Abstract
Porcine epidemic diarrhea virus (PEDV) infection leads to diarrhea and subsequently to decreased feed efficiency and growth in weaned pigs. Given that few studies have addressed the host-virus interaction in vivo, this study focused on endoplasmic reticulum (ER) stress and unfolded protein response (UPR) in jejunal epithelial cells during PEDV infection. Eight-week-old pigs (n = 64) were orally inoculated with PEDV IN19338 strain (n = 40) or sham-inoculated (n = 24) and analyzed for PEDV viral RNA shedding using reverse transcription–quantitative polymerase chain reaction and for viral antigen within enterocytes using immunohistochemistry (IHC). ER stress was analyzed in a subset of 9 PEDV-inoculated pigs with diarrhea, detectable viral RNA, and viral antigen (PEDV-immunopositive pigs). Compared with control pigs, PEDV-immunopositive pigs had a reduced ratio of villus height to crypt depth in the jejunum (P = .002, n = 9 per group), consistent with intestinal injury. The protein levels of ATF6, IRE1, PERK, XBP1u, ATF4, GRP78, and caspase-3 were assessed in jejunal epithelial cells at the villus tips via IHC. Both ER stress and UPR were demonstrated in PEDV-immunopositive pigs by the increased expression of ATF6 (P = .047), IRE1 (P = .007), and ATF4 (P = .001). The expression of GRP78 (P = .024) and caspase-3 (P = .004) were also increased, indicating an accompanying increase in ER protein folding capacity and apoptosis. Overall, these results reveal that PEDV infection induces ER stress and UPR in intestinal epithelial cells of weaned pigs.
Keywords
The endoplasmic reticulum (ER) is the primary site of protein synthesis and folding in eukaryotic cells. Regardless of physiological or pathological factors, disrupted ER homeostasis leads to ER stress, which is an accumulation of unfolded proteins in the ER lumen and simultaneously invokes the unfolded protein response (UPR). The UPR is a series of signal transduction pathways that may eliminate the deleterious effects of ER stress, including lipid accumulation, cell death, and inflammation. 39 UPR signaling pathways are mediated by 3 protein sensors on the ER membrane, including activating transcription factor 6 (ATF6), inositol-requiring transmembrane kinase/endonuclease 1 (IRE1), and pancreatic ER eIF2α kinase (PERK). 39 Under ER stress, the ER chaperone glucose-regulated protein 78 (GRP78, also known as BiP) dissociates from the intraluminal domains of the 3 protein sensors, thereby activating UPR and downstream signaling. 4,40 The released ATF6 fragment translocates to the nucleus to stimulate UPR gene transcription and the increased abundance of X-box binding protein 1 (XBP1) and GRP78. 1,52 Thereafter, activated IRE1 splices XBP1 mRNA and generates a functionally active isoform of XBP1 (XBP1s), a transcription factor for most UPR target genes. 51,52 Unspliced XBP1 mRNA encodes the unspliced form of XBP1 (XBP1u), which functions as a UPR inhibitor. 53 IRE1 is also required for autophagy activation, a mechanism to remove unnecessary or dysfunctional components in cells. 32 Activated PERK phosphorylates the α subunit of eukaryotic translation initiation factor 2 (EIF2α), which enhances the translation of ATF4. 41 ATF4 is a transcription factor that regulates the expression of genes implicated in protein folding and oxidative stress response. 27 Taken together, via the signal transduction pathways of ATF6, IRE1, and PERK, the UPR facilitates adaptation by either increasing ER protein folding capacity or activating ER-associated degradation (ERAD). 35 Also, in cells under irremediable ER stress, the UPR is able to trigger apoptosis. 27 –29
Porcine epidemic diarrhea virus (PEDV) is an enveloped single-stranded RNA virus of the Coronaviridae family that infects pigs at all ages via fecal-oral transmission and replicates in small intestinal epithelial cells. 12,13,42 Viral infection in the small intestine contributes to degeneration and death of intestinal epithelial cells, leading to a decrease in the ratio of villus height to crypt depth (VH:CD) and later villous atrophy. 13 During replication, the virus induces the formation of double-membrane vesicles (DMVs), which are directly derived from the ER, leading to morphologic and functional changes in the ER. 55 The depletion of ER membrane due to viral assembly and budding also contributes to coronavirus-induced ER stress. 14 In Vero cells, PEDV has been shown to induce ER stress and UPR via the modulation of PERK and ATF6 pathways. 49,50,58 Additionally, autophagy and apoptosis are observed in PEDV-infected cells, and apoptosis plays a vital role in PEDV pathogenesis. 11,16,20,22,58 These findings suggest that ER stress and UPR might constitute a significant aspect of the observed PEDV-host interaction.
Although in vitro studies reveal that PEDV-infection results in ER stress and UPR, these changes have not been thoroughly examined in vivo. Given that we have previously reported that epithelial-mesenchymal transition (EMT) in the small intestine of PEDV-infected nursery pigs is a consequence of chronic ER stress, 8 the present study aimed to characterize ER stress and UPR in jejunal enterocytes of weaned pigs that are immunopositive for PEDV antigen. The a priori hypothesis was that PEDV infection induces ER stress and initiates UPR via either ATF6, IRE1, or PERK pathways and subsequently increases ER protein folding capacity and apoptosis.
