Abstract
In a retrospective study of a western pygmy marmoset (Cebuella pygmaea) colony, postmortem examination of 1/8 juvenile and 29/47 adult animals identified vascular, cardiac, and renal lesions consistent with systemic hypertension. This included frequent renal arteriolar hypertrophy, hyaline and proliferative arteriolosclerosis, fibrinoid necrosis of arterioles, glomerulosclerosis, and nephrosclerosis. Affected animals ranged from 0.6 to 12 years of age (mean 6 years) and had an observed male predominance. Genealogical relatedness was evident in several breeding pairs and spanned multiple generations. Concurrent cardiac and renal disease was commonly identified, although frequently subclinical, and both were important causes of morbidity and mortality in affected animals. Cardiomegaly and hypertrophy were typical features and were accompanied by left atrial thrombosis in 10 animals. Signs of heart failure included chronic pulmonary edema in 20 cases and body cavity effusions in 17. In the kidneys, 19 cases had glomerular disease and hypertensive vasculopathy, and 26 cases had nephrosclerosis or glomerulosclerosis. Common extrarenal secondary causes of hypertension were excluded by necropsy examination. The pathogenesis is suggested to involve primary hypertension leading to renal and cardiac disease. Elevated sympathetic activity might be an underlying factor in the frequent development of primary systemic hypertension in the pygmy marmoset, as for the owl monkey.
Keywords
Cardiovascular disease has long been recognized in managed populations of primates in zoological or biomedical research facilities as an important and persistent cause of noninfectious morbidity as well as mortality. 18,23,26,29,40,43 The latter often occurs unexpectedly without clinical signs, or clinical signs were not manifested until disease was advanced. 23,43 Relatively few studies on spontaneous systemic hypertension as the cause of cardiovascular disease in primates in managed care have been conducted. The most notable previous studies in primates include those of spontaneous hypertension in the African green monkey (Chlorocebus aethiops sabaeus), 36 owl monkey (Aotus sp.), 40 and woolly monkey (Lagothrix lagotricha). 6,14 All of these species experience mortality associated with lesions suggestive of systemic hypertension. In all 3 of these species, direct blood pressure measurements and clinical and pathologic research have established evidence of systemic hypertension. The African green monkey and owl monkey have been used as models of systemic hypertension.
Of current interest, however, is the pygmy marmoset (Cebuella pygmaea), 3 a small New World primate with an average weight of 97.1 ± 22.2 g 15 native to the Western Amazon in Ecuador, Colombia, Bolivia, Brazil, and Peru. 16 A previous comprehensive review of infectious and noninfectious diseases of the Callitrichidae that included pygmy marmosets did not include any reference of cardiovascular disease or hypertension, 35 but a colony of pygmy marmosets at the University of Wisconsin was identified with evidence of spontaneous systemic hypertension and cardiovascular disease based on gross necropsy, and histopathologic and electron microscopic findings. The specific aims of this study were to describe the gross and microscopic findings of presumptive primary systemic hypertension in pygmy marmosets. In considering the pathogenesis, we discuss classical and evolving determinants of hypertension, particularly primary hypertension, and controllers of those main determinants including the proposed centrality of sympathetic overactivity and the potential relevance to these pygmy marmosets.
Materials and Methods
The University of Wisconsin Department of Psychology western pygmy marmoset colony was established in 1973 and directed by Professor Charles T. Snowdon from wild-caught monkeys imported from Peru by the National Institutes of Health. Additional animals were obtained by breeding within the colony and by breeding loan or trade from facilities accredited by the Association of Zoos and Aquariums as well as research facilities. The colony disbanded in the early 1990s. All animals were housed as pairs or in family groups in enclosures within one large room. The 6 cages were large with floor space ranging from 3.9 to 12.1 m2, extended 3 m high and were constructed from wood and wire mesh. Later cages were constructed from anodized aluminum and mesh. Extensive environmental enrichment was provided and included an intricate trail system of branches and ropes with multiple feeding platforms and nest boxes to mimic the arboreal habitat of these animals. Since pygmy marmosets gouge trees and extract sap as part of their natural foraging behavior, wood structures and extensive branches allowed animals to mimic this behavior. The colony was maintained on an artificial 12-hour day-night cycle using full-spectrum (including ultraviolet B) lights. All animals in the large colony room had visual, olfactory, and auditory contact with each other. Animals were fed a varied diet 3 times a day consisting of marmoset diet (ZuPreem Marmoset Diet) supplemented with vitamin C, high-protein cereal (Gerber), live culture yogurt, drop vitamins (EVSCO Hi-Vite), taurine, fresh fruit and vegetables, cottage cheese, eggs, chicken, nuts, and mealworms. The colony was created for noninvasive studies of pygmy marmosets and was designed to provide the animals with an enriched physical and social environment to facilitate studies in vocal communication and reproductive biology. 41,46 All research reported in this article was retrospective and adhered to the American Society of Primatologists Principles for Ethical Treatment of Non-Human Primates, as well as all local, state, and national regulations. Research protocols were evaluated and approved by the University of Wisconsin, College of Letters and Science Animal Care and Use Committee, and followed the USPHS and ILAR guidelines for research with nonhuman primates applicable during the duration of the colony.
Animals were examined from 1985 to 1999 and included wild-caught, F1, F2, and F3 generations. Complete necropsy examinations including gross and histologic examination of all visceral organs, brain, eyes, bone marrow, skeletal muscle, and skin were performed on 68 animals and consisted of the following: 13 infants (defined as less than 84 days old, based on weaning between 10 and 12 weeks), range 1 to 70 days old; 8 juveniles (defined as <1 year old, based on puberty at 15–17 months), range 89 to 217 days old; and 47 adults, range 1.2 to 14.5 years old. Veterinary care was provided by veterinarians from the Wisconsin National Primate Research Center or by the University of Wisconsin School of Veterinary Medicine veterinary specialists. Physical examinations were completed including attempts to obtain accurate and repeated indirect blood pressure measurements. However, due to the small size of the subjects, the smallest available commercial neonatal occlusive cuff was too large for limb or tail measurements. In addition, these animals were not easily habituated to handling by humans resulting in labile measurements, similar to what has been observed in other animal hypertension studies. 4
Necropsy examinations were performed by University of Wisconsin School of Veterinary Medicine pathologists; 57 of the 68 necropsies were performed by one pathologist (AJC). During necropsy examination, particular attention was given to the heart, vasculature, and kidneys. To facilitate gross morphologic comparisons among individual animals, the majority of hearts were bisected in a so-called echo-plane section or 4-chamber view method to allow visualization of both left and right atria and ventricles. The hearts were fixed prior to being halved. Samples of numerous tissues were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned at 3 to 5 µm, and stained with hematoxylin and eosin (HE). Selected sections were stained with periodic acid–Schiff (PAS) and Gomori trichrome aldehyde-fuchsin (GTAF; elastic fibers—dark purple; sulfated mucins—purple; collagen—green). Particular attention in histopathologic examination was given to kidneys, adrenal glands, thyroid glands, brain, eyes, and heart. The hearts of 6 animals (cases 61, 66, 67, 68, 69, 72) were carefully removed, flushed with water, and heart weight to body weight ratios were determined. These were compared to the allometric predictive heart weights concerning expectations of organ weight to body weight. 42
Tissues for electron microscopy were fixed in cold modified Karnovsky’s fixative, washed overnight in 0.1 M phosphate buffer (pH = 7), and postfixed in 1% osmium tetroxide for 1.5 hours, followed by dehydration in increasing concentration of ethanol and propylene oxide. Subsequently, the tissues were embedded in Epon-Araldite epoxy resin. Thick sections were cut at 1.5 µm, stained with methylene blue, and examined for selection of ultrathin sections (30–70 nm thick) for electron microscopy. Ultrathin sections were mounted on copper grids and stained with 2% uranyl acetate and 0.4% lead citrate and examined with a Philips transmission electron microscope.
