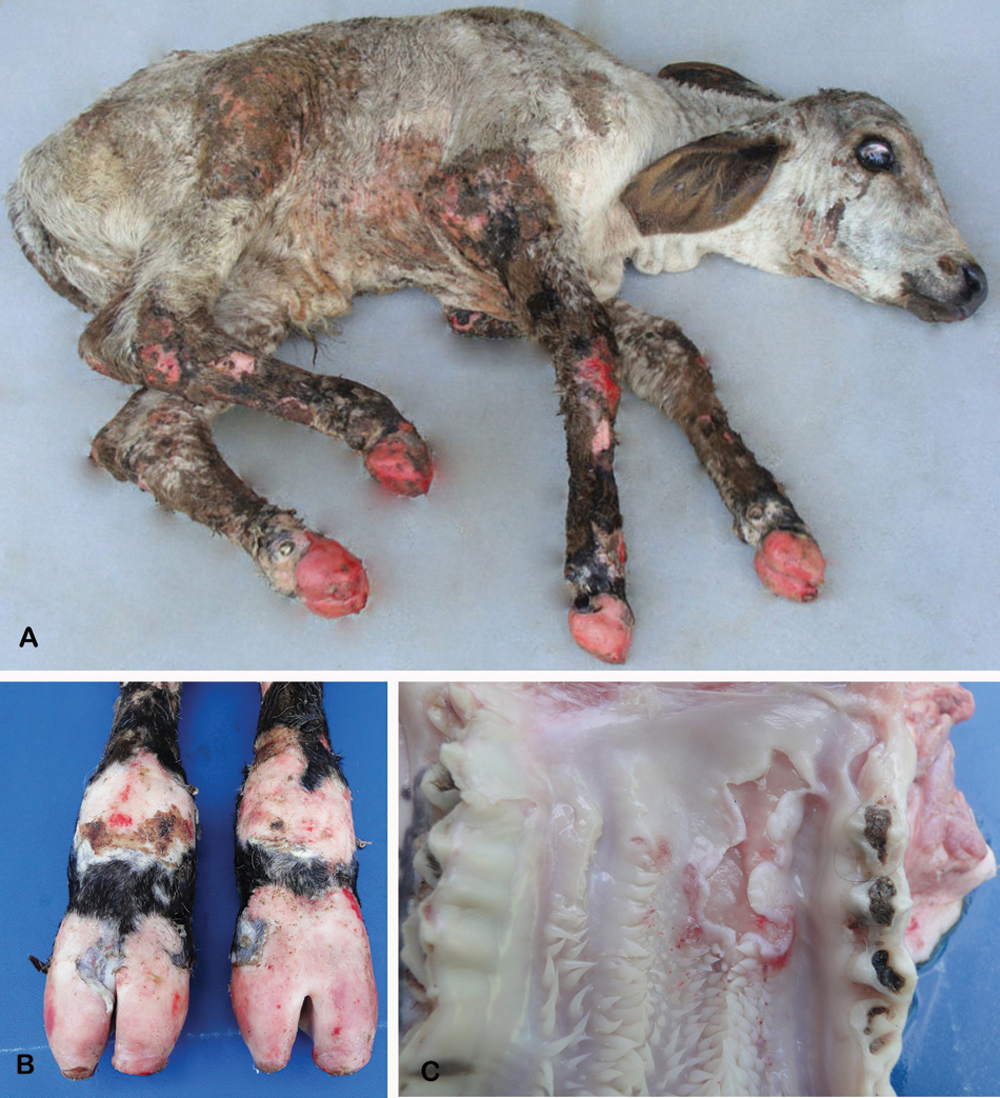
Calf affected with junctional epidermolysis bullosa.
Tissue samples from liver, spleen, kidney, adrenal, lymph nodes, thymus, lung, heart, skeletal muscles, thyroid, central nervous system, esophagus, fore stomachs, abomasum, large and small intestine, oral mucosa, tongue, and skin were fixed in 10% buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin, periodic acid–Schiff, and Gomori trichome. Histologically, there was subepidermal separation (Fig. 2), forming blisters filled occasionally with clear eosinophilic fluid, cellular debris, or neutrophils. Epidermal cells showed degeneration evolving to necrosis. The dermal papillae were edematous with marked degeneration. In more advanced lesions, there were erosions covered by crusts, with perivascular edema and infiltration by neutrophils and mononuclear cells in the underlying dermis. Similar clefts, occasionally filled with eosinophilic fluid lesions, were observed in the oral mucosa and tongue. Areas of total loss of the oral and lingual epithelium were also observed, leading to necrosis, extensive ulceration, and infiltration by neutrophils and hemorrhages.
