Abstract
Proventricular infection with the anamorphic, ascomycetous yeast Macrorhabdus ornithogaster and disease associated with infection (macrorhabdiosis) have been widely reported in budgerigars (Melopsittacus undulatus). Proventricular adenocarcinoma has also been previously reported in this species. In a retrospective archival search of clinical cases submitted to a zoo animal and exotic pet pathology service between 1998 and 2013, a total of 28 128 avian submissions were identified, which included 1006 budgerigars kept in zoos or aviaries or as pets. Of these budgerigars, 177 were identified histologically as infected with M. orthithogaster at the time of necropsy. Histologic examination of tissues from budgerigars infected with M. orthithogaster identified an apparent continuum in the development of proventricular isthmus lesions associated with M. ornithogaster that included inflammation, mucosal hyperplasia, glandular dysplasia, and adenocarcinoma. Proventricular adenocarcinoma was identified histologically in 21 budgerigars. Budgerigars with proventricular adenocarcinoma were significantly more likely to have macrorhabdiosis than budgerigars without proventricular adenocarcinoma. Based on odds ratios for archival data, budgerigars were 41 times (95% confidence interval [CI], 33–52) more likely to have macrorhabdiosis than other avian species and 19 times (95% CI, 11–33) more likely to have proventricular adenocarcinoma than other birds. Budgerigars were 323 times (95% CI, 42–2490) more likely to be affected by both diseases simultaneously compared with other avian species. These findings suggest that macrorhabdiosis may be an associated factor for proventricular adenocarcinoma, although the explanation for this statistically significant correlation remains unknown and further investigation is warranted.
Infection with Macrorhabdus ornithogaster in birds was first recognized in 1980 and is common in budgerigars (Melopsittacus undulatus). 2,3,5,7,12,14,15,19,23 The organism is rod shaped, microaerophilic, Gram positive, argyrophilic, and periodic acid–Schiff (PAS) positive and is the only species of this genus in the phylum Ascomycetes. 4,14,15,23,26,28 The prevalence of macrorhabdiosis in budgerigar aviaries where the disease is enzootic ranges from 27% to 64% based on fecal shedding rates and pathologic findings. 3,15 Acute and chronic clinical manifestations have been described in the budgerigar. 7,15,27 In the chronic form, clinical signs are more common in middle-aged to older birds. 3,15 Infected birds may be asymptomatic with no associated lesions observed at necropsy, or birds may have clinical signs ranging from mild to severe gastrointestinal disease, including weight loss, regurgitation, diarrhea, and melena. 3,4,7,15,26,27
Reported pathologic findings include proventricular and ventricular ulceration, isthmus gland atrophy or necrosis, goblet cell hyperplasia, inflammation, koilin disruption, and gastric perforation. 7,9,12,14,18,19,26 In 1 retrospective study, 89 of 175 (50.9%) budgerigars with gastric lesions were infected with M. ornithogaster at the time of necropsy. 20 The organisms are often present in greatest numbers in the superficial mucosal glands of the proventricular isthmus. 9,14,23 The isthmus marks the transition zone between the oxynticopeptic glands of the proventriculus and the straight, koilin-producing ventricular glands.
Neoplasia is common in budgerigars compared with other avian species. 1,16,19,21,22,24,29 While the overall prevalence of neoplasia in birds was just 3.8% in 1 study, 21 the prevalence rates of neoplasia reported in budgerigars are much higher, ranging from 15.8% to 34%. 13,17,19 Adenocarcinoma is the most common gastric neoplasm in birds and generally affects the proventriculus more often than the ventriculus. 10 Proventricular adenocarcinoma has been reported in budgerigars, gray-cheeked parakeets (Brotogeris pyrrhopterus), cockatiels (Nymphicus hollandicus), Amazon parrots (Amazona spp.), macaws (Ara spp.), lovebirds (Agapornis spp.), and lories. 11,18,20,25,26 In a recent retrospective review, 19 gastric carcinomas and adenocarcinomas represented 51 of 454 (11.2%) neoplastic lesions identified in budgerigars. Similar to macrorhabdiosis, proventricular adenocarcinoma most commonly occurs at the gastric isthmus between the proventriculus and ventriculus. 10,18,20,26,27 Clinical and diagnostic findings include lethargy, inappetence, weight loss, weakness, polyuria, maldigestion, melena, anemia, hypoproteinemia, and a positive fecal occult blood test. 10,18,27,29
The purpose of this cross-sectional retrospective study was to evaluate the relative prevalence of proventricular adenocarcinoma and infection with M. ornithogaster in budgerigars through review of archived necropsy data. The hypotheses being tested in this study were that (1) budgerigars are significantly more likely to have M. ornithogaster and proventricular adenocarcinoma than other avian species and that (2) the histologic presence of M. ornithogaster at the time of necropsy is associated with concurrent preneoplastic change or proventricular adenocarcinoma in budgerigars.
