Abstract
Porcine epidemic diarrhea virus (PEDV) was first recognized in North America in April 2013 and has since caused devastating disease. The objective of this study was to characterize disease and viral detection associated with an original North American PEDV isolate inoculated in neonatal piglets. Thirty-six 1-day-old cesarean-derived and colostrum-deprived piglets were randomly assigned to the control (n = 16) or challenged group (n = 20); the latter were orogastrically inoculated with 1 ml of US/Iowa/18984/2013 PEDV isolate titered at 1 × 103 plaque-forming units per milliliter. Rectal swabs were collected from all piglets prior to inoculation and every 12 hours postinoculation (hpi) thereafter, with 4 control and 5 challenged piglets euthanized at 12, 24, 48, and 72 hpi. One piglet had a positive real-time quantitative polymerase chain reaction test on rectal swab at 12 hpi, and all remaining piglets were positive thereafter, with highest viral quantities detected at 24 and 36 hpi. Diarrhea was evident in 30% and 100% of challenged piglets at 18 and 24 hpi, respectively. Viral antigen was detected in enterocytes by immunohistochemistry in the duodenum and ileum of piglets euthanized at 12 hpi and was apparent throughout the small intestine of all piglets thereafter, with villus height:crypt depth ratios consistently below 4:1. Viremia was confirmed in 18 of 20 pigs at euthanasia. Clinical disease was severe and developed rapidly following infection with an original North American PEDV isolate, with lesions, viremia, and antigen detection possible by 12 hpi.
Keywords
Porcine epidemic diarrhea virus (PEDV) was first detected in North America 19 in April 2013 and has since infected numerous production facilities throughout the United States, Canada, and Mexico. PEDV is an enveloped single-stranded RNA virus of the Coronaviridae family 1 that was first reported in the United Kingdom, 21 but this alphacoronavirus can now be found in multiple countries worldwide. 18 Viral sequencing information from these initial North American breaks indicate >99% homology with 2012 Chinese isolates entered into GenBank. 6,19 More recently, North American PEDV sequences have revealed a second and distinct variant virus. 20
PEDV is transmitted by the fecal-oral route, and it causes watery diarrhea, anorexia, lethargy, and vomiting in swine of all ages, with either acute or endemic disease presentations. Enterocytes are the target cell of PEDV replication and undergo necrosis following replication, resulting in severe villous atrophy and malabsorptive diarrhea. 17 Clinical signs and microscopic lesions are indistinguishable from those of transmissible gastroenteritis virus (TGEV), another swine alphacoronavirus, but it has been suggested that some strains of PEDV may be less severe. 16 Both PEDV and TGEV can be associated with high morbidity and mortality, especially in neonatal pigs without lactogenic immunity 17 ; however, these 2 viruses are antigenically distinct and do not cross-react serologically. 15
Due to a lack of available scientific information regarding the newly introduced PEDV isolates in North America, the objective of this study was to characterize disease caused by experimental infection with an original US PEDV isolate (US/Iowa/18984/2013) 4 in cesarean-derived and colostrum-deprived (CDCD) neonatal piglets by documenting clinical responses, fecal shedding, microscopic intestinal lesions, and immunohistochemical antigen detection within enterocytes.
Materials and Methods
Animals
Healthy pregnant commercial cross-bred second- and third-parity sows that had the same artificial insemination date and were confirmed negative for PEDV, TGEV, and porcine reproductive and respiratory syndrome virus antibodies were delivered to Iowa State University 1 week prior to expected farrowing. Neonatal piglets used in this experiment were CDCD on day 113 of gestation. All CDCD piglets were triaged at birth and prophylactically given an antibiotic injection (Excede; Zoetis, Florham Park, NJ, USA) per labeled dose and route.
Experimental Design and Housing
Thirty-six CDCD piglets from 4 litters were randomly divided without concern for sex or litter into 2 groups: negative control (n = 16) and challenged (n = 20). Piglets were individually housed in 18-gallon plastic totes (Rubbermaid, Port Washington, NY, USA) for the study period, and groups were kept in a biosafety level 2 animal facility separated by room, entry, and ventilation system. Room temperature was maintained at 29°C with supplemental heat lamps placed overhead to achieve a microenvironment temperature of approximately 35°C. All piglets were fed 30 ml of milk replacer (Esbilac; Pet-Ag, Hampshire, IL, USA) 3 times daily by gavage using an 8F catheter (Sovereign; Tyco/Healthcare, Mansfield, MA, USA).
Piglets were either sham inoculated with virus-free cell culture media or challenged via gastric gavage with 1 ml of cell culture–derived US/Iowa/18984/2013 PEDV isolate at 1 × 103 plaque-forming units per milliliter (PFU/ml) approximately 5 hours after cesarean section. Four control and 5 challenged piglets per time point were randomly selected and euthanized at 12, 24, 48, and 72 hours postinoculation (hpi). The study design was approved by the Iowa State University Institutional Animal Care and Use Committee (protocol log No. 6-13-7593-S).
PEDV Inoculum
Plaque-cloned isolate US/Iowa/18984/2013 (GenBank accession No. KF804028) was propagated in Vero cells (ATCC CCL-81) and titrated by plaque assay. 4,5 The PEDV isolate originated from naturally infected neonatal piglets as described elsewhere and was documented to cause severe disease in postweaned pigs. 4,11 Challenge inoculum was polymerase chain reaction (PCR) negative for TGEV, porcine respiratory coronavirus, porcine rotaviruses (serotypes A, B, C), porcine circoviruses (types 1 and 2), porcine parvovirus, porcine reproductive and respiratory syndrome virus, porcine enterovirus, porcine teschovirus, porcine hemagglutinating encephalomyelitis virus, porcine deltacoronavirus, and swine hepatitis E virus. Testing was done according to the Iowa State University Veterinary Diagnostic Laboratory standard operating procedure for each specific agent.