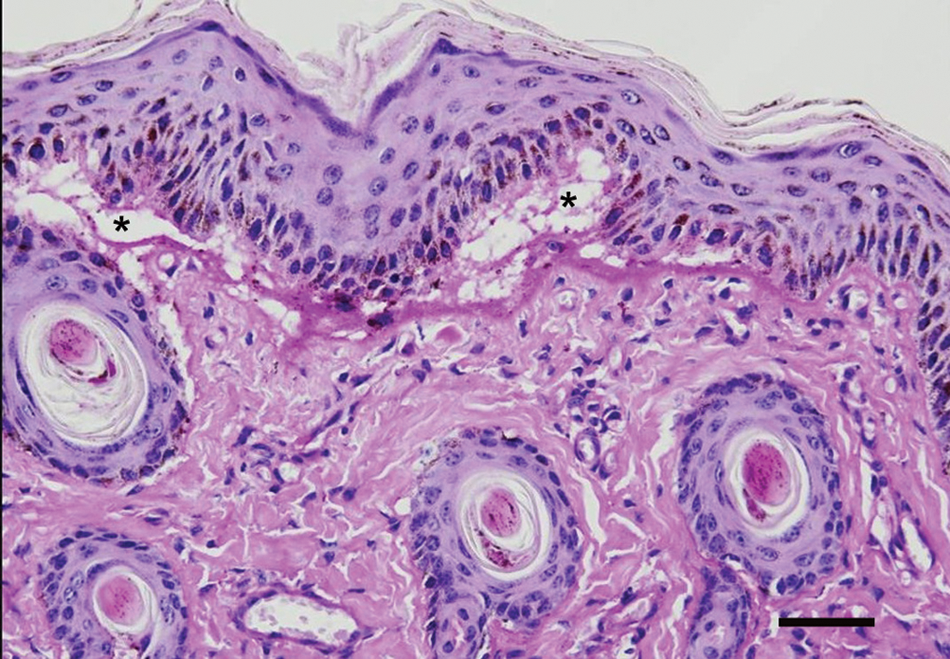
Skin; calf. The epidermis is detached from the dermis, forming a subepidermal cleft (*). Hematoxylin and eosin. Bar = 50 µm.
For ultrastructural examination, small fragments of skin were fixed in 2% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4) Blocks were postfixed in 1% osmium tetroxide buffered in 0.166 M sodium cacodylate (pH 7.4), and embedded in resin. a One-micron, methylene blue–stained sections were used to evaluate histological changes and to select the areas to be examined ultrastructurally (Fig. 3, inset). Then, selected sections were cut at 70–80 nm, stained with uranyl acetate and lead citrate solutions, and examined with an electron microscope. b Samples of the skin of a normal control calf of similar age were collected and processed in the same way. Ultrastructurally, at the epidermal–dermal interface, the basal cell layer was detached from the dermis at the level of the lamina lucida (Fig. 3). Further, the lamina densa was attached to the dermal papillae (Fig. 4). The hemidesmosomes were small and without clear demarcation when compared with the control calf.