Materials and Methods
Animals and Experimental Design
All experimental protocols were approved by the Institutional Animal Care and Use Committee at Iowa State University (IACUC# 8-17-8580-S). A total of 64 eight-week-old, commercial, cross-bred gilts were selected from a PEDV-negative herd. Animals were randomly allotted to either PEDV-challenged (n = 40) or sham-inoculated (control; n = 24) treatment groups. Each treatment was housed separately across multiple rooms in a BSL2 facility with strict biosecurity and biocontainment. Animals were given free access to water and ad libitum fed a diet that met or exceeded NRC (2012) requirements for this size of pig throughout the study period. 30 Pigs were allowed to acclimatize to the facility for 4 days before inoculation. On day 0, pigs in the PEDV-challenge group were orally inoculated with 10 mL of 103 TCID50/mL PEDV isolate (USA/IN19338/2013) as previously described, 6,46 while control pigs received 10 mL of sterile medium. At each time point (ie, 2, 4, 6, and 10 days post inoculation [DPI]), 6 control pigs and 10 PEDV-inoculated pigs were randomly chosen and euthanized for analysis of PEDV load by reverse transcription–quantitative polymerase chain reaction (RT-qPCR), histopathology, and analysis of PEDV antigen by immunohistochemistry (IHC). To evaluate the relationship between PEDV infection and ER stress, a subset of 9 PEDV-inoculated pigs was selected for further evaluation of ER stress (Fig. 1). These pigs had diarrhea and detectable viral RNA and viral antigen and are referred to as “PEDV-immunopositive pigs” hereafter. PEDV-immunopositive pigs were from DPI 4 (n = 3), 6 (n = 4), and 10 (n = 2). For comparison, equal numbers of control pigs from DPI 4 (n = 3), 6 (n = 4), and 10 (n = 2) were selected.
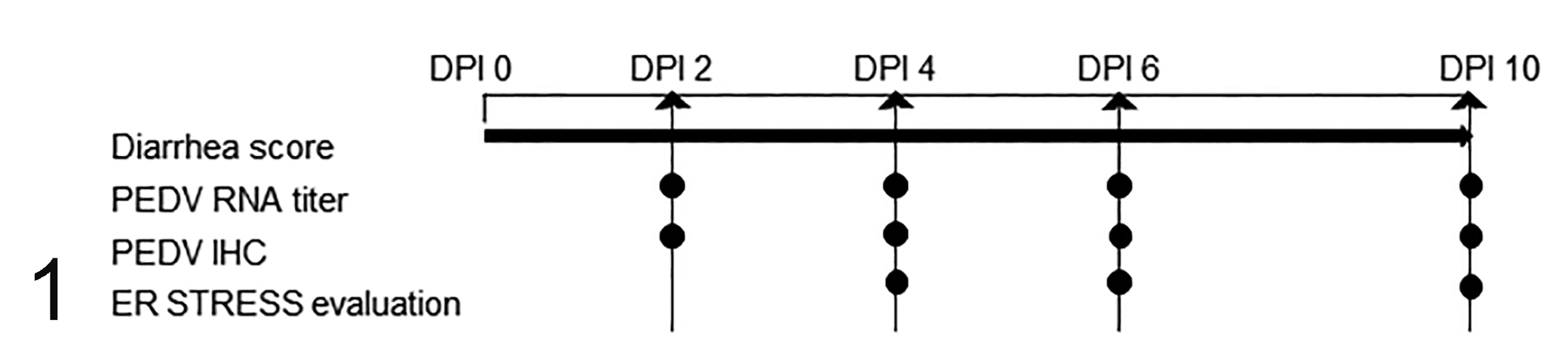
Experimental design. Pigs were assigned to porcine epidemic diarrhea virus (PEDV)-inoculated (n = 40) and control (n = 24) groups and evaluated for daily diarrhea score. At 2, 4, 6, and 10 days post inoculation (DPI), 10 PEDV-inoculated pigs and 6 control pigs were euthanized and analyzed for PEDV RNA titer and PEDV antigen via IHC. Based on the result of IHC, a total of 9 PEDV-immunopositive pigs from DPI 4 (n = 3), 6 (n = 4), and 10 (n = 2) and an equal number of control pigs were chosen for further evaluation of ER stress.
Clinical Evaluation, Fecal PEDV RNA Shedding, and Histopathology
Changes in fecal consistency in both treatments were evaluated daily throughout the study period by the same individual. Fecal consistency scores were assigned using a 4-point scale, as follows: 0 = normal feces; 1 = soft feces; 2 = loose and shapeless diarrhea; 3 = watery diarrhea.
Rectal swabs were collected before euthanasia at dpi 2, 4, 6, and 10 from pigs and submitted to the Iowa State University Veterinary Diagnostic Laboratory (Ames, IA) for RT-qPCR of PEDV in feces using a routine assay targeting the N gene. 9,10 A cycle threshold (Ct) of ≥36 was considered negative and <36 considered positive for PEDV.
Necropsy was performed immediately following euthanasia. Jejunum (∼1.5 m distal to the stomach) was collected from each pig and placed in 10% neutral buffered formalin for no more than 48 hours. Fixed tissues were routinely processed, embedded in paraffin, and sections of 4-µm thickness were stained with hematoxylin and eosin (HE) at the Iowa State University Veterinary Diagnostic Laboratory (Ames, IA).
Immunohistochemistry
Assessment of PEDV antigen was performed via IHC at the Iowa State University Veterinary Diagnostic Laboratory (Ames, IA) using a PEDV-specific mAb (BioNote). 6,26 The positive control was PEDV-infected pigs from clinical cases, which were known as PEDV positive by PCR, while the negative control was IHC with omission of the primary antibody.
