Abstract
Glomerular lipidosis (GL) is characterized by dilated glomerular capillary loops containing lipid-laden cells (foam cells). Previously, GL was considered to be an incidental finding because affected dogs were typically not azotemic. However, the International Renal Interest Society staging system for canine chronic kidney disease has increased the awareness of other clinical parameters (eg, proteinuria and hypertension) that should be included in the assessment of renal function. As such, the aim of this study was to determine clinical abnormalities and concurrent renal lesions in dogs with GL. GL was identified in renal biopsies from 46 dogs evaluated by the International Veterinary Renal Pathology Service. GL was the sole diagnosis in 5 of 46 cases (11%), all of which were proteinuric. All 5 dogs had at least 1 additional clinicopathologic abnormality consistent with renal disease, including hypertension (4), azotemia (3), and/or hypoalbuminemia (2). The remaining 41 dogs had GL in combination with other glomerular lesions, the most common being focal segmental glomerulosclerosis (16, 35%), lesions consistent with juvenile nephropathy (8, 17%), and glomerular amyloidosis (5, 11%). Overall, dogs with severe GL were younger than were those with mild GL (P < .001). The percentage of glomeruli affected by GL differed by concurrent diagnoses (P = .034), with the highest percentage of affected glomeruli in dogs with GL alone or those with concurrent juvenile nephropathy. These findings suggest that GL should be a recognized histologic phenotype of glomerular injury associated with clinical renal dysfunction and/or juvenile nephropathies.
Glomerular lipidosis (GL) was first described in dogs using light microscopy (LM) and transmission electron microscopy (TEM). It was determined to be a segmental change in mesangial cells of the glomerular tuft with aggregation of lipid-laden foam cells; this lesion did not appear to affect overlying podocytes. 22 Shortly thereafter, a study using rabbits fed high-cholesterol diets determined that glomerular foam cells were derived from migrant blood monocytes and mesangial cells but not from glomerular endothelium. 18 These foam cells could be observed in capillary lumina or within the mesangium, and the lipid was present as non–membrane-bound cytoplasmic droplets. 18 These morphologic studies did not attribute clinical significance to the foam cells.
Since these early descriptions, only a few isolated case studies and descriptions of GL as a component of other renal diseases have been published. 11,16,23 Macrophages with cytoplasmic lipid have also been described in glomeruli of cats with primary hyperlipoproteinemia. 21 Spontaneous GL was described in a young Syrian hamster that had lipid droplets in mesangial cells as well as lipid emboli within glomerular capillaries, which was also considered to be related to hyperplidemia. 10 Most texts reiterated the previous assumption that GL was an incidental finding and not associated with functional impairment. 21 Similar findings of glomerular foam cell accumulation have been described in cases of canine renal dysplasia and were interpreted as a degenerative, nonspecific lesion secondary to dysplasia in these dogs. 11 Thus, GL is described in pathology textbooks merely as “an occasional incidental finding in dogs.” 9,21 In other species, lipid-laden macrophages in glomeruli have been associated with hyperlipidemia, 3 and we recently described glomerular foam cell accumulation in a hyperlipidemic Miniature Schnauzer and a hypercholesterolemic Shetland Sheepdog. 4,8
The aims of this retrospective study were to use the database of the International Veterinary Renal Pathology Service (IVRPS) to determine the frequency of GL in dogs undergoing renal biopsy, to describe concomitant glomerular lesions in dogs with GL, and to determine whether GL is associated with clinical evidence of renal dysfunction in dogs, including proteinuria, hypoalbuminemia, azotemia, and hypertension.
Materials and Methods
The database of the IVRPS was searched for the terms “lipid” or “foam cell” in the morphologic diagnosis, in the histologic description, or within the “Comments” section of all canine renal biopsy reports from 2005 to 2014. Materials (glass slides and TEM images, when available) from all identified cases were reexamined to confirm the presence of GL. Research materials can be accessed by contacting the author (R. E. Cianciolo).
Routine evaluation of renal biopsy specimens by the IVRPS has been described previously. 2 Briefly, histopathology samples are cut at 3-μm thickness and stained with hematoxylin and eosin, periodic acid–Schiff, Masson’s trichrome, and Jones methenamine silver method. When indicated, specimens were cut at 8 μm and stained with Congo red. TEM specimens were prepared as described previously 2 at Texas Heart Institute (Houston, TX), at Athens Diagnostic Laboratory (Athens, GA), or at the Comparative Pathology & Mouse Phenotyping Shared Resource Main Laboratory (Columbus, OH). Direct immunofluorescence (IF) was also performed as described previously. 2 FITC-conjugated polyclonal goat anti-dog IgG, IgM, IgA, and C3 antibodies (Bethyl Labs, Montgomery, TX), as well as FITC-conjugated rabbit anti-human C1q, kappa light chain, and lambda light chain antibodies (Dako North America, Carpinteria, CA), were used to detect components of immune complexes on frozen tissue sections.