Materials and Methods
Case Selection
A retrospective archival study was performed on all avian cases submitted to a zoo animal and exotic pet pathology service (Northwest ZooPath [NWZP], Monroe, WA) between 1998 and 2013. Data sets included species, age, sex, clinical findings, gross necropsy findings, and histologic findings. All birds were kept in zoological collections, aviaries, or as companion birds. Birds either died spontaneously or were euthanized at the discretion of the attending clinicians. Clinical findings were reported by the submitting veterinarians. Gross necropsies and tissue prosections were performed by the submitting veterinarians or by one of the authors (M.M.G.). Subjects that lacked adequate samples of the proventriculus (including cardia, fundus, and isthmus) and ventriculus were excluded from the study.
Histopathology
All tissues were preserved in 10% neutral buffered formalin for routine sample processing. Paraffin-embedded tissues were sectioned at 5 microns, mounted on glass slides, stained with hematoxylin and eosin (HE), and examined by light microscopy. Because the organism has unique morphology in HE sections, fungal and bacterial stains were not applied to these tissues. A mucicarmine stain was used to demonstrate mucin in neoplastic and normal proventricular mucosal glands. Available archived avian cases internally coded as macrorhabdiosis, proventriculitis, or proventricular adenocarcinoma were collated and reread by 1 pathologist (M.M.G.) to standardize fungal and lesion morphology (Suppl. Table S1). Histologic sections of gastric tissues stained with HE were evaluated for the presence of M. ornithogaster. Available proventricular sections (cardia, fundus, and isthmus) were evaluated for pathologic changes, including inflammation, glandular changes (ectasia, necrosis, and abscessation), mucosal erosion and hyperplasia, dysplasia, neoplasia, and vascular invasion. Mucosal erosion and hyperplasia were graded on an ordinal scale of 0 (absent) to 3 (severe). Additional tissues, if available, were examined for evidence of distant metastasis and for other disease processes.
Statistical Methods
The distribution of continuous data was evaluated using the Shapiro-Wilk test, skewness, kurtosis, and q-q plots. A Mann-Whitney test was used to determine if age in years was associated with the presence of proventricular adenocarcinoma, infection with M. ornithogaster, or both conditions; only birds with known ages were included in the analysis. Odds ratios and 95% confidence intervals (CIs) were calculated for each of the binomial proportions. Fisher exact tests or χ2 (r × c) were used to determine if there was an association between outcome and sex, year of diagnosis, age, and pathologic findings, where outcome included cases of M. ornithogaster infection only, proventricular adenocarcinoma only, or both conditions. When significance was found, follow-up 2 × 2 χ2 tests or Fisher exact tests were done to determine which outcomes differed. Logistic regression modeling of the data was attempted, but the model fit was poor because of the small sample sizes of 2 groups (birds with proventricular adenocarcinoma but without M. ornithogaster infection [n = 9] and birds affected by both conditions [n = 12]), and thus univariate statistics were reported. A commercial statistical software (SPSS 23.0; SPSS, Inc, an IBM Company, Chicago, IL) was used to analyze the data. P ≤ .05 was used to determine statistical significance.
Results
Clinical Findings
Clinical findings reported by submitting veterinarians varied widely, precluding statistical evaluation. Unexpected death was the primary finding reported by clinicians for many budgerigars determined to have M. ornithogaster infection or proventricular adenocarcinoma. Lethargy, reduced or absent appetite, weight loss, and emaciation were also commonly reported with both conditions. Regurgitation, diarrhea, maldigestion, coelomic distension, palpable coelomic mass, vent soiling, tachypnea or dyspnea, wing droop, ataxia, and seizures were also reported findings for both diseases.