To evaluate the relationship between PEDV infection and ER stress, PEDV-immunopositive pigs in PEDV treatment were selected and IHC of ER stress and UPR marker proteins were evaluated. The primary antibodies raised against ATF6, IRE1, PERK, XBP1u, ATF4, GRP78, and caspase-3 are listed in Table 1, and these antibodies are valid for porcine tissues. 19,24,33,37,38,57 GRP78, as an ER protein chaperone, is an indicator of ER stress and increased ER protein folding capacity, 17,48 whereas caspase-3 as the ultimate executioner is an indicator of apoptosis. 36 Sections of jejunum were cut and placed onto positively charged slides, deparaffinized, and rehydrated in a series of ethanol dilutions. Antigens were retrieved by 20 minutes of steam treatment in an autoclave. After 15 minutes of cooling, slides were incubated in 3% hydrogen peroxide in methanol for 20 minutes to suppress endogenous peroxide. Slides were blocked with 10% normal goat serum for 1 hour and then incubated with the primary antibody at the dilutions listed in Table 1. The secondary antibody, peroxidase-conjugated goat anti-rabbit IgG (ThermoFisher Scientific), was applied for 1 hour. Slides were then incubated in metal-enhanced 3,3′-diaminobenzidine (DAB) substrate (ThermoFisher Scientific) for 30 minutes. Later, slides were counterstained with hematoxylin for 10 minutes followed by a 2-minute Scott’s tap water bath. Positive controls were porcine tissues containing the target molecules in their known locations described in publications, while IHC staining with the primary antibody omitted was applied as the negative control.
Antibody source, dilution, and antigen retrieval for immunohistochemistry.
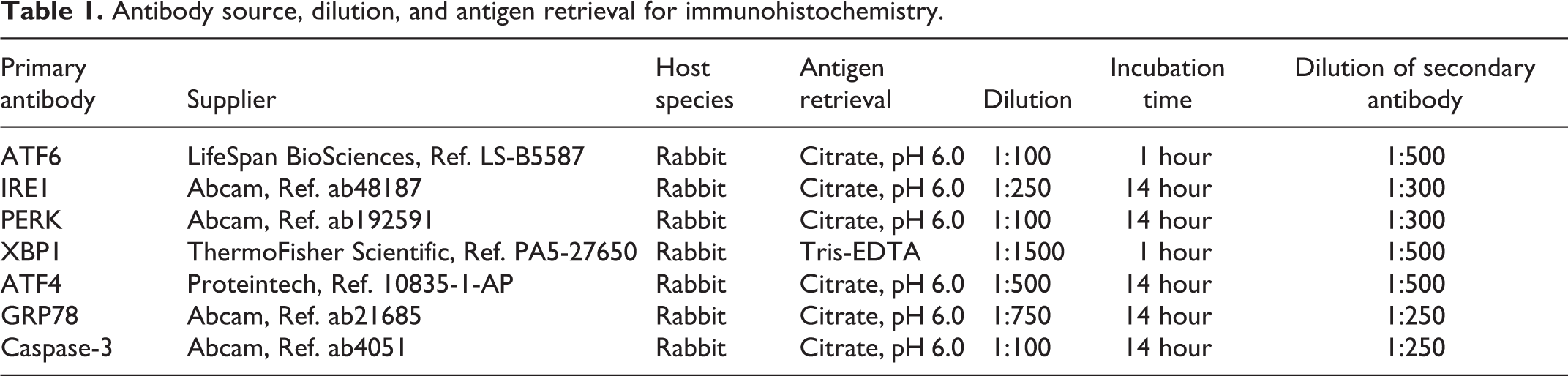
Image Analysis
All HE and IHC sections were examined and images captured using an Olympus BX40 microscope (Olympus Optical) equipped with an Olympus DP26 camera and CellSens Standard software (Olympus Optical).
To assess the effect of PEDV on jejunal morphology, villus height and crypt depth were measured blindly without knowledge of infection status or time point. The average measurement of 10 naturally oriented villus and crypt units was presented as VH:CD ratio per pig. PEDV antigen-positive enterocytes were estimated and assigned scores as follows: 0 = negative; 1 = 1% to 10% of cells immunopositive; 2 = 11% to 50% of cells immunopositive; 3 = more than 50% of cells immunopositive.
To evaluate PEDV-related ER stress, IHC of ATF6, IRE1, PERK, XBP1u, ATF4, GRP78, and caspase-3 was semiquantitatively estimated via the Area Quantification module v1.0 within the HALO image analysis platform (v2.0.1145.19, Indica Labs). Given that PEDV mainly infects the enterocytes on villus tips in jejunum, enterocytes on the villus tips were manually selected for evidence of ER stress. At least 10 villi, well-orientated and lined by a single layer of epithelial cells, were selected (Suppl. Fig. 1). Results for each antibody are presented as mean immunopositive area (μm2) per micrometer length of the epithelium per pig.
Statistical Analysis
All data were analyzed using JMP software 15 (SAS Inst Inc), and each pig was considered an experimental unit. Nonparametric statistical tests, including the Wilcoxon rank sum test and Spearman’s rank-ordered correlation, were performed on the IHC results. A P value ≤.05 was considered statistically significant. The data analyzed in this study are available as Supplemental Materials.
Results
Clinical Assessment, Gross Pathology, and Fecal PEDV RNA Shedding
During the entire experiment, control pigs did not develop clinical signs of porcine epidemic diarrhea. From DPI 1 to 10, PEDV-inoculated pigs exhibited varying degrees of diarrhea (Fig. 2). Watery diarrhea was recorded predominantly from DPI 6 to 10 (Suppl. Table S1). At necropsy, PEDV-inoculated pigs had minimal to marked fecal staining in the perineal region, flaccid and thin-walled intestines, and watery intestinal contents. All control pigs appeared grossly normal.
