Abstract
The clinical, hemodynamic, and pathologic features of hypertensive encephalopathy in two cats with reduced renal mass are described. The cats developed a progressive syndrome of lethargy, ataxia, blindness, stupor, and seizures following an abrupt increase in blood pressure associated with a surgical reduction in renal mass. The cats had severe gross brain edema, evidenced by cerebellar changes of caudal coning and cranial displacement over the corpora quadrigemina and cerebral changes of widening and flattening of the gyri. Histologically, interstitial edema was most pronounced in the cerebral white matter. Hypertensive vascular lesions were present as hyaline arteriolosclerosis in one cat and hyperplastic arteriolosclerosis in the other. Rare foci of parenchymal microhemorrhages and necrosis were also observed. Systemic hypertension (especially severe or rapidly developing) accompanied by neurologic signs and the pathologic findings of diffuse brain edema with cerebral arteriolosclerosis are consistent with an etiologic diagnosis of hypertensive encephalopathy.
Hypertensive encephalopathy is an acute neurologic syndrome precipitated by an abrupt, sustained rise in systemic arterial blood pressure (BP). 3, 5, 9, 32, 44 It has been well described as a complication of systemic hypertension in people in which associated clinical signs include decreased alertness, headache, nausea and vomiting, seizures, and visual disturbances. 3, 5, 8, 9, 13, 23, 27, 38 The typical neuroimaging lesion associated with hypertensive encephalopathy in people is bilateral, white matter edema of the posterior cerebrum, and this lesion is reversible with appropriate antihypertensive therapy 8, 13, 27 Although reversible if recognized and treated promptly, hypertensive encephalopathy can progress to coma and death if treatment is delayed. 40 Although hypertensive encephalopathy can occur in people with primary hypertension, it more commonly occurs when both hypertension and renal disease are present, and it is a recognized complication of human renal transplantation. 9, 23, 39
A clinically similar neurologic syndrome has been described in cats with systemic hypertension and renal disease and in cats following renal transplantation. 10, 12, 19, 21 However, little is known about the pathologic features of this disease in cats. This report describes the clinical, hemodynamic, and pathologic features of hypertensive encephalopathy in two cats with reduced renal mass.
Materials and Methods
Animals
Four female domestic short-hair cats aged between 6 and 12 months were studied. The four cats described in this report were part of a previously reported study designed to test the hypothesis that amlodipine besylate would decrease blood pressure (BP) and reduce the rate of complications in cats with induced renal insufficiency. 22 The four currently reported cats had surgically induced renal insufficiency but did not receive antihypertensive therapy (untreated control group). All animal experimentation has been previously described and was conducted in accord with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and was approved by the Institutional Animal Care Committee. 22
Renal mass was reduced by partial nephrectomy, as previously described. 22 Briefly, on day 0 the left kidney was partially infarcted by ligation of selected interlobular arteries, and 14 days later the right kidney was removed, resulting in a net reduction of approximately 11/12 of total renal mass. This degree of renal mass reduction resulted in moderate azotemia with a mean serum creatinine in the original study of 3.5 mg/dl. 22
At the time of partial renal infarction (day 0), a radiotelemetric BP sensing implant (Data Sciences International, St. Paul, MN) was placed in the right femoral artery of each cat as previously described. 22 This radiotelemetry system allows for continuous measurement of BP, pulse rate, and physical activity. 22 All radiotelemetry measurements were recorded as an average value over 10-second intervals every 5 minutes for 24 hours daily throughout the study. Twenty-four-hour ambulatory BP measurement is considered to provide the most accurate assessment of BP. 28
Pathology
Cats were euthanatized by administration of an overdose of sodium pentobarbital, and brain from each of the four cats was examined grossly and microscopically. After initial in situ gross examination, the brains were removed and placed in 10% neutral formalin. After fixation, the brains were sagittally sectioned and evaluated grossly. Consecutive 4-mm transverse slices were made through the brain, embedded in paraffin, sectioned at 5-μm, and stained with HE. Selected sections were stained with periodic acid–Schiff hematoxylin (PASH). Immunohistochemistry was performed using formalin-fixed, paraffin embedded 4- to 6-μm-thick sections. Briefly, sections were heated for 30 minutes in a 60°C oven, deparaffinized, and stained using a TechMate 500 Immunostainer (BioTek Solutions, Inc., Santa Barbara, CA) with antibodies against smooth muscle actin (Dako Corporation, Carpinteria, CA).