Gross Necropsy Findings
Emaciation was a common gross finding for budgerigars that had either M. ornithogaster infection or proventricular adenocarcinoma. Other reported findings were variable but included (for both diseases) an enlarged, distended, or discolored proventriculus; proventricular erosion, ulceration, or hemorrhage; proventricular wall thickening; nodular mucosal changes at the proventricular isthmus; excessive proventricular mucus accumulation; discolored or distended intestines; and darkened or discolored lungs. Birds (44%; 95% CI, 11–76; n = 4/9) found to have a grossly visible gastric mass were significantly (P = .021) more likely to have proventricular adenocarcinoma and no histologic evidence of M. ornithogaster infection compared to birds affected with both conditions (0%; 95% CI, 0–26; n = 0/12) at the time of necropsy (Figs. 1–4).
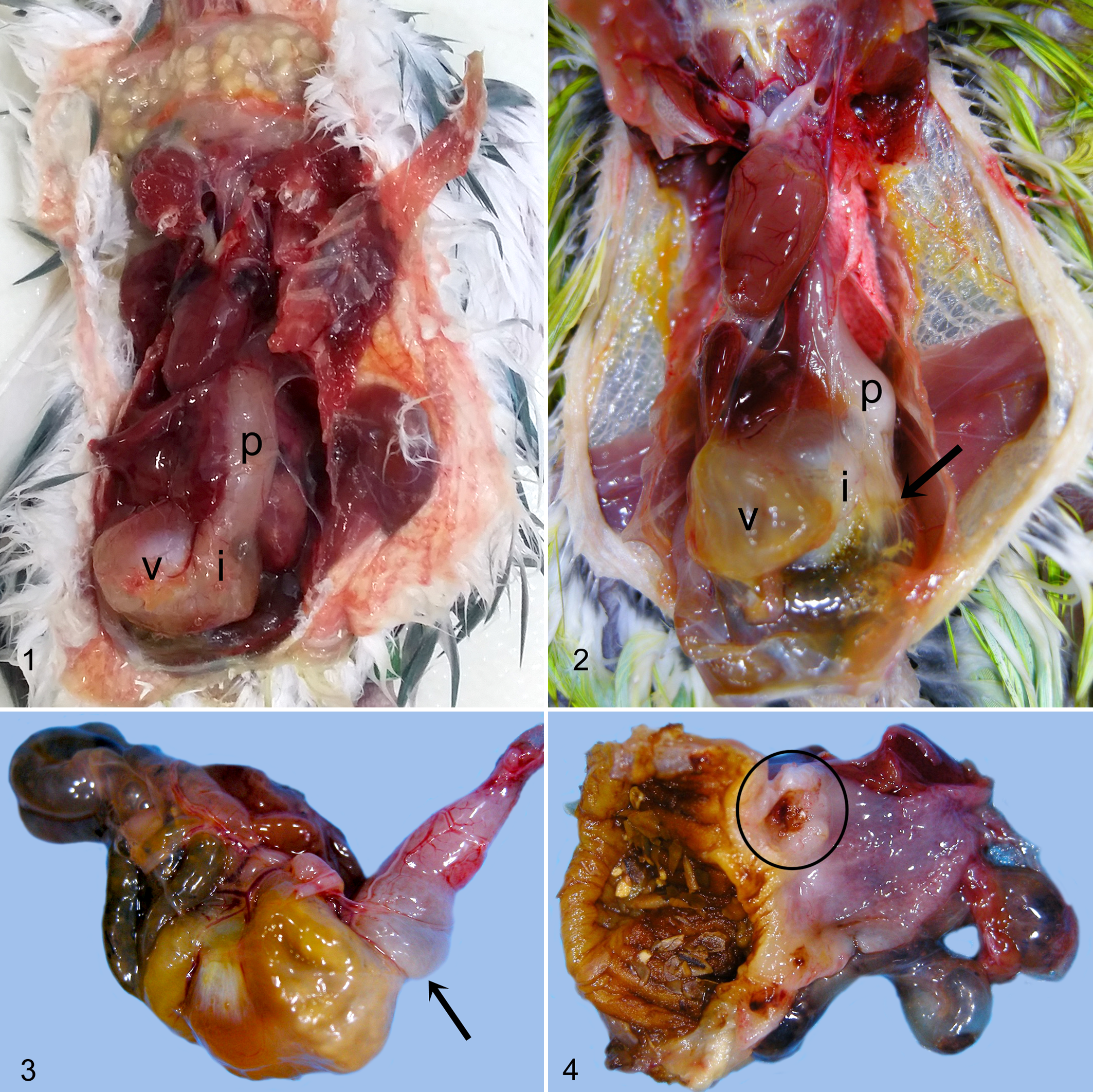
Normal proventriculus (p), proventricular isthmus (i), and ventriculus (v), budgerigar.
