Abstract
Porcine epidemic diarrhea virus (PEDV) induces an often fatal gastrointestinal disease in piglets. In this study, we performed a PEDV infection experiment with the Microminipig, the smallest of experimental minipigs, as a novel small animal model. We orally inoculated a neonatal Microminipig with an intestinal homogenate of a PEDV-infected pig and housed it in a small cage originally designed for rats in an animal biosafety level 2 facility. The infected Microminipig showed the typical signs of porcine epidemic diarrhea (PED), such as watery diarrhea, loss of appetite and weight loss. We also recognized a high amount of excreted PEDV in its rectal swabs and villus atrophy of the small intestine. These results suggest that the Microminipig is a good small animal model for PED, which may contribute to a better understanding of the pathogenesis of PEDV.
To understand the pathogenesis of porcine epidemic diarrhea virus (PEDV), in vivo animal experiments are indispensable. Because the clinical signs of porcine epidemic diarrhea (PED) can be evaluated only in young piglets, suckling piglets are often used for infection experiments of PEDV. 9 However, such experiments are labor intensive, are expensive, and need special equipment, such as an animal biosafety level 2 (ABSL2) facility for large animals.
The Microminipig (MMPig; Fuji Micra, Fujinomiya, Japan) is the smallest minipig among those for experimental use (eg, Clawn, Göttingen, and Yucatan minipigs) and are genetically similar to conventional pigs. 3 –5,11 The body weight (BW) of neonatal MMPigs is approximately 400 g, and thus the neonates can be housed in isolation cages designed for rats, 5 so the MMPig might be an effective small animal model for PED and may overcome the disadvantages of using mouse models of PED. 7 In this study, to develop a novel small animal model for PED, we infected a neonatal MMPig with PEDV and pathologically examined the infected pig.
The sow was impregnated by natural mating. Two colostrum-deprived male MMPig neonates were delivered by cesarean section at day 114 of gestation with BW of 356 and 353 g. After delivery, the neonatal MMPigs were immediately moved to an ABSL2 facility that included the cages (dedicated isolation chamber) designed for rats, 605 × 370 × 330 mm (depth × width × height), and were maintained at a temperature of 28 ± 2°C and a relative humidity of 50% ± 20%, with a 12-hour light/dark cycle. Both pigs were fed every 3 to 4 hours with commercial milk replacer (HP Kobuta-etsuke; Scientific Feed Laboratory Co., Ltd., Tokyo, Japan) that did not contain PEDV antibodies (Fig. 1B). Both piglets voluntarily drank milk using 1-ml syringes with the double-gauze-covered tips. No antibiotics were given in this study. All protocols were approved by the Ethics Committee of Animal Care and Experimentation, Kagoshima University (VM14034) and the Food Safety and Consumer Affairs Bureau, Ministry of Agriculture, Forestry and Fisheries, Japan (approval number: 26-3784). The research was performed according to the Institutional Guidelines for Animal Experiments and was in compliance with the Japanese Law Concerning the Protection and Control of Animals (Law No. 105 and Notification No. 6).
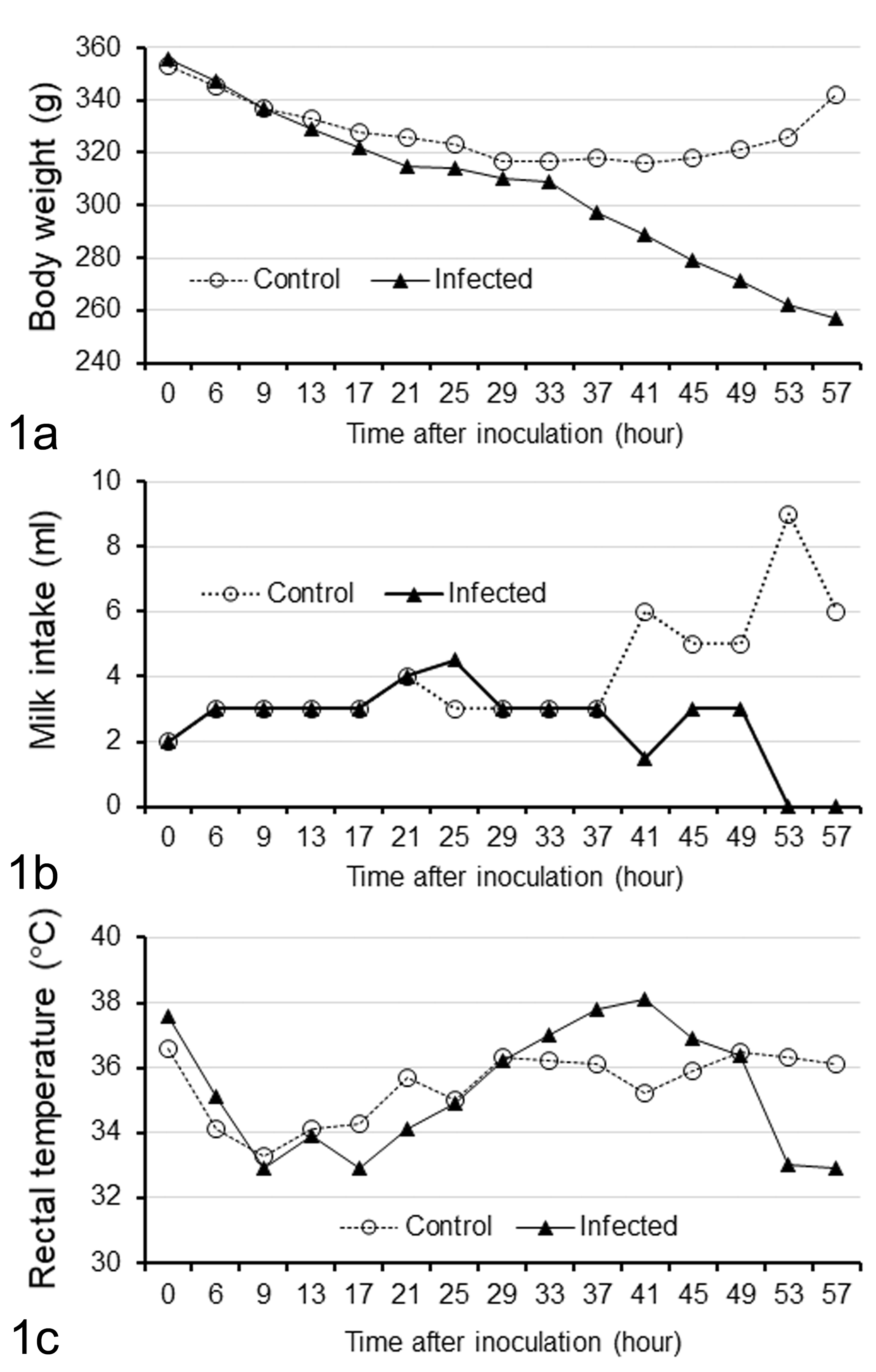
Changes in body weight (a), milk intake (b), and rectal temperature (c) after challenge of a Microminipig with intestinal homogenate containing porcine epidemic diarrhea virus compared to a control uninfected Microminipig.
Small intestinal homogenate from a PEDV-infected domestic pig was kindly provided from the Kagoshima Central Livestock Hygiene Service Center (Kagoshima, Japan). We isolated the PEDV strain KU-KGS1/14, 1 which belongs to the North American clade, from this specimen after this study was conducted. The homogenate was centrifuged at 3000 × g for 10 minutes at 4°C to remove debris, and then the supernatant was filtrated with a 0.22-μm PVDF membrane filter (Millipore, Burlington, MA) to remove bacteria from the supernatant.
For quantitative polymerase chain reaction (qPCR), RNA was extracted from 140 μl of the inoculum or the rectal swab suspensions (QIAamp Viral RNA Mini Kit; QIAGEN, Hilden, Germany), and complementary DNA (cDNA) was synthesized (Verso cDNA Synthesis Kit; Thermo Fisher Scientific, Waltham, MA) using a random hexamer. qPCR was performed using Brilliant III Ultra-Fast SYBR Green QPCR Master Mix (Agilent Technologies, Santa Clara, CA) with the primers 5′-ACTGTGCCAAAGTCTGATCC-3′ and 5′-TCAACAGCTGTGTCCCATTC-3′, which were designed based on highly conserved regions in PEDV N genes. The real-time PCR conditions were as follows: first denaturation at 95°C for 3 minutes, then 40 cycles at 95°C for 5 seconds and 60°C for 5 seconds. Viral gene copy numbers were calculated using a standard calibration curve generated from a serially diluted plasmid control (1–7 log10 copies/reaction) containing a PEDV N gene sequence. The detection limit of this assay was approximately 2.23 log10 copies/μl sample. In this study, virus titers are shown as log10 genomic equivalent (GE)/ml. 2
Each neonatal piglet at approximately 1 hour after birth was orally inoculated with 200 μl of the filtered small intestinal homogenate containing 7.87 log10 GE of PEDV (infected piglet) or with 200 μl of Eagle minimum essential medium (control piglet) in the safety cabinet of the ABSL2 facility. The experimental period was set at 4 days, during which time the general condition, milk intake volume, BW, and rectal temperature measurements were recorded at 0, 6, and 9 hours postinoculation (hpi) and every 4 hours afterward. Swabs of rectal content were prepared for quantification of the viral titers by suspending the rectal thermometer in 1 ml of Eagle’s minimal essential medium supplemented with 0.5% bovine serum albumin, 2 mM L-glutamine, 10 000 U/ml penicillin, 10 mg/ml streptomycin, 0.3 mg/ml gentamicin, and 2.5 μg/ml amphotericin B. Fecal consistency was scored as described previously 8 : 0, firm feces; 1, soft feces; 2, mild diarrhea; and 3, severe diarrhea. The infected piglet suddenly became worse in condition with hypothermia and immobilization and was sacrificed in extremis by exsanguination under anesthesia at 58 hpi, and the control piglet was sacrificed at 79 hpi after cleaning and disinfecting the necropsy room.
At necropsy of the infected piglet, the parts of duodenum, jejunum, and ileum that included contents were collected, frozen with liquid nitrogen, and stored at –80°C. These samples tested negative by PCR for transmissible gastroenteritis virus (TGEV), porcine deltacoronavirus (PDCoV), and rotavirus (SMC Corporation, Tokyo, Japan).
The small intestine (duodenum, jejunum, and ileum) and large intestine (cecum, colon, rectum; except the above frozen samples) were fixed in 10% phosphate-buffered formalin for 1 week, then cut in sagittal section; rolls of the whole intestine were embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin. Liver, spleen, testes, heart, lung, gallbladder, kidney, cerebrum, cerebellum, brainstem, and spinal cord were similarly prepared. The villous height to crypt depth ratio was assessed in both animals to identify villous atrophy. 6 Liver sections of all animals were stained with routine periodic acid–Schiff (PAS) to evaluate glycogen.
Immunohistochemistry (IHC) was carried out (Envision; Dako Cytomation Co., Ltd., Kyoto, Japan) on paraffin sections of intestines and organs. The deparaffinized sections were heated by microwave for 25 minutes for antigen retrieval and incubated overnight with a primary antibody against PEDV (1:1 concentration, mouse monoclonal anti-PEDV antibody 3F.12; MEDIAN Diagnostics, Gangwon, Korea) at 4°C, detected with 3,3′-diaminobenzidine tetrachloride, and counterstained with hematoxylin. Reactivity of the antibody was confirmed by immunocytochemistry using Vero cells infected with a PEDV field isolate that was genetically similar to the inoculated virus.
During the experimental period, the control piglet did not show any clinical signs. The infected piglet then developed mild diarrhea (score 2) at 33 hpi and yellowish watery diarrhea (score 3) from 37 to 57 hpi (the end point of the experiment). The BW of the infected piglet progressively decreased from 37 hpi to the end of experiment (Fig. 1A). The milk intake was less than that of the control from 41 hpi to the end of the experiment (Fig. 1B). The rectal temperature of the infected piglet reached a maximum of 38.1°C at 41 hpi and was higher than that of the control piglet from 33 to 45 hpi, then drastically decreased to 32.9°C at 53 hpi (Fig. 1C).
In the infected piglet, the viral titer in rectal content based on qPCR was 9.1, 10.7, 8.4, 8.9, 8.6, and 8.6 log10 GE/ml at 37, 41, 45, 49, 53, and 57 hpi, respectively. No PEDV nucleic acid was detected from any swab samples of the control piglet.
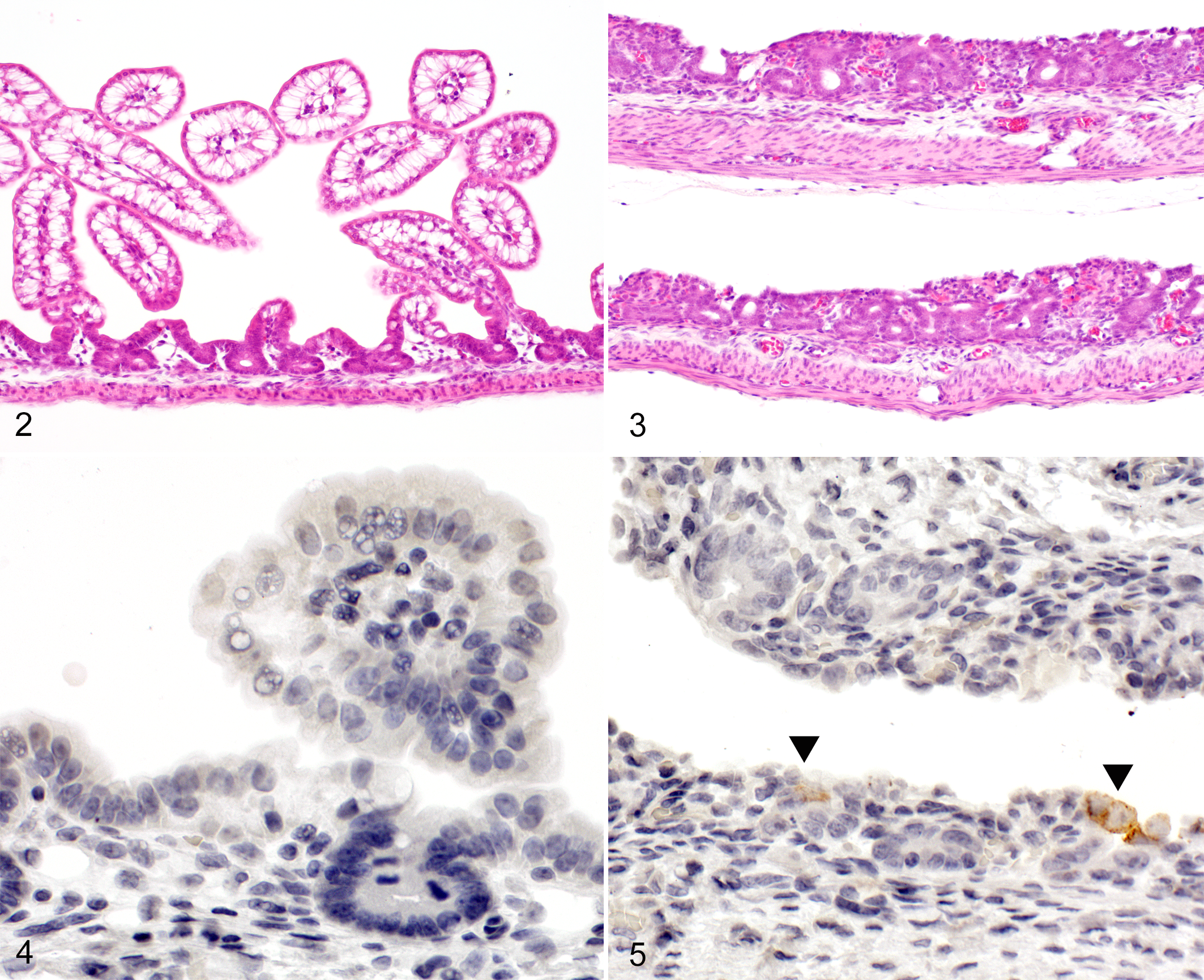
The infected piglet showed thinning of the small intestinal wall (duodenum, jejunum, and ileum) macroscopically and severe villus atrophy histologically (Figs. 2, 3), while the large intestines showed no pathological changes. The villous height to crypt depth ratio in the small intestine of the infected piglet was markedly decreased compared with the control (Table 1). By contrast, the control piglet showed no lesions in the intestine. The PAS reaction showed a marked reduction in hepatocellular glycogen in the infected piglet. The other organs exhibited no pathological changes.
Small Intestinal Villous Height (VH, μm), Crypt Depth (CD, μm), and VH/CD Ratios in a Control Microminipig and 1 Infected With Porcine Epidemic Diarrhea Virus.

By IHC, scattered individual epithelial cells (around crypt) in the duodenum, jejunum, and ileum of the infected piglet were found to express PEDV antigen (Figs. 4, 5). All other tissues of the infected piglet as well as intestines and tissues of the control animal were negative for PEDV antigen.
The clinical signs in the PEDV-infected MMPig were very similar to those observed in conventional piglets infected with recently isolated highly virulent PEDV strains. The infected MMPig showed severe watery diarrhea accompanied by poor appetite and progressive weight loss. However, the onset of clinical signs was delayed compared with other experiments. While the infected MMPig showed diarrhea from 33 hpi, several other studies have reported the onset of clinical signs in PEDV-infected piglets at approximately 24 hpi. 8 –10 This may be because of the lower virus titer in our inoculum. We inoculated 7.87 log10 GE, but many other studies have used more than 10 log10 GE. 8 –10 However, in 1 study, pigs developed signs from 25 to 46 hours after being inoculated with 6.3 to 9.0 log10 GE of PEDV. 2 This may be due to the differences of inocula: while we used intestinal homogenate from a PEDV-infected pig, the other study used fecal suspensions as inocula. 2,8 –10 Because it was highly probable that our inoculum contained a large amount of infected cell-derived RNA, including viral subgenomic RNA, the amount of infectious virus may have been lower than those used in other studies, even if the GE in the inocula were comparable. Further studies are needed to assess this issue.
The lesions in the infected piglet were similar to those in conventional piglets. 2 By IHC, we found that only some epithelial cells in the small intestine were positive for PEDV antigen. It is likely that most PEDV-infected epithelial cells were sloughed and the remaining and regenerating epithelial cells were less susceptible to virus infection.
In this study, we found that PEDV infection of a MMPig was clinically, macroscopically, and histopathologically similar to infection of conventional pigs. These results indicate that the MMPig may be a useful small animal model for PED. To our knowledge, this is the first report showing that neonatal MMPigs are suitable for an infection experiment in small isolation cages in an ABSL2 facility. Because some other infectious diseases, such as TGEV infection, neonatal Escherichia coli diarrhea, and clostridial enteric infection, are also important in young piglets, our protocol may also be suitable for investigations of such pathogens.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by a Health Labor Science Research Grant (No. 33361105) from the Ministry of Health, Labor, and Welfare of Japan (to AT) and the Adaptable and Seamless Technology Transfer Program (A-Step No. AS2316907E) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (to AT and HK). This work was also partly supported by JSPS KAKENHI Grants 16H05176 (to AT), 25450426 (to HK), and 16K08023 (to HK).