Results
Normal 24-hour mean systolic BP obtained by telemetry from normal cats in this laboratory is 125.1 ± 10 mm Hg. 22 All four cats had mean 24-hour systolic BPs greater than 140 mm Hg on day 3 after the unilateral renal infarction, with the highest systolic BPs in cats No. 1 and 3 (Fig. 1). From day 3 to day 14, cats No. 3 and 4 had a progressive decline in systolic BP, decreasing to normal in cat No. 4, and remaining mildly elevated in cat No. 3. The systolic BP in cat No. 2 remained relatively stable and by day 10, was similar to that of cat No. 3. In contrast, the mean systolic BP in cat No. 1 remained at or above 180 mm Hg on days 6 through 14. Following the second reduction in renal mass (unilateral nephrectomy on day 14), cat Nos. 1 and 2 had a marked abrupt increase in BP (Fig. 1, cat Nos. 1 and 2). Over 48 hours, the mean 24 hr systolic BP in cat Nos. 1 and 2 increased from 184.6 mm Hg and 138.2 mm Hg to 234.4 and 198.2 mm Hg, respectively, representing a 40–50 mm Hg elevation in BP. Within 12–18 hours of this increase in BP, both cats developed severe ataxia that progressed over a period of 8 hours to blindness, stupor, and seizures. Cat Nos. 1 and 2 were euthanatized approximately 8 hours after the onset of neurologic signs. For comparative purposes, we studied two cats that were subjected to the same procedures but did not develop neurologic signs (Fig. 1, cats 3 and 4). Although the mean 24-hour systolic BP in cat Nos. 3 and 4 also increased following the unilateral nephrectomy, the magnitude of the increase was small compared with the BP increases in cat Nos. 1 and 2. The mean systolic blood pressure in cat Nos. 3 and 4 increased from 156 mm Hg and 136.2 mm Hg to 170 and 155 mm Hg, respectively, representing a 14–19 mm Hg elevation in BP following the unilateral nephrectomy. Neurologic abnormalities were not observed in cat Nos. 3 and 4, and they were euthanatized at the completion of the study approximately 21 days later.
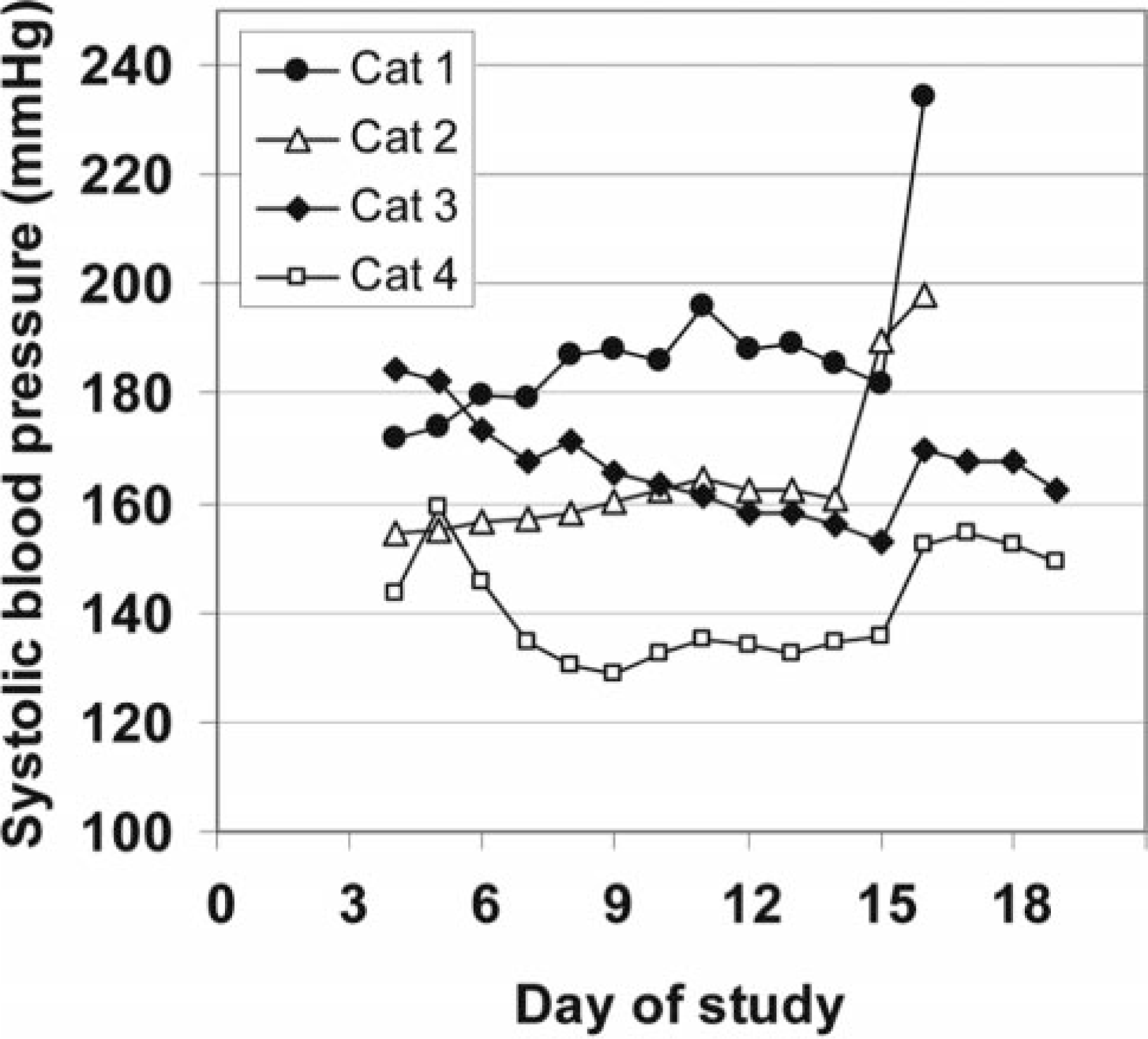
Cats. Mean 24-hour systolic blood pressure values.
