Abstract
A multiorgan infection with a Coxiella-like organism was determined to be the cause of death of a female eclectus parrot(Eclectus roratus). The diagnosis was based on gross lesions, histopathology, Gimenez and Gram special stains, immunohistochemistry, electron microscopy, and polymerase chain reaction amplification and sequencing of a bacterial 16s rRNA gene fragment isolated from hepatic and cardiac tissue. Gross postmortem examination revealed multifocal to coalescing foci of hepatic necrosis. The most significant histologic lesions included multifocal lymphohistiocytic necrotizing hepatitis, locally extensive lymphoplasmacytic myocarditis, and myocardial degeneration and necrosis. Intralesional cytoplasmic organisms were identified in cardiomyocytes, biliary epithelium, and pancreatic exocrine cells. This is the first description of a Coxiella-like organism with wide-ranging cellular tropisms in a psittacine bird. In addition, lymphoplasmacytic neuritis, myositis, splenitis, airsacculitis, and enteritis were detected. It is also the first report of a Coxiella-like infection in an eclectus parrot.
Coxiella burnetii, the etiologic agent of Q fever, infects a variety of animal species and humans.1,6,9,10 It is an obligate intracellular Gram-negative bacteria that is phylogenetically related to Legionella, Francisella, and Rickettsia spp and resides within a parasitophorous vacuole in the cellular cytoplasm. 10 C. burnetii has been identified in many species of mammals, birds, reptiles, and arthropods. 3 Illness associated with bacterial infection is well documented and varies greatly between and within species, ranging from inapparent to severe disease.1,9,10 Avian Coxiella-like infections have been described recently in a toucan and 8 psittacines, including 3 lorikeets.9,10 Clinical signs of the affected birds ranged from inapparent illness to severe lethargy and weakness, altered mental status, seizures, and sudden death.9,10 Gross findings in the affected birds included emaciation with hepatomegaly and splenomegaly.9,10 Histologic examination of the liver revealed multifocal necrosis of hepatocytes with infiltration of a mixed population of inflammatory cells, including lymphocytes, heterophils, plasma cells, and macrophages. Infected macrophages often contained basophilic intracytoplasmic inclusions. Increased numbers of macrophages were present in the spleens of affected birds and contained similar intracytoplasmic basophilic inclusions. 9 A similar inflammatory infiltrate was present in multiple anatomic locations, including the epicardium, endocardium, pulmonary interstitium, kidney, adrenal and thyroid glands, intestine, brain, bursa of Fabricius, and bone marrow.9,10 Histologically affected avian tissues were negative for C. burnetii antigen expression by immunohistochemistry (IHC), but ultrastructural morphology consistent with Coxiella spp was detected by electron microscopy. Polymerase chain reaction (PCR) amplification of the bacterial 16s rRNA gene, performed with genomic DNA isolated from brain and liver tissues and subsequent sequencing, revealed a 97.3% identity with an unnamed Coxiella sp isolated from Rhipicephalus sanguineous and 97% identity with the sequence of C. burnetii isolate.9,10 This avian isolate has been referred to as a Coxiella-like microorganism.9,10
This report describes a fatal systemic infection in a captive eclectus parrot by a Coxiella-like organism that was morphologically similar to the agent reported to cause infection in several avian species.9,10 In addition, a portion of the 16s rRNA gene was determined to be genetically identical to the same agent. However, unlike the previous reports documenting a disseminated intrahistiocytic infection, the Coxiella-like agent in this eclectus parrot had a unique cellular tropism that, in addition to macrophages, included cardiac myofibers, biliary epithelium, hepatocytes, and pancreatic exocrine cells.
Anamnesis
A 2-year-old female eclectus parrot was submitted to the California Animal Health and Food Safety Laboratory at the University of California, Davis, after being found dead in its cage. The bird had been owned for 11 months and was the only bird on the property. Other animals on the premises included a single cat, 3 dogs, a fish, and 3 horses. The parrot was kept in a large cage measuring 1.2 × 0.9 × 0.9 m, with newspaper bedding that was changed daily. The cage was placed next to the window, which occasionally remained open. The bird was fed a variety of fruits, vegetables, and cooked meats in addition to commercial mixed seeds. Normally, the parrot was loquacious, actively mimicking numerous sounds. Four weeks prior to death, these vocalizations ceased. Two weeks prior to death, the bird adopted a squatting posture and failed to perch. A few days prior to death the bird became lethargic but continued to eat.
