Abstract
Xanthogranulomatosis is an inflammatory lesion characterized by lipid-containing macrophages, extracellular lipid, hemorrhage, and necrosis. We describe disseminated intracoelomic xanthogranulomatosis in 5 eclectus parrots (Eclectus roratus) and 2 budgerigars (Melopsittacus undulatus). Postmortem, clinicopathologic, and historical case material was reviewed. Ages ranged from 3 to 24 years; there were 5 males and 2 females. Table food was included in the diet of 3/5 cases, and animal products were included in 2/3 cases. Common clinicopathologic abnormalities included leukocytosis (4/5 cases) and elevated concentrations of bile acids (3/4 cases) and cholesterol within 6 months prior to death (2/4 cases). At postmortem examination, all 7 birds had grossly visible, irregular, soft, tan to yellow, amorphous plaques distributed on the surfaces of the viscera and body wall. Histologic evaluation and oil red O stain revealed xanthogranulomatous inflammation with phagocytized and extracellular lipid, necrosis, cholesterol clefts, fibrosis, and mineralization. Infectious agents were not identified with special stains in all cases. Concurrent hepatobiliary disease was present in 6/7 cases, and 6/7 had lipid accumulation within the parenchyma of various visceral organs. Five cases had atherosclerosis of great vessels. We describe a unique form of disseminated coelomic xanthogranulomatosis in 2 psittacine species. This condition should be recognized as a differential diagnosis in cases of disseminated coelomic mass formation and coelomic distension in psittacine birds, particularly in eclectus parrots and budgerigars.
Keywords
Xanthogranulomas are accumulations of cholesterol, cholesterol esters, and other lipids that result in a granulomatous inflammatory reaction; these lesions can be locally invasive. 1,10,11,20,23,26,31,33 In the avian literature, xanthoma, xanthomatosis, and xanthogranulomatous inflammation have been inconsistently defined. 26,33 Avian xanthoma is most frequently reported in psittacine and gallinaceous birds, and typically is attributed to trauma and chronic hemorrhage. Xanthomas form discrete, yellow, friable to firm masses in the skin and are composed of lipid-laden macrophages, multinucleate giant cells, free cholesterol, and fibrosis. 11,20,26,27,32,33 Commonly reported cutaneous sites in birds are the neck, wattles, back, distal wings, over the sternum, tibial region, and around the uropygial gland. 11,28,31 If possible, surgical removal is the treatment of choice. 23,26,33 While cutaneous and subcutaneous lesions are most commonly reported in birds, additional documented sites include the oral cavity, 29 conjunctiva, 36 tissues surrounding joints and bone, 9,16,28,38 and the viscera. 26,30 The term xanthomatosis indicates a less discrete mass; however, it is often also present within the skin and subcutaneous tissues. 11,23,26,27,33 Xanthogranulomas have characteristics of xanthomas in addition to more prominent granulomatous inflammation. 26,28,33 Because of the considerable morphologic overlap between these 3 terms, and to reduce confusion and inconsistency, some authors propose replacing xanthoma, xanthomatosis, and xanthogranuloma with a single term, “xanthogranulomatosis,” which is defined as a lesion in avian tissues containing macrophages with intracytoplasmic lipid vacuoles. 26,33
Xanthogranulomatous inflammation is reported in many other species, including humans, rabbits, cats, dogs, horses, amphibians, and reptiles. 1,8,10,12,14,15,18,21,25,34 In humans, xanthomatous conditions encompass a wide spectrum of diseases and include those with and without hyperlipidemia. 10,12,19,24,26 Some authors hypothesize that almost all types of xanthogranulomatous reactions are caused by lipid phagocytosis resulting from hemorrhage, necrosis, or suppurative inflammation, with the exception of hyperlipemic xanthomas derived from leakage of plasma lipoproteins and immunologic or storage disorders. 10 Regardless of the underlying cause, once macrophages undergo phagocytosis of lipid, the cause and pathogenesis of the subsequent inflammatory response is difficult to discern. 10,12,24,26 Although avian xanthogranulomatosis has been attributed to hyperlipidemia in some cases, 20,23,36 xanthoma and xanthogranulomatous inflammation involving the skin and subcutis, viscera, or periosseous regions is also documented in normolipemic birds. 11,26,28,33
Cytologically, xanthogranulomatosis is characterized by lipid-containing macrophages and small numbers of other inflammatory cells, including lymphocytes, eosinophils, neutrophils/heterophils, and extracellular or phagocytized rhomboid, pale-staining cholesterol crystals. 1,11 Histologic lesions include granulomatous inflammation with variably sized macrophages and multinucleated giant cells with intracytoplasmic lipid, amorphous extracellular lipid deposits, cholesterol clefts, and variable pleocellular inflammation. 1,10,14,32
The objective of this study was to describe disseminated coelomic xanthogranulomatosis in 5 eclectus parrots (Eclectus roratus) and 2 budgerigars (Melopsittacus undulatus).