Sample Collection and Clinical Observations
Fecal swabs were collected from all piglets prior to inoculation and at 12, 24, 36, 48, 60, and 72 hpi. Diarrhea, vomiting, lethargy, loss of condition, and dehydration were recorded with knowledge of infection status for each piglet prior to inoculation and every 12 hours thereafter until euthanasia. Tissues collected at necropsy included fresh and formalin-fixed stomach, small intestine, colon, cecum, mesenteric lymph node, tonsil, lung, heart, liver, spleen, and kidney. Intestinal contents were also obtained from all pigs at necropsy. Sections of fixed small intestine for microscopic evaluation included standardized locations of duodenum, proximal jejunum, midjejunum, distal jejunum, and ileum. 11
Real-time Reverse Transcription PCR
Swabs, tissue homogenates, and sera were processed on the day of collection. Briefly, fecal swabs were added to tubes containing 1 ml of 0.01M phosphate-buffered saline (pH 7.4) without calcium and magnesium (GIBCO/Life Technologies, Carlsbad, CA, USA) and vortexed. Tissue sections of colon, heart, kidney, lung, mesenteric lymph node, small intestine, spleen, and stomach weighing approximately 1 g were minced and homogenized in 20 ml of Hank’s balanced salt solution (GIBCO/Life Technologies) using a Stomacher Biomaster machine (Seward Laboratory Systems Inc, Davie, FL, USA). Serum separator blood collection tubes were centrifuged at 1000 × g for 10 minutes to collect sera. All samples were stored at –80°C until viral RNA was extracted with the MagMAX Viral RNA Isolation Kit (Life Technologies, Carlsbad, CA, USA) using a modified high-volume extraction protocol per manufacturer instructions. Reverse transcription quantitative PCR was performed as previously described 11 alongside viral standards with known infectivity titers (PFU) for quantification. Each cycle threshold value was then converted to viral titer, PFU equivalent per milliliter, estimated from a standard curve generated from the viral standards of known titers.
Histology, Intestinal Morphometry, and Immunohistochemistry
Tissues samples were collected in 10% neutral buffered formalin, allowed to fix for 48 hours, and then transferred to 100% alcohol for 7 days before routine histologic processing and staining with hematoxylin and eosin. Nonintestinal tissue sections were evaluated for evidence of disease and/or inflammation. For each location of small intestine (1–5), 4 serial sections were trimmed into 1 paraffin block. The mean length and depth of 3 perceived full-length villi and crypts per location were obtained using a computerized image system (Olympus DP72 camera, cellSens digital imaging software) operated by a veterinary pathologist blinded to the treatment group. Mean villus length and crypt depth were used for statistical analysis and for determining villus height:crypt depth ratios. 11
Immunohistochemical staining for PEDV antigen was performed on duodenum, proximal jejunum, midjejunum, distal jejunum, and ileum sections from all pigs. Antigen labeling in each section was semiquantitatively assessed on a scale ranging from 0 to 3, where 0 signifies no signaling detected and 3 indicates that >50% of villous enterocytes were observed with positive staining. 11 All scores were recorded by a single pathologist blinded to individual piglet identification and treatment group. In addition, mesenteric lymph node, colon, spleen, stomach, heart, spleen, liver, and kidney were evaluated for PEDV antigen by immunohistochemistry (IHC).
Statistical Analysis
Mean intestinal villus height and crypt depth values and ratios were analyzed by a commercial statistical software program (SAS 9.3; SAS Institute, Cary, NC, USA) using linear mixed models. Treatment group, necropsy time point, small intestinal location, and their interactions were fixed effects, while piglet was the random effect. Mean measurements were evaluated between groups by location and necropsy time point, as well as between necropsy time points by group and location. Statistical significance was set at P < .05 for all analyses.
Results
Clinical Observations
Control piglets did not develop clinical signs of porcine epidemic diarrhea for the duration of the experiment. Mild diarrhea and lethargy in PEDV-challenged piglets were first observed at 18 hpi in 6 of 20 (30%), and 20 of 20 (100%) challenged piglets developed diarrhea by 24 hpi, with severe watery diarrhea observed in all remaining piglets at 36 hpi. Diarrhea continued thereafter for all time points, with vomiting observed in 1 or 2 piglets at 24, 36, 48, and 60 hpi. Loss of body condition and dehydration appeared moderate and then became more severe in all challenged pigs at 48 and 72 hpi, respectively. Three of the 5 remaining challenged pigs at 60 hpi were severely dehydrated, which required intraperitoneal treatment with 30 ml of phosphate-buffered saline.
Detection of Virus and Viral RNA
Viral shedding in feces from challenged piglets is summarized in Table 1, with first detection at 12 hpi in a single piglet. All challenged piglets were positive for PEDV RNA in feces thereafter with highest viral loads detected at 24 hpi.
PCR Detection of Porcine Epidemic Diarrhea Virus RNA in Rectal Swabs From Cesarean-Derived and Colostrum-Deprived Piglets Challenged With US/Iowa/18984/2013.
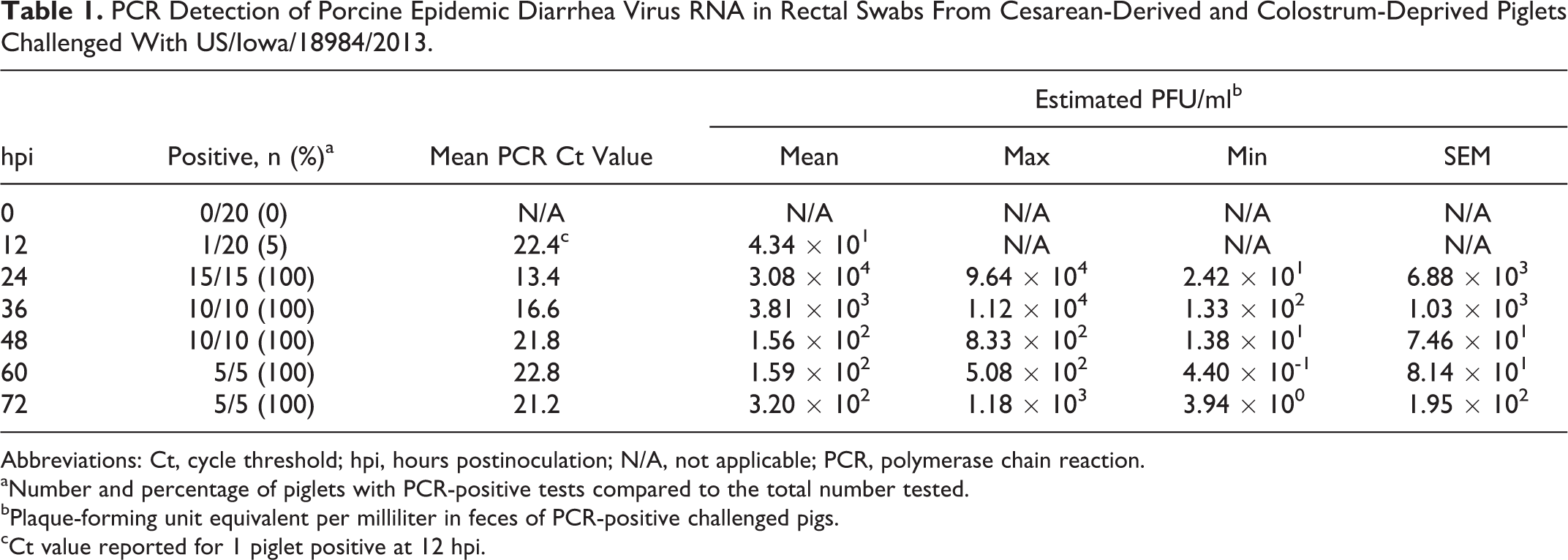
Abbreviations: Ct, cycle threshold; hpi, hours postinoculation; N/A, not applicable; PCR, polymerase chain reaction.
aNumber and percentage of piglets with PCR-positive tests compared to the total number tested.
bPlaque-forming unit equivalent per milliliter in feces of PCR-positive challenged pigs.
cCt value reported for 1 piglet positive at 12 hpi.
At necropsy 18 of 20 (90%) challenged piglets had measurable viral RNA in serum. Two piglets were nonviremic at necropsy: 1 at 12 hpi and 1 at 72 hpi (Table 2). All assayed tissues were reverse transcription quantitative PCR positive for PEDV RNA regardless of necropsy time point. Trends for the quantity of PEDV RNA in tissue were as follows, in descending order: small intestine, colon, mesenteric lymph node, spleen, stomach, kidney, lung, and heart (Table 2).
PCR Detection of Porcine Epidemic Diarrhea Virus RNA in Sera and Tissue From Cesarean-Derived and Colostrum-Deprived Piglets Challenged With US/Iowa/18984/2013.a
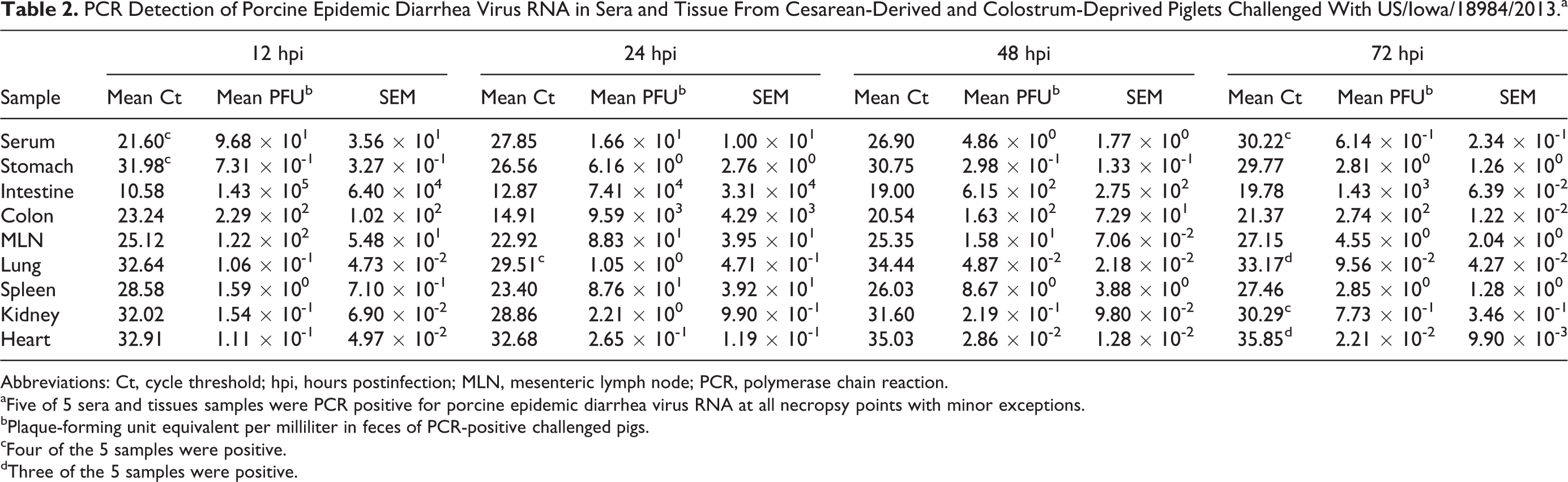
Abbreviations: Ct, cycle threshold; hpi, hours postinfection; MLN, mesenteric lymph node; PCR, polymerase chain reaction.
aFive of 5 sera and tissues samples were PCR positive for porcine epidemic diarrhea virus RNA at all necropsy points with minor exceptions.
bPlaque-forming unit equivalent per milliliter in feces of PCR-positive challenged pigs.
cFour of the 5 samples were positive.
dThree of the 5 samples were positive.
PEDV RNA was not detected by reverse transcription quantitative PCR testing of feces of any piglet prior to inoculation or of serum, feces, and tissues of control piglets at any time during the study.
Histology, Intestinal Morphometry, and IHC
Stomach, cecum, colon, mesenteric lymph node, tonsil, lung, heart, liver, spleen, and kidney sections from all piglets were histologically unremarkable. Duodenal, jejunal, and ileal sections were within normal limits for control piglets euthanized at 12, 24, 48, and 72 hpi. Acute enterocyte swelling and vacuolation with minimal subjacent collapse of lamina proprial tissue were evident in 3 of 5 (60%) challenged piglets euthanized at 12 hpi. Two of these piglets had lesions limited to the duodenum, while lesions were apparent in the duodenum, proximal jejunum, midjejunum, and distal jejunum of the third piglet. At 24, 48, and 72 hpi, all challenged piglets had severe atrophy and variable fusion of villi with enterocyte necrosis, degeneration, attenuation, or exfoliation and collapsed lamina proprial tissue containing karyorrhectic debris. Minimal lamina proprial hemorrhage, villous tip erosion with neutrophil exudation into the lumen, and/or enterocyte syncytial cell formation were also noted in challenged piglets euthanized at 24 and 48 hpi (Fig. 1).
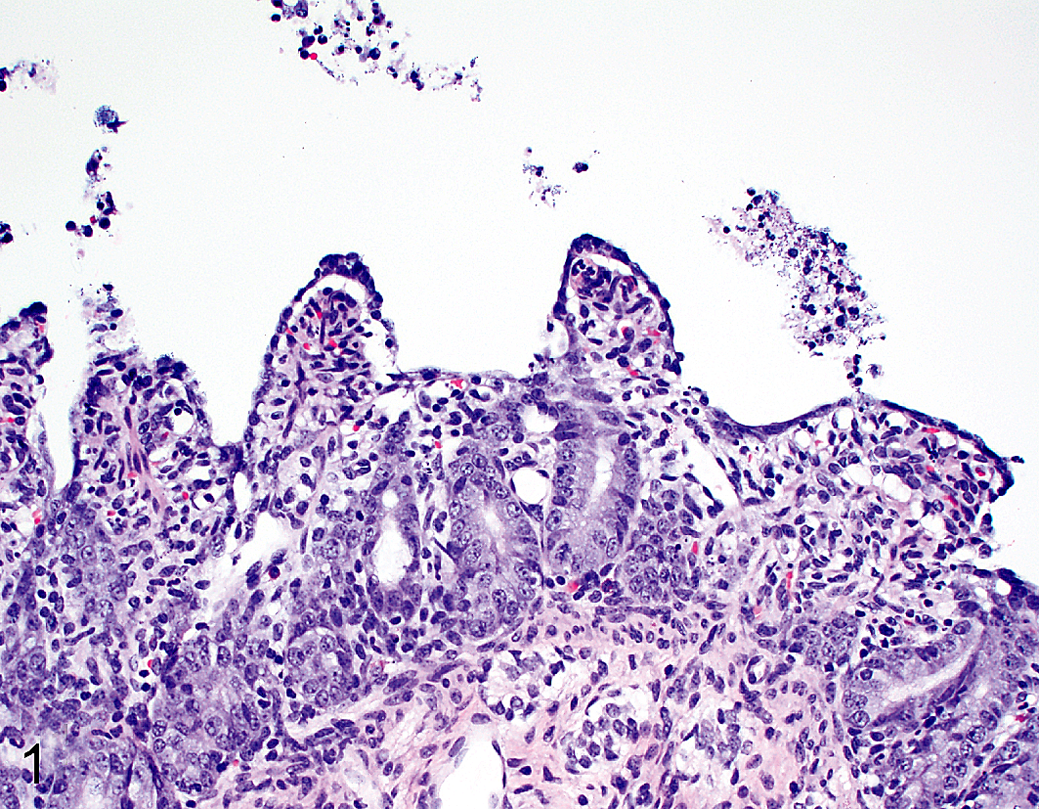
Porcine epidemic diarrhea, piglet, proximal jejunum. Cesarean-derived and colostrum-deprived animal 24 hours postinoculation. Villous atrophy with lamina proprial collapse and attenuation of enterocytes. Hematoxylin and eosin.
Villi were significantly shorter in the sections of duodenum and ileum (P < .01), but not jejunum, at 12 hpi in PEDV-challenged piglets compared to controls. Mean villus height in all 5 intestinal sections evaluated in the challenged group was significantly decreased compared to controls (P < .01, all analyses) at 24, 48, and 72 hpi (Figs. 2–5). Crypt hyperplasia, as measured by crypt depth, was significantly increased in duodenal sections from the challenged group at 48 and 72 hpi and the proximal jejunum at 72 hpi compared to controls (Figs. 6–9). The mean villus height:crypt depth ratios in the duodenum and proximal, mid-, and distal jejunum were ≥9.5:1 for the control group at all time points, and those in the ileum were ≥8.9:1 for the entire study. In contrast, the corresponding ratios in the challenged group were slightly lower at 12 hpi, <4:1 at 24 hpi, and diminished to approximately 2:1 in all intestinal sections thereafter (Table 3).
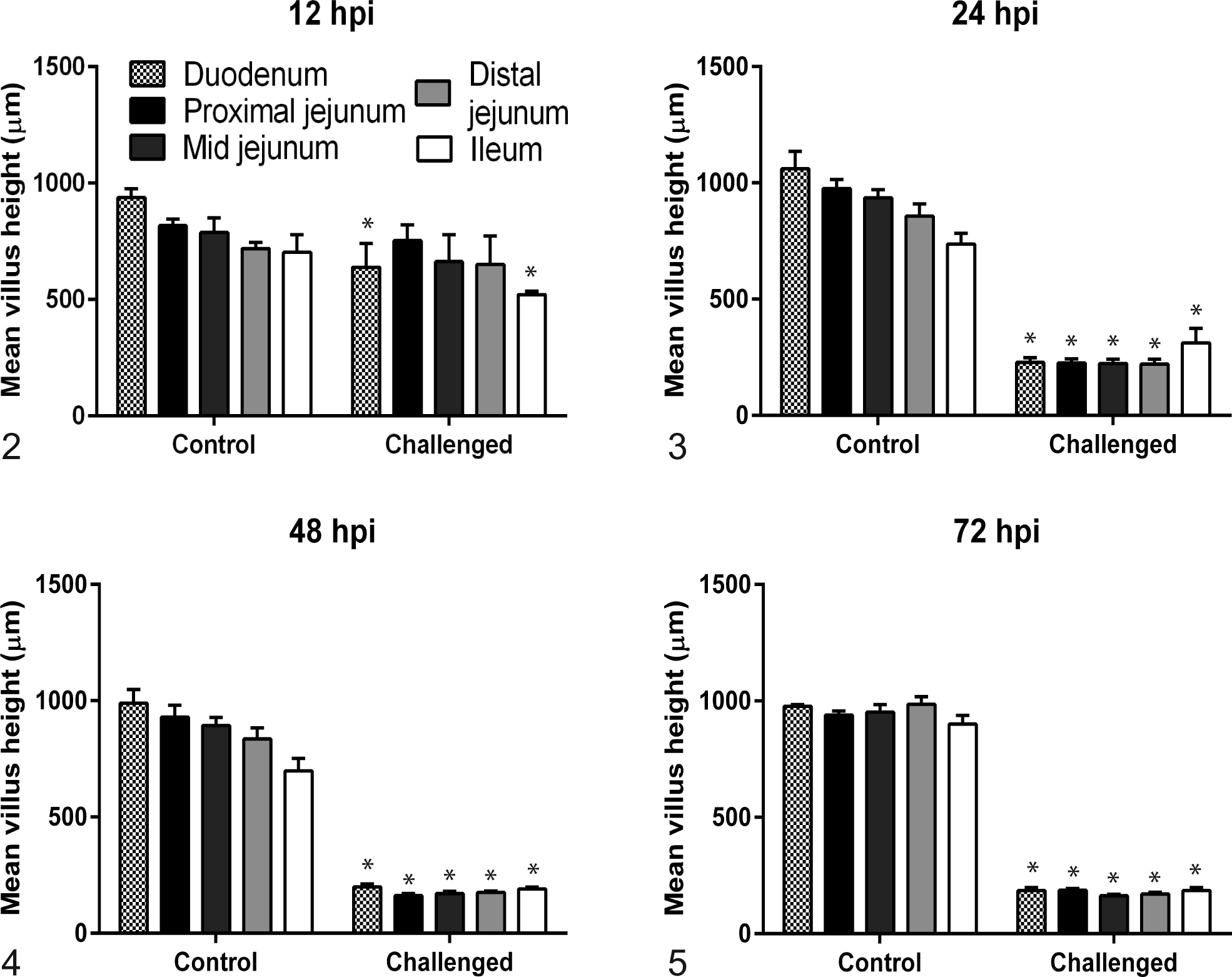
Mean villus height for duodenum; proximal, mid-, and distal jejunum; and ileum from 4 negative control and 5 porcine epidemic diarrhea virus euthanized piglets at varying number of hours postinoculation (hpi).
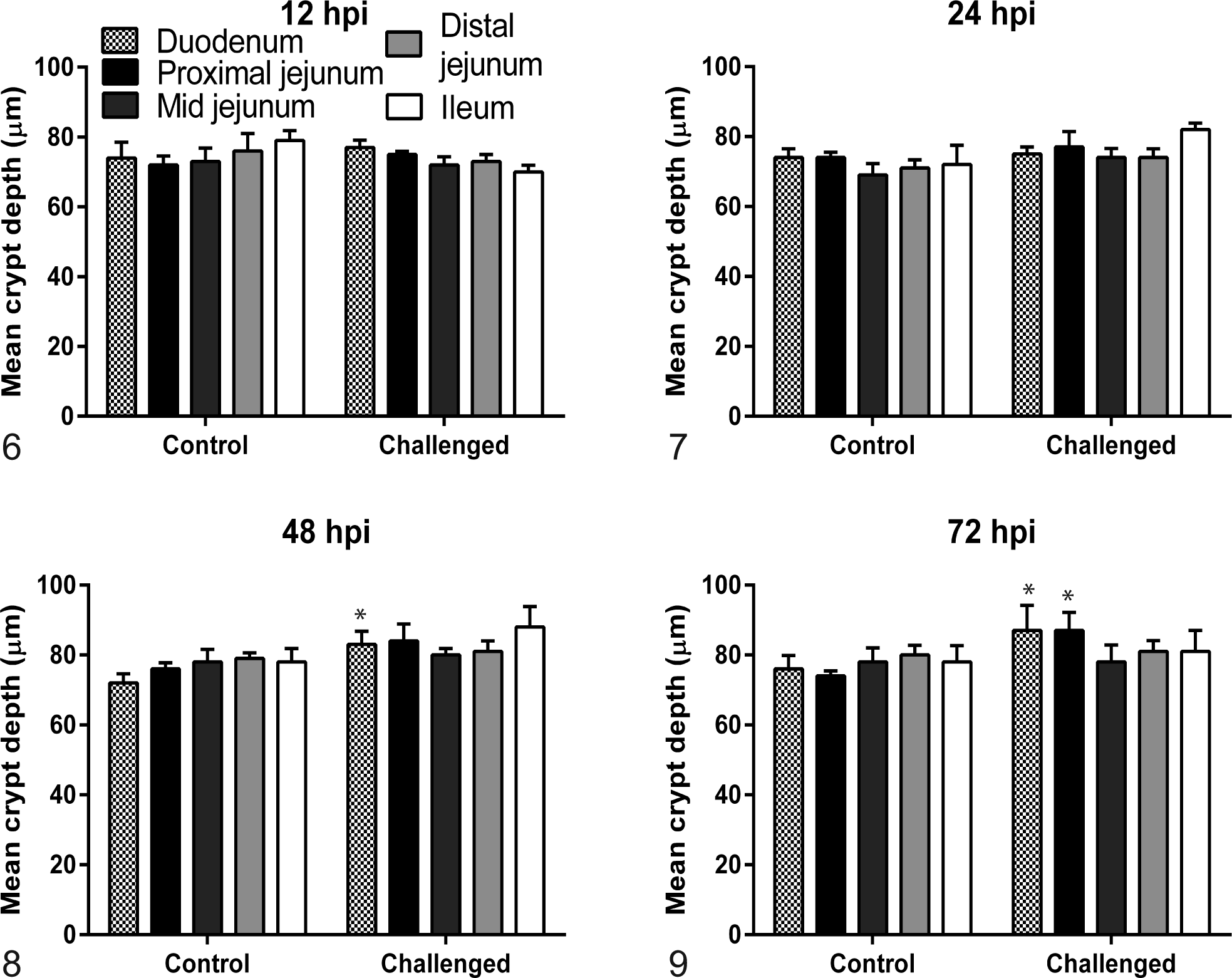
Mean crypt depth for duodenum; proximal, mid-, and distal jejunum; and ileum from 4 negative control and 5 porcine epidemic diarrhea virus piglets euthanized at varying number of hours postinoculation (hpi). Crypt depth measurements of porcine epidemic diarrhea virus–challenged piglets were not different at 12 and 24 hpi (Figs. 6, 7) but were significantly (P ≤ .02) elongated in duodenum and proximal jejunum at 48 (Fig. 8) and 72 (Fig. 9) hpi.
Mean Villus Height:Crypt Depth Ratio at Necropsy of 4 Negative Control and 5 Porcine Epidemic Diarrhea Virus–Challenged Cesarean-Derived and Colostrum-Deprived Piglets.
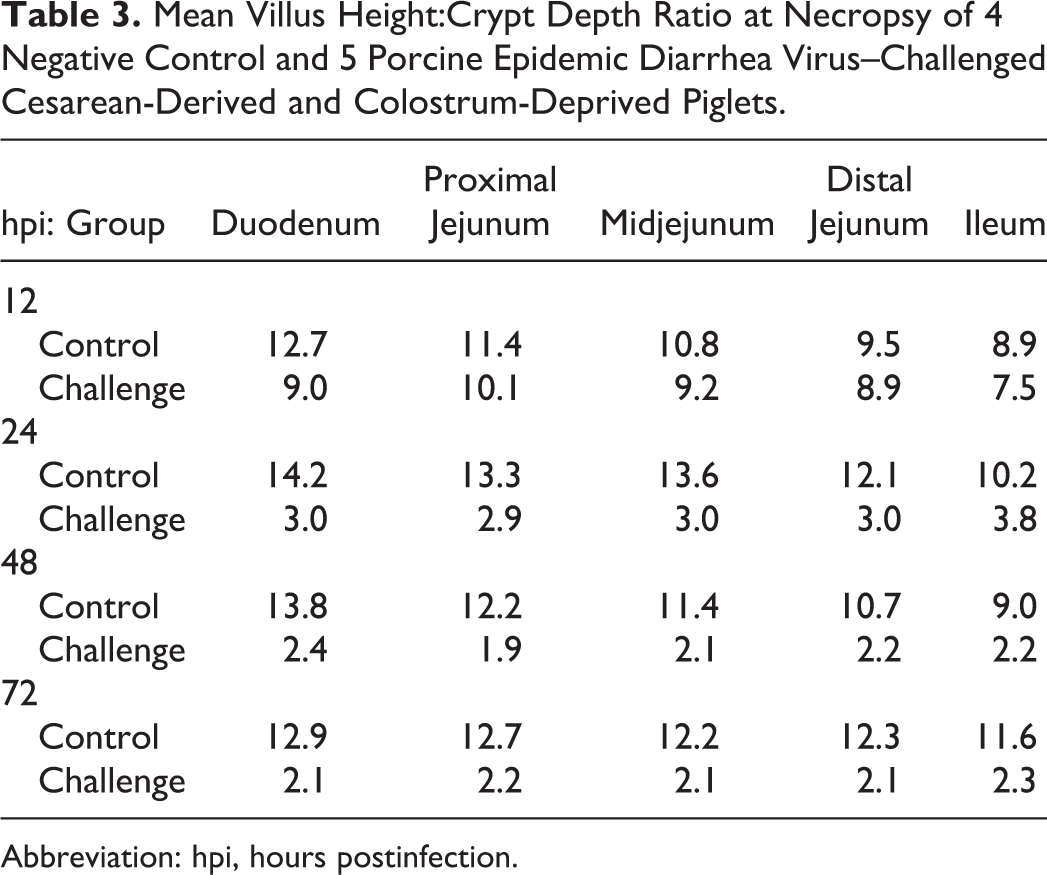
Abbreviation: hpi, hours postinfection.
PEDV antigen detection was detected by IHC in villous enterocytes of all challenged piglets euthanized at 12 hpi (Fig. 10). Staining was marked (score = 3) within duodenal and proximal and midjejunal sections and diminished to a few (score = 1) or no positive cells within the distal jejunum or ileum depending on the individual piglet. Immunolabeling of enterocytes in the distal jejunum and ileum was first visualized at the villus-crypt interface (Fig. 11). At 24 hpi, antigen was detected at moderate (score = 2) to marked amounts in all sections. At 48 hpi, PEDV antigen was detected by IHC in the duodenum of 3 of 5 piglets and the proximal jejunum of 4 of 5 piglets, respectively. At 72 hpi, all piglets and all intestinal segments were positive for PEDV antigen, with mild to marked staining depending on the section.
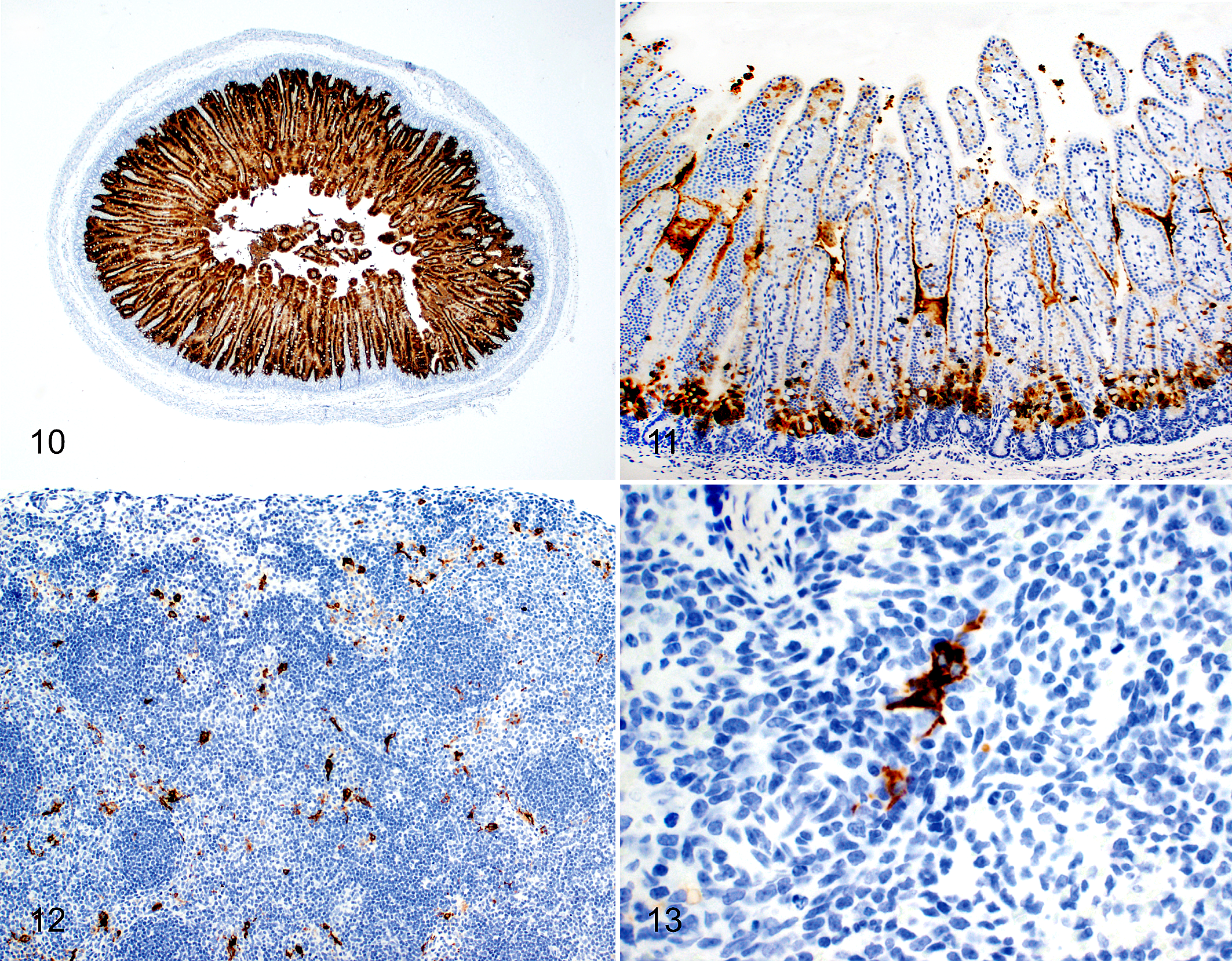
Porcine epidemic diarrhea, cesarean-derived and colostrum-deprived piglet. Immunohistochemistry for porcine epidemic diarrhea virus (PEDV).
PEDV antigen was not found in sections of lung, heart, kidney, liver, and stomach of any challenged pigs, regardless of time point. Positive staining was evident in 18 of 20 (90%) lymph node sections, 6 of 20 (30%) colon sections, and 3 of 20 (15%) splenic sections from challenged pigs. In lymph node and spleen, IHC staining was present in cells interpreted to be macrophages (Figs. 12, 13). Figure 14 demonstrates the mean IHC score by intestinal location for each time point for PEDV-challenged piglets.
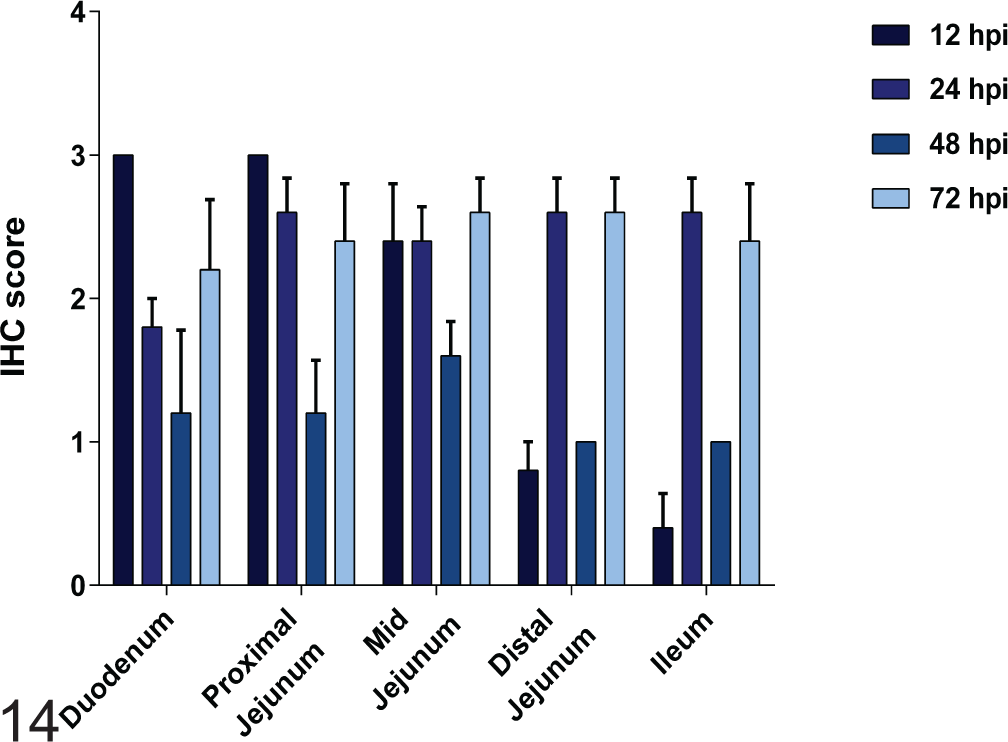
Scores for detection of porcine epidemic diarrhea virus antigen detection by immunohistochemistry (IHC) in duodenum; proximal, mid-, and distal jejunum; and ileum from porcine epidemic diarrhea virus–challenged cesarean-derived and colostrum-deprived piglets euthanized at varying number of hours postinoculation.
Discussion
PEDV unexpectedly entered the North American swine population in 2013, resulting in widespread and devastating disease. Understanding this virus and its infection characteristics has proven difficult. Specific objectives of this study were to determine the onset, progression, and degree of disease induced by infection with a prototype North American isolate of PEDV in neonatal CDCD piglets. Study outcomes broadly align with other previously published PEDV and TGEV challenge studies in neonatal swine 2,8,9 ; however, differences were observed among older reports suggesting that virulence can vary among PEDV isolates and that porcine epidemic diarrhea can be as severe as transmissible gastroenteritis in naïve neonates.
Onset of disease following PEDV inoculation in naïve neonatal swine is rapid. Virus isolate CV777 was the first PEDV characterized and evaluated in pathogenesis studies. 2,14 This isolate shares 96.9% nucleic acid homology with US/Iowa/18984 of 2013 and resulted in diarrhea 22 to 36 hpi in 2- to 3-day-old CDCD piglets when challenged oronasally with 2 ml of 104 pig-infectious doses, with antigen-positive intestinal cells and villus atrophy noted 18 and 24 hpi, respectively. 2 In a more recent investigation, 2 ml of Korean isolate SNUVR971496 (106.5 TCID50, orally dosed) produced diarrhea at 12 hpi, but initial IHC detection of antigen was not reported until 24 hpi. Differences in villus measurements between challenge and control piglets were also not observed until 24 hpi with this Korean PEDV isolate. 9 Results from the present study (103 PFU) differ in that both villous atrophy and antigen detection were observed 12 hpi with intense IHC staining in the upper small intestine of all examined piglets (Fig. 10). Test sensitivity, antibody affinity, antigen retrieval methods for IHC, and viral dose are all factors that may have affected these IHC findings, as methods differed among studies; however, clinical disease and microscopic lesions both developed earlier in the course of infection with PEDV isolate US/Iowa/18984 of 2013 compared to reported studies with either CV777 or SNUVR971496. This conceivable heightened replication of the North American PEDV isolate suggests the possibility of increased virulence compared to the historic CV777 European PEDV or some Asian viruses. Furthermore, US/Iowa/18984 of 2013 induces similar clinical signs and severity of disease as reported with recent Chinese PEDV strains, which have exhibited enhanced disease in endemic regions with increased morbidity and mortality. 10 Historical observations along with the results from this study imply virulence differences among PEDV isolates, similar to what has been reported for TGEV. 8
PEDV has been described as less virulent than TGEV 9,16 ; however, studies reporting reduced morbidity and mortality were conducted prior to the identification of the highly virulent Chinese PEDV strains documented in 2010. 10 Our results suggest similarities between TGEV and PEDV in terms of clinical disease and microscopic lesions. Specifically, diarrhea was reported earlier (18 hpi) in the current study compared to what was observed in the study conducted with 3 different TGEV isolates. 8 By 36 hpi in both studies, all piglets had severe diarrhea with anorexia and lethargy. Interestingly, vomiting was not reported in the previous TGEV study 8 but was apparent following PEDV inoculation in this experiment. A 75% reduction in villus height was noted at 24 hpi in both studies; however, at 12 hpi, the greatest reduction in villus height was in the jejunum of TGEV-infected piglets, in contrast to the duodenum and the ileum in the present study of PEDV (Fig. 2). These generated experimental data are in agreement with field reports from North America suggesting that lesions of PEDV and TGEV infection are indistiguishable. 19
PEDV replication in mature enterocytes is rapid, with previous investigations demonstrating antigen or detecting nucleic acid 24 hpi. 2,7,11 In the current study, PEDV antigen and RNA were detected at 12 hpi. It is possible that PEDV RNA detected by fecal swab collection at 12 hpi represented the inoculum as it passed through the intestine. However, more than 1 piglet would have been expected to be positive if this were true. This is the earliest report of detection of fecal RNA and intestinal immunolabeling of antigen. A limitation of these findings would be that PEDV disease progression and severity in the CDCD piglet model could be different from other models or field infection.
With regard to PEDV IHC labeling, Debouck et al showed a decrease in PEDV antigen-positive enterocytes from 12-45 hpi that subsequently increased thereafter. 2 Our data support this change in detection of PEDV antigen over time, diminishing from 12 to 48 hpi and then increasing at 72 hpi. This second peak of PEDV replication may represent infection of recently matured enterocytes derived from the crypts that are susceptible to PEDV entry and replication. 12 Interestingly, in the distal jejunum and ileum of PEDV-challenged piglets, enterocytes at the villus-crypt interface were the first cells to show positive staining in acute infection (Fig. 11). As the duration of infection increased, antigen-positive enterocytes within the intestine were observed toward the luminal surface of infected villi rather than the villus-crypt interface. It is unknown if enterocyte maturity affects infection or viral replication or if in the neonate has specific receptors located on recently matured enterocytes. Further studies are needed to understand if there are differences between enterocyte location and maturity that may influence PEDV entry or replication in these cells.
PEDV viremia was present in a high proportion (90%) of PEDV-challenged piglets at the time of euthanasia. The finding could be a consequence of gut permeability in 1-day-old piglets; however, it was recently reported that PEDV viremia can occur in the acute stage of infection of young inoculated gnotobiotic pigs and naturally infected 3- to 5-month-old commercial swine, thus suggesting that the young age of piglets in this study was not the reason for the high prevalence of viremia. 7 Detection of PEDV RNA in nonenteric tissue samples has not been reported and may well be a function of viremia. Although the identification of PEDV in extraintestinal tissues suggests the potential for avenues of virus transmission other than the fecal-oral route, this has yet to be demonstrated. 3,13 Beyond small intestinal samples, colon and mesenteric lymph node contained more viral RNA and were variably IHC positive, suggesting that the colon is a potential site of minor viral replication; furthermore, there is considerable immune cell phagocytosis along the intestinal tract, followed by antigen presentation in associated lymphoid tissue. PEDV antigen has been reported in the colon and mesenteric lymph node. 2,9
This report describes PEDV disease in neonatal piglets from an original North American isolate that is consistent with the severe death loss of naïve neonatal and weaning pigs due to PEDV infection noted in the field. When the current study results are compared with previous reports, these data suggest that clinical differences may exist among PEDV isolates and that porcine epidemic diarrhea has the potential to be as severe as transmissible gastroenteritis in neonates. Clinical disease associated with either TGEV or PEDV should be considered indistinguishable based on the findings of this study and what has been described by others.
Footnotes
Acknowledgements
We are grateful to the Iowa State University Veterinary Diagnostic Laboratory staff for their assistance with sampling and testing, the Laboratory Animal Resources staff for animal care, and numerous Iowa State University veterinary students for assisting with the animal procedures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding provided by the Iowa Pork Producers Association.