Histologic Findings
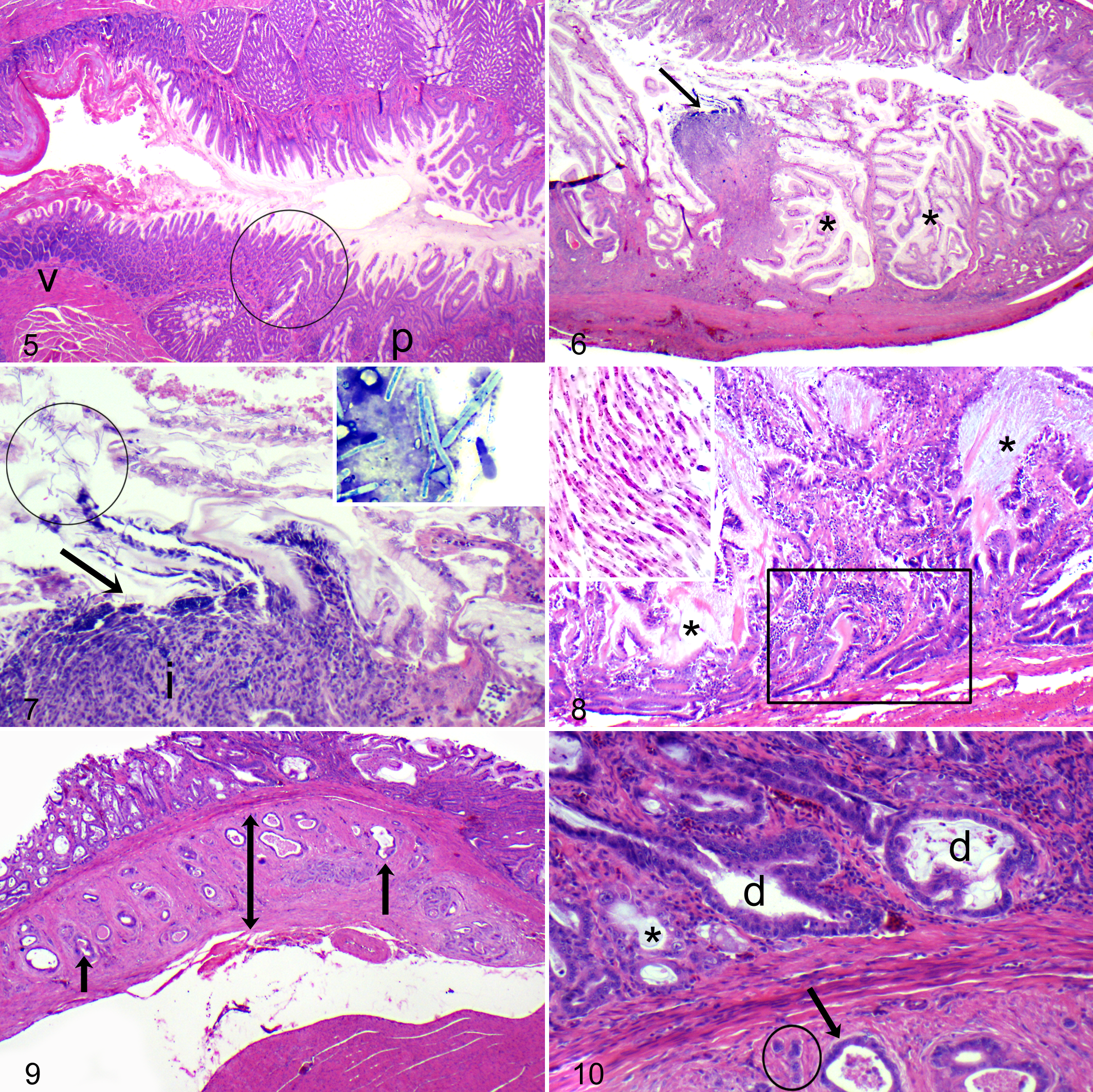
Birds with M. ornithogaster infection generally had few to many elongated yeasts aligned in parallel rows in the superficial or deep glandular lumina of the isthmus, rarely extending into the glandular structures of the immediately adjacent fundus or along the surface of the ventriculus. Yeasts had typical morphology as previously described, stained pale basophilic with HE, were approximately 200–250 microns × 5–10 microns, and were uninucleate and spherical in transverse section. 4,14,15,23,26 Individual or clustered organisms were sometimes present in the lumen of the proventricular isthmus and ventriculus. Inflammation in affected budgerigars was primarily lymphoplasmacytic and histiocytic throughout the lamina propria and, when severe, also extended into the submucosa or was transmural. Inflammation was frequently accompanied by erosion or ulceration at the isthmus, and frank or digested blood was sometimes present along the surfaces of the proventriculus and ventriculus. Koilin in the ventriculus was often degenerate or lined with blood or bile.