Materials and Methods
The Animal Medical Center (New York, NY) postmortem database was searched for psittacine cases diagnosed with xanthogranulomatous coelomitis or xanthogranulomatosis, which yielded 4 cases involving 3 eclectus parrots and 1 budgerigar. The coauthors (MMG, DP, DR, SM, SC, CE) identified 3 additional cases of disseminated xanthogranulomatous inflammation in eclectus parrots or budgerigars from 3 other institutions; no cases of disseminated xanthogranulomatous coelomitis were identified in any other psittacine species at these 4 institutions. Material from the 7 cases was reviewed including history, dietary information, imaging findings, clinicopathologic findings, and gross and histologic lesions. Diagnostic imaging findings from 4 of these cases are described elsewhere. 17
Results
Clinical Findings
Signalment, diet, and clinicopathologic findings are summarized in Table 1. Disseminated coelomic xanthogranulomatosis was identified in 5 eclectus parrots and 2 budgerigars. Five cases were male and 2 were female. Ages ranged from 3 years to 24 years, with 1 bird of unknown age.
Signalment, diet, and clinicopathologic findings in 5 eclectus parrots and 2 budgerigars with xanthogranulomatosis.
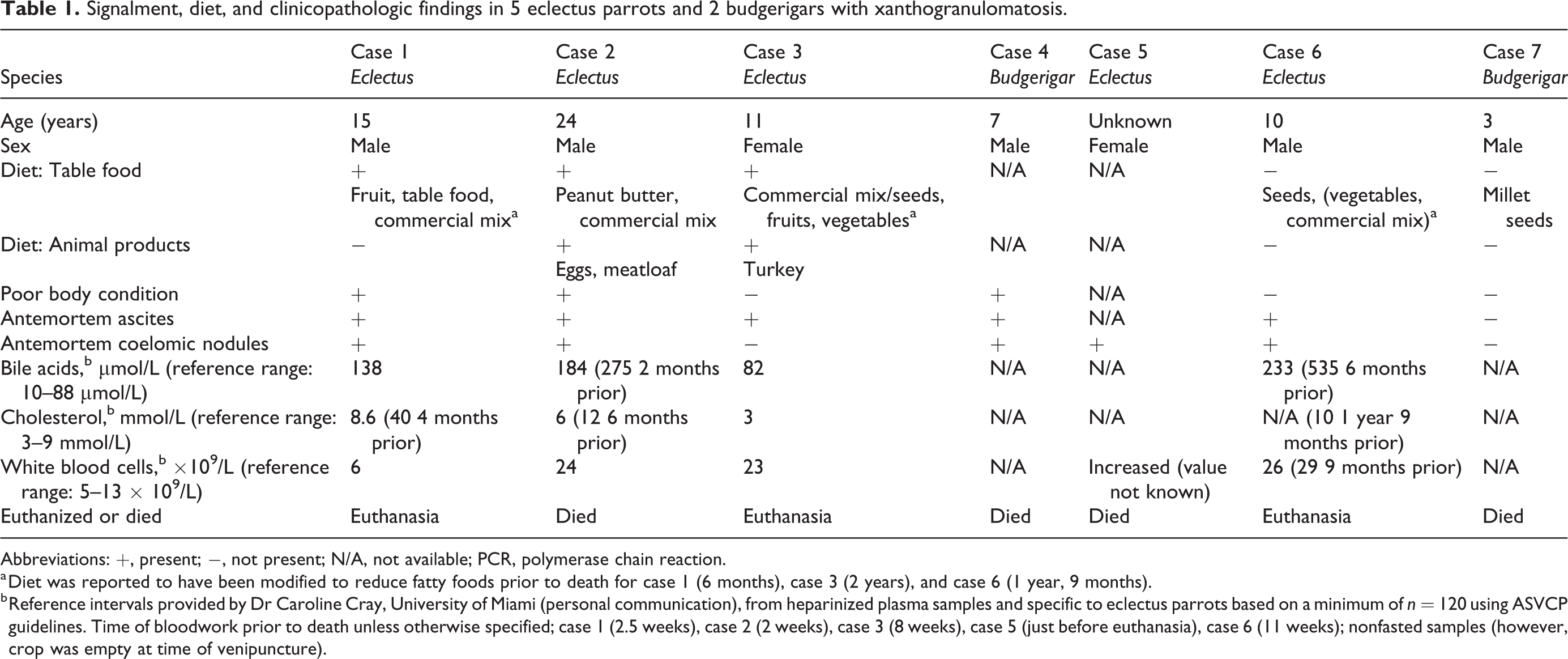
Abbreviations: +, present; −, not present; N/A, not available; PCR, polymerase chain reaction.
a Diet was reported to have been modified to reduce fatty foods prior to death for case 1 (6 months), case 3 (2 years), and case 6 (1 year, 9 months).
b Reference intervals provided by Dr Caroline Cray, University of Miami (personal communication), from heparinized plasma samples and specific to eclectus parrots based on a minimum of n = 120 using ASVCP guidelines. Time of bloodwork prior to death unless otherwise specified; case 1 (2.5 weeks), case 2 (2 weeks), case 3 (8 weeks), case 5 (just before euthanasia), case 6 (11 weeks); nonfasted samples (however, crop was empty at time of venipuncture).
Of the 5 cases in which dietary history was available, table food was given to 3 cases (all of which were eclectus parrots; Table 1). In 2 of these 3 cases, animal-based food (eggs or meat) was listed as a portion of the diet. The percentage of table foods was only known for case 3, for which table food including fruit and vegetables comprised 40% of the diet. The diets of 3 cases (all eclectus parrots) had been historically modified to reduce fatty foods including seeds. One bird (a budgerigar) had a diet consisting mainly of millet seeds.
Body condition was listed as poor based on palpation of the pectoral musculature in 3 of 6 birds for which body condition was described. Two cases had a previous history of obese body condition. Five of 7 cases presented with ascites and hepatic or coelomic nodules were identified antemortem in 5 cases. Dyspnea was reported in 4 cases, with regurgitation in 1 case. Nonspecific signs included decreased appetite, lethargy, and weight loss in 3 birds (detailed case information available in Supplemental Table S1). Clinical information for 4 birds is detailed elsewhere. 17
Antemortem Case Investigations
Partial or complete hematology data were available for 5 birds (Table 1). Case 1 had a weak positive antibody titer for Aspergillus (Aspergillus antibody 1.4 weak positive, galactomannan negative; Avian and Wildlife Laboratory, University of Miami, Miami, FL, USA). Two cases had radiographic and ultrasonographic evidence of intestinal dilation and were positive on fecal polymerase chain reaction (PCR) testing for avian bornavirus (Veterinary Molecular Diagnostics Laboratory, Milford, OH, USA), whereas 1 case was negative. Clostridial-like organisms were identified on a fecal Gram stain in 2 cases. Case 6 tested negative for Mycobacterium sp on PCR testing of coelomic fluid, blood, and feces.
Cytologically, coelomic effusion from case 6 was a modified transudate containing vacuolated macrophages and fewer heterophils and lymphocytes; no crystals were observed. A liver biopsy from the same case (case 6) consisted of inflamed granulation tissue with granulocytes and foamy macrophages, necrotic tissue (loss of nuclear detail), karyorrhectic cell debris, erythrocytes, and mineralization, but viable hepatic tissue was not observed.
Postmortem Findings
Three birds were euthanized and 4 birds died, one of which was clinically normal before death (case 7). All 7 cases had disseminated inflammation throughout the coelomic cavity, characterized by accumulations of irregular, soft, tan to yellow, amorphous material distributed over the visceral surfaces of the coelomic organs and parietal surfaces of the body wall (Figs. 1–6). This material was interpreted to represent the coelomic mass lesions identified with imaging for five of the cases. Subcutaneous masses were soft to firm, tan to yellow, and ranged from 1.5 to 3.0 cm in diameter. Ascites was present in 3 of 6 cases.
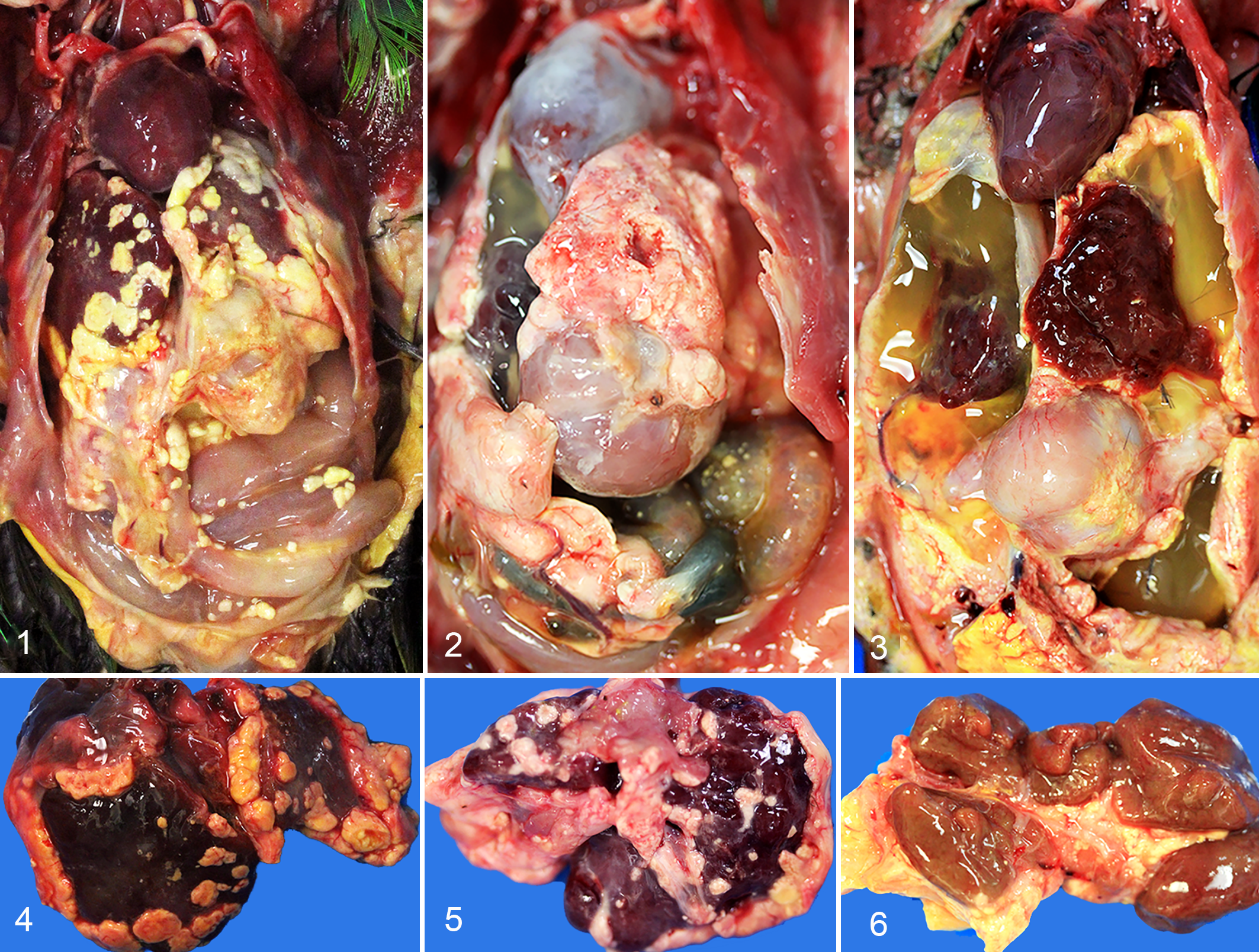
Disseminated coelomic xanthogranulomatosis, eclectus parrots and budgerigars.
Cytologic evaluation of postmortem impression smears of the coelomic plaques and subcutaneous masses was performed in cases 1 to 3. Cytologic features included large numbers of refractile, rectangular, and acicular crystals (Fig. 7), the morphologies of which were not consistent with cholesterol crystals. Numerous macrophages contained well-demarcated, round vacuoles (consistent with lipid; Fig. 7) and fewer additional inflammatory cells (lymphocytes, plasma cells, and heterophils) were interspersed with blood. Mild polychromasia of erythrocytes was present in case 3.
Histologically, xanthogranulomatous inflammation involved the surfaces of multiple coelomic organs and parietal peritoneum (Figs. 7–15, Table 2). Accumulations of macrophages contained discrete, round vacuoles (consistent with lipid) and were intermixed with amorphous, light pink, acellular material and karyorrhectic cell debris (necrosis), cholesterol clefts, and fragmented, basophilic, acellular material (mineral) that was confirmed by Von Kossa stain in cases 1 to 3 (Figs. 7–15). Eosinophilic fibrous connective tissue with a hyaline appearance was present in 3 cases (Fig. 9), all of which were negative for amyloid with Congo red stains. Intermixed with the macrophages were fewer inflammatory cells (heterophils, lymphocytes, plasma cells). The presence of intracellular and extracellular lipid was confirmed with oil red O stain performed on formalin fixed, nonprocessed, frozen tissues in 5 of 7 birds (Fig. 14). Gomori’s methenamine silver stain for fungi and Ziehl-Neelsen or Fite faraco stain for acid-fast bacteria were negative in all 7 birds. In the 2 budgerigars, visceral plaques predominantly comprised sheets of foamy, lipid-containing macrophages (Fig. 14, inset).
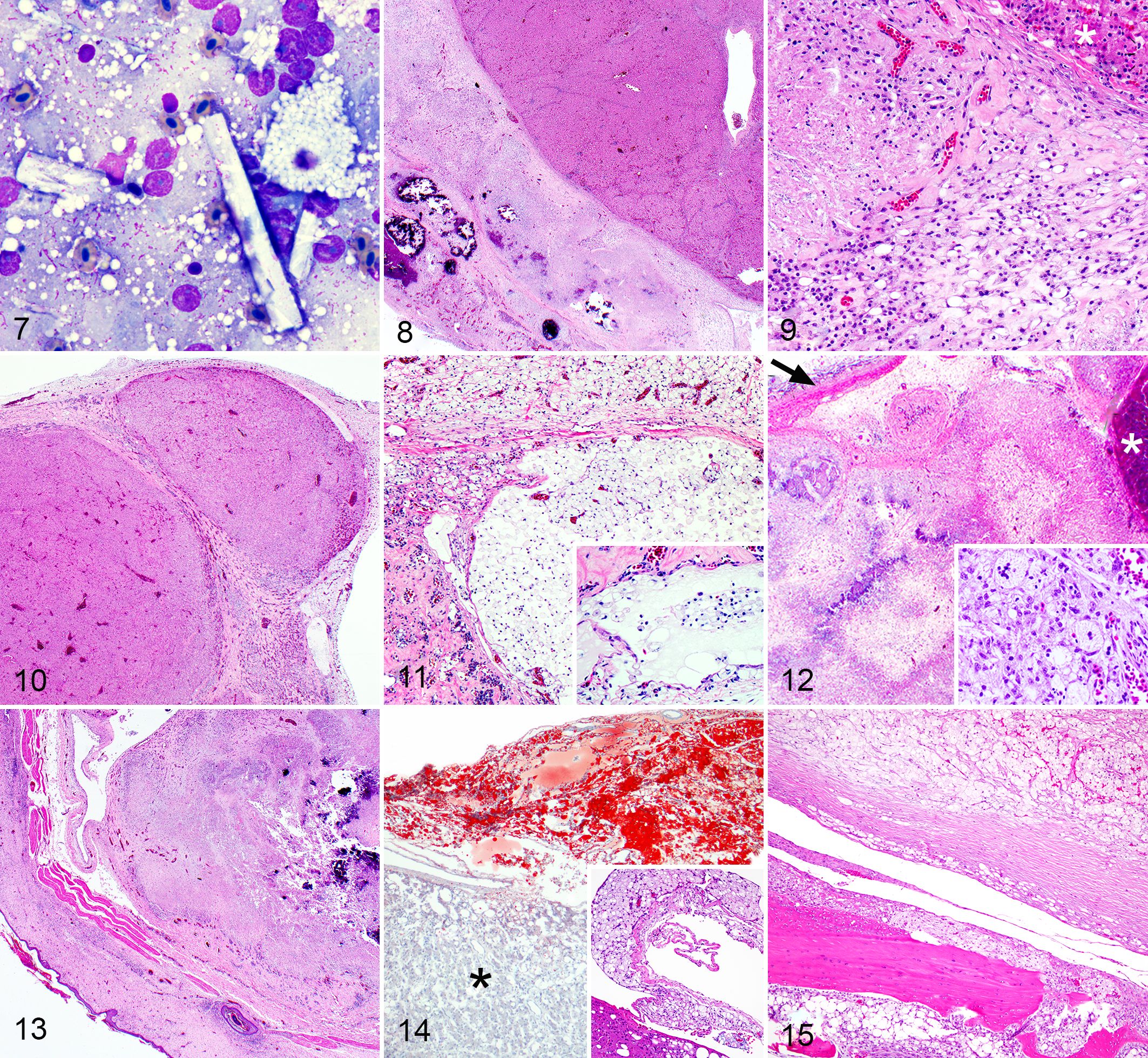
Xanthogranulomatous coelomitis, eclectus parrots and budgerigars.
Histologic findings in 5 eclectus parrots and 2 budgerigars with xanthogranulomatosis.

Abbreviations: +, present; −, not present; N/A, not available; LP, lymphoplasmacytic; H, heterophilic; L, lymphocytic.
One budgerigar (case 4) had cysts within or beneath the hepatic capsule that were bordered by fibrous connective tissue and contained numerous foamy macrophages with intracytoplasmic, discrete, round vacuoles (lipid, Fig. 11), and structures interpreted as veins or lymphatics contained similar foamy macrophages (Fig. 11, inset). In case 5, a cystic structure adhered to the capsule of the liver contained vacuolated macrophages and a large central core of acicular (cholesterol) clefts. Subcutaneous nodules (cases 2 and 7) were histologically similar to the visceral plaques. Case 7 (a budgerigar) had a large subcutaneous mass affecting the ventral abdomen that had central necrosis bordered by fibrous connective tissue septa. Lipid-containing macrophages infiltrated into the subjacent sternum with bone lysis and reactive woven bone formation (Fig. 15). Xanthogranulomatous inflammation adhered to the sternum in case 6.
Lipid accumulation was present in the parenchyma of some visceral organs including adrenal cortical cells in 4/7 birds, the lungs (parabronchial epithelium and macrophages) in 4/7 birds, the kidneys (proximal tubules) in 3/7 birds, the liver (hepatocytes) in 2/7 birds, the spleen (macrophages within the capsule and parenchyma) in 2/7 birds, and the gastrointestinal tract (lamina propria macrophages, vessels, smooth muscle) in 1/7 birds.
Concurrent atherosclerosis was present in the great vessels in 5 of 7 birds (Table 2), assessed as grade IV in 2 cases, grade III in 1, grade II in 1, and grade I in 1 case. 5 The most common comorbidity involved the liver. Specifically, cholangitis or cholangiohepatitis in 6 birds (Table 2) and end-stage hepatic disease with inflammation, fibrosis, and nodular regeneration were present in 1 case (Fig. 10). In case 2, the pancreas was obscured by the coelomic xanthogranulomatous inflammation. The pancreas was normal in 5 other birds and was not evaluated in 1 bird. Inflammation within the intestinal wall including the autonomic nervous system was present in case 1 (which tested positive for avian bornavirus); however, primary ganglioneuritis could not be distinguished from extension of coelomic inflammation into the ganglia. The cause of death or decision for euthanasia was attributed to xanthogranulomatosis in all cases.
Discussion
In this case series, we describe a unique form of disseminated coelomic xanthogranulomatosis in 7 psittacine birds including 5 eclectus parrots and 2 budgerigars. The gross, cytologic, and histologic appearances were similar in all cases, consisting of yellow to tan plaques or masses formed by lipid-containing macrophages with variable amounts of necrotic cell debris, cholesterol clefts, and mineral. Specials stains were negative for infectious agents in all 7 cases. Avian practitioners and veterinary pathologists should be made aware of coelomic xanthogranulomatosis in these psittacine species, as this disease has nonspecific clinical signs and imaging findings. 17 Features that were shared by many of the cases were antemortem ascites (5/7 birds), antemortem identification of coelomic nodules (5/7 birds), concurrent hepatic disease involving the biliary tree (6/7 birds), high concentrations of bile acids (3/4 birds), cholesterol (2/4 birds), leukocytosis (4/5 birds), and atherosclerosis (5/7 birds). Coelomic xanthogranulomatosis resulted in morbidity and was interpreted as the major contributor or cause of death or decision for euthanasia in all cases. End-stage hepatic disease contributed to the death of 1 budgerigar.