A complete necropsy was performed, and a complete set of tissues was collected, including liver, kidney, heart, spleen, sciatic nerve, trachea, skeletal muscle (pectoral and quadriceps), thyroid gland, thymus, air sac, ovary, oviduct, adrenal gland, multiple sections of gastrointestinal tract, and brain. The tissues were fixed in 10% neutral buffered formalin and processed routinely for histologic examination. Aerobic bacterial culture was performed on the pulmonary and hepatic tissue samples, and Salmonella enrichment culture was performed on feces. Tracheal swabs were submitted for avian paramyxovirus 1 and avian influenza virus matrix gene screening by real-time PCR according to National Veterinary Services Laboratory protocols (AV1505.05 and AVPRO1510.04). In addition, cytologic impression smears of liver, air sac, and spleen were screened for Chlamydophila antigens by florescent antibody according to the manufacturer instructions (D3 DFA Chlamydia Reagent, catalog No. 01-045005, Diagnostic Hybrids, Athens, OH) and examined with a fluorescence microscope. An additional Gimenez-stained impression smear was screened for Chlamydophila and Coxiella-like organisms. Virus isolation was performed on a tissue pool of lung, liver, kidney, spleen, and brain, as previously described. 9 IHC assays for detection of Chlamydophila psittacii and C. burnetii (Integrated Diagnostics, Inc., Baltimore, MD) were performed on paraffin-embedded sections as previously described, except for the addition of avidin–biotin blocking solution according to the manufacturer’s instructions (Vector Laboratories Inc., Burlingame, CA) after the 0.5% casein-blocking step. 10 Immunohistochemical analyses for West Nile Virus 2 (WNV) antigen and in situ hybridization for psittacine polyomavirus nucleic acid were performed on heart and kidney. 7 A panel of special stains was performed on multiple tissue sections: modified Steiner’s, tissue Gram, periodic acid–Schiff, Gimenez, Hall’s bile, and Ziehl-Neelsen stains. For transmission electron microscopy, thin sections of formalin-fixed hepatic and cardiac tissues were postfixed (approximately 1 week following the initial formalin fixation) in a modified Karnovsky’s fixative, 1.5% osmium tetroxide, stained en bloc with uranyl acetate, processed routinely, and examined with a Leo 906E transmission microscope (Leo Electron Microscopy, Oberkochen, Germany).
Genomic DNA was isolated from paraffin-embedded cardiac and hepatic samples using a commercial DNA extraction kit (QIAamp DNA FFPE Tissue Kit, Qiagen, Valencia, CA). PCR was performed utilizing previously described primers and method (forward primer: 5′-GTAGTCCACGCTAACGA; reverse primer: 5′-CTGTCACTCGGTTCCA). 10 Cycling conditions were as follows: 95°C for 2 minutes, followed by 25 cycles of 95°C for 15 seconds, 56°C for 30 seconds, 72°C for 30 seconds, and a final elongation step at 72°C for 5 minutes. Genomic DNA isolated from an IHC-confirmed case of a C. burnetii infected goat placenta was used as positive control, while molecular-grade water and DNA isolated from a pool of 3 tissues from an unaffected eclectus parrot were utilized as negative controls. The amplified DNA was gel isolated (Agarose Gel Isolation Kit, Thermo Fisher Scientific, Waltham, MA) and sent for sequencing through a local vendor (Davis Sequencing, Davis, CA).
Diagnostic Investigations
The bird was in good postmortem condition, weighing 350 g with moderate atrophy of the pectoral musculature. The bird’s weight several months prior to death was essentially the same. The liver was of normal size and shape with widespread, randomly distributed yellow irregular necrotic foci (diameter, 1–2 mm) scattered throughout the hepatic parenchyma (Fig. 1 ). The spleen was small and light pink. No other gross lesions were observed.