Glandular lesions in affected budgerigars occurred primarily in the deep glandular regions and included dilatation, tortuosity, rupture, hyperplasia, dysplasia, and neoplastic transformation (Figs. 5–8). Hyperplasia was characterized by hypertrophy and pseudostratification or stratification of the lining columnar epithelium and an increase in the number of mitotic figures in the glands and crypts. Dysplastic changes typically occurred in the deep glands and crypts of those birds with concurrent hyperplasia and included tortuosity and branching of the deep glands, asymmetrical crowding or stacking of the lining epithelium, and cellular atypia that included a loss of nuclear polarity, anisokaryosis, increase in the nuclear to cytoplasmic ratio, and enlarged or multiple nucleoli.
Normal proventriculus (p), isthmus (circle), and ventriculus (v), budgerigar. Hematoxylin and eosin (HE).
Neoplastic transformation of glandular epithelium was diagnosed when glands or cellular aggregates with atypical morphology invaded the tunica muscularis with or without extension to the serosa (Figs. 9, 10). Neoplastic cells were individualized or clustered into glandular formations and frequently were accompanied by a scirrhous response and inflammation, perforation of the bowel at the site of invasion, coelomitis, and coelomic adhesions. In budgerigars, Macrorhabdus yeasts were often present in the lumina of dysplastic glands but were not seen in the neoplastic tissue. Two neoplastic variants were observed. The most common form had glandular architecture with mucinous differentiation or accumulation of mucin in glandular lumina. Less common were “solid” tumors formed by compact nests or sheets of epithelial cells. Some overlap of these features was seen in few tumors. Macrorhabdus yeasts were associated with proventricular adenocarcinoma in only 1 other species, a single zebra finch (Taeniopygia guttata).
Epidemiologic Findings
The numbers of budgerigar and nonbudgerigar M. ornithogaster infection and proventricular adenocarcinoma cases are reported in Table 1. The overall prevalence of M. ornithogaster infection in birds submitted to NWZP between 1998 and 2013 was 1.1% (95% CI, 0.51%–0.69%; n = 317/28 128). Budgerigars (17.6%; 95% CI, 15.2%–20.0%; n = 177/1006) were found to be 41.1 (95% CI, 32.6–51.9) times more likely to be diagnosed with M. ornithogaster infection than nonbudgerigar avian species (0.5%; 95% CI, 0.4%–0.6%; n = 140/27 122) at the time of necropsy. The overall prevalence of proventricular adenocarcinoma in birds was 0.18% (95% CI, 0.12%–0.22%; n = 52/28 128). Budgerigars (2.1%; 95% CI, 1.2%–3.0%; n = 21/1006) were found to be 18.6 (95% CI, 10.7–32.5) times more likely to be diagnosed with proventricular adenocarcinoma than all other bird species (0.1%; 95% CI, 0.06%–0.14%; n = 31/27 122).
Frequency at Necropsy of Macrorhabdiosis (MO) and Proventricular Adenocarcinoma (PA) in Budgerigar and Nonbudgerigar Birds.

Of the 186 budgerigars diagnosed with either proventricular adenocarcinoma or M. ornithogaster infection at the time of necropsy, 12 (7.0%) were diagnosed with both conditions simultaneously. Budgerigars with proventricular adenocarcinoma were significantly (χ2 = 24.38, P = .000001) more likely to have M. ornithogaster infection (57%; n = 12/21) than budgerigars without proventricular adenocarcinoma (16.7%; 95% CI, 14.4%–18.9%; n = 165/985). Budgerigars (1.2%; 95% CI, 0.5%–1.9%; n = 12/1006) were found to be 323 (95% CI, 42–2490) times more likely to be diagnosed with both proventricular adenocarcinoma and M. ornithogaster infection simultaneously than all other avian species (0.00004%; 95% CI, 0.00003%–0.00011; n = 1/27 122).