Following euthanasia, the brain was removed from all four cats and examined grossly and microscopically. In cat Nos. 1 and 2, the middle vermis of the cerebellum was displaced caudally into the foramen magnum. In cat No. 1, the meninges overlying the herniated middle vermis were diffusely hemorrhagic (Fig. 2). The brains from all four cats were sectioned longitudinally after fixation in formalin (Fig. 3). In cat Nos. 1 and 2, the cerebellum exhibited caudal coning or “lipping” through the foramen magnum and cranial displacement above the corpora quadrigemina (Fig. 3A, B). The expansion of the cerebellum resulted in marked distortion of the contour of this structure compared with the oval contour of the normal cerebellum. There was also widening and flattening of the cerebral gyri, resulting in attenuation of the sulci. In contrast, cat Nos. 3 and 4 did not have gross cerebellar or cerebral abnormalities (Fig. 3C).
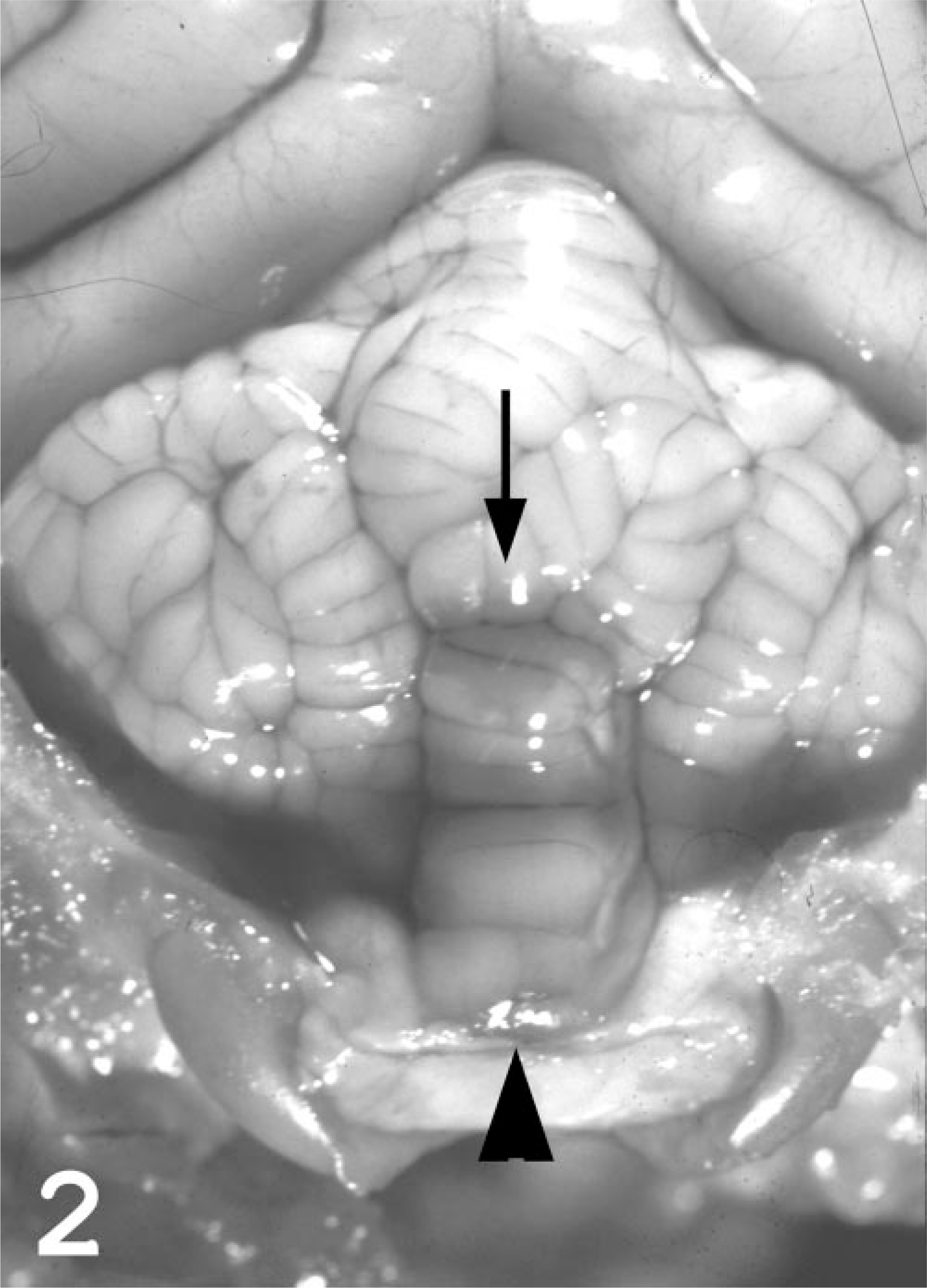
Brain in situ, skull cap removed; cat No. 1. Herniation of the middle cerebellar vermis through the foramen magnum (arrowhead) with congestion and focal meningeal hemorrhage (arrow).