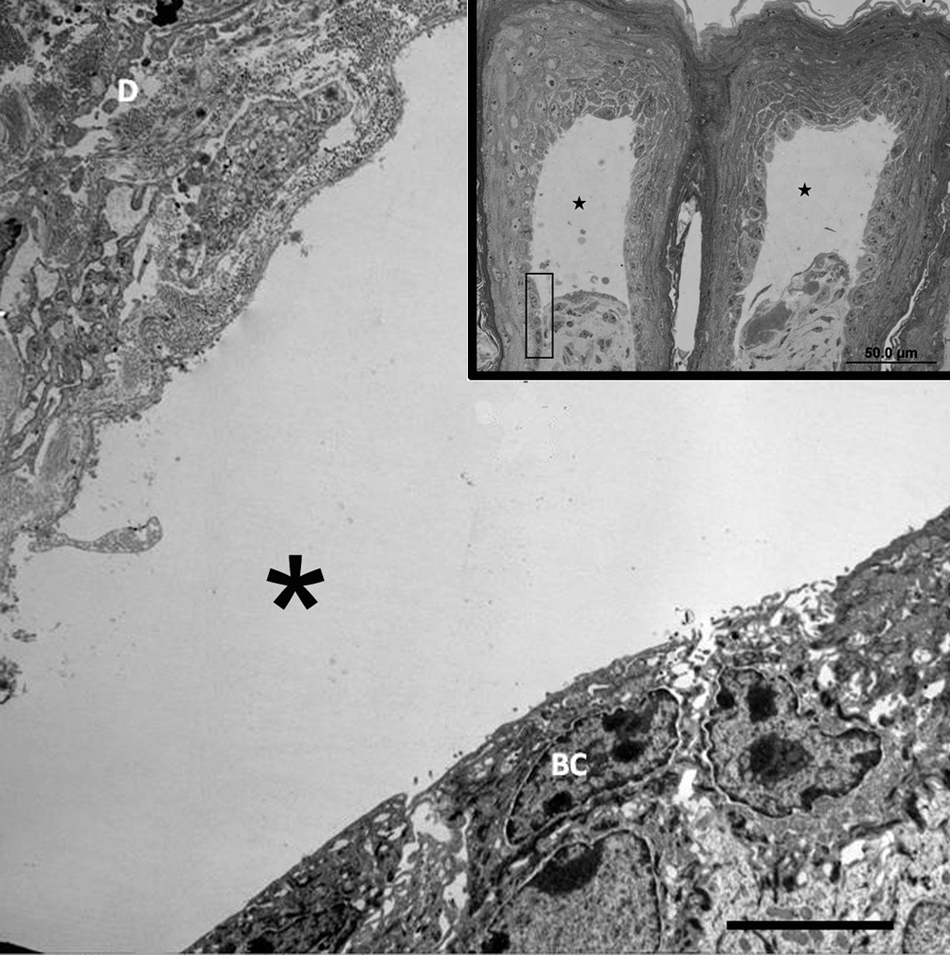
Transmission electron micrograph; skin; calf. Epidermal–dermal junction. Separation of the basal cell layer (BC) from the dermis (D) is observed at the lamina lucida with vesicle formation (*). Bar = 5 µm. Inset: Semi-thin section showing the local of separation observed on electron microscopy (rectangle). Note vesicle formation (black stars). Bar = 50 µm.
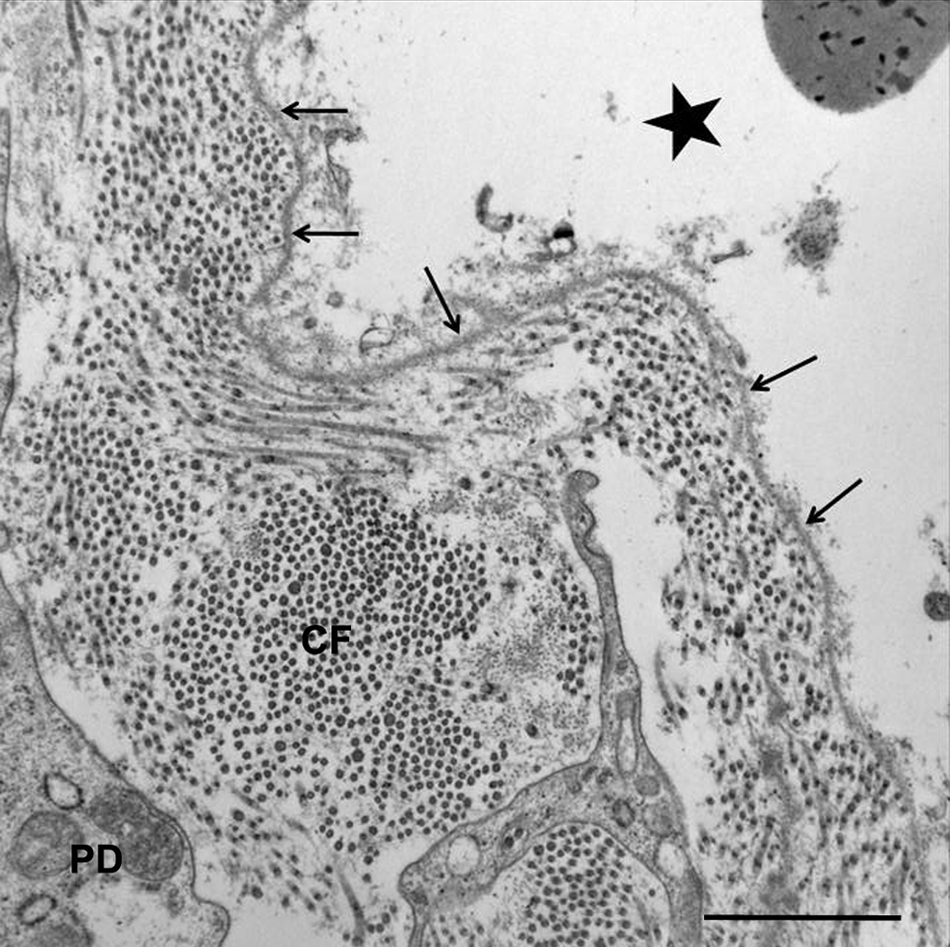
Transmission electron micrograph; skin; calf. Epidermal–dermal junction, dermis at separation from the basal cells: lamina densa (arrows) stays attached to the papillary dermis (PD). Note 20-nm collagen fibers (CF) within an edematous dermis. The vesicle is also observed (black star). Bar = 1 µm.
Clinical signs and pathology observed in the present case are characteristic of EB in animals and human beings.3,4,8 The ultrastructural findings showed that the separation occurred at the lamina lucida in the epidermal basement membrane zone, consistent with the diagnosis of JEB. Ultrastructurally, the epidermal basement membrane zone is divided into 3 areas: hemidesmosomes, lamina lucida, and lamina densa (Fig. 5). The hemidesmosomes are composed of an inner and an outer plaque. The inner plaque is composed of cytoplasmic hemidesmosomal proteins HD1/plectin and BP230. The outer