For reporting of the morphologic necropsy findings, lesions are presented first with respect to expected correlations and consistent sequelae of hypertension. Therefore, an emphasis is placed on vascular, cardiac, and renal lesions. Animals often had coexisting sporadic findings. Some variance in nomenclature occurred due to the preference of different pathologists such as inclusion or absence of cardiomegaly as a morphologic diagnosis, or conflation of cardiac hypertrophy and hypertrophic cardiomyopathy. Some of these minor variances have been reconciled by review of case descriptions and archived materials. The terms benign and malignant for subcategories of hypertension were not applied in this study. This concept has been previously applied in humans to subdivide hypertension of longer duration that is largely asymptomatic (benign hypertension) from hypertension that is clinically severe and typically develops rapidly (malignant hypertension). Since cardiovascular disease was not attributed to hypertension by blood pressure measurements antemortem and some animals presented in extremis, progression from milder forms of hypertension to more severe hypertension was not studied, and the spontaneous deaths reported in affected animals imply a severe form of the disease. This was often exemplified by multi-organ damage, particularly involving kidney and heart. In some instances, brain, eyes, and other visceral organs were documented as affected. Particularly, presumed hypertension cases exemplified by chronic renal failure with glomerulosclerosis, interstitial fibrosis, and tubular atrophy are believed to be a consequence of the renal arteriolar changes subsequent to hypertension, so-called hypertensive renovascular disease, that have been noted to be a significant cause of end-stage renal failure in humans 10,17,27,31,39 rather than a sequela of chronic renal disease. With respect to nomenclature, nephrosclerosis as a general morphologic diagnosis may include hyaline arteriolosclerosis (particularly affecting glomerular afferent arterioles), glomerular obsolescence (interpreted as a consequence of ischemia), and subsequent tubulointerstitial fibrosis. Proliferative or hypertrophic arteriolosclerosis may result in segmental or global glomerulosclerosis that may include an increase of mesangial matrix with minor increased mesangial cellularity and capillary basement membrane thickening, speculated as a consequence of increased pressure at the afferent arteriolar level. These changes may progress until the entire glomerulus is sclerotic and subsequent ischemia and hypoxia may further account for tubular atrophy and interstitial fibrosis. 17,31
Results
This retrospective anatomic pathological study spanned 15 years and included 68 animals. Overall, 1 juvenile and 29 adult pygmy marmosets had vascular, cardiac, and renal lesions consistent with hypertension, and the pathologic findings are summarized in Table 1 and detailed in Supplemental Table S1. The 21 neonates/juveniles in the study did not have any clinical signs or lesions of cardiovascular or renal disease, with the exception of one 7-month-old animal with cardiac and renal lesions consistent with hypertension and clinical evidence of left heart failure. Of the 47 adults in the study, 29 had gross and histologic cardiovascular and renal lesions suggestive of hypertension. The 30 animals with lesions suggestive of hypertension ranged from 7 months to 12 years of age (mean 5.8 years, median 6.4 years, SD 2.4 years). The analysis included 3 wild-caught animals (cases 5, 14, 15) introduced into the colony as young adults that had been in managed care for 7 to 10 years. Inclusion in calculations was justified based on longevity in the colony. Three animals were excluded from the analysis of ages. Case 48, 1.3 years old, had significant cardiomegaly and left atrial thrombosis, but also necrotizing myocarditis. However, in this case the inflammatory disease of the heart may have been preceded by hypertension. An accurate date of birth was not available for 2 animals (cases 20, 10); the latter was wild-caught and had been in the colony only a short time.
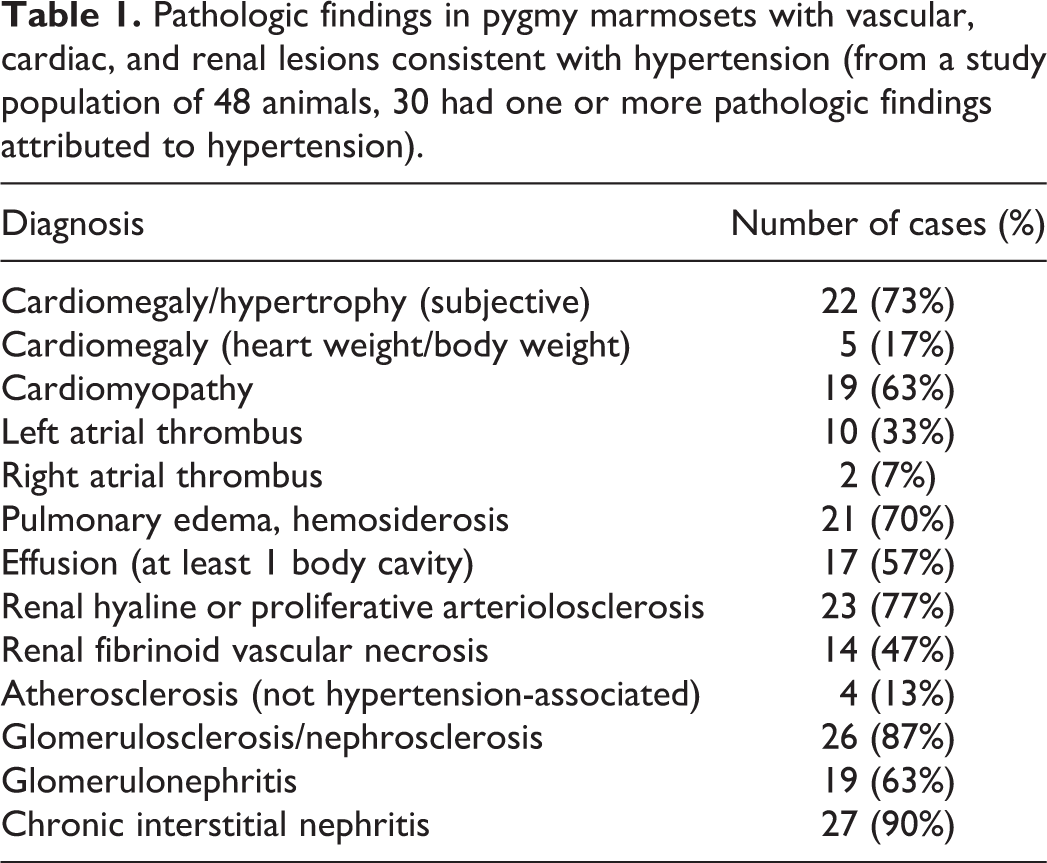
Pathologic findings in pygmy marmosets with vascular, cardiac, and renal lesions consistent with hypertension (from a study population of 48 animals, 30 had one or more pathologic findings attributed to hypertension).
The sex distribution of pygmy marmosets with vascular, cardiac, and renal lesions suggestive of systemic hypertension had an observed male prevalence. The 30 affected animals included 21 males (of 42 males necropsied) and 9 females (of 23 females necropsied). All 4 wild-caught males had lesions suggestive of hypertension.
A high prevalence was identified in the progeny of several breeding pairs, often spanning multiple generations (Figs. 1, 2). The genealogy identified that 7 affected siblings (6 sons, 1 daughter) occurred in one breeding pair, AN (F) × 73 (M); and 10 affected descendants occurred in another breeding pair, AL (F) × FE (M), including 1 son and 1 daughter, 3 grandsons and 2 granddaughters, and 2 great grandsons and 1 great granddaughter. Among all affected animals, 4 twin pairs with both animals affected were identified. Three breeding pairs with affected descendants included one wild-caught male. One of these affected wild-caught males (case 10) had one affected descendent, a son. A second affected wild-caught male (case 15) had 6 affected descendants including 1 son and 1 daughter, and 3 grandsons and 1 granddaughter. A third affected wild-caught male (case 14) had 8 affected descendants including 3 sons and 2 daughters, and 2 grandsons and 1 granddaughter. All 3 of these wild-caught males were subsequently necropsied and verified as affected. One additional affected wild-caught male had no documented progeny.