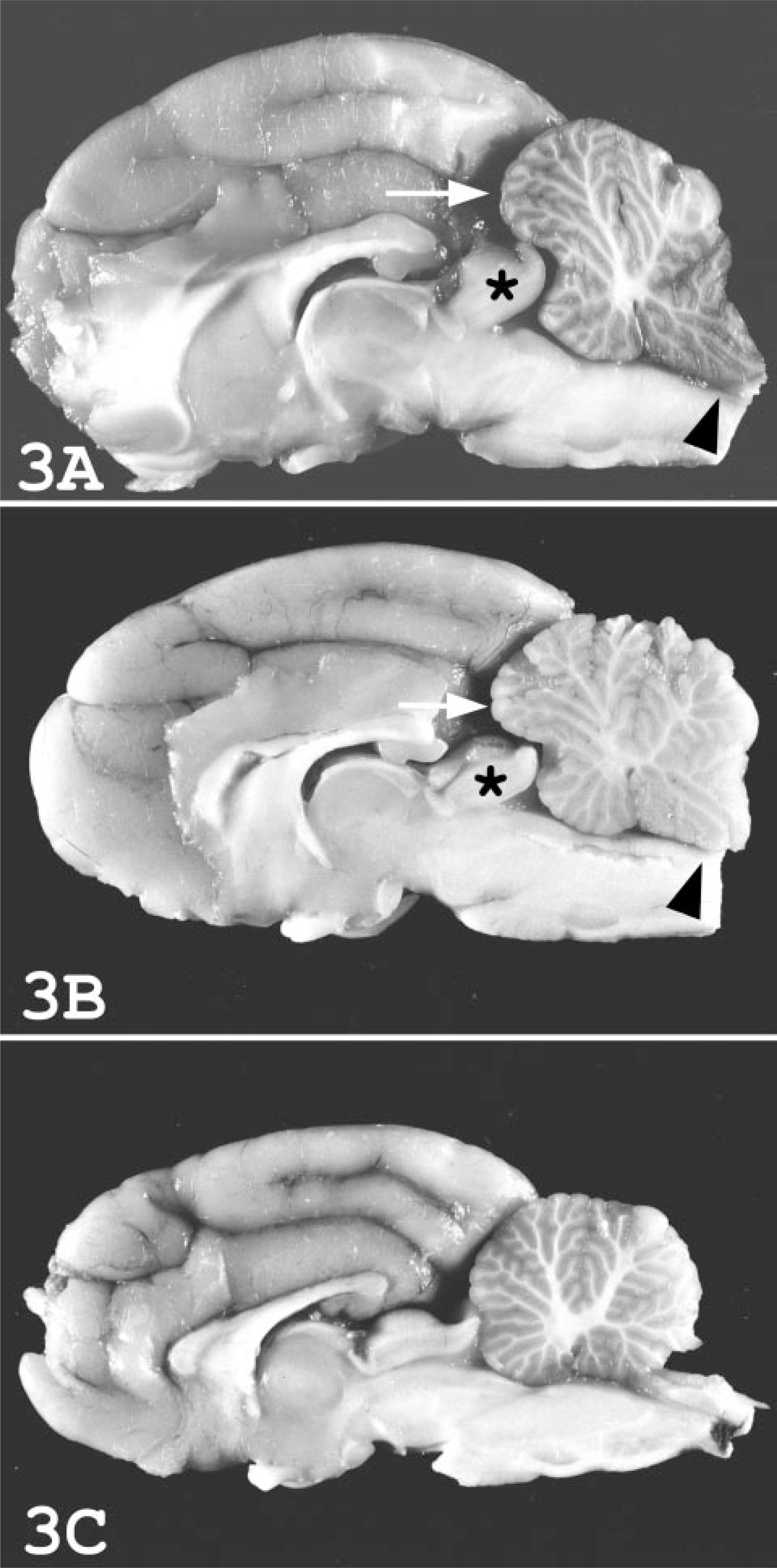
Brain in sagittal plane; cat.
In cat Nos. 1 and 2, microscopic brain lesions consisted primarily of marked pallor of the cerebral white matter; equivocal vacuolization of the white matter was present in the cerebellum and brain stem. There was marked separation and accentuation of the linear myelin sheaths by interstitial edema (Fig. 4A). Marked widening of the spaces around arterioles in severely hypertensive cats was attributed to the accumulation of clear edema fluid. Variably sized, periodic acid–Schiff-positive protein droplets were occasionally present within this perivascular edema (Fig. 5A). Narrower clear spaces were also present around blood vessels in cat Nos. 3 and 4 and were interpreted to be normal fixation artifacts (Fig. 5B). Protein droplets were not present in the perivascular space.
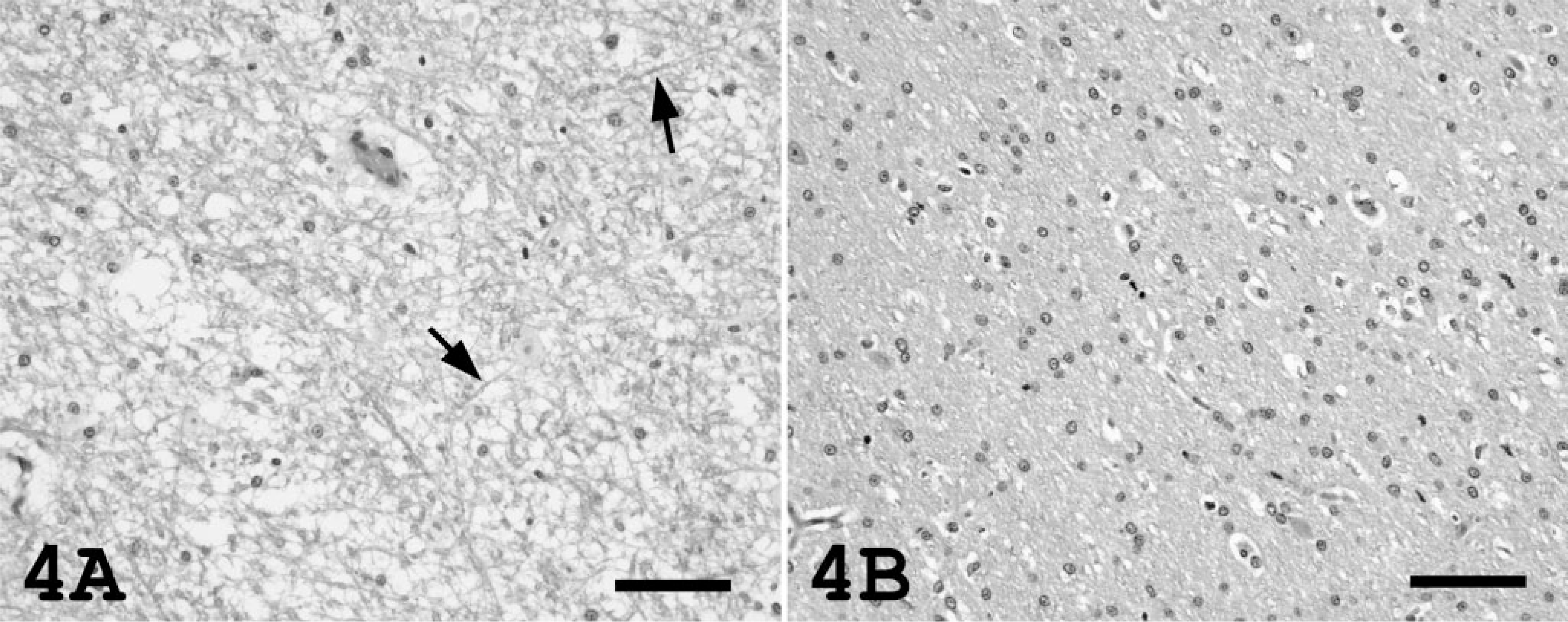
Cerebral white matter; cat.
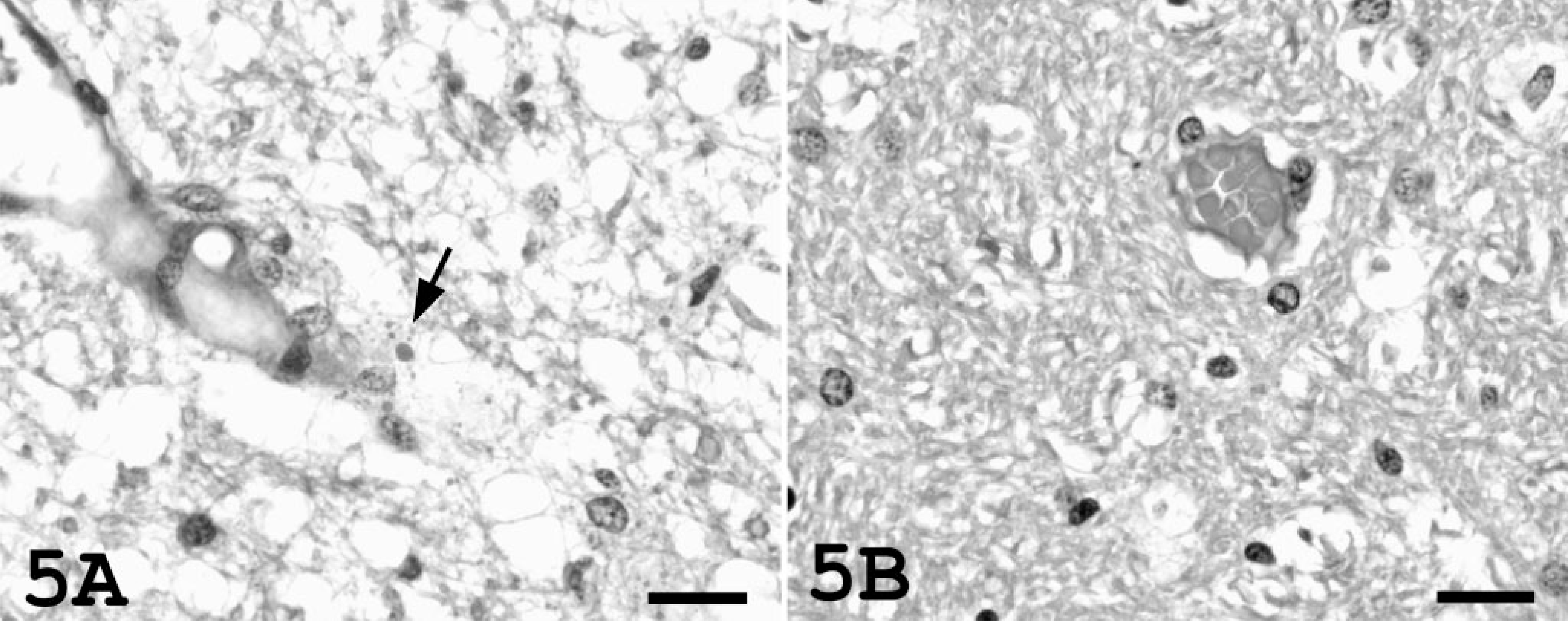
Cerebral white matter; cat.