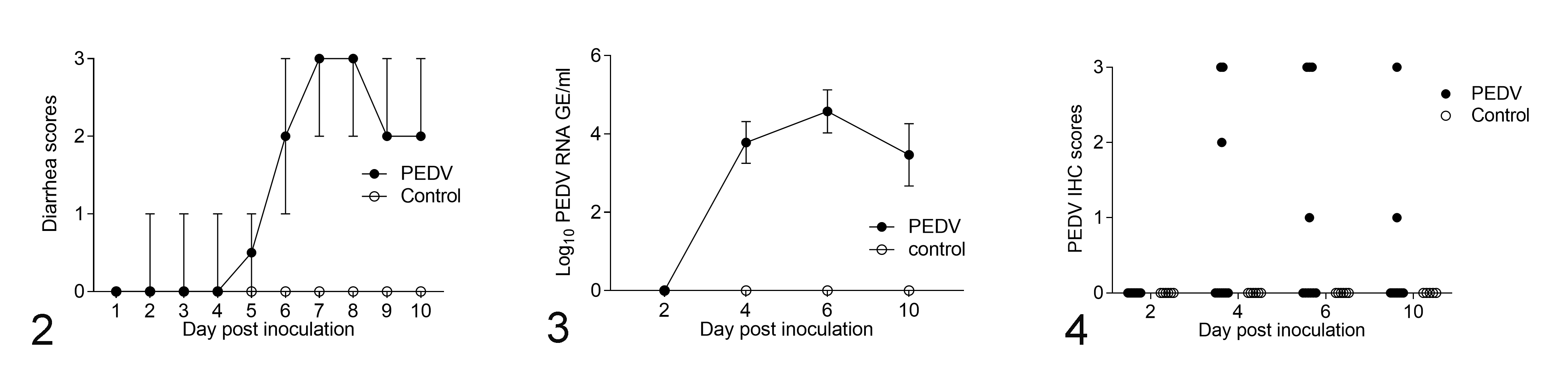
Changes over time in weaned pigs inoculated with porcine epidemic diarrhea virus (PEDV; n = 40) and control pigs (n = 24).
Using RT-qPCR, the controls had no detectable PEDV RNA in feces, nor did the PEDV-inoculated pigs at DPI 2. The PEDV-inoculated pigs at DPI 4, 6, and 10 exhibited low to moderate viral RNA shedding ranging from 2.3 to 7.1 log10 GE/mL (Suppl. Table S2). Fecal PEDV RNA shedding peaked at DPI 6, when the mean PEDV RNA titer was 4.6 log10 GE/mL (Fig. 3).
PEDV IHC and Histopathology
All jejunum sections from control pigs were negative for PEDV antigen via IHC. However, PEDV antigen was detected in the jejunum sections of PEDV-inoculated pigs from DPI 4 to 10 (Fig. 4). At DPI 2, none of PEDV-inoculated pigs were PEDV-immunopositive. Within the PEDV-inoculated pigs, 3/10 pigs at DPI 4, 4/10 pigs at DPI 6, and 2/10 pigs at DPI 10 were positive for PEDV antigen.
Histologically, in comparison with the controls (Fig. 5a), jejunum in PEDV-immunopositive pigs showed villous atrophy, villous fusion, and multifocal attenuation in which the degenerate epithelial cells were hypereosinophilic and vacuolated with a basally located nucleus (Fig. 6a). No PEDV antigen was detected within enterocytes of the controls (Fig. 5b). In PEDV-inoculated pigs, PEDV antigen was discontinuously located within epithelial cells of villi, and the morphologically degenerate epithelial cells were often negative for PEDV antigen (Fig. 6b).
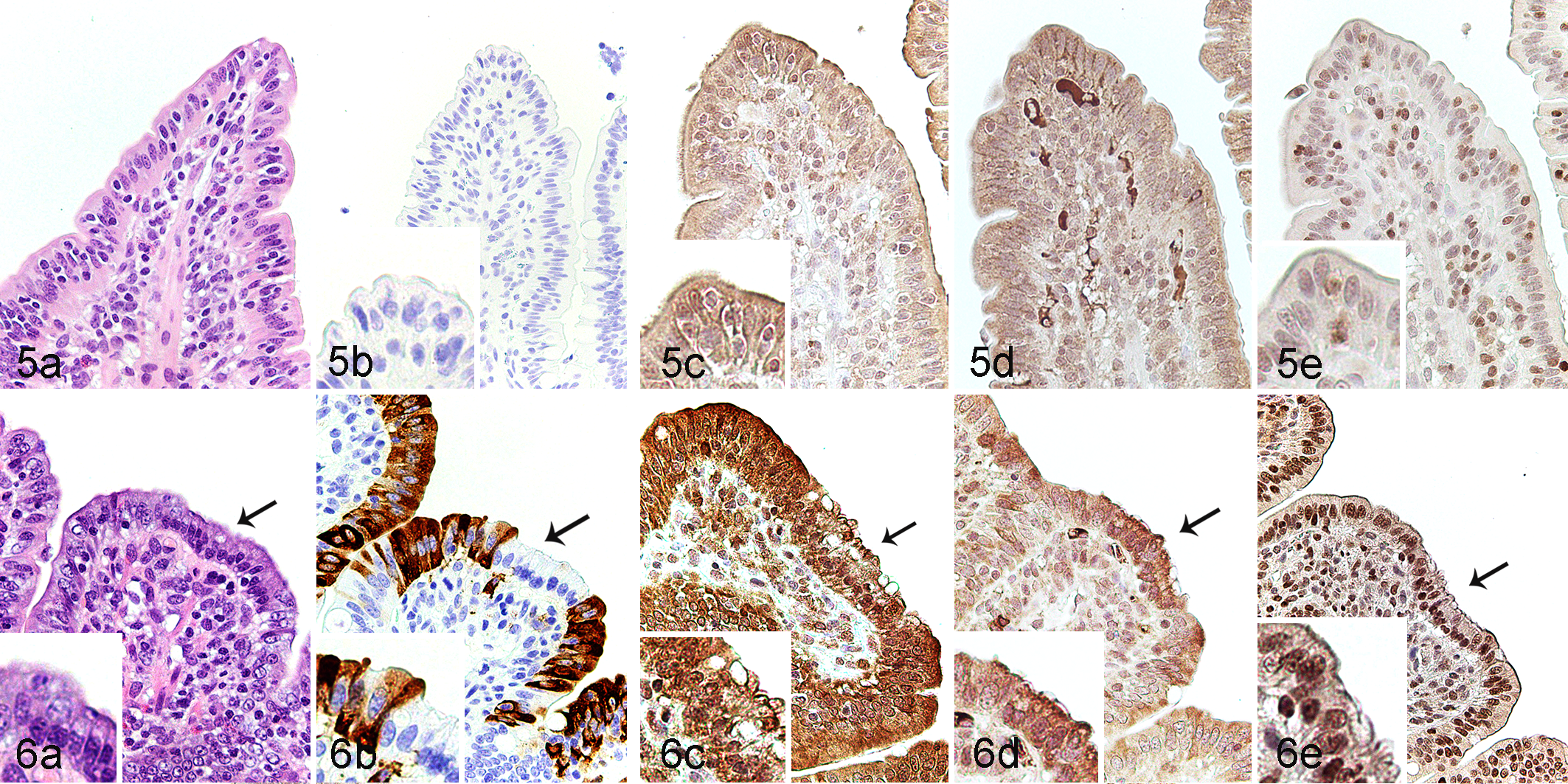
Porcine epidemic diarrhea virus (PEDV) infection, jejunum, pigs.
ER Stress, UPR, and Apoptosis in Jejunal Epithelial Cells
To test the hypothesis that PEDV infection results in ER stress and activation of the UPR in villus epithelial cells, ATF6, IRE1, and PERK stress sensor pathway markers and related downstream signals (XBP1u, ATF4, GRP78, and caspase-3) were assessed via IHC. In the controls, ATF6 and PERK were diffusely expressed in the cytoplasm of jejunal epithelial cells (Fig. 5c and d), while IRE1 was expressed in both the nuclear membrane and cytoplasm (Fig. 5e). On the other hand, in the PEDV-immunopositive pigs, PERK was diffusely expressed in the cytoplasm, while ATF6 and IRE1 were expressed in both the nuclear membrane and cytoplasm of jejunal epithelial cells (Fig. 6a–e). The morphologically degenerate epithelial cells had a weak signal expression of ATF6, but a strong expression of PERK.
UPR’s downstream signal factors and apoptosis were then evaluated through the expression of ATF4, XBP1u, GRP78, and caspase-3 in the jejunal epithelial cells via IHC. In the controls, ATF4 was expressed in both the nuclear membrane and cytoplasm of epithelial cells (Fig. 7a), while XBP1u, GRP78, and caspase-3 were expressed in the cytoplasm (Fig. 7b–d). GRP78 was particularly noted in the apical cytoplasm. In PEDV-immunopositive pigs, ATF4 and caspase-3 were expressed in both the nuclear membrane and cytoplasm (Fig. 8a and d), while XBP1u and GRP78 were diffusely expressed in the cytoplasm (Fig. 8b and 8c).
In comparison with the controls, morphological assessment showed that PEDV-immunopositive pigs had a lower VH:CD ratio (P = .002; Fig. 9; Suppl. Table S2). Additionally, the scores of PEDV IHC positively correlated with viral RNA fecal shedding (P < .001) and negatively correlated with the VH:CD ratios (P < .001; Table 2). In comparison with the controls, PEDV-immunopositive pigs had increased expression of ATF6 (P = .047; Fig. 10) and IRE1 (P = .007; Fig. 11), while PERK expression was not different between treatments (Fig. 12).
Correlations between PEDV RNA titer and other study variables. The data show Spearman’s rank-ordered correlation coefficients.
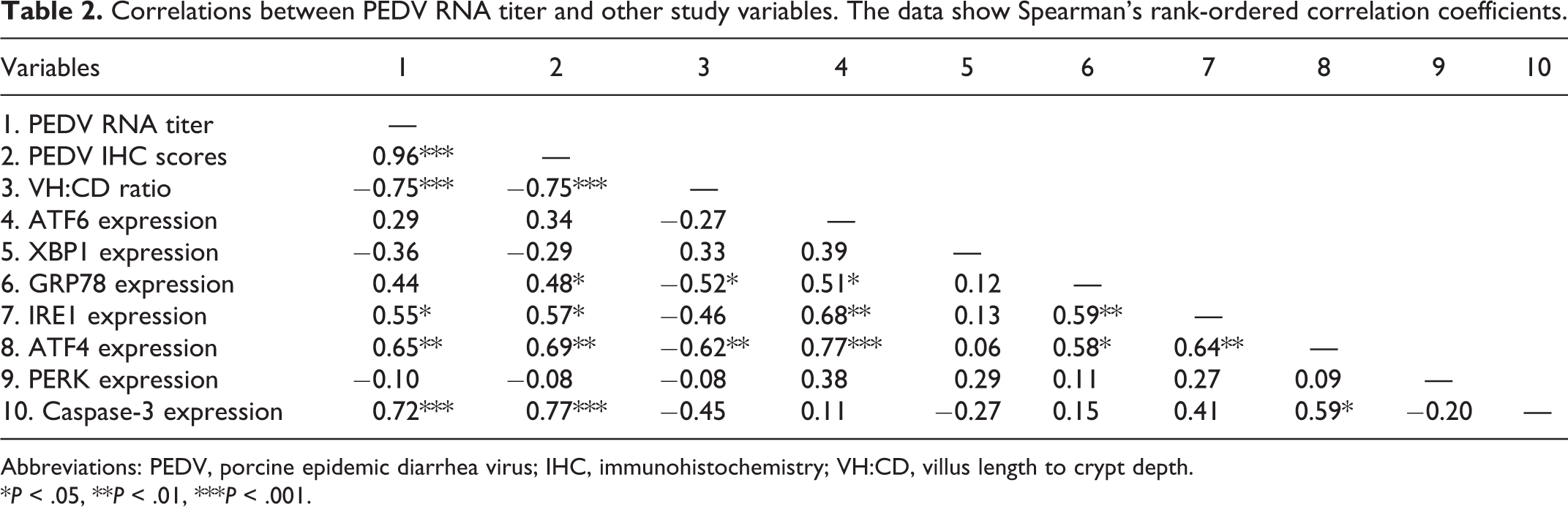
Abbreviations: PEDV, porcine epidemic diarrhea virus; IHC, immunohistochemistry; VH:CD, villus length to crypt depth.
*P < .05, **P < .01, ***P < .001.
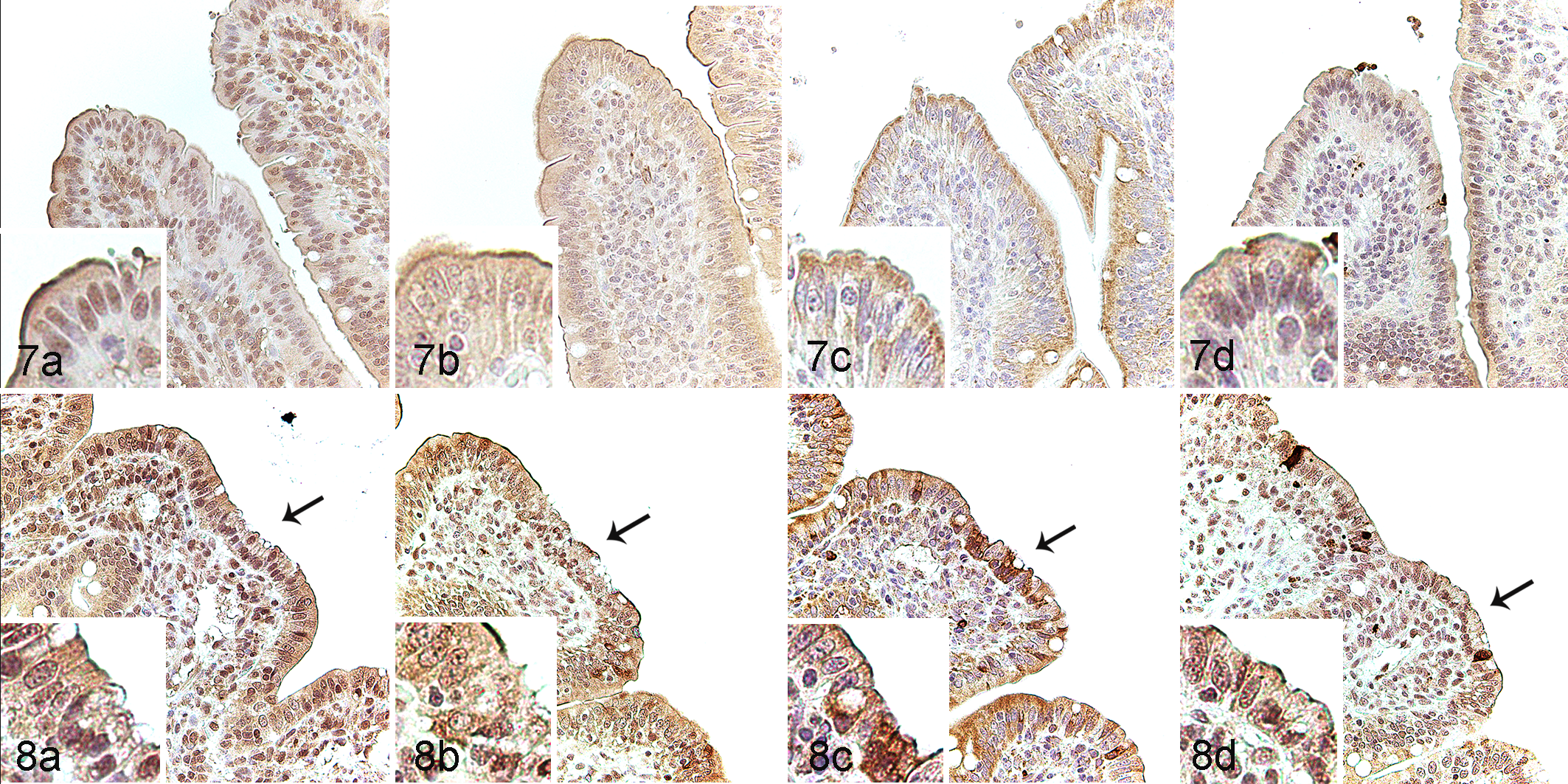
Porcine epidemic diarrhea virus (PEDV) infection, jejunum, pigs.
In comparison with controls, PEDV-immunopositive pigs had increased expression of ATF4 (P = .001; Fig. 13), GRP78 (P = .024; Fig. 14), and caspase-3 (P = .004; Fig. 15), while XBP1u expression was not different between groups (Fig. 16). Furthermore, ATF4 expression was positively correlated with PEDV RNA shedding (r = 0.65; P = .004), and with the expression of ATF6 (r = 0.77; P < .001), IRE1 (r = 0.64; P = .004), GRP78 (r = 0.58; P = .011), and caspase-3 (r = 0.59; P = .011; Table 2). GRP78 expression was positively correlated with ATF6 (r = 0.51; P = .030) and PEDV IHC scores (r = 0.48; P = .045; Table 2). Both ATF4 (r = −0.62; P = .006) and GRP78 (r = −0.52; P = .027) were negatively correlated with VH:CD ratio (Table 2).
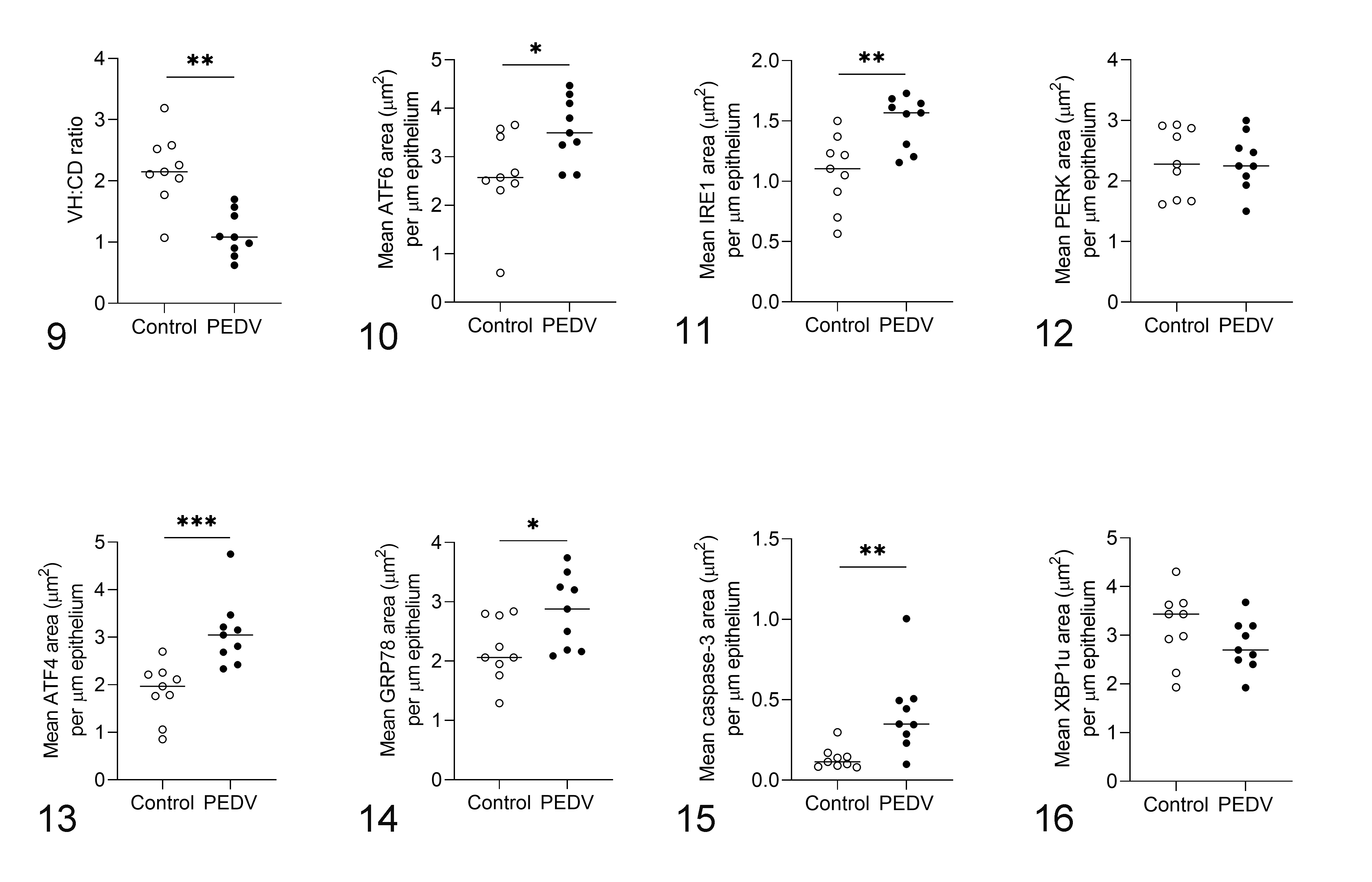
Porcine epidemic diarrhea virus (PEDV) infection, jejunum, pigs. Bars show the mean values for each group, including the control (n = 9) and PEDV-immunopositive (n = 9) groups. *P < .05, **P < .01, ***P < .001.
Discussion
Our previous studies showed that PEDV alters mucosal innate immunity in 4-week-old weaned piglets, including decreased lysosome expression in ileal Paneth cells at DPI 2, increased ileal pIgR expression at DPI 4, and a decreased number of villous and Peyer’s patch M cells in both the jejunum and ileum at DPI 6. 7 In these 4-week-old pigs, watery diarrhea peaked at DPI 5, and the fecal viral RNA titer ranged from 5.1 to 9.8 log10 GE/mL at DPI 2, 4, and 6 when all PEDV-inoculated pigs were positive for PEDV antigen. Compared with younger pigs from our previous PEDV experiment, weaned pigs at 8 weeks of age showed clinical signs later, with watery diarrhea peaking at DPI 8, lower PEDV RNA titers (2.3–7.1 log10 GE/mL), lower PEDV IHC scores, and milder intestinal injury. These findings are consistent with the concept that PED severity is age-related, whereby older pigs have relative resistance to PEDV. 18,46 Nevertheless, in the present study, 9/40 (22.5%) PEDV-inoculated pigs showed both PEDV immunopositivity and decreased VH:CD ratio in the jejunum, indicating that PEDV does cause disease in older weaned pigs. Decreased commensal bacteria and gut microbiota changes have also been observed in PEDV-infected pigs that were less than 3 months old, implying the disruption of intestinal homeostasis. 21 Although it is unclear if PEDV infection in older weaned pigs results in similar diminished growth performance as observed in nursing and 4-week-old piglets, 2,10 the intestinal damage and dysbiosis suggest that PEDV infection in older weaned pigs still warrants investigation.
Our previous study demonstrated EMT as a consequence of chronic ER stress in jejunal epithelial cells after PEDV infection. 8 Therefore, our objective was to assess the relationship between PEDV infection and ER stress. Herein we focused on the jejunum of PEDV-immunopositive pigs. Our previous studies revealed that in PEDV infection the jejunum had more severe villus damage, including a decreased VH:CD ratio and subsequent EMT expression, compared to the ileum. 7,8 Accordingly, only the jejunum was evaluated in the present study. Furthermore, the evidence of ER stress was specifically pursued in the epithelial cells at the villus tips, which is the primary location of PEDV replication and EMT presentation.