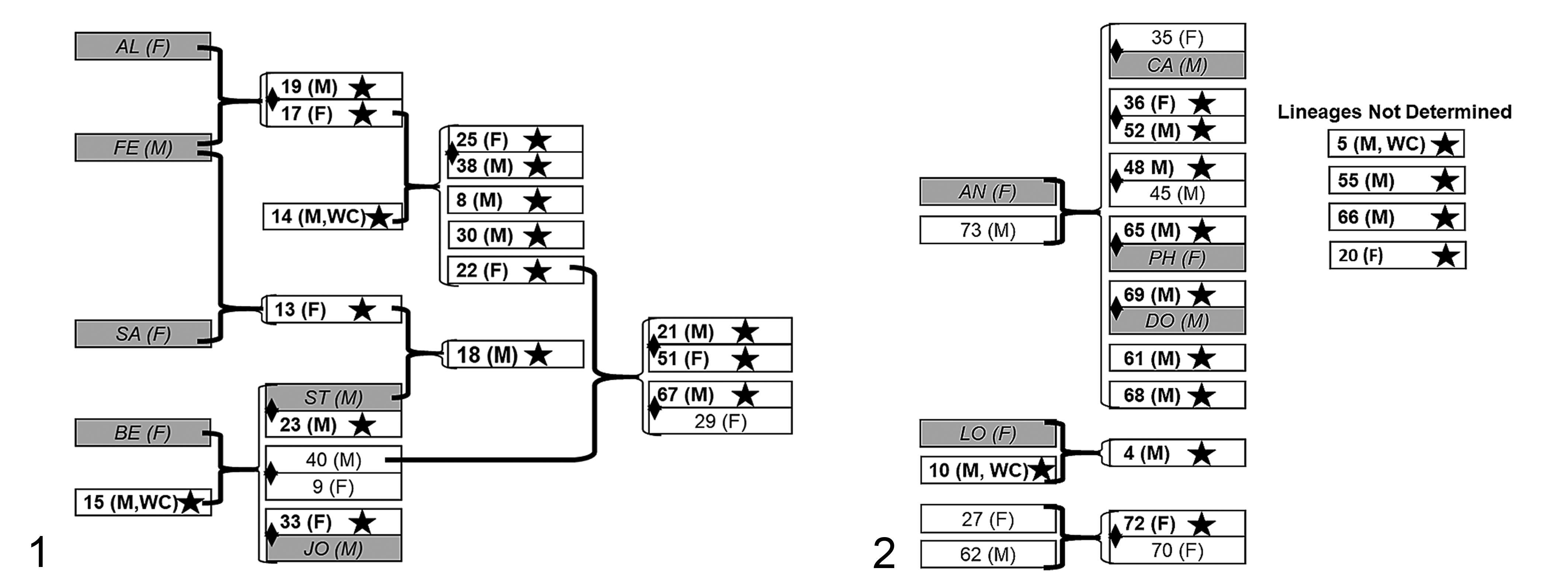
Genealogy of animals with lesions attributed to systemic hypertension (bold text, star and white blocks), pygmy marmosets. White blocks and number indicate animals with postmortem data. Shaded boxes and italicized letters are animals with no postmortem examination. Breeding pairs (heavy brackets) and the subsequent progeny (thinner brackets) including some multiple generations are indicated; respective sexes are in parentheses (M, male; F, female). The diamond spanning 2 blocks represents a twin pair. WC, wild-caught.
Histopathologically, the kidneys in affected pygmy marmosets had the clearest vascular lesions of hypertension. Renal vascular lesions that could be attributed to hypertension occurred in 23 animals. These lesions usually affected interlobular (cortical radial) arteries, frequently merging with afferent arterioles; however, changes could also be identified, rarely, in interlobar and arcuate arteries. Some medium arteries and, more commonly, arterioles had prominent thickening of the tunica media attributed to concentric hypertrophy/hyperplasia, often resulting in apparent concentric “onion skin-like” proliferation of cells in the tunica media (Figs. 3, 4). In some instances, a GTAF stain revealed reduplication of elastic fibers in the intima with prominent endothelial cells along the intimal margin, mildly increased cellularity in the thickened intima, and apparent fragmentation of the multilayered elastic lamina (Fig. 3). Obscuration of the expected red staining of the smooth muscle cells of the tunica media in the GTAF-stained histologic section was attributed to sulfated mucins suggestive of mucoid extracellular matrix accumulation. Vacuoles among smooth muscle cells of the lamellated tunica media occurred intermittently (suggestive of medial vacuolar degeneration; Fig. 4). Intimal proliferation resulted in apparent narrowing of the arterial lumen (Fig. 4).
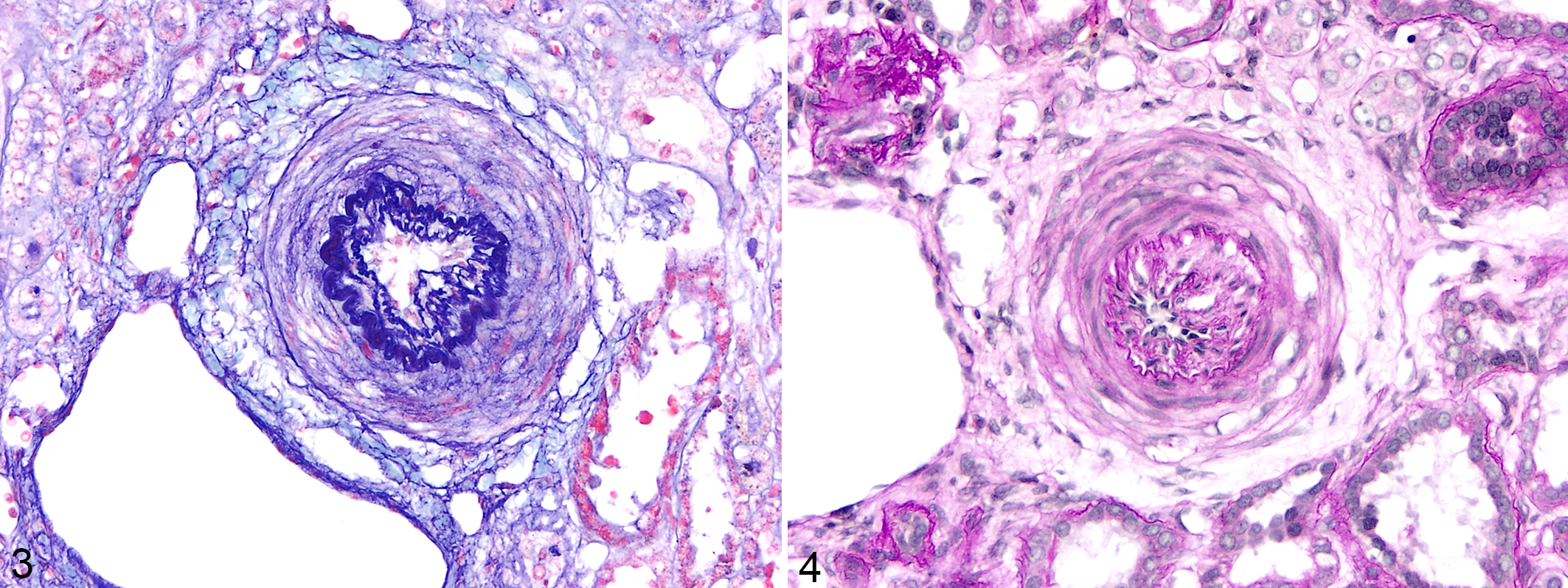
Hypertensive renovascular disease, kidney, pygmy marmoset (case 5). Lamellated renal arteries with “onion skin-like” change of the tunica media and intimal proliferation.