Vascular lesions were present in both hypertensive cats. There was a single focus of arteriolar hyalinosis in cat No. 2, characterized by eosinophilic hyaline material within the vessel wall of a cerebral surface arteriole (Fig. 6A). The perivascular space contained fine strands of fibrin and small numbers of macrophages and neutrophils. There was focal ischemia and inflammation in the underlying outer cortical lamina. More pronounced vascular changes associated with hypertension were present in cat No. 1. Many pial arterioles exhibited hyperplastic arteriolosclerosis, characterized by onion-skin laminated thickening of the walls of the arterioles with luminal narrowing (Fig. 6B). Cells within the thickened vessel wall stained intensely with antibodies against smooth muscle actin (Fig. 6C). These hyperplastic vascular changes were associated with rare ischemic changes in the brain. An arteriole in the one focal ischemic area was markedly thickened due to smooth muscle hypertrophy and mural edema, and there was mural dissection with mural and peri-vascular edema, hemorrhage, and fibrin exudation (Fig. 6D). The surrounding neuropil was necrotic, and there was nuclear pyknosis. Rare focal microhemorrhages were present within the neuropil in cat Nos. 1 and 2. There were no vascular lesions observed in the brains of cat Nos. 3 and 4 (Fig. 6E).
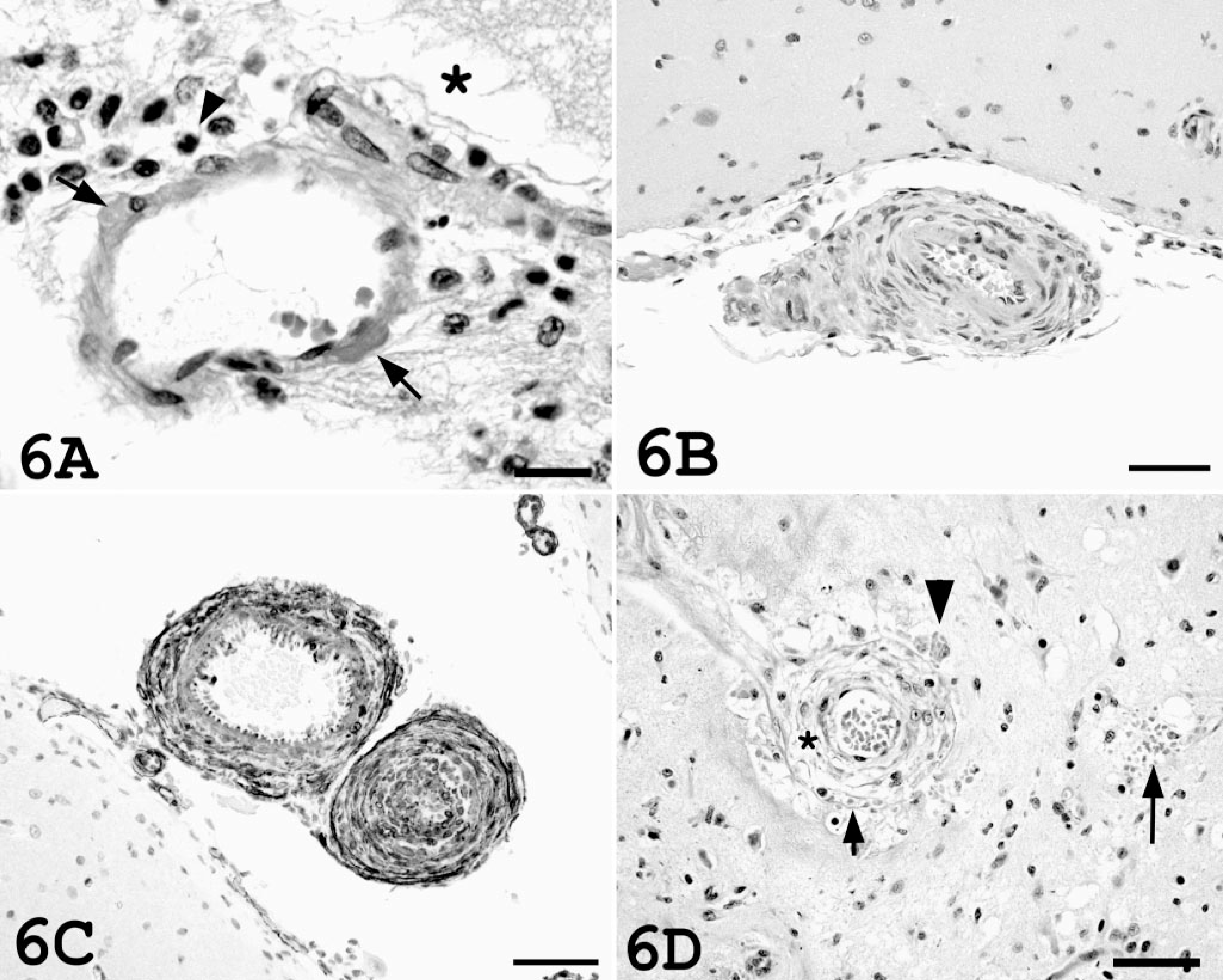
Brain, vascular histology; cat.
Discussion
Cerebral arterial blood flow is normally maintained at a relatively constant level despite changes in BP, thereby preventing barotrauma and edema during periods of elevated BP and ischemia during periods of low BP. 26 This “autoregulation” of cerebral blood flow occurs by constriction of small resistance cerebral arteries and arterioles when BP is elevated and by vasodilation when BP falls. 15, 26 As BP approaches the upper limit of cerebral autoregulation, pial arterioles develop a sausage-string appearance. 7, 15 The constricted segments represent areas in which the normal autoregulatory response is maintained, whereas the dilated segments represent areas of autoregulatory failure and forced vessel overdistension. 5, 7, 15 Vessel over-distension causes breakdown of the blood-brain barrier, with opening of the endothelial tight junctions and leakage of plasma proteins into the extracellular space (vasogenic edema). 7, 11, 15, 20 When mean BP rises further or is sustained at high levels, the segmental dilation becomes diffuse, leading to transmission of the elevated pressure to the capillary lumen. This alteration in Starling forces leads to generalized, interstitial, cerebral edema. 7, 11, 20
Hypertensive encephalopathy occurs following a precipitous and sustained rise in BP that exceeds the upper limit of autoregulation. 5, 44 In cats, systemic hypertension is most commonly associated with chronic kidney disease, and the end-organ effects of hypertension on the eyes and heart are being increasingly recognized. 4, 12, 17, 19, 21, 24, 31, 35 An association between seizures and other neurologic signs in cats with chronic kidney disease and uncontrolled hypertension has also been noted. 12, 18, 19 In two retrospective studies, most hypertensive cats had evidence of renal dysfunction and 29–46% had neurologic signs compatible with hypertensive encephalopathy. 19, 21 Up to 21% of cats develop seizures, ataxia, stupor, and/or blindness after renal transplantation due to posttransplantation hypertension. 10, 18 Prophylactic treatment of cats with antihypertensive medications following renal transplantation largely prevents the development of these neurologic complications. 2, 18 In people, hypertensive encephalopathy is also commonly associated with renal failure and is a recognized complication of renal transplantation. 23, 34, 39
The common clinical features in people with hypertensive encephalopathy include headache, altered sensorium, confusion, seizures, vomiting, and visual disturbances. 5, 9, 13, 27 Seizures are often a presenting clinical abnormality and may be preceded by visual auras and hallucinations consistent with an occipital lobe origin for the seizures. 8, 32 Visual disturbances, including cortical blindness, may be present. 8, 32 Many patients also have hypertensive retinal lesions consisting of retinal hemorrhages, exudates, and papilledema. 3, 5, 9, 32 Clinical signs present in the hypertensive cats in this report included ataxia, lethargy, seizures, stupor, coma, and blindness. In other cats that have developed hypertensive encephalopathy following renal mass reduction, polyphagia, abnormal vocalization, photophobia, frequent blinking, head pressing, and extensor rigidity have also been observed (S. Brown, personal observation). Although ophthalmologic examinations were not performed on the cats in this study, the blindness observed in some cats with hypertensive encephalopathy may be cortical in nature as evidenced by the rapid (hours) return of vision following control of BP (S. Brown, personal observation). As in people, cats with hypertensive encephalopathy often have ocular manifestations of hypertension, including retinal detachment and/or intraocular hemorrhage, which may also cause severe visual deficits. 19, 21 These visual deficits may also be reversible following normalization of BP, but return of vision occurs over a much longer (weeks) period of time. 12
The primary gross and microscopic brain lesion in the cats with hypertensive encephalopathy in this report was interstitial edema. This cerebral edema was clearly evident on gross examination of the brains, particularly when the brains were sectioned longitudinally. The microscopic features of cerebral edema were much more subtle than the gross changes, underscoring the importance of gross evaluation of the brain for the presence of edema. 36 In this study, the edematous brains were characterized histologically by vacuolization of the neuropil with myelin separation. In people with hypertensive encephalopathy the edema is present primarily within the white matter; this was also the case in affected cats of the present study. 8 The predilection of the white matter to develop interstitial edema has been attributed to its relatively loose composition of myelinated fiber tracts in a matrix of glial cells, arterioles, and capillaries. 8, 16
In addition to the diffuse white matter edema, scattered focal hypertensive vascular lesions were observed in both cats. Two forms of small blood vessel disease, arteriolar hyalinosis and hyperplastic arteriolosclerosis, were present in the hypertensive cats in this study. The lesion of arteriolar hyalinosis occurs in people with benign hypertension and is thought to reflect hemodynamic injury to the vascular endothelium with leakage of fibrinoid plasma components into the vessel wall. 11, 33 This lesion was also observed in one of the hypertensive cats. Arteriolar hyalinosis has also been observed in areas of forced vascular distension in acutely hypertensive rats and may precede the development of vascular fibrinoid necrosis. 11 Neutrophils and macrophages were present around affected vessels in the cat with arteriolar hyalinosis; similar cerebrovascular inflammation has been attributed to increased adhesion molecule and cytokine expression by hypertension-activated endothelial cells in hypertensive rats. 37 Hyperplastic arteriolosclerosis in people is most often associated with malignant hypertension 33 and was present in the cat with more severe hypertension in this report. The lesion is characterized by concentric thickening of the arteriolar wall by hyperplastic smooth muscle cells with attenuation of the vascular lumen. 33 Changes of fibrinoid vascular necrosis, consisting of mural hyaline deposits with necrosis, are frequently present along with these hyperplastic changes in people but were not observed in the cat in this report. 33 Both cats had rare parenchymal microhemorrhages. In people, primary brain microhemorrhages are most commonly caused by hypertension and the resultant angiopathy. 6, 14 The focal area of hemorrhage and necrosis in one cat was associated with a focal dissecting mural lesion within a hyperplastic arteriole. Similar vascular dissection has been reported in the aorta of a hypertensive cat. 43 Parenchymal microhemorrhages have also been noted in cats dying with neurologic signs, hypertension, and chronic renal disease. 19 Likewise, vascular lesions, including fibrinoid necrosis, mural fibrosis, and mononuclear perivascular cuffs with focal parenchymal necrosis and hemorrhage, have been described in people with fatal hypertensive encephalopathy. 3 Vascular cerebral lesions were not observed in the two cats that did not develop a marked, abrupt increase in BP following unilateral nephrectomy and did not develop signs of hypertensive encephalopathy. These cats remained clinically normal and continued to be moderately hypertensive (systolic BPs between 150 and 170) during the additional 3-week study period, so it is considered unlikely that cerebral vascular lesions were initially present and subsequently resolved.
In people, the clinical signs and lesions associated with hypertensive encephalopathy are, at least initially, reversible. 3, 8, 9 With appropriate antihypertensive treatment the reversal of the central nervous system (CNS) symptoms in people is rapid, typically occurring within hours to days. 3, 32, 44 This rapid reversal of clinical signs following antihypertensive therapy is considered the best clinical criterion for confirming the diagnosis of hypertensive encephalopathy. 9 Likewise, the neurologic signs may be prevented or occasionally reversed (S. Brown, unpublished observations) with appropriate antihypertensive therapy in cats. 18, 22 For example, complete resolution of clinical signs with antihypertensive therapy was observed in a cat that developed severe hypertension (systolic BP > 300 mm Hg) following renal mass reduction. This cat originally exhibited head pressing and seizures that progressed to coma but became clinically normal within 6 hours following the administration of an antihypertensive agent (S. Brown, personal observation). Similar resolution has been reported in posttransplantation cats following antihypertensive therapy. 18 However, with severe or prolonged hypertensive encephalopathy, additional focal vascular lesions may develop, which may result in focal, irreversible, neurologic defects. 8, 44
Symptoms of hypertensive encephalopathy occur at lower BPs when hypertension develops rapidly. 9 Women with eclampsia or children with acute glomerulonephritis, both of which are associated with sudden rises in BP, may exhibit neurologic signs consistent with hypertensive encephalopathy at pressures of 160/100 mm Hg, whereas chronically hypertensive people rarely develop hypertensive encephalopathy at pressures less than 250/150 mm Hg. 9 Chronic hypertension leads to structural adaptation of the cerebral resistance vessels and an elevation in the BP limits for cerebral autoregulation. 1, 25 Similarly, in cats made acutely hypertensive by the administration of angiotension II, brain edema developed at mean BPs of 170 mm Hg, indicating that the upper limit of cerebral autoregulation had been reached. 7 In contrast, only 11 out of 24 cats with chronic hypertension developed hypertensive encephalopathy despite having a mean systolic BP of 233 mm Hg. 19 The two cats in the present report developed signs of hypertensive encephalopathy at systolic pressures of 194 and 233 mm Hg. This variability in susceptibility to the development of hypertensive encephalopathy is most likely attributable, in part, to elevations in the limits of autoregulation with chronic hypertension in cats. It is important to recognize that normotensive cats that become acutely hypertensive may exhibit hypertensive encephalopathy at lower BPs than do chronically hypertensive cats. Interestingly, the presence of the hyperplastic vascular changes in the cat in this study with sustained hypertension suggests that vascular remodeling, and presumably resetting of autoregulation, can occur within 14 days of the onset of hypertension.
Early reports of seizures in people with chronic kidney disease attributed these neurologic complications to uremic toxins, CNS infections, and steroid administration. 23, 29, 30 However, hypertensive encephalopathy is now believed to be the most frequent cause of seizures in people with severe chronic kidney disease. 23, 41, 42 Evidence from published veterinary clinical reports is consistent with the assertion that hypertensive encephalopathy is an important cause of neurologic disease in cats with chronic kidney disease. Systemic hypertension (especially severe or rapidly developing) accompanied by neurologic signs, in conjunction with the pathologic findings of diffuse brain edema, cerebral arteriolosclerosis, and parenchymal microhemorrhages, is consistent with an etiologic diagnosis of hypertensive encephalopathy.