There was no significant difference in findings by sex (χ2 = 3.67, P = .452) or by year of diagnosis (χ2 = 38.76, P = .085). Age was significantly (P = .003) associated with the final diagnosis in budgerigars: the median age of budgerigars with proventricular adenocarcinoma and no M. ornithogaster infection (n = 8) was 6.0 years (25%–75%: 5.0–7.75; minimum–maximum: 5.0–10.0), while birds with both conditions (n = 9) had a median age of 4.0 years (25%–75%: 3.0–5.0; minimum–maximum: 3.0–5.0).
There was a significant difference in fundic mucosal hyperplasia between the different groups of budgerigars in tissues available for rereview (χ2 = 20.416, P = .002), with hyperplasia being significantly more likely in birds with both proventricular adenocarcinoma and M. ornithogaster infection (PA/MO group) (P = .001) and in birds with only proventricular adenocarcinoma (PA/no MO group) (P = .05) compared with birds that only had infection with M. ornithogaster at the time of necropsy (no PA/MO group) (Table 2). There was no difference between the 2 groups of birds with proventricular adenocarcinoma (PA/MO vs PA/no MO). Significant differences in mucosal hyperplasia (χ2 = 28.682, P = .0001), mucosal erosion in the isthmus (χ2 = 6.804, P = .033), and dysplasia (χ2 = 30.0, P = .0001) were also noted between the different