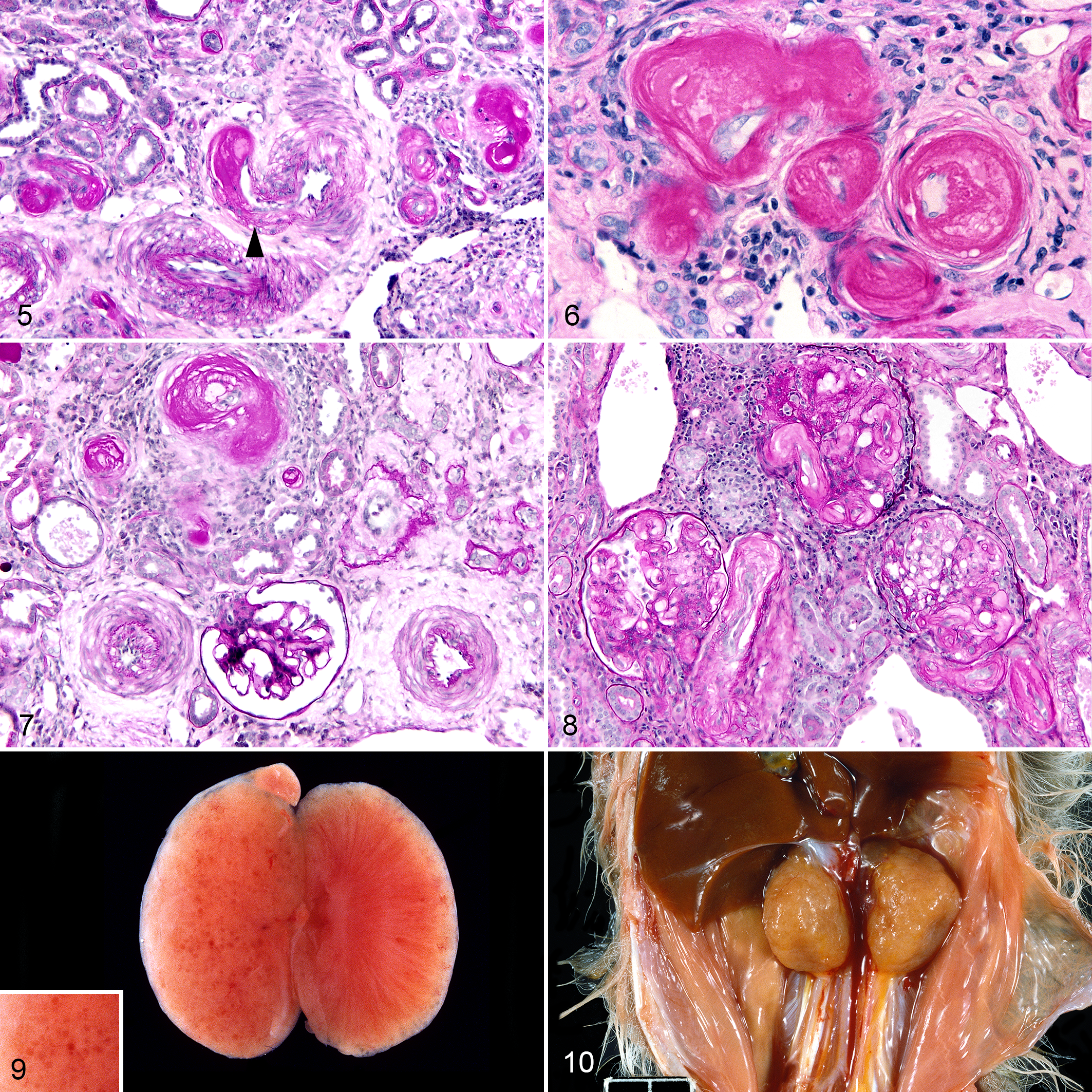
Hyaline arteriolosclerosis accompanied this hypertrophic or proliferative arteriolosclerosis and, additionally, fibrinoid necrosis of arterioles was a frequent finding, prominently evident in at least 14 cases. These lesions were prominently manifested in the kidneys (Figs. 5 –8). The mildest arterial change was hypertrophy of the tunica media and hyalinization, evidenced by deposition of homogenous eosinophilic material in HE-stained slides, which was often intensely PAS-positive (Figs. 5 –8). The homogeneous eosinophilic hyaline material in the tunica media of renal interlobular arteries and afferent arterioles often had variable clefts or vacuoles. Hyalinized arterioles often had a fibrillary quality and these zones occurred concurrently with the smudgy hyaline regions. The most severely affected arterioles stained intensely PAS-positive, which has been interpreted as more consistent with fibrinoid necrosis. 45 Tortuosity of these arterioles was evidenced by the variable profiles of contiguous arteriolar segments with hyaline arteriolosclerosis (Fig. 6). In transverse sections, the endothelium was often retained and concentric adventitial fibrosis was evident (Fig. 6). In the kidney, this hyaline arteriolosclerosis was typically accompanied by some surrounding fibrosis and locally mild to moderate infiltrates of lymphocytes and plasma cells (Fig. 7). Arteriolar hyalinosis often involved the vascular pole of the glomerulus (Fig. 8). Electron microscopic examination of selected renal arterioles revealed amorphous granular deposits interspersed with electron-dense cellular debris that were subendothelial as well as within the tunica media. In severely affected arterioles, the effaced tunica media was replaced by this granular amorphous material with suspended fragments of degenerating and necrotic smooth muscle cells.
Hypertensive renovascular disease, kidney, pygmy marmoset.
Hyalinization and hypertrophy of arterioles and small arteries were identified variably in other tissues including small pulmonary arteries in 5 cases, small intramural coronary arteries of the heart in 5 cases, small visceral arteries of the stomach in 3 cases, splenic pulp arterioles in 2 cases, and a cerebral arteriole in the brain in 1 case. Accompanying fibrinoid necrosis, as previously described in the kidneys, was identified in affected small arteries in the heart and lungs. Vascular necrosis in the thalamus of the brain in one animal with subsequent malacia and hemorrhage may have been primarily a consequence of a thromboembolus. Prior weakening of the arterial wall as a contributing factor could not be excluded. Histologic examination of the eyes in several animals revealed hypertrophy and hyalinization of inner retinal vessels, a feature enhanced with PAS reaction. Incidentally, in 4 animals, atherosclerosis was found, chiefly in the aorta and major branches, although this was not attributed to hypertension. The renal artery was not affected by atherosclerosis in any of these cases.
In addition to frequent and often severe renal vascular lesions, there was often marked involvement of renal parenchyma, particularly glomeruli and tubules. Grossly, kidneys were often pale tan and had a finely granular capsular surface, frequently punctuated by uniform cysts of <1-mm-diameter (Figs. 9, 10). This often correlated with variable cystic dilatation of the glomerular capsule. Other glomeruli had wrinkling of the glomerular basement membrane and apparent reduplication on PAS reaction, and mesangial cell proliferation (Fig. 11). Some glomeruli were completely hyalinized and this was often accompanied by hypertrophy and hyalinization or fibrinoid necrosis of the afferent arteriole (Fig. 12). Mild to moderate periglomerular infiltrates of predominantly lymphocytes and plasma cells encircled sclerotic glomeruli. Intermittent renal parenchymal atrophy correlated with the slightly granular appearance of the capsular surface seen grossly. The region of the juxtaglomerular apparatus at the juncture of the altered afferent arteriole and distal tubule was often enlarged and expanded. With electron microscopy, prominent heavily granulated cells with abundant membrane-bound round to rhomboidal electron dense granules were found, typical of a hyperplastic juxtaglomerular apparatus. Selected glomeruli had irregular thickening and lack of uniformity of the glomerular basement membrane and a band of amorphous granular material in the subendothelial zone. Endothelial processes extended into a region of amorphous granular material and coalescing vacuoles in the basement membrane in 1 case (Fig. 13). No electron-dense deposits suggestive of immune complex deposition were identified. Podocytes were irregular and variably fused (Fig. 13). In condensed and hyalinized sclerotic glomeruli, irregular bands of material of basement membrane origin were interspersed with cellular debris and occasional randomly intermingled viable cells (Fig. 14). Based on longitudinal histologic sections of kidneys, even in severely affected animals fewer than one third of glomeruli were abnormal. Features of nephrosclerosis or glomerulosclerosis were identified in at least 26 animals. Of the 26 animals with glomerulosclerosis or nephrosclerosis, 20 had evidence of hyaline or proliferative arteriolosclerosis or fibrinoid necrosis, and 16 had histologic evidence of glomerulonephritis. Lymphoplasmacytic interstitial inflammation (typically mild) and tubular atrophy were similarly prevalent in the affected zones in at least 27 animals.