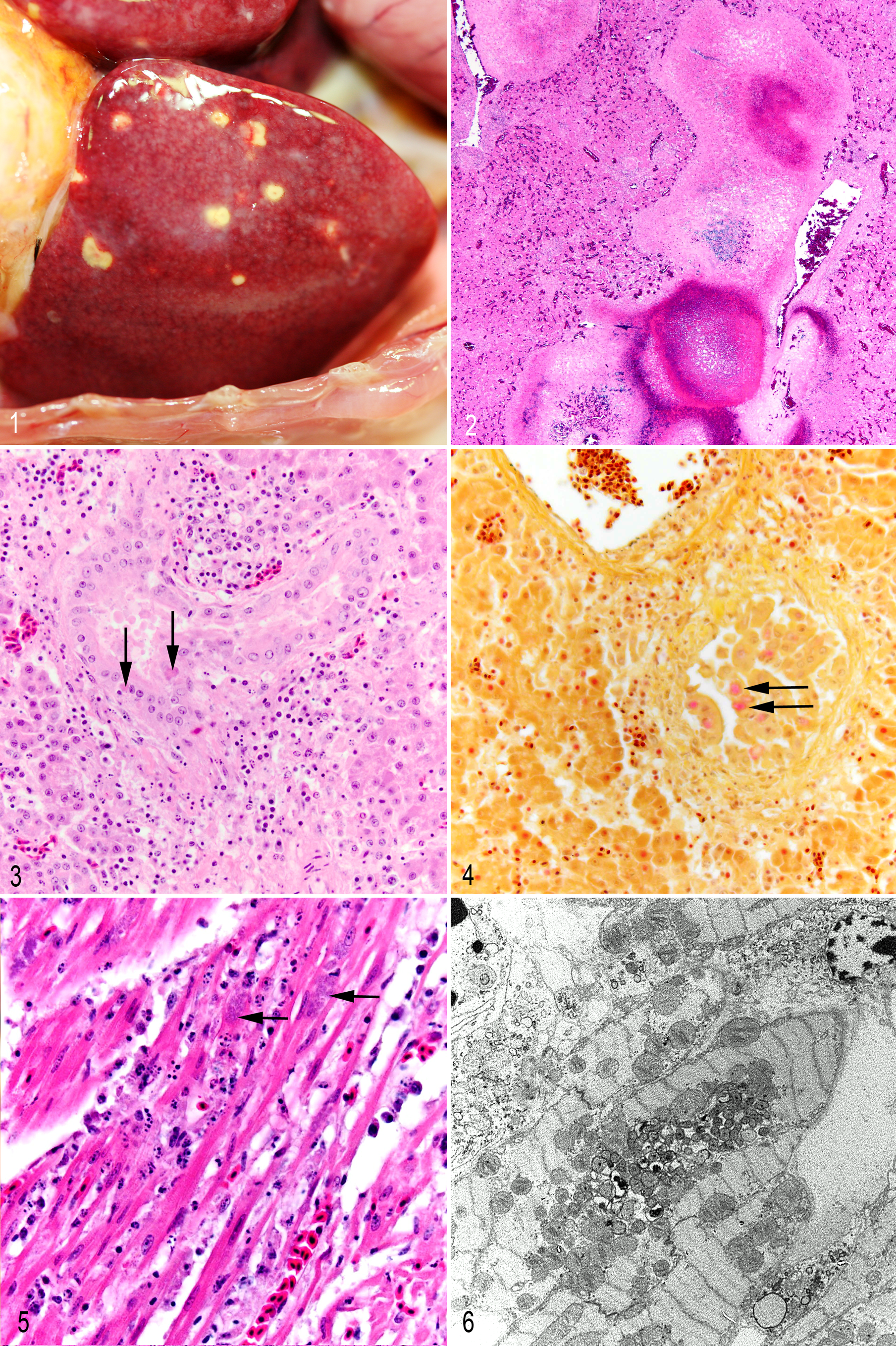
Microscopically, the hepatic parenchyma had prominent diffuse mixed inflammatory cell infiltrates consisting predominantly of macrophages, with smaller numbers of plasma cells, lymphocytes, and heterophils. The leukocytes dissected along perisinusoidal spaces formed dense focal infiltrates within the parenchyma and infiltrated throughout portal tracts and portal veins (vasculitis). Large irregular foci of hepatic caseous necrosis, often with central foci of hemorrhage and fibrin deposition, were rimmed by infiltrates of macrophages and/or epithelioid macrophages admixed with heterophils, lymphocytes, and plasma cells (Fig. 2). At the periphery of these foci of necrosis, there often were multiple biliary duct profiles surrounded by edematous, fibrotic stroma with variable numbers of mixed inflammatory cell infiltrates. The biliary epithelium was often disorganized, hyperplastic, and hypertrophied (biliary regeneration; Fig. 3). Inflammatory cells cuffed bile ducts but were not found in epithelium or lumen. A mixed mononuclear inflammatory cell infiltrate was present in the mucosa and muscularis of a single major bile duct. Random biliary epithelial cells contained granular eosinophilic intracytoplasmic inclusions, variably sized to large (diameter, 8–10 μm); with special stains, these inclusions demonstrated an identical staining pattern to the microorganisms identified within cardiac myofibers (Fig. 4).