plaque contains the proteins α6β4 integrin and BP180. The lamina lucida is formed by the plasma membrane, the subdesmosomal dense plate, and the anchoring filaments. The lamina densa is composed by type IV collagen and anchoring fibrils (type VII collagen). Anchoring filaments are composed of BP180 and laminin 5, which connect hemidesmosomes to type VII collagen of anchoring fibrils (Fig. 5).3,8 In the current case, electron microscopic data indicated abnormalities in the hemidesmosome-anchoring filaments complex, leading to separation at the lamina lucida. In human beings, JEB is the most genetically heterogeneous form of EB, caused by 6 different recessive genes that encode the proteins laminin 5 (α3, β3, and γ2 subunits) and BP180. The lack of expression of these genes leads to 6 subtypes of JEB, but the most lethal and most common variant is the Herlitz JEB (HJEB). 8
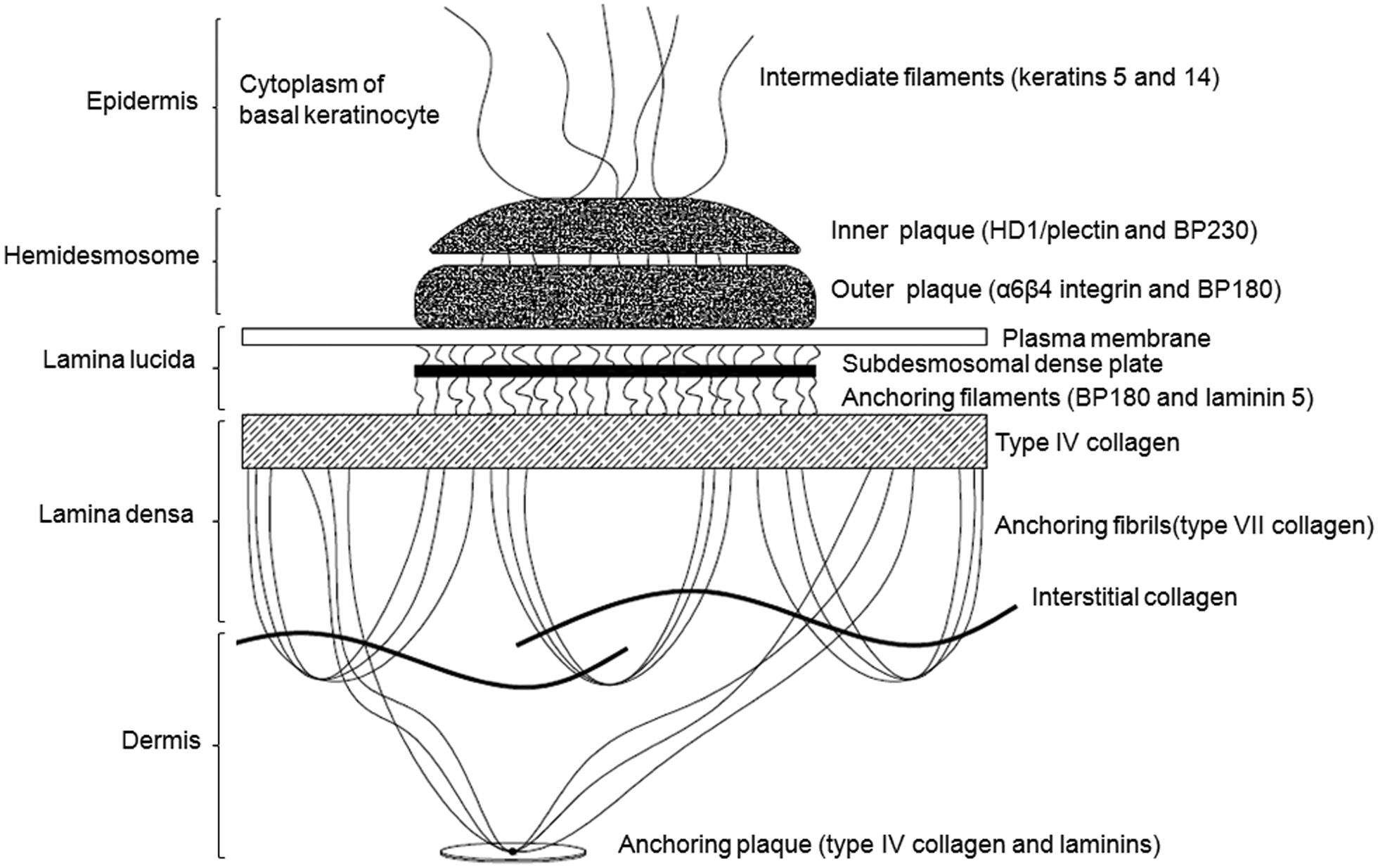
Schematic representation of the ultrastructure of the epidermal basement membrane zone.
Junctional epidermolysis bullosa has been diagnosed in horses of various breeds.5,6,9 In 2 studies in foals, the diagnosis of HJEB was based on clinical, histological, ultrastructural, and molecular examinations. A mutation in the LAMC2 gene causing the failure in the expression of laminin 5 (γ2 subunit) was identified.6,9 The distribution of lesions in human neonates and foals with HJEB, as well as ultrastructural changes, had the same pattern observed in the current study, suggesting that in this calf, the epidermis–dermis separation occurred due to failure in the expression of laminin 5.
Previous reports of cattle with EB were described based only on clinical and histopathological findings.1,2,10 In a 2009 report, 8 cases of EBS in calves were diagnosed by ultrastructural examination in the United Kingdom. 4 A case of JEB in a Charolais calf was registered in France, the diagnosis was supported by histopathological examination, and antigen immunomapping of frozen skin sections using antibodies to the major components of the human cutaneous basement membrane revealed an intralamina lucida cleavage; ultrastructural examination was not performed (Guaguere E, Berg K, Degorce-Rubiales, et al.: 2004, Junctional epidermolysis bullosa in a Charolais calf with deficient expression of integrin α6β4. Vet Dermatol 15 (Suppl. 1):28. Abstract). Further studies are needed to investigate possible modes of inheritance and the molecular characterization of JEB.
Footnotes
a.
EMBED 812, Electron Microscopy Sciences, Hatfield, PA.
b.
JEOL 1200EX II, JEOL Ltd., Tokyo, Japan.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