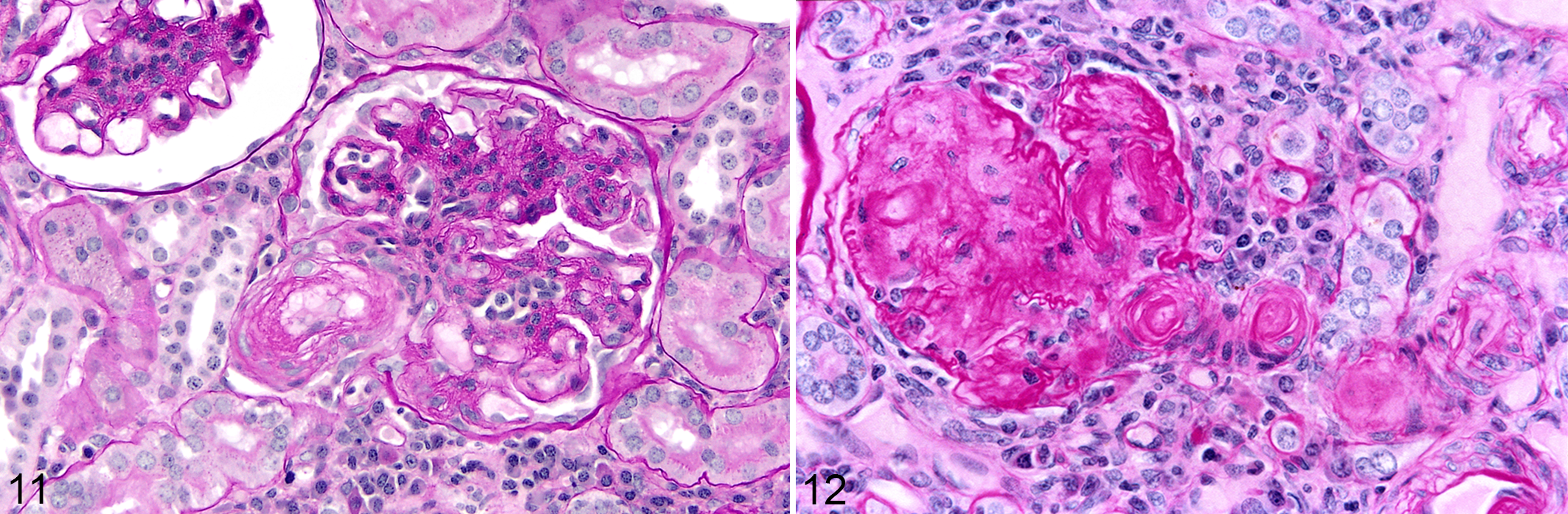
Hypertensive renovascular disease, kidney, pygmy marmoset.
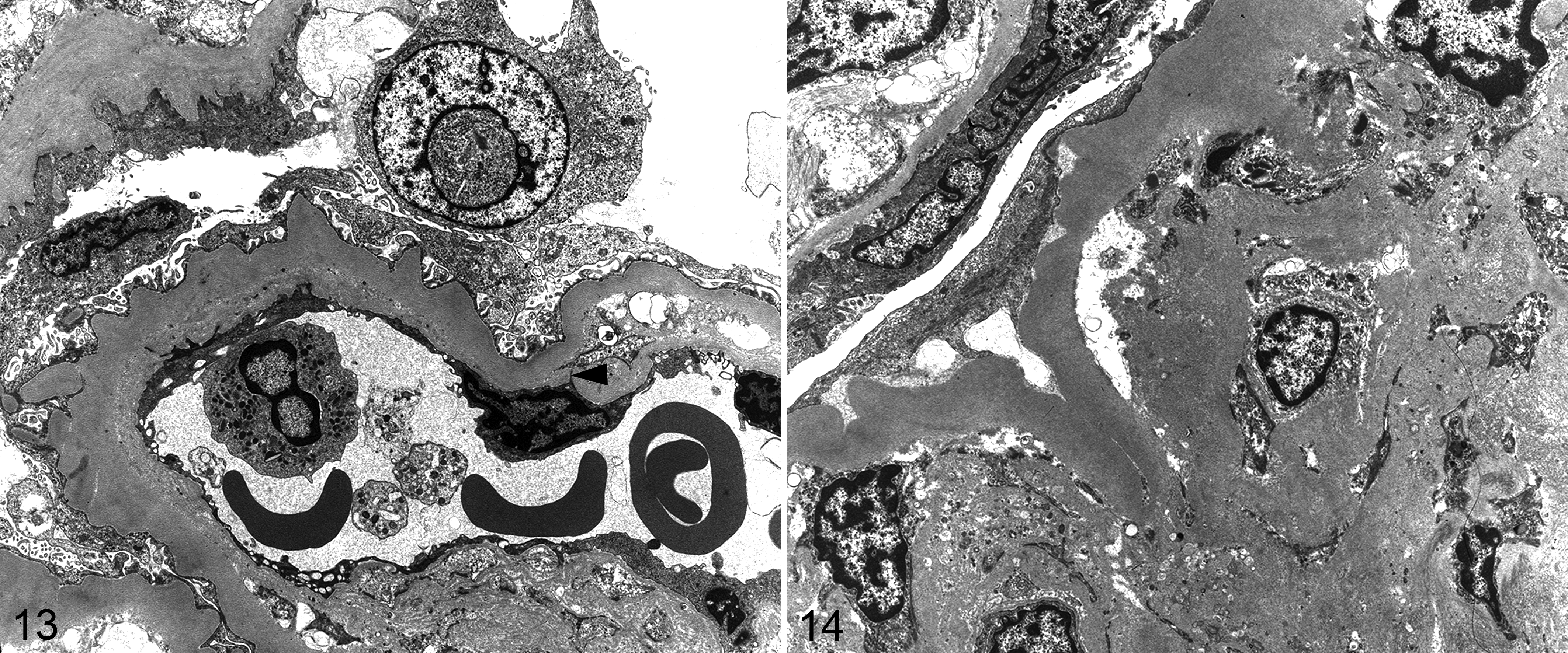
Hypertensive renovascular disease, kidney, pygmy marmoset. Case 15. Transmission electron microscopy.
Fourteen of the total pygmy marmosets necropsied were identified with renal glomerular or interstitial lesions that did not manifest additional cardiac and vascular lesions suggestive of hypertension. The most prevalent diagnosis in these cases was amyloidosis that typically was systemic with concomitant involvement of the kidneys, affecting 6 adult animals. Mild to occasionally moderate interstitial nephritis similar to that coexisting in some of the animals with hyaline or proliferative arteriolosclerosis was found in 4 adult animals. Three adults had glomerulonephritis without other evidence of hypertension. Animals with glomerulonephritis also had multifocal necrotizing vasculitis (1 animal), severe necrotizing pancreatitis associated with Proteus mirabilis bacteremia (1 animal), and pyelonephritis with intralesional bacteria (1 animal). One adult had glomerular thrombosis, hepatitis with microthrombi, enterocolitis, and bacterial endocarditis with intralesional gram-negative bacteria.
The heart in pygmy marmosets with evidence of hypertension had variable degrees of left ventricular hypertrophy (often referred to as concentric hypertrophy) and cardiomegaly. Compared with cases that did not have lesions of hypertension (such as case 60, Fig. 18, a 4-year-old female with generalized multi-organ amyloidosis and chronic pyelonephritis), 10 of the 30 cases with lesions of hypertension also had the most subjectively convincing cardiomegaly and cardiac hypertrophy. All 10 of these had left atrial thrombosis at necropsy (Figs. 15 –17), and these were clearly adherent thrombi with evidence of early organization at the endocardial junction with the thrombus. Two additional animals with lesions of hypertension had evidence of right atrial thrombosis, although one of these was interpreted as of short duration and was not associated with cardiomegaly or cardiac hypertrophy. One additional animal had a left atrial thrombus associated with a small mitral valve thrombus suggestive of endocarditis. The thrombus yielded no growth on bacterial culture. Of the 30 cases with lesions of hypertension, an additional 10 animals had variable subjective cardiomegaly and hypertrophy (Figs. 19, 20). Thus, overall, 22 of the 30 cases with lesions of hypertension were considered to have some degree of cardiomegaly and hypertrophy. Histopathologically, in multiple cases, the cardiac myofibers had variation in myofiber size and nuclear size, and intermittent degeneration, necrosis, and atrophy, and there was interstitial fibrosis. Scattered coronary arteries had hyalinization of the tunica media.
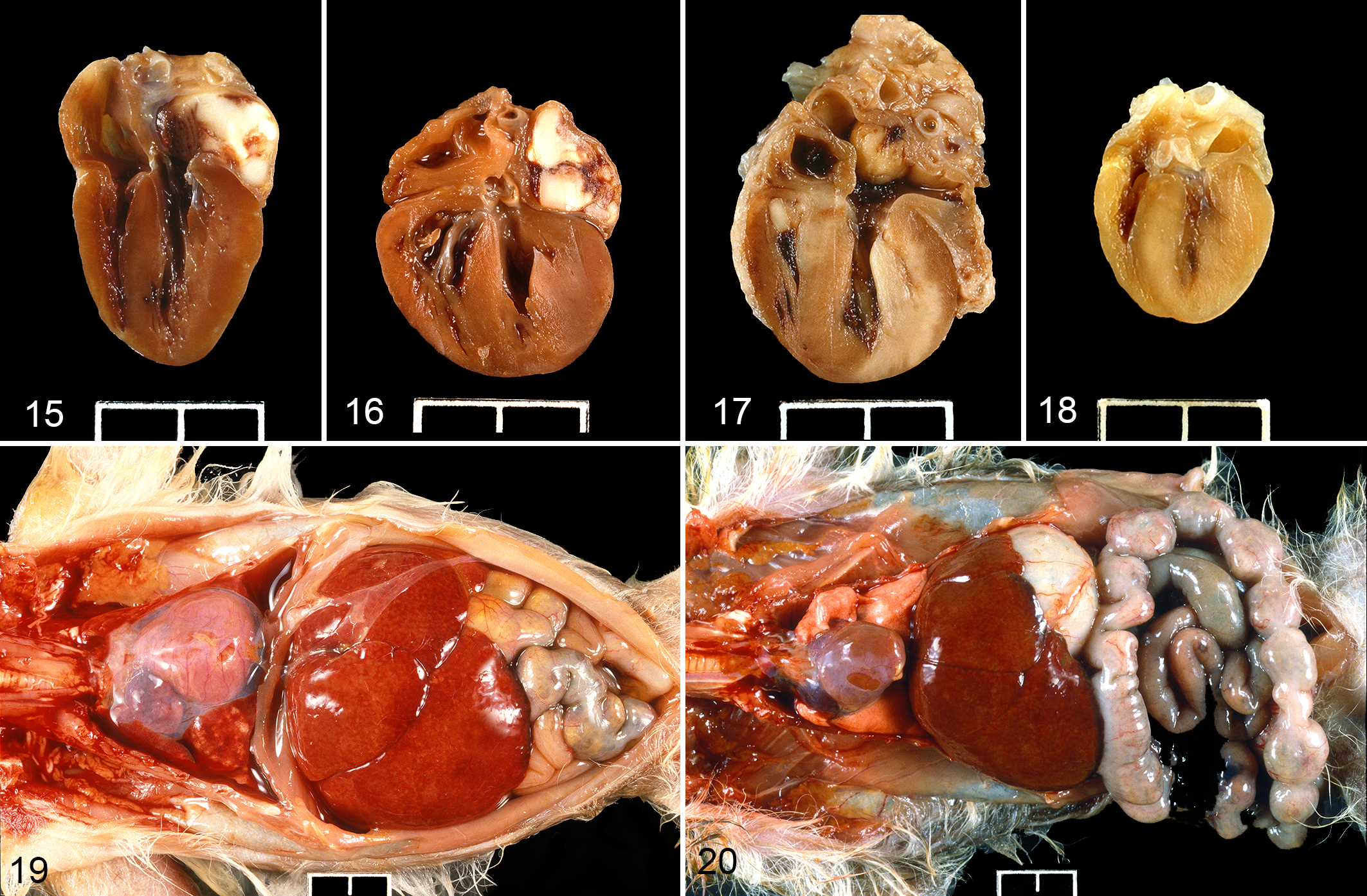
Hypertensive disease, heart, pygmy marmoset. Case 14 (Fig. 15), case 68 (Fig. 16), and case 48 (Fig. 17) have marked cardiomegaly, cardiac hypertrophy, and left atrial thrombosis. Case 60 (Fig. 18) is a reference case without morphologic evidence of hypertension. Figures 15–18: Scale bar = 1 cm.
Heart weight and body weight at necropsy were available for 6 animals (cases 61, 66, 67, 68, 69, 72). Five of these cases had subjective enlargement of the heart and thickening of left ventricular free walls or biventricular hypertrophy on gross examination. In 2 of these 5 cases (cases 67, 68), a left atrial thrombus was identified. The heart weight/body weight ratios for the 6 cases, respectively, were 1.27%, 0.56%, 1.32%, 1.27%, 0.90%, and 1.01%, whereas this ratio was 0.56% for the case in which cardiomegaly was not identified grossly (case 66). Using allometry based on comparisons to data on primate organ weights obtained from published reports and additional data points from the Oregon Regional Primate Research Center, predictive values for organ weights were determined. 42 A prediction equation generated from primate data that Stahl provided for primates is as follows: Organ (Heart) Weight (grams) = aMb , where M = body mass (kg), a = 5.2, and b = 0.97. The values of the constants are referable to primate hearts, and derivations are provided in Stahl. 42 Based on this formula basis, the predicted heart weight for case 66 was 0.932 g; the observed heart weight was 0.95 g (ie, indicating the heart was not enlarged, consistent with gross observations). For the other 5 animals, the observed heart weights were 209 ± 31% (mean ± SD) of the predicted heart weights based on body mass (consistent with the gross diagnoses of cardiomegaly and hypertrophy). The observed values may have been partially skewed in the 2 cases with left atrial thrombosis (Suppl. Table S1).
Pulmonary edema with low-protein intra-alveolar fluid interspersed with hypertrophied alveolar macrophages containing hemosiderin (so-called heart failure cells) were found in 21/30 animals with lesions of hypertension. Grossly, the lung was turgid and had moderate brown discoloration in some animals, attributed to the presence of siderophages. In these 21 cases with lung lesions, 10 had left atrial thrombi (often organizing) and 15 had excessive pleural, pericardial, or abdominal fluid. The 8 animals with bi-cavitary and 5 animals with tri-cavitary effusions had cardiomegaly and often hepatomegaly consistent with chronic passive congestion of the liver (Fig. 19). For the 8 animals with significant hepatomegaly, one had concomitant severe hepatic amyloidosis which likely contributed to hepatic enlargement.
No gross morphologic or histologic lesions were identified in the adrenal or thyroid glands. In the brain, 1 case had laminar cortical necrosis attributed to ischemia, and another had focal malacia and hemorrhage in the thalamus associated with vascular necrosis and thrombosis.
Discussion
The frequent vascular, cardiac, and renal lesions identified in spontaneous deaths in this colony of western pygmy marmosets were consistent with the underlying diagnosis of hypertension. This included frequent arteriolar hypertrophy (proliferative arteriolosclerosis) including “onion skin-like” change, hyalinization (hyaline arteriolosclerosis), and fibrinoid necrosis in arteries and arterioles, particularly in the kidneys; as well as nephrosclerosis or glomerulosclerosis and subjectively enlarged juxtaglomerular apparatus with prominent granular cells in kidneys; and variable cardiomegaly and cardiac hypertrophy attributed to pressure overload, frequently associated with left atrial thrombosis. Due to the small size of the animals and the inability to find a neonatal occlusive cuff that could be used with these animals, definitive confirmation by blood pressure measurements could not be achieved.