diseased groups of budgerigars (Table 3). Of budgerigars infected with M. ornithogaster, hyperplasia was significantly more likely in those with proventricular adenocarcinoma compared to those without (P = .0001 for the PA/MO group and P = .04 for PA/no MO group). Of budgerigars with proventricular adenocarcinoma, there was a similar prevalence of hyperplasia in infected and uninfected birds. Of budgerigars infected with M. ornithogaster, erosions in the isthmus were significantly more likely in those with proventricular adenocarcinoma compared to those without (P = .015). Finally, the no PA/MO group had significantly more dysplasia than the PA/no MO group (P = .0001) or PA/MO group (P = .0001). There were no significant differences in pathology of the cardia between the different groups (all P > .30).
Relationship of Proventricular Mucosal Hyperplasia in the Fundus and Isthmus With Macrorhabdiosis (MO) and With Proventricular Adenocarcinoma (PA) in Budgerigars.
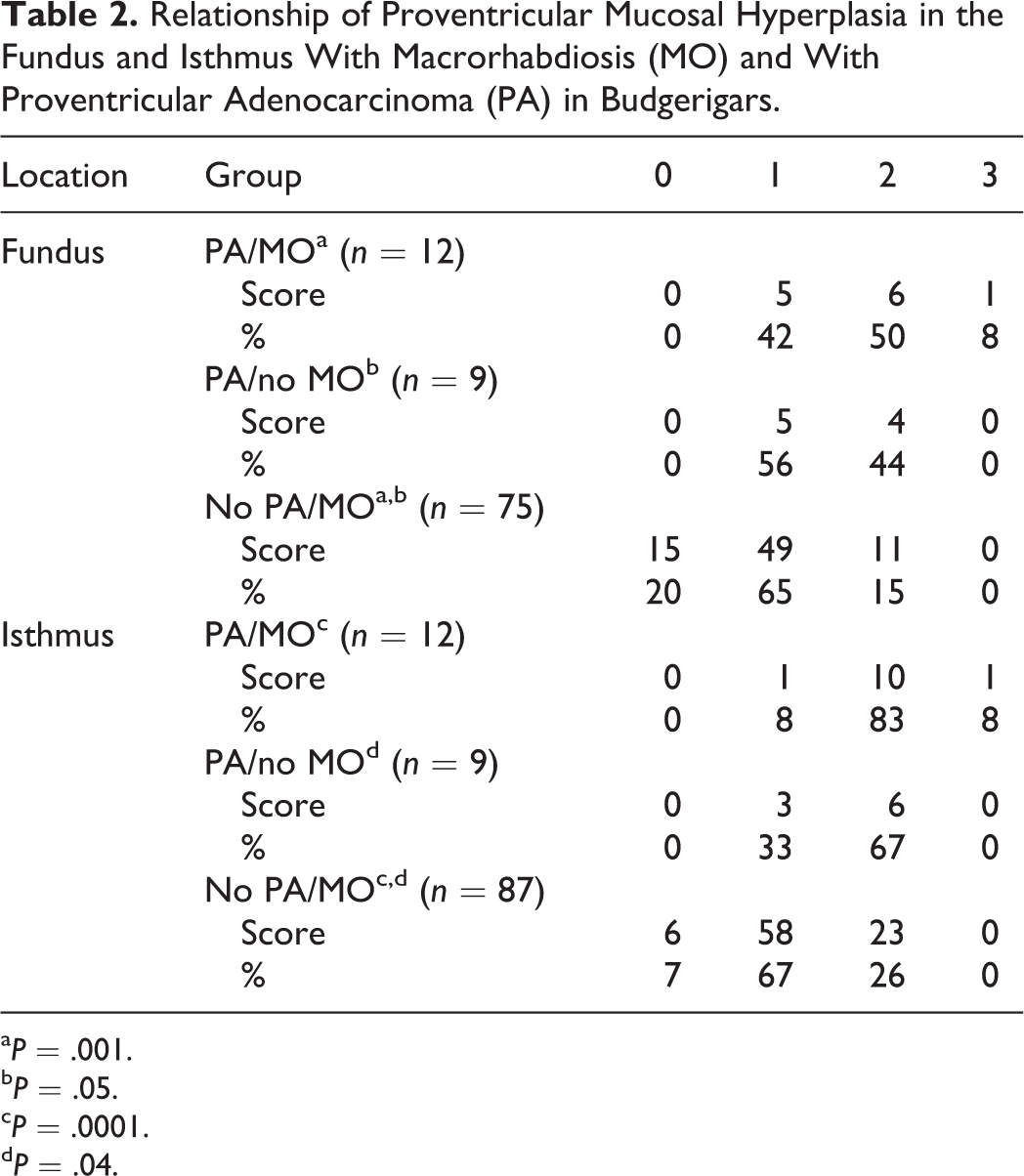
a P = .001.
b P = .05.
c P = .0001.
d P = .04.
Relationship of in Proventricular Mucosal Erosion of the Isthmus or Proventricular Dysplasia With Macrorhabdiosis (MO) and With Proventricular Adenocarcinoma (PA) in Budgerigars.
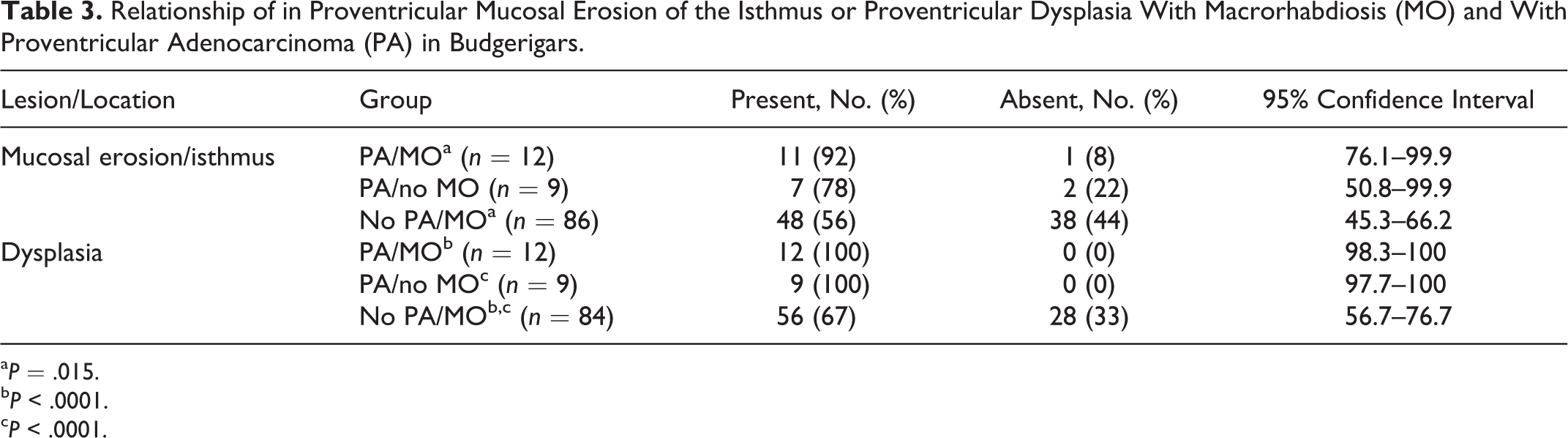
a P = .015.
b P < .0001.
c P < .0001.