The epicardium and subepicardial myocardium were multifocally infiltrated by histiocytes admixed with scattered plasma cells, discrete focal collections of lymphocytes, and rare heterophils. The remaining myocardium had a mild locally extensive interstitial histiocytic infiltrate. The subepicardial myocardium was mildly edematous; myofibers were often atrophied and degenerate with a fine granular eosinophilic sarcoplasm lacking cross striations. Rare myofibers demonstrated segmental necrosis with pyknotic or karyorrhectic nuclei. Numerous cardiomyocytes contained large elliptical perinuclear cytoplasmic inclusions (diameter, 8–10 μm) comprising fine, granular, lightly basophilic organisms estimated to be less than 1 μm in diameter (Fig. 5). With a tissue Gram stain, the inclusions stained light pink, negative to light pink with a Gimenez stain, and dark black with a modified Steiner silver stain and did not stain with periodic acid–Schiff. In contrast to the tissue sections, the organisms did stain positive with a Gimenez stain of liver impression smears. In the pancreas, random individual exocrine cells were found with pale eosinophilic intracytoplasmic inclusions that displaced zymogen granules and resembled the inclusions seen in biliary epithelium, and there was a very light accompanying interstitial infiltrate of macrophages with similar intracytoplasmic inclusions.
In skeletal muscle, random focal interstitial lymphoplasmacytic infiltrates surrounded blood vessels and nerves and dissected between the myofibers. In the pectoral muscle, there also were rare, segmentally necrotic myofibers with fractured segments of hyalinized sarcoplasm often admixed with variable intracellular macrophage infiltrates. The quadriceps musculature was histologically normal. Similar mixed inflammatory cell infiltrates—predominantly comprising macrophages, lymphocytes, and plasma cells with variable numbers of heterophils—were detected in multiple organs, including spleen, ventriculus, proventriculus, ovary, and air sac. Random peripheral nerves were also infiltrated by macrophages with accompanying axonal degeneration. Mild infiltrates of macrophages and plasma cells with focal myelopoiesis were present in the kidney. Prominent numbers of plasma cells infiltrated the oviduct and vessels surrounding the outer muscularis of the proventriculus and gizzard. The white matter of the cerebellum was focally necrotic with neuropil rarefaction, hypereosinophilia, and scattered capillaries containing marginated leukocytes.
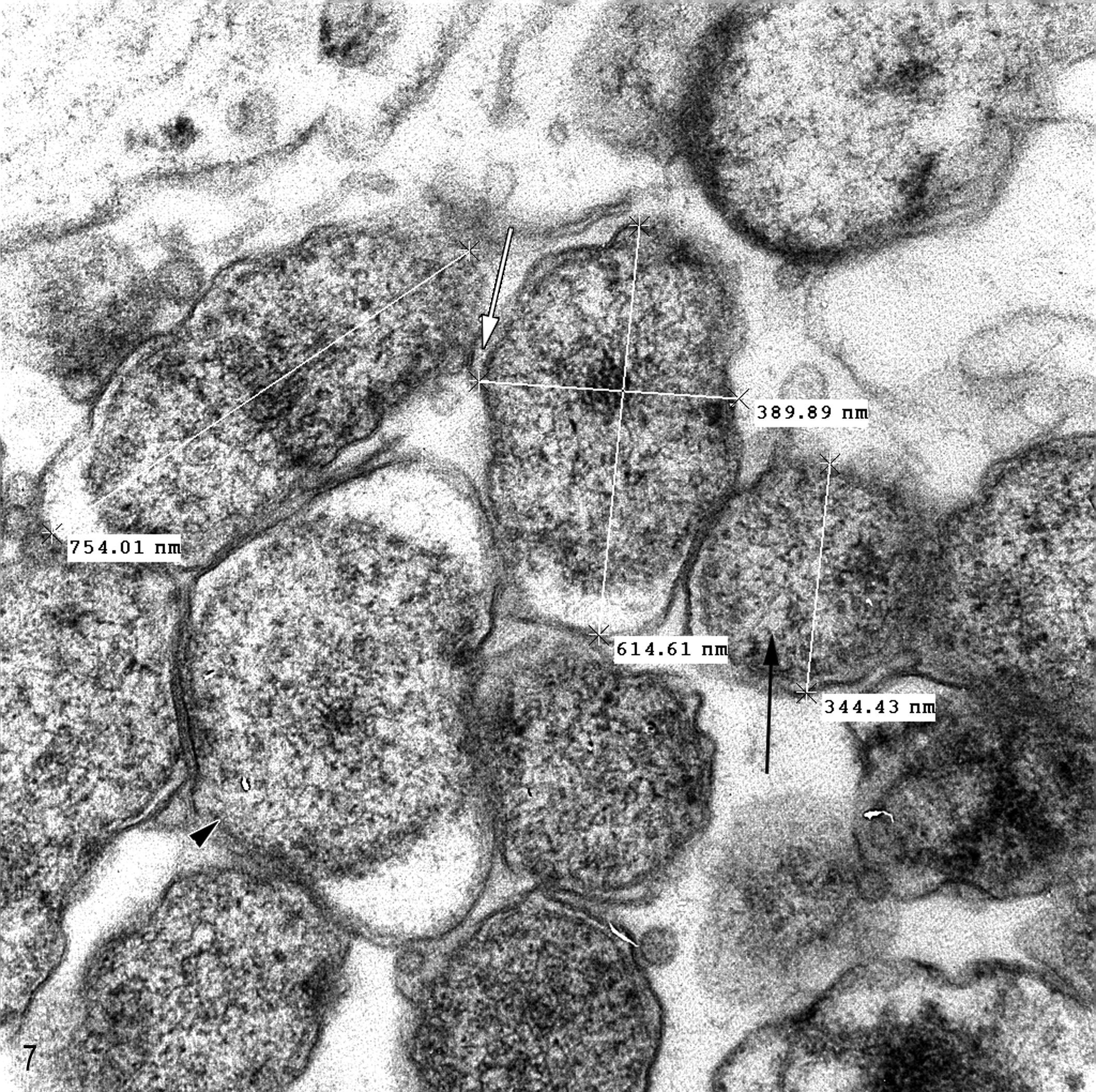
By transmission electron microscopy of the cardiac myofibers, hepatocytes, biliary epithelium, and histiocytes, microorganisms ultrastructurally similar to Rickettsia or Coxiella were found within membrane-bound vacuoles. The organisms were round to elongate with an outer and inner trilaminate membrane separated by a variable but often limited periplasmic space with a central to eccentric nucleoid. Larger organisms had a more abundant, lightly granular cytoplasm with more prominent periplasmic space as compared to smaller and often elongate forms with reduced and electron-dense granular cytoplasm. These 2 morphologic stages were comparable to large and small cell variants described for C. burnetii 5,9 and ranged from 304 to 446 nm in diameter and up to 1200 nm in length (Figs. 6, 7). Small numbers of very weakly staining microorganisms were detected by C. burnetii IHC within the perinuclear cytoplasmic inclusions within cardiac myofibers, hepatocytes, biliary epithelial cells, pancreatic exocrine cells, and histiocytes in several tissues, including liver, lung, spleen, air sacs, gastrointestinal tract, and peripheral nerves. Interestingly, on WNV IHC assays, which utilized a polyclonal antisera, occasional Coxiella-like organisms were also faintly positive; however, all parenchymal cells were negative.