Hypertension is defined as persistent elevated blood pressure of ≥140/90 mm Hg in humans. 1 Systemic hypertension is typically considered as primary or essential hypertension, which implies that blood pressure is persistently elevated with no identifiable cause, or secondary hypertension associated with some underlying abnormality. Although a single cause is not established for primary hypertension, numerous factors can contribute to the elevated blood pressures. In humans, primary or essential hypertension accounts for greater than 95% of cases of elevated blood pressure and this develops gradually over a period of years. 38
Secondary hypertension is associated with specific identifiable causes and accounts for 5% to 10% of human hypertension patients. 28,37 These causes include renal disease, hyperadrenocorticism, pheochromocytoma, primary hyperaldosteronism, thyroid gland disease such as hyperthyroidism, congenital vascular defects, some brain tumors, certain drugs, obstructive sleep apnea, obesity, and pregnancy-induced hypertension. 9,37 The majority of these specific causes are excluded by complete necropsy examination of the pygmy marmosets. In particular, no evidence of pheochromocytoma was found in any of the animals in the adrenal glands. Previous reviews of pheochromocytoma in callitrichids found myocardial degeneration and fibrosis and coronary artery hyalinization and medial thickening, interpreted as consistent with hypertension in a cotton-top tamarin (Saguinus oedipus). 32 Similarly, myocardial fibrosis and variable arteriolosclerosis (possibly due to hypertension) were reported in 6 New World primates with pheochromocytomas, including 2 golden lion tamarins (Leontopithecus rosalia) and 2 cotton-top tamarins. 20
The vascular changes that were prevalent in the kidneys of these pygmy marmosets involved interlobar, arcuate, and interlobular (cortical radial) arteries and afferent arterioles to variable degrees. The vascular lesions are not a usual consequence of end-stage renal failure or other primary renal disease. Therefore, we interpreted the hypertension—evidenced by hyaline and proliferative arteriolosclerosis and fibrinoid necrosis—as a cause of the renal lesions rather than a sequela of chronic renal disease. This argument has been discussed in human studies. 17,27,31,39 As early as 1937, significant intrarenal arteriolar disease was recognized in as high as 95% of hypertensive humans. 33 Tisher and Brenner discussed the history of human hypertension research and what he considered, at the time, to be a “dominant central theme” of hypertension research: whether alterations of the renal vasculature are the result of elevated blood pressure or the initiator of hypertension. 44
The nature and severity of the renal vascular disease described in these pygmy marmosets suggest primary hypertension as the initiating event driving renal parenchymal disease—so-called hypertensive renovascular disease or hypertensive nephrosclerosis. 10,17 However, these are terms that are a pathologic description, lacking the specificity of an actual disease. 31 Interstitial inflammatory fibrosis is a main feature of nephrosclerosis, which in turn can result in entrapment of peritubular capillaries and renal ischemia associated with obsolescent glomeruli. In other instances, the pathogenesis may involve loss of autoregulation of renal blood flow associated with alterations of smooth muscle of afferent arterioles resulting in hypertrophied glomeruli and focal segmental glomerulosclerosis. Both consequences to glomeruli can be seen as morphologic findings in the pygmy marmosets, although glomerulosclerosis was more typically global as also noted in human texts. 10
The argument might be made that alterations in glomeruli have obscured the primary initiating renal disease, which in turn resulted in the now-predominant changes in the renal vasculature. However, we think that the different glomerular morphologies—including normal glomeruli, ischemic fibrotic obsolescent glomeruli, and hypertrophied glomeruli with segmental or diffuse glomerulosclerosis—suggest that the glomerular lesions are a response to vascular disease secondary to hypertension. This issue cannot be resolved in a retrospective postmortem study of animals that died spontaneously, and we acknowledge this limitation. However, following the exclusion of commonly occurring causes of secondary hypertension and considering the nature of the renal disease in these animals and the absence of clinical renal failure and generalized end-stage renal disease, we consider that secondary hypertension caused by chronic renal disease is not likely.
Renal and cardiovascular disease have not been common topics of publication in callitrichids, unlike Old World primates or Great Apes, although both have been described in some references to incidental spontaneous or background lesions of the cardiovascular system, mostly in common marmosets (Callithrix jacchus) and tamarins. 8,21 Clinically evident congestive heart failure was seen in significant numbers of tamarins, with mention of some myocardial fibrosis or of cardiac dilatation with minimal histologic change. 12 Progressive glomerulonephropathy in common marmosets has been well studied, but the publications do not indicate vascular disease suggestive of hypertension. 19,47 A comprehensive review of callitrichid diseases included a reference to IgM mesangial nephropathy in a colony in Germany. 5,35 Of the 213 tamarins and marmosets studied, 2 of the animals were pygmy marmosets diagnosed with tubulointerstitial nephritis and mesangial proliferation, but absence of renal failure. No report was made of vascular or cardiac lesions in any of the marmosets or tamarins. Padilla and colleagues 34 described a high prevalence of glomerulonephritis in adult pygmy marmosets in the Smithsonian National Zoological Park that could progress in the end stages of the renal disease to a hypertensive syndrome with cardiovascular disease. The authors speculated that immune-mediated mechanisms subsequent to chronic infections or parasitism eventuated in chronic renal disease followed by cardiovascular disease. The authors did not provide clinical support for this pathogenetic sequence and did not state the number of animals progressing to a hypertensive syndrome. Alternatively, we propose, and have previously suggested, 11 that primary hypertension is predominant in pygmy marmosets and leads to secondary renal and cardiovascular disease. However, no definitive clinical support for primary hypertension was documented in the present study, as well. Sporadic animals had biochemical profiles that did not indicate hyperglycemia, although mild proteinuria was occasionally noted on urinalysis. One animal was diagnosed with worsening cardiomegaly based on repeat thoracic radiographs.
As mean arterial pressure equals cardiac output times total peripheral resistance, elevated blood pressure can stem from increased cardiac output, increased total peripheral resistance, or both. 38,44 Cardiac output is determined by heart rate and stroke volume, which is determined by both the size of the ventricular compartment and myocardial contractility; and peripheral resistance is determined by both anatomic changes in and function of the arteries and arterioles. The definitive explanations for primary hypertension remain elusive.
The determinants of hypertension in this pygmy marmoset colony require further investigation. The high prevalence of pygmy marmosets with vascular, cardiac, and renal lesions suggestive of hypertension occurred in several breeding pairs spanning multiple generations, as well as in twin pairs born in this colony. This corresponds to the familial basis for essential hypertension in humans that has been estimated as high as 60%. 38 A genetic factor in these pygmy marmosets is further supported by the occurrence in 3 wild-caught animals, all of which sired progeny with hypertension. The male predominance of systemic hypertension is also similar to what has been observed in humans. Other determinants of hypertension in humans include renal mechanisms involving excess intake of sodium and pressure natriuresis or the excretion of sodium in the urine, vascular mechanisms concerning endothelial cell dysfunction involving the nitric oxide pathway, and hormonal mechanisms involving the renin-angiotensin-aldosterone system (RAAS). 38
In recent years, sympathetic nervous system overactivity has emerged as a potential controller of the main determinants of hypertension and is proposed as a central factor in primary hypertension associated with a wide spectrum of mechanisms or contributory factors. This is associated with disturbed basal sympathetic tone rather than simply sympathetic activity. There is direct sympathetic innervation of both the vasculature and the myocardium. The initiating site of this sympathetic activity is the hypothalamus, subsequently controlled by the cerebral cortex through cortical hypothalamic connections. This implies that hypertension is not solely a disease of the circulatory system. 38 The importance of autonomic dysfunction in essential hypertension has been noted by multiple authors. 7,22,36,40 However, acknowledgment must be made that in a major review of animal models of hypertension from the American Heart Association in 2019, the pathogenesis of human hypertension was cited as obscure and the statement was made that “the primary cause of elevated blood pressure is not apparent in the vast majority of affected individuals.” 25 Therefore, any suggestion of the cause of apparent hypertension in pygmy marmosets in which clinical substantiation of elevated blood pressure is lacking is speculative.
Hypertension is recognized as an important risk factor for cardiovascular disease and, specifically, heart failure. Elevated left ventricular ejection force may be associated with sympathetic overactivity. 39 Cardiac pressure overload is a sequela of elevated peripheral vascular resistance, as is left ventricular hypertrophy. Several pygmy marmosets had cardiomegaly and often left ventricular or biventricular hypertrophy. Furthermore, morphologic alterations in the myocardium may have contributed to diminished compliance. Coronary artery disease is another contributing factor and some affected animals had hyaline arteriolosclerosis or fibrinoid necrosis of coronary vasculature. Last, cardiac arrhythmias may be involved, although no electrocardiograms were obtained. Several of these contributors to heart failure may occur in conjunction with left atrial thrombosis which was documented in 10 animals.