Markers of all 3 sensors of UPR on the ER were evaluated in this study. Under nonstressed conditions, both ATF6 and PERK are observed in the ER membrane, whereas IRE1 is located in the ER membrane and inner nuclear membrane. 34 In humans, ATF6, PERK, and GRP78 are diffusely expressed in the cytoplasm of hepatocytes, while IRE1 is present in both the nuclear membrane and cytoplasm of breast cancer cells. 47,54 In the present study, upon PEDV infection (and presumably PEDV-induced ER stress), ATF6 protein expression was increased and further redistributed in both the cytoplasm and nuclei, indicating that the activated ATF6 translocated into the nucleus to regulate gene expression. Consistent with ATF6 activation, GRP78 expression was increased, indicating the GRP78 gene was upregulated by activated ATF6. Induction of GRP78 transcription indicates ER stress and increased ER protein folding capacity in PEDV-immunopositive pigs. Additionally, unlike GRP78 expression in the controls, which was located primarily at the apical cytoplasm where the ER is also located, GRP78 in PEDV-immunopositive pigs was diffusely distributed. The wide distribution is likely attributed to the overexpression of GRP78 which could localize in the cytoplasm, mitochondria, mitochondria-associated membrane, or even the cell surface. 5,25
The expression of IRE1 was also modulated by PEDV exposure in the jejunum of pigs. To our knowledge, this is the first report that the IRE1 pathway participates in the PEDV-induced ER stress. Activated IRE1 can stimulate XBP1s protein expression as a potent transcriptional activator of UPR genes that regulate autophagy. 52 PEDV-induced autophagy has been observed in vitro, 16,22,58 and our findings further suggest that autophagy in PEDV infection is related to IRE1 pathway in vivo. We also examined XBP1u as an inhibitor of UPR. The XBP1u protein had no difference between treatments, implying that PEDV-induced ER stress is not affected by XBP1u during viral infection. One of the limitations in our study is that the XBP1s protein as a transcription factor for UPR target genes was not examined. Therefore, it is undetermined if PEDV induces the translation of XBP1s.
The activation of PERK in PEDV-induced ER stress has been observed in vivo; 49,58 however, this was not confirmed in our study. Interestingly, ATF4, which is a downstream factor of PERK activation, was increased in our PEDV-immunopositive pigs. Among the 3 UPR sensors, PERK is generally believed to be activated first in response to ER stress. 44 In infectious bronchitis virus infection, PERK phosphorylation is triggered at the early stage and diminishes quickly after that. 3 Therefore, the unremarkable change of PERK expression between treatments in our study may be attributed to the selected time points of tissue collection. However, independent ATF4 induction without activated PERK has also been reported in ER stress induced by tunicamycin, a classical ER stressor, suggesting PERK and ATF4 regulate UPR through separate mechanisms. 23 Therefore, additional investigation is warranted to determine if PERK is activated in PEDV-infected pigs. We also found that caspase-3 expression was increased in PEDV-immunopositive pigs, indicating that PEDV induces apoptosis in epithelial cells at the villus tips. It has been demonstrated that PEDV induces apoptosis in vitro to benefit viral replication. 16 Under ER stress, all 3 UPR signal pathways can contribute to apoptosis. 45 In the present study, caspase-3 expression positively correlated with ATF4, suggesting ATF4 stimulates apoptosis in PEDV-immunopositive pigs.
PEDV antigen is detected discontinuously in epithelial cells at the villus tips, and the border between antigen-positive and antigen-negative cells is often distinct. 26,31,42,43 We further observed that some immunonegative cells at the villus tips appear shrunken and degenerate with cytoplasmic vacuolation and hypereosinophilia. Consistent with this finding, ER stress-related markers in our study also showed a patchy distribution between immunopositive and immunonegative cells, especially GRP78. Compared to adjacent immunopositive cells, the immunonegative cells appear to have strong GRP78 expression. Some viruses, such as porcine reproductive and respiratory syndrome virus and porcine circovirus type 2, can regulate GRP78 expression to benefit viral replication and survival. 15,56 However, it is unclear if PEDV directly modifies the expression of ER stress-related proteins between PEDV antigen-positive and antigen-negative cells, and further investigation is warranted.
One potential limitation is the lack of assessment of ER stress between DPI 4, 6, and 10 in this study. PEDV-immunopositive pigs at different time points were grouped together due to a limited number of immunopositive pigs overall. A larger study with more animals is needed for temporal assessment of these changes.
In conclusion, ER stress and UPR induced by PEDV infection is demonstrated in jejunal epithelial cells by increased protein levels of ATF6, IRE1, and ATF4. Additionally, the expression of GRP78 and caspase-3 was enhanced, indicating increased ER protein folding capacity and apoptosis. These findings provide insight into the pathogenesis of PEDV in weaned pigs.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211048622 - Porcine epidemic diarrhea virus infection induces endoplasmic reticulum stress and unfolded protein response in jejunal epithelial cells of weaned pigs
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211048622 for Porcine epidemic diarrhea virus infection induces endoplasmic reticulum stress and unfolded protein response in jejunal epithelial cells of weaned pigs by Ya-Mei Chen, Nicholas K. Gabler and Eric R. Burrough in Veterinary Pathology
Footnotes
Acknowledgements
This study was funded by a USDA AFRI grant (# AH10072632017). We thank the staff of the Livestock Infectious Disease Isolation Facility for their assistance and Shelby (Curry) Ramirez and Wesley Schweer for their help with the animal challenge. We appreciate Jennifer M. Groeltz-Thrush and staff from the Iowa State University Veterinary Diagnostic Laboratory for slide preparation and technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a USDA AFRI grant (# AH10072632017).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