Heart; eclectus parrot. Higher magnification of the intracellular organisms shown in Figure 7. Note the characteristic Gram-negative bacteria with trilaminal cell wall. EM.
PCR analysis resulted in the amplification of 232-nucleotide-long amplicons from the cardiac and hepatic tissues. Both amplicon sequences were the same and were 100% identical to the 16S rRNA sequence of the Coxiella-like organism previously isolated from the hepatic tissue of the affected parrots (GenBank accession Nos. EU143669 and EU143670). 9 No amplicons were evident in samples derived from the negative control tissues and water template. Bacterial cultures as well as IHC for WNV antigen and in situ hybridization for polyomavirus nucleic acid were negative. Occasional renal tubular epithelial cells contained intranuclear basophilic inclusion bodies compatible with adenoviral inclusion bodies. In situ hybridization on renal tissue was negative for polyomavirus.
Discussion
The microorganism responsible for the systemic infection in this parrot appears to be very similar, if not identical, to the recently reported Coxiella-like organism in psittacines, with 100% identity of a portion of the 16s rRNA amplicon.9,10 In the eclectus parrot described here, this infectious agent demonstrated a wide range of tropism for cardiac myofibers, biliary epithelium, and pancreatic exocrine cells, in addition to the histiocytes and hepatocytes previously reported. Whether the unique host cell tropism detailed here is due to differences among the various psittacine host species or the result of the dose of the infections agent is not determined.
It was not determined how this bird acquired the Coxiella-like bacteria. It is possible that some incidental contact with wild birds occurred, as the cage was located adjacent to an intermittently open window. C. burnetii seropositivity has been reported in a variety of wild and domestic birds. 4 In addition, Coxiella spp have been demonstrated to be carried by more than 40 species of ticks. 8 Although no ticks were detected on the bird at the time of the postmortem examination, previous tick exposure cannot be ruled out.
Foci of hepatic necrosis often centered on portal tracts with proliferative and disrupted biliary ducts. Although the pathogenesis for this necrosis is unknown, the peribiliary fibrosis and hyperplastic bile duct profiles suggest the possibility of Coxiella-mediated biliary destruction leading to parenchymal bile leakage and hepatocellular necrosis. Alternatively, they may possibly have resulted from ischemia secondary to the portal vasculitis. Occasional positivity of Coxiella-like organisms on the WNV IHC assay is most likely a result of nonspecific polyclonal antisera binding. There was no detectable inflammation associated with the small numbers of intranuclear (adenoviral-like) inclusions in renal tubular cells. These were considered an incidental finding. In our experience, renal tubular adenoviral inclusions can be found in psittacines with concurrent systemic infections.
Molecular analysis appears to be a specific and sensitive method to detect the Coxiella-like organism in affected tissues. In contrast to the other reports,9,10 the Coxiella IHC assay in this case was very weakly positive, possibly reflecting some variability in reactivity of specific polyclonal human antisera used in the assay. However, this staining reaction was inconsistent with each IHC run, and a positive reaction was detected only in small numbers of organisms within large inclusions. In our opinion, the reaction was not sensitive enough to be relied on as a routine diagnostic test. To fulfill Koch postulates and establish disease etiology, it will be necessary to reproduce the disease by experimental inoculation of birds, which would require isolation of the organism.
To our knowledge, this is the third report of a Coxiella-like organism associated with severe disease in a psittacine species. This report should raise pathologists’ awareness of this emerging infectious agent in birds, along with an appreciation of the variability of tissue tropism and nature of the lesions produced.
Footnotes
Acknowledgements
We would like to thank the California Animal Health and Food Safety Laboratory–Davis Histology Laboratory, Boynton Donna, as well as Dr Jerry D. Long for their assistance and expertise. We would also like to thank Dr Kirsten Murphy for review and editing of this manuscript and John Duval for assistance with the figures.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