The chronic progressive nature of xanthogranulomatosis combined with radiographic evidence of organomegaly, coelomitis, and/or the presence of intracoelomic soft tissue masses, coupled with a significant leukocytosis, are findings that might be associated with other chronic inflammatory diseases including aspergillosis, mycobacteriosis, egg yolk peritonitis, and diffuse neoplastic disease. The only way to definitively rule out these other differential diagnoses is by obtaining biopsies with microscopic examination of the affected tissues.
The cytologic and histologic features were compatible with the term xanthogranulomatosis because of the presence of widely disseminated phagocytized lipid within macrophages. 26,33 Cytologic examination of impression smears collected at necropsy allowed identification of lipid-containing macrophages, along with crystalline material, even in autolyzed tissue. Notably, the crystalline material was not cytologically consistent with cholesterol crystals, despite the presence of cholesterol clefts in the histologic specimens. This may indicate that the crystalline material does not represent cholesterol or is an atypical manifestation of cholesterol accumulation occurring with disseminated xanthogranulomatosis.
The histologic pattern of inflammation differed between the 2 species. In the eclectus parrots, the macrophages were interspersed with large amounts of necrotic cell debris, which contained cholesterol clefts and dystrophic mineralization. In contrast, coelomic material from the 2 budgerigars predominantly consisted of sheet-like aggregates of lipid-containing macrophages, with less necrotic cell debris and an absence of dystrophic mineralization. In 1 budgerigar, a large subcutaneous nodule exhibiting central coagulative necrosis with lysis and infiltration into the sternum may more accurately represent a necrotic infiltrative lipoma or liposarcoma as opposed to disseminated xanthogranulomatosis.
In cases 4 (a budgerigar) and 5 (an eclectus parrot), hepatic cysts were observed. Therefore, visceral xanthogranulomatosis can result in cyst formation that may potentially be visualized with antemortem diagnostic imaging. Lipid-containing macrophages were rarely present within the lumen of vessels in 1 case, suggesting that thromboembolism is a potential consequence of xanthogranulomatosis, although thrombosis or infarcts were not present in any of the cases. Lipid accumulation was most prominent in the grossly observed tan to yellow aggregates in coelomic tissues; however, some birds also showed lipid accumulation within parenchymal cells (lipidosis) of visceral organs, the most frequent of which were the adrenal glands, lungs, and kidneys. It is important to note that there are additional causes of adrenal gland vacuolation and that oil red O stains did not specifically confirm the presence of lipid within these parenchymal cells (as opposed to the coelomic material). Lipidosis in the liver was only present in 2 cases.
A specific cause for the disseminated coelomic xanthogranulomatosis in these 7 cases was not identified. Avian xanthomatosis is also of uncertain etiology 11,20 but is reported to result from hypercholesterolemia and hypertriglyceridemia, 23,36 fat-related toxic hydrocarbons in feed, 31 trauma, 11,26 and infection. 35 There was no evidence for an infectious cause in these cases. A species predisposition to disseminated coelomic xanthogranulomatosis cannot be determined in this series due to the small case numbers. Since many other psittacine species are prone to lipid-related disorders, additional investigation regarding possible susceptibility of these 2 species may be indicated. 2,3,7,13,32 Given the small case numbers, no conclusions can be drawn about sex predominance. In avian and other species, both females and males are reported to be predisposed to lipid disorders, including atherosclerosis, lipidosis, and xanthogranulomatosis. 3,13 –15,32 For example, a predisposition to xanthoma formation in the skin has been noted in female budgerigars, 32 lipidosis in organs, and atherosclerosis has been noted in female budgerigars, 13,32 while male Quaker parrots (Myiopsitta monachus) have been reported to be more susceptible than females to other lipid-related disorders including atherosclerosis and hepatic lipidosis. 7 Additional information is needed to determine whether the male or female sex is a possible risk factor for coelomic xanthogranulomatosis in eclectus parrots or budgerigars.
Cholangitis or cholangiohepatitis was prevalent in this group of birds and 1 had lesions of end-stage hepatic disease. Some of these cases also had high plasma concentrations of bile acids and cholesterol (species-specific reference ranges provided by Dr Caroline Cray, personal communication). Hepatobiliary disease in birds can lead to changes in the plasma lipid fractions. In avian species, the liver synthesizes apolipoproteins and acts as a source of cholesterol and lipid. 2 Biliary obstruction in birds is also known to increase plasma cholesterol levels. Although speculative, especially given the variations in severity, hepatobiliary disease may have contributed to dyslipidemia in these birds.
Four cases had leukocytosis, possibly resulting from hepatobiliary and/or xanthogranulomatous inflammation. In 1 bird, a suspected Aspergillus infection may have exacerbated the leukocytosis; however, no gross or histologic evidence of fungal infection was present at postmortem examination.
Atherosclerosis was present in 5 birds of this case series, graded as at least III/VII in 3 cases. 5 Atherosclerosis is common in birds, with a high incidence of spontaneous lesions in psittaciform species kept as household pets. 3 –6 The great vessels and carotid arteries are most commonly affected. 3 In 2 large-scale studies in psittacine birds, risk factors for development of advanced atherosclerotic lesions (graded IV–VI based on an adaptation of a human classification system) 5 were age, female sex, reproductive diseases, hepatic disease, and species (Amazon parrots, African grey, and cockatiels). 3 –5 In that study, eclectus parrots and budgerigars were not evaluated. 3 The presence of atherosclerosis in addition to widespread xanthogranulomatosis in these cases suggests a possible association or shared etiopathogenesis for these 2 conditions. The combination of xanthogranulomatosis and atherosclerosis have been reported in animal models of human diseases with genetic susceptibility to cholesterol-induced atherosclerosis, including quail and some strains of mice. 20,37
Review of the dietary history revealed that 3 of 5 birds were eating human table foods including animal products in 2 cases (eg, eggs and meat), with efforts made to alter the diets by reducing fatty food in 3 cases as a result of clinical obesity. Fatty foods comprised seeds in 3 cases, and in 1 case that died suddenly, the diet consisted almost entirely of Millet seeds. Dyslipidemia is defined as any abnormality of the plasma lipid profile, which in birds can manifest with increases of high-density lipoproteins (HDLs), the main carrier lipoprotein (as opposed to low-density lipoproteins [LDLs] in humans). 2,3 Familial dyslipidemias in humans are often the result of defective LDL receptors, enzymes, or apolipoproteins. Experimentally induced xanthogranulomas have been reported in birds, rabbits, and mice deficient in apolipoprotein E or LDL receptors when fed a diet high in cholesterol. 1,10,12,16,22,25,37 Although the presence of elevated cholesterol and triglyceride levels in some of these cases suggests dyslipidemia, HDL levels were not specifically assessed, and therefore a definitive evaluation of the lipid fraction profile in these birds was not possible. Given the natural and experimental associations with high dietary lipid or cholesterol intake, high body fat, and xanthoma formation in birds and other species, 1,10,12,14,16,25 suboptimal diet may be a possible predisposing or contributing factor in the development of xanthogranulomatosis in these 2 avian species.
Conclusions
This study characterizes a unique form of disseminated coelomic xanthogranulomatosis in eclectus parrots and budgerigars. Shared features included ascites, coelomic nodules, concurrent hepatobiliary disease, atherosclerosis, and inflammatory leukograms. Avian clinicians and pathologists should be aware of this condition as a differential diagnosis for birds with ascites and coelomic nodules and strive for a consensus of terminology for lipid-containing inflammatory lesions so that this unusual disease can be further characterized.
Supplemental Material
Supplemental Material, sj-docx-1-vet-10.1177_03009858211045931 - Disseminated coelomic xanthogranulomatosis in eclectus parrots (Eclectus roratus) and budgerigars (Melopsittacus undulatus)
Supplemental Material, sj-docx-1-vet-10.1177_03009858211045931 for Disseminated coelomic xanthogranulomatosis in eclectus parrots (Eclectus roratus) and budgerigars (Melopsittacus undulatus) by Taryn A. Donovan, Michael M. Garner, David Phalen, Drury Reavill, Sebastien Monette, Alexandre B. Le Roux, Melissa Hanson, Sue Chen, Cyndi Brown, Catalina Echeverri and Katherine Quesenberry in Veterinary Pathology
Footnotes
Acknowledgements
We thank Ms Amanda Ramkissoon, Ms Kim McBride, and Ms Jacqueline Candelier for data collection and for performing special stains. We also thank Dr Timothy Wright, Department of Biology, New Mexico State University, for historical dietary information; Cathy Minogue of Northwest ZooPath for data retrieval; and Leroy Brown of Histology Consulting Service, Everson, WA, for slide preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