In each histologic sample, glomeruli containing foamy macrophages were counted to determine the percentage affected by GL for that sample. The samples were then scored as follows: mild GL (≤25% of glomeruli affected) and severe GL (>25% of glomeruli affected). Diagnoses of lesions and disease processes followed the guidelines recommended by the World Small Animal Veterinary Association Renal Standardization Study Group. 2
Signalments and clinical histories were evaluated. Clinicopathologic data were collected when available, including serum creatinine, urea, albumin, and cholesterol; urine protein:creatinine ratio (UPC); urine-specific gravity; and systolic arterial blood pressure. Hypoalbuminemia was defined as a serum albumin ≤2.5 g/dL. Criteria for the diagnosis of hypertension were arterial systolic pressure >150 mmHg or treatment with antihypertensive medications such as benazepril, enalapril, and amlodipine. All cases were staged and substaged according to International Renal Interest Society (IRIS) criteria. 6 Clinicians were contacted to obtain follow-up clinical information on treatment and patient progress.
Statistical Analysis
Age is reported as a median (range) and was compared between dogs with mild GL and severe GL using a Wilcoxon rank sum test. The median percentage of GL and interquartile range (IQR) is reported for all samples with GL detected. The median percentage of GL is reported for the renal pathology diagnoses that were identified in >3 dogs. The Kruskal-Wallis test was used to compare the percentages according to the diagnostic category. The statistical analyses were performed using the R software for statistical computing (R Development Core Team, Vienna Austria), and P < .05 was considered significant.
Results
Signalment
Fifty samples from 46 dogs had at least 1 glomerulus with foam cell accumulation either within dilated capillary loops or within mesangial zones. These represented 3.8% of the canine submissions (total N = 1,210) during the study period. The median percentage of glomeruli affected for all samples was 9% (IQR, 4%–32%). In total, 38 samples (76%) were classified as mild GL and 12 (24%) as severe GL. GL was diagnosed as the sole histologic lesion in 5 dogs; these dogs had a median age of 1.5 years (range, 0.7–12.1 years) at the time of renal biopsy and included 2 Golden Retrievers, a Soft Coated Wheaten Terrier, an Alaskan husky, and a Newfoundland. The remaining 41 dogs had GL as well as a concurrent renal histologic diagnosis; these dogs had a median age of 7.9 years (0.7–12.4 years), and the most common breeds were Golden Retriever (4), Labrador Retriever (3), Scottish Terrier (3), and Shetland Sheepdog (3). Dogs with severe GL (affecting >25% of glomeruli in the sample) were significantly younger than were those with mild GL, with median ages of 3.5 years (0.7–7.3 years) and 8.4 years (0.8–12.4 years), respectively (P < .001) (Fig. 1).
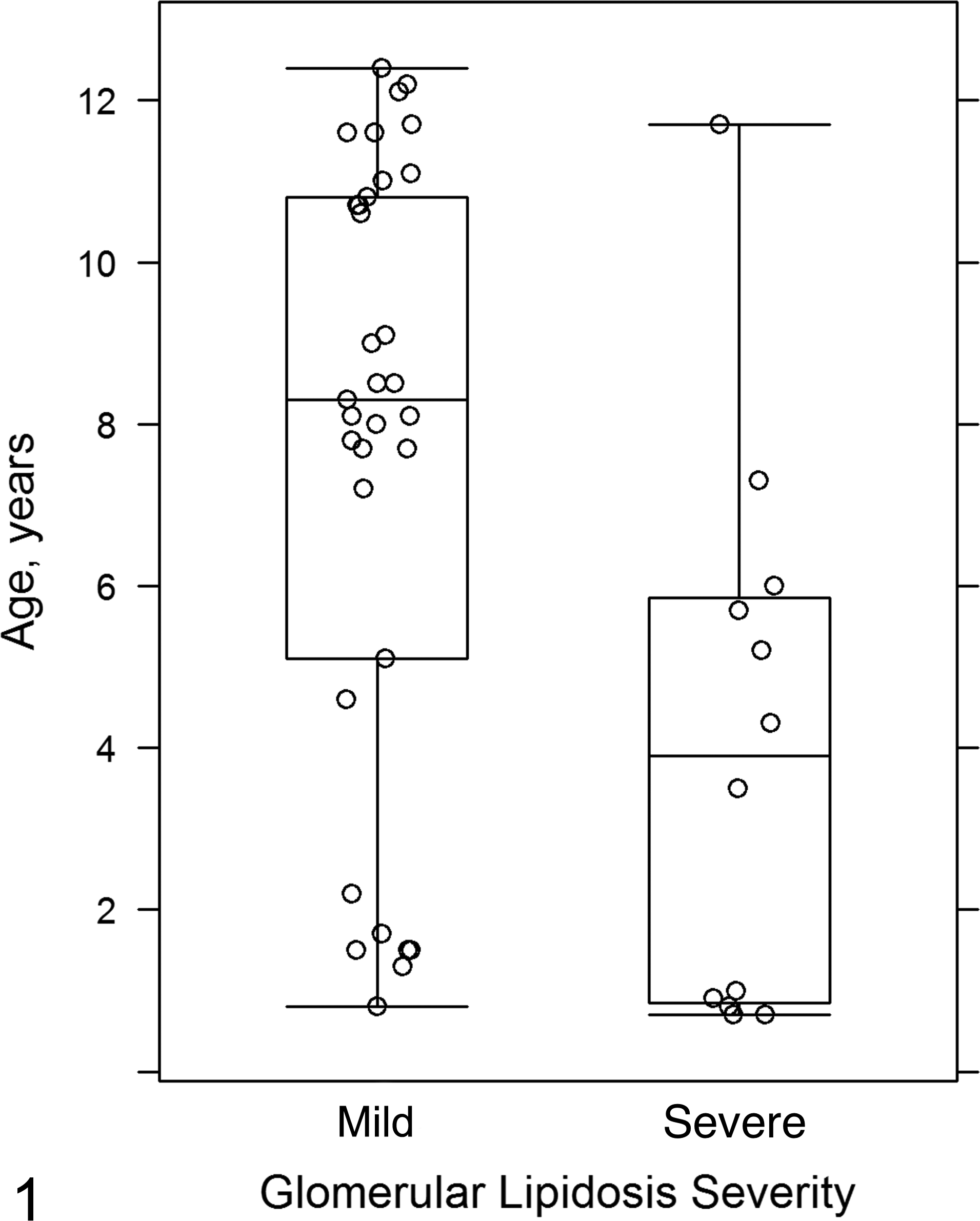
Percentage of glomeruli affected by glomerular lipidosis (glomerular lipidosis severity) compared to the age of dog affected. Box and whisker plot representing interquartile range of data; the horizontal line represents the median and the whisker bars extend to 1.5× the interquartile range. Dots represent individual dogs. A higher percentage of glomeruli is affected in younger dogs than in older dogs (P < .001).
Diagnostic Categories
Of the 46 dogs, 41 had concurrent diagnoses of renal disease in addition to GL. Diagnostic categories for concurrent lesions included both immune complex–mediated processes and non–immune complex–mediated disease. A minority of dogs had concurrent immune complex–mediated glomerulonephritis (ICGN) (n = 5; 11%). One other dog had equivocal evidence of immune deposition, whereas the remaining 40 dogs did not have ultrastructural or IF evidence of immune complex disease. The most common concurrent diagnosis in our cohort was focal segmental glomerulosclerosis (FSGS) (n = 16). Other diagnoses in order of decreasing prevalence included lesions consistent with juvenile nephropathy (n = 8), glomerular amyloidosis (n = 5), abnormalities of the glomerular basement membrane (n = 2), glomerulocystic atrophy (n = 1), glomerular atheroemboli (cholesterol and lipid within glomerular capillaries, n = 1), acute tubular necrosis (n = 1), and collagenofibrotic glomerulonephropathy (n = 1).
The percentage of glomeruli affected by GL varied significantly by diagnosis (P = .034) (Fig. 2), with the highest median percentage identified in those dogs with only GL (50% [range, 3%–100%]) and in dogs with GL and concurrent juvenile nephropathy (15% [range, 1%–100%]). In 4 dogs, all of the sampled glomeruli had evidence of foam cell accumulation. The degree to which a glomerular tuft was distorted by foam cells varied from segmental accumulation (Fig. 3) to global effacement by foam cells (Fig. 4).
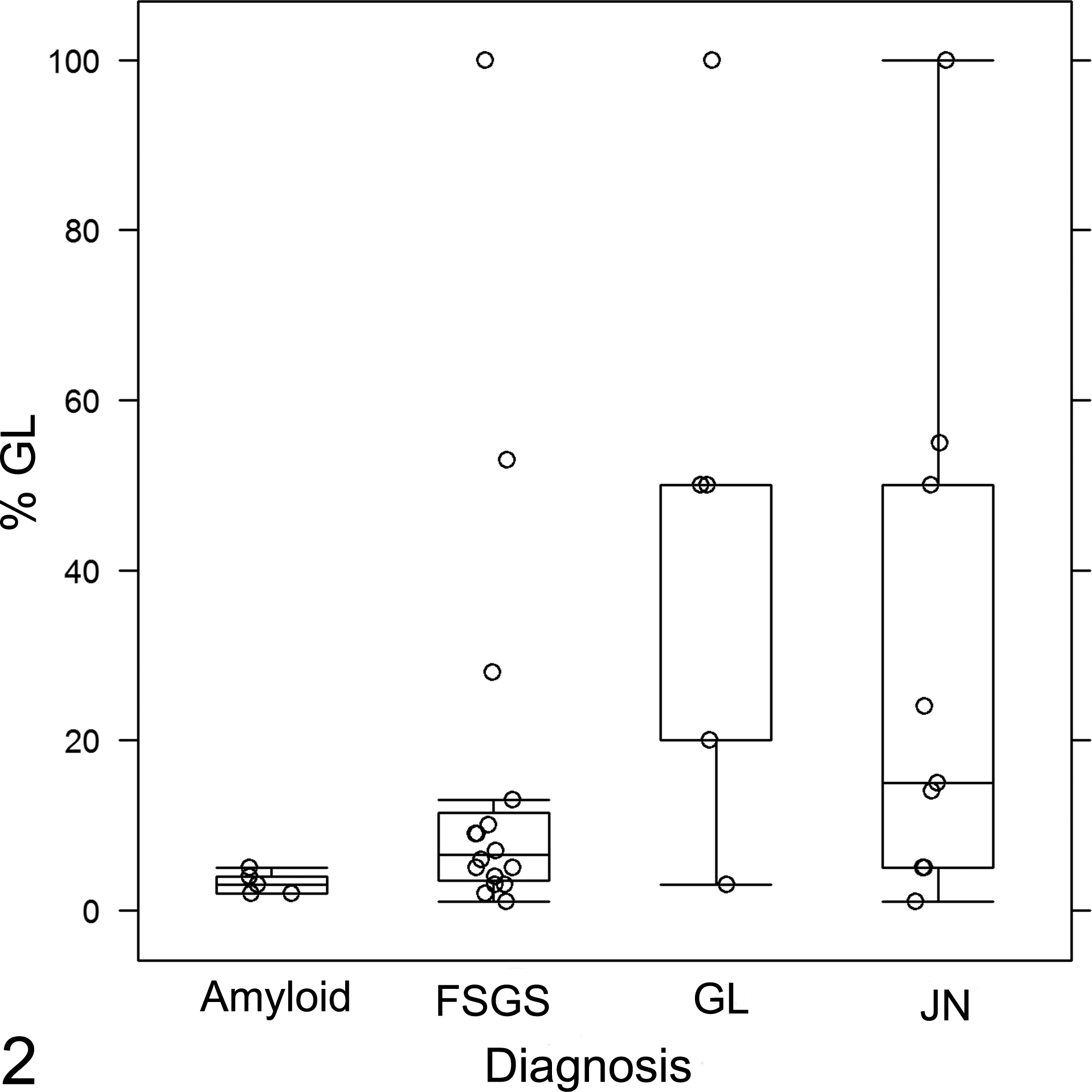
Percentage of glomeruli affected by glomerular lipidosis (%GL) in each major diagnostic category. Box and whisker plot representing the median and interquartile range. Dots represent individual dogs, and the dots that fall outside of the whisker bars represent outliers. The median %GL is highest for dogs with juvenile nephropathy (JN) or glomerular lipidosis as the only lesion (GL), compared to dogs with amyloidosis or focal segmental glomerulosclerosis (FSGS) (P = .034).
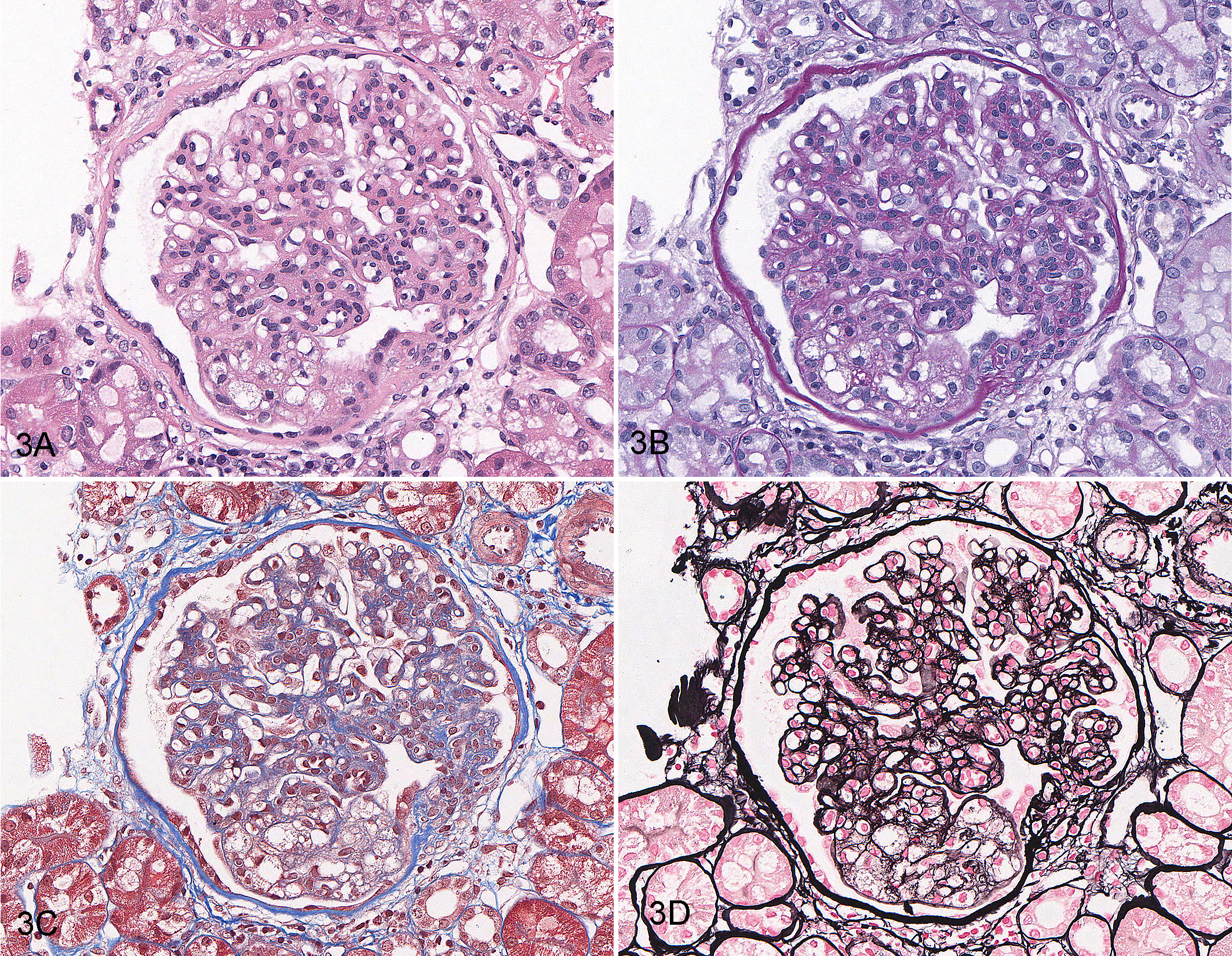
A–D. Mild segmental glomerular foam cell accumulation, glomerulus, dog. Glomerular capillary loops in a single segment are dilated and contain lipid-laden cells. The same glomerulus is shown in each figure. (A) Hematoxylin and eosin (HE). (B) Periodic acid—Schiff. (C) Masson’s trichrome. (D) Jones methenamine silver method.
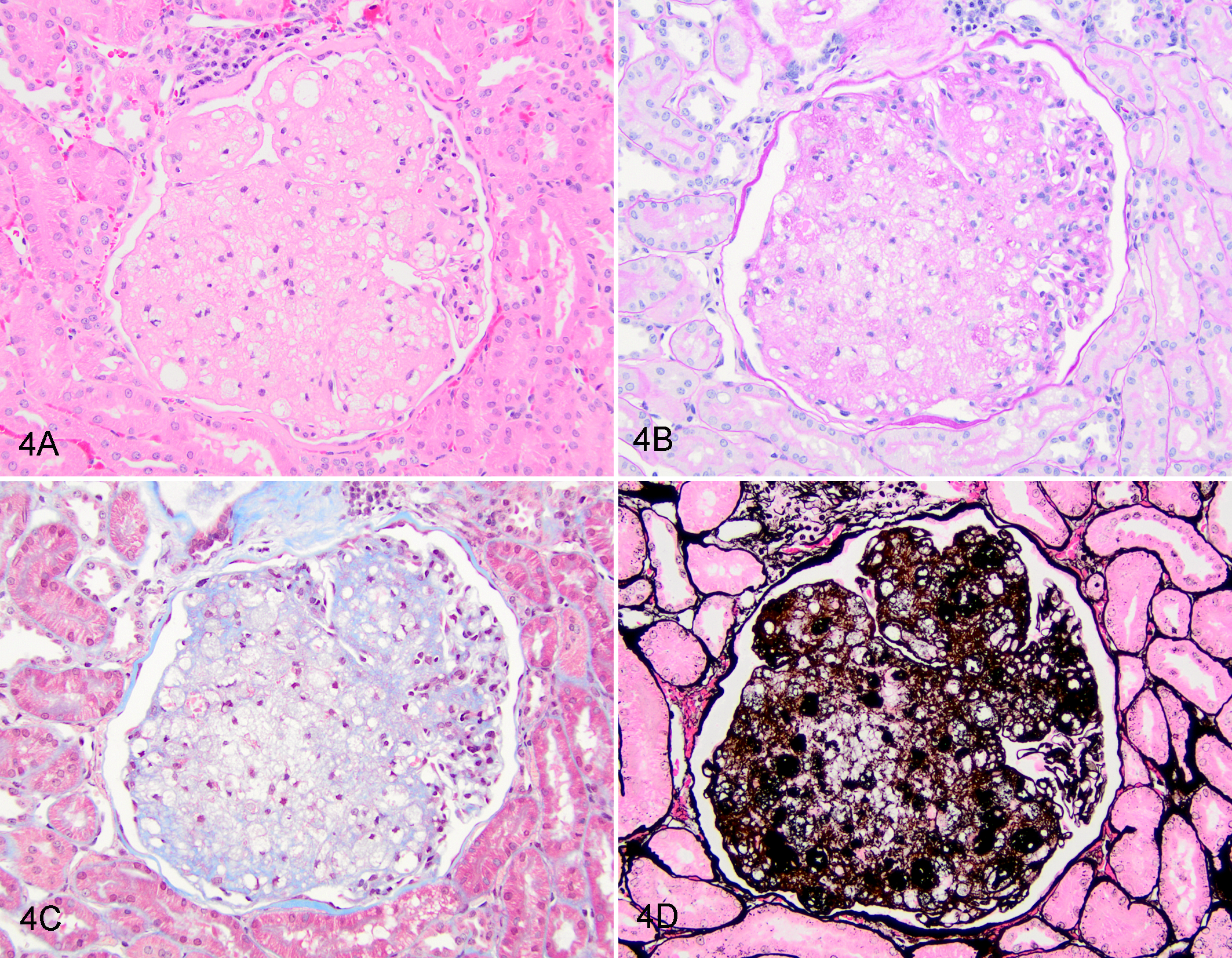
A–D. Severe global glomerular foam cell accumulation, glomerulus, dog. Nearly all glomerular capillary loops of the tuft are dilated and contain lipid-laden cells. The same glomerulus is shown in each figure. (A) Hematoxylin and eosin (HE). (B) Periodic acid–Schiff. (C) Masson’s trichrome. (D) Jones methenamine silver method.
IF and Electron Microscopy
Definitive immune complexes (based on IF and TEM results) were identified in 5 dogs, whereas 1 additional dog had a few scattered small electron–dense deposits in mesangial zones in addition to equivocal-to-weak IF labeling. Foam cells, when present in the IF sample, demonstrated auto-fluorescence. Samples were determined to have positive IF labeling only if there was granular labeling of capillary walls and/or mesangial zones.
Ultrastructurally, glomeruli with GL had osmophilic globular material consistent with lipid in the cytoplasm of mesangial cells and in intravascular foam cells. In 2 cases, GL was present only in the TEM sample and was not identified in the LM specimen.
Clinicopathologic Findings
Of the 46 dogs, 45 had sufficient clinical data for IRIS staging of chronic kidney disease with substaging by proteinuria. Of the 5 dogs that were diagnosed with GL only, 2 had IRIS stage 1 subclinical disease and 1 each had IRIS stages 2 to 4. The dog with IRIS stage 2 disease had polydipsia and incontinence, but clinical signs were not reported for the dogs with IRIS stages 3 and 4 disease. Peak UPC in this group ranged from 4.0 to 18.8 (mean, 7.4). Of these 5 dogs with only GL, 2 were hypoalbuminemic, 3 were hypercholesterolemic, and 4 were hypertensive. Two cases had mild GL, despite GL being the only renal lesion, and 3 had severe GL.
Of the 40 dogs with GL and a concurrent renal histologic diagnosis and sufficient clinical data for staging, 1 dog (3%) was staged as at risk, or not azotemic with no signs of chronic kidney disease, and 20 dogs (50%) were IRIS stage 1. Of the 21 dogs staged as at risk or IRIS stage 1, 15 (71%) were reported to have subclinical proteinuria, meaning that they were being clinically evaluated for reasons other than renal disease (Supplemental Table S1). Seven dogs (18%) were classified as IRIS stage 2, 9 (23%) as IRIS stage 3, and 3 (8%) as IRIS stage 4.
A UPC was provided for 36 of the 40 dogs with GL and concurrent renal disease; all were proteinuric (UPC > 0.5) with a median UPC of 5.4, including 33 (92%) with a UPC > 2. Of the 37 dogs for which serum albumin was reported, 14 (38%) were hypoalbuminemic. Cholesterol levels were reported for 11 dogs, 9 (82%) of which were hypercholesterolemic, ranging from 248 to 777 mg/dL. Information on systolic blood pressure was available in 32 dogs; 23 (72%) were hypertensive.
Follow-up Data
For 5 dogs, repeat specimens were submitted for follow-up evaluation. Though all of these cases represent animals with histologic evidence of progressive disease, only 1 dog had GL as the sole diagnosis in the original tissue sample. Case 24, a 1-year-old spayed female Golden Retriever, presented with subclinical IRIS stage 2 disease and a UPC of 3.1. The initial renal biopsy revealed severe GL as the sole diagnosis, involving all glomeruli in the sample. There was also mild remodeling of the glomerular basement membrane. The clinical disease progressed, and 6 months later the dog was euthanized. The autopsy renal specimens had segmental glomerulosclerosis affecting 80% of glomeruli, with 20% of glomeruli affected by global glomerulosclerosis and <10% of glomeruli with GL. In this case, GL progressed rapidly to end-stage kidney disease. The other 4 dogs had GL with concurrent lesions of FSGS or juvenile nephropathy on initial biopsy, and both disease processes were suspected to have played a role in the progression of disease. Of the 9 dogs for which follow-up clinical pathologic data were available, 4 demonstrated worsening azotemia and 3 remained moderately proteinuric, with follow-up intervals ranging from 1 month to 2.5 years (Supplemental Table S2).
Discussion
GL is characterized by accumulation of foam cells within dilated capillary loops of the glomerular tuft. There are limited descriptions of foam cells in veterinary diseases. In this study, we sought to characterize the presence of GL in canine glomerular disease. We identified 46 dogs that had GL either as the sole lesion or concurrent with other disease processes. The majority of our cohort were IRIS stage 1, proteinuric, and hypertensive dogs. In these animals, FSGS was a predominant lesion, occurring in 35% of dogs. Follow-up clinical data demonstrated progression of renal disease in 4 of 9 dogs; however, almost all dogs for which follow-up data were collected had concurrent glomerular disease (GL with FSGS). Whether the presence of GL led to a more rapid progression of the concurrent renal disease is difficult to determine, especially since therapeutic interventions and time frame of follow-up data varied widely.
The pathogenesis of glomerular foam cell accumulation remains unknown and is likely multifactorial. In our cohort, GL was noted as a concurrent lesion with several types of glomerular disease. While it was most often identified in the context of FSGS, GL in this group was typically mild. The most severe GL lesions were noted in dogs with GL as the sole diagnosis and in dogs with concurrent juvenile nephropathy.
Overall, concurrent immune complex disease was uncommon in our GL cohort. This was surprising given the relative frequency of ICGN in dogs that have been biopsied for investigation of proteinuria. Approximately 48% of North American dogs biopsied for proteinuria have definitive evidence of immune complex deposition, whereas only 13% of dogs with GL had immune deposits. 13 Although the criteria for inclusion differed between the 2 studies, all dogs in the GL cohort with a reported UPC were proteinuric. The presence of GL in a biopsy sample does not rule out the possibility of ICGN, but it is more likely indicative of glomerular damage that was not driven by immune complex deposition.
Notably, glomerular foam cell accumulation differs from accumulation of foam cells in the renal interstitium, which can be observed in various human renal diseases, including Alport syndrome, membranous nephropathy, IgA nephropathy, diabetic nephropathy, and FSGS. 3,20 In humans, segmental glomerulosclerosis was more commonly noted in cases with interstitial foam cells, with the supposition that the foam cells are secondary to long-standing high levels of proteinuria. 20
The mechanistic link between glomerular foam cell accumulation and progressive glomerular disease has not been thoroughly investigated. One study of renal interstitial foam cell accumulation suggests these cells contribute to progressive glomerular injury and are associated with a higher prevalence of concurrent FSGS in people. 20 Murine models of hyperlipidemic renal injury develop progressive glomerular foam cell accumulation; however, whether or not this progresses to end-stage kidney disease is unknown. 14 A recent study utilized a model of hyperlipidemic mice crossed with the NEP25 model of podocyte depletion induced by LMB (an immunotoxin with specificity to CD25). These mice develop significant podocyte loss preferentially in glomeruli affected by foam cell accumulation. 5 While the mechanism is unknown, it was theorized that initial podocyte injury leads to disordered lipid metabolism, with endothelial and mesangial production of pro-inflammatory cytokines that recruit macrophages and foam cells to the site. 5 Monocytes in glomerular tufts have been demonstrated to contribute to focal segmental endocapillary proliferation, a lesion that can precede FSGS in some cases. 12 Interestingly, 1 young dog in our cohort had an initial diagnosis of severe GL that progressed to FSGS with mild GL, suggesting that GL preceded the podocyte injury and segmental sclerosis. It is possible that the relationship between GL and podocyte injury is bidirectional.
Data from human disease also suggest that foam cell accumulation may be a result of dyslipidemia. 1,3,17,19 For example, in humans, the lesion has been seen with immune-mediated acquired lecithin-cholesterol acyltransferase (LCAT) deficiency and glomerulopathy due to homozygous mutation in apolipoprotein E. 7,15 The ultrastructural appearance of lipid in LCAT deficiency is a distinct lesion in humans, but similar lesions were not observed in these cases in dogs.
The link between lipid or cholesterol disorders and foam cell accumulation is not well understood in dogs. We had limited data regarding cholesterol levels for the dogs in our cohort. However, 2 dogs (cases 35 and 37) developed hypercholesterolemia during the course of their renal disease. In addition, case 2 was hypercholesterolemic at the time of initial biopsy and progressed from rare glomeruli with GL to having 33% of sampled glomeruli affected by GL on the repeat specimen 3 years later. Unfortunately, follow-up cholesterol levels for this dog were not available. Our cohort included 3 Shetland Sheepdogs and 2 Miniature Schnauzer dogs, breeds known to be predisposed to dyslipidemia. 4 The serum cholesterol concentration was reported only for 1 of these 5 dogs, and it was hypercholesterolemic. This particular dog also had glomerular atheroemboli noted on the biopsy. 8 In a series of Miniature Schnauzer dogs, hyperlipidemia was associated with the presence of glomerular foam cells (observed with TEM), but glomerular lipid thromboemboli were more common than were dilated loops filled with foam cells. 5 Further studies are required to determine the association between hyperlipidemia and glomerular foam cell accumulation in dogs.
The main limitation of this study is the incomplete follow-up data for the majority of our cohort. The renal biopsy service predominantly interacts with referral centers, and many dogs are lost to follow-up. However, the available follow-up data support our conclusions that GL can occur in dogs with proteinuric glomerular disease, mild GL often occurs in the context of FSGS, and GL is present in cases that demonstrate clinical or histologic evidence of progressive renal disease. GL is also noted in early stages of renal disease and in young dogs, may be severe in juvenile nephropathy cases, and should not be ignored as an incidental lesion in this context, even as a singular lesion.
Footnotes
Acknowledgements
We would like to thank Alan Flechtner and Anne Saulsbery (Comparative Pathology & Mouse Phenotyping Shared Resource Main Laboratory, The Ohio State University, Columbus, OH), Dr Fred J. Clubb Jr (Department of Veterinary Pathobiology, Texas A&M University, College Station, TX), Ralph Nichols (Texas Heart Institute, Houston, TX), and Mary Sanders (Department of Small Animal Clinical Sciences, Texas A&M University, College Station, TX) for their expert technical assistance, without which this project could not have been completed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for E. Furrow was provided by the Office of the Director, National Institute of Health (NIH), under award No. K01OD019912. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