Although sympathetic overactivity is an important contributor to hypertension and can explain these cardiovascular changes, sympathetic overactivity can have consequences independent of hypertension. 22 Coexistent with the detrimental consequences related to hypertension and sometimes independent of primary hypertension are the effects of sympathetic overactivity on chronic kidney disease. This includes (a) direct stimulation by the sympathetic nervous system of β1 adrenergic receptors on the juxtaglomerular apparatus resulting in increased renin secretion, which leads to elevated plasma angiotensin II levels that contribute to peripheral vasoconstriction; (b) feedback of chemoreceptors and baroreceptors richly supplied in the kidneys to the brain contributing to regulation of sympathetic outflow and, subsequently, systemic blood pressure; (c) renal origin of enzymes (such as renalase) that degrade activated catecholamines to decrease blood pressure; and (d) elevation of the primary endogenous nitric oxide synthase inhibitor in chronic renal disease. Therefore, based on these findings, hypertension and chronic renal disease may be linked partly by a cause-and-effect relationship or by a more complex relationship related to sympathetic overactivity that contributes to both.
Based on the most recent reviews of hypertension in humans, sympathetic overactivity is an important unifying hypothesis as a major direct or indirect causal factor in the development of primary hypertension. 7,38 The centrality of sympathetic overactivity thus becomes an important risk factor for cardiovascular and renal disease. In turn, this may explain the spectrum of pathologic changes in the heart and kidneys and the probable hypertension in this colony of pygmy marmosets, although there is no confirmatory proof.
Spontaneous hypertension has been recognized in other primate species. The owl monkey (Aotus sp.) is the biomedical research model most closely satisfying the hypothesis of the centrality of sympathetic overactivity. 40 These monkeys experience extremely high systolic blood pressures ranging from 120 to 150 mm Hg at rest to 220 to 259 mm Hg with minor disturbance, as determined either under ketamine anesthesia or via implanted aortic pressure transducers. Increased cardiovascular mortality, cardiomegaly, and cardiomyopathy are observed in captive animals. Electrical stimulation of the perifornical/lateral hypothalamic area produces elevated blood pressure and heart rate, and neurosurgical ablation of this structure resulted in a significant decrease or elimination of the hypertensive response to routine laboratory procedures. This region of the brain appears critical as a neural control in the initiation or exacerbation of cardiovascular responses contributing to primary hypertension. 40 These findings support the role of sympathetic tone in primary hypertension and suggest that owl monkeys may experience hyperreactive responses to seemingly normal environmental events such as feeding, cleaning, and cage opening. These findings might be applicable to hypertension in pygmy marmosets.
The African green monkey, Chlorocebus aethiops sabaeus, 36 is another species of interest. African green monkeys introduced in the West Indies as an invasive species develop spontaneous hypertension in both free-ranging and managed care animals. Hypertension is defined similarly to that in humans as systolic blood pressure ≥140 mm Hg and borderline hypertensive at ≥121 to 139 mm Hg. In captive African green monkeys, 37% of the 424 animals studied were assessed as hypertensive, 36 corresponding to the estimate of the American Heart Association that 34% of Americans have hypertension. 1 Increased age was associated with a higher risk of hypertension, as in humans. Hypertensive monkeys had a higher heart rate correlating with essential hypertension in humans with increased sympathetic drive. Changes in monkey renal vasculature and glomeruli correlate with those in humans with essential hypertension. Genetic factors have been implicated by examination of data in the African green monkeys that develop spontaneous hypertension, 24 similar to genetic factors in humans.
The woolly monkey, Lagothrix lagotricha, also develops spontaneous hypertension based on physiologic, clinical, and pathologic studies. 6,14 These animals developed hyaline arteriolar nephrosclerosis and death associated with presumed complications of hypertension including congestive heart failure, renal failure, and acute cardiovascular accidents. In a case report, cardiomyopathy and spontaneous intracardiac thrombi were found at necropsy in a captive woolly monkey. 2 Similar to the pygmy marmoset, woolly monkeys are classified as vulnerable in the wild, poorly amenable to captivity based on failure of captive breeding, and ethically inappropriate as an experimental hypertensive model species.
The documentation of morphologic findings consistent with hypertension in pygmy marmosets is not meant to suggest that this is an appropriate or ethical comparative hypertension model. However, these primates are found in zoological collections and the propensity for certain diseases should be part of the scientific record and used to modify husbandry practices to ensure the well-being of this species. Research in Ecuador found that pygmy marmosets vacated human-disturbed habitats and were, thus, susceptible to being lost in local regions in which they were indigenous. 13 Unlike more generalized and flexible species, 30 such as the common marmoset, pygmy marmosets are more specialized marmosets, and, subsequently, vulnerable to changes in habitat.
Our findings suggest that hypertensive renovascular disease with concurrent or subsequent cardiovascular disease is a common necropsy finding in the pygmy marmoset and is an important underlying cause of morbidity and mortality. This conclusion is based entirely on morphology and is evidenced by vascular and glomerular lesions including hyaline or proliferative arteriolosclerosis, fibrinoid necrosis, and glomerulosclerosis that is global or segmental consistent with hypertensive renovascular disease described in human texts. 10,44 Cardiovascular and renal disease are typically not manifested until severe target organ damage occurs, often evident as heart failure or multi-organ failure, consistent with the observation that prey species are very good at hiding clinical signs until animals are in extremis. Based on recognition in humans that hypertension-induced renal disease may include arteriolosclerosis, interstitial fibrosis, and glomerulosclerosis and that persistent hypertension is a leading cause of cardiac injury and congestive heart failure, 25 the lesions in this colony of pygmy marmosets are consistent with systemic hypertension. We recognize that hypertension may not be the only cause of these lesions and comorbidities such as glomerulonephritis may occur and complicate the disease. The severe and advanced arteriolar lesions in the kidneys are unlike the lesions more commonly encountered in the hypertension models that have a short lifespan or limited duration of hypertension (such as the rat and mouse) and these lesions have not been reported in the multiple morphological studies of common marmosets such as that reporting progressive glomerulonephropathy. 47 The current study involved a wide age range of affected pygmy marmosets, and the study findings suggested a genetically influenced failure of adaptation, and sex differences in prevalence. In summary, the study findings suggest that the severe and prevalent vascular disease in these animals may be the result rather than the cause of hypertension and the hypertension may be primary rather than secondary, resulting in hypertensive renovascular disease and congestive heart failure.
The findings of this study suggest that spontaneous hypertension may be prevalent in the pygmy marmoset, as it is in the owl monkey, African green monkey, and woolly monkey. Similar to the owl monkey and possibly African green monkey, 25 sympathetic overactivity is hypothesized as a major factor in the development of hypertension in pygmy marmosets. We speculate that elevated sympathetic nervous activity may be attributed to elevated levels of stress in pygmy marmosets, even though these animals are seemingly adjusted to routine husbandry practices. Given the small size of pygmy marmosets, they live under constant threat of predation in the wild and their strategy to avoid this is to either freeze or hide or flee. In most managed care facilities, pygmy marmosets do not have the option to flee or may not feel sufficiently “hidden” when humans enter their space. Given the size of humans in comparison to pygmy marmosets, this suggested level of constant stress when exposed to humans might contribute to the elevation of sympathetic activity similar to that documented in the Aotus due to perceived routine events triggering hypertension. Therefore, although the present study did not measure sympathetic activity or blood pressures, we speculate that elevated sympathetic activity might be causing hypertension and the resulting pathologic changes in the kidneys and heart that predominated in this colony of pygmy marmosets. Because of the high rate of hypertension in this group of captive pygmy marmosets and their vulnerable conservation status in the wild, we recommend that careful consideration is given when determining the suitability of this species for long-term managed care or captive research programs. Rather than solely maintaining a genetically viable, managed care population, efforts should be focused on conserving this species in the wild and mitigating long-term habitat loss to prevent extinction of this species.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211052664 - Vascular, cardiac, and renal lesions attributed to primary systemic hypertension in western pygmy marmosets (Cebuella pygmaea)
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211052664 for Vascular, cardiac, and renal lesions attributed to primary systemic hypertension in western pygmy marmosets (Cebuella pygmaea) by Avery James Cooley, Anne Savage and Charles T. Snowdon in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