Distant metastasis was observed in 6 of 21 budgerigars with proventricular adenocarcinoma (29%; 95% CI, 9.3%–47.9%). There was no statistical difference (P = .50) in prevalence of distant metastasis in birds with (n = 3/12; 25%) or without (n = 3/9; 33%) concurrent M. ornithogaster infection. Only 1 bird affected concurrently by both conditions demonstrated evidence of vascular invasion.
Concurrent Diseases
Of 186 budgerigars diagnosed with M. ornithogaster infection and/or proventricular adenocarcinoma, 109 had additional concurrent conditions, including adenovirus infection (n = 18; 16.5%; 95% CI, 9.5%–23.5%), proventricular cryptosporidiosis (n = 9; 8.3%; 95% CI, 3.0%–13.5%), hepatitis (n = 9; 8.3%; 95% CI, 3.0%–13.5%), gastrointestinal protozoal infections (n = 6; 5.5%; 95% CI, 1.5%–9.5%), interstitial nephritis (n = 6; 5.5%; 95% CI, 1.5%–9.5%), ingluvial candidiasis (n = 5; 4.6%; 95% CI, 0.5%–8.5%), urate stasis (n = 4; 3.7%; 95% CI, 0.1%–7.4%), pituitary gland carcinoma (n = 4; 3.7%; 95% CI, 0.1%–7.4%), and enteritis (n = 3; 2.8%; 95% CI, 0.1%–5.9%).
Discussion
Gross and histologic findings in affected budgerigars were similar to those in previous reports for both M. ornithogaster infection and proventricular adenocarcinoma. 6,10,14,15,18,20,26 There appeared to be no sex predilection for either condition. However, birds diagnosed with both M. ornithogaster infection and proventricular adenocarcinoma had a shorter median life span than those diagnosed only with neoplasia (4.0 vs 6.0 years), suggesting that changes related to this comorbidity may affect longevity, such as inanition, sepsis, peritonitis, and anemia. At 16.2% of all budgerigar cases submitted during the study period, infection with M. ornithogaster was a common finding, but the prevalence was lower than in previous reports. 3,15,20 However, previous studies evaluated budgerigars in aviaries in which infection was endemic, 3,15 or prevalence rates were reported only for birds with gastric lesions. 20
A positive and significant correlation between M. ornithogaster infection and proventricular adenocarcinoma in budgerigars was observed in this study. Because of the design of this retrospective study, the explanation for this association was not determined. While infection with M. orthithogaster may induce preneoplastic gastric changes, it is also possible that preneoplastic changes may provide a more suitable environment for infection by the yeast.
Mucosal hyperplasia of the proventricular fundus and isthmus was commonly observed in budgerigars with proventricular adenocarcinoma (with or without M. ornithogaster infection). In this regard, hyperplasia may be a preneoplastic event in these birds. Proventricular dysplasia was more often observed in birds with M. ornithogaster infection and no adenocarcinoma than in birds with proventricular adenocarcinoma and no M. ornithogaster infection. The reason for this difference is not known, but it is likely that this result reflects differences in sectioning or tissue collection. Proventricular erosion was more often observed in birds with M. ornithogaster infection without proventricular adenocarcinoma than in birds affected by both conditions. The reason for this difference is not known, but based on the pattern of lesion development in these birds, it is likely that erosions were not always in the plane of histologic section.
A grossly visible gastric mass was significantly more likely to occur in birds with only proventricular adenocarcinoma than in budgerigars affected by both diseases. The reason for this difference is not known, but possibilities include previous treatment for M. ornithogaster infection, autolysis of the gastric mucosa, and differences in gross sectioning techniques. Although distant metastasis of proventricular adenocarcinoma was common in this study group, the frequency of metastasis did not appear to be different in birds with or without M. ornithogaster infection at the time of death.
The role of infectious agents in carcinogenesis is becoming increasingly recognized. The exact mechanisms by which gastric cancer develops as a result of infection have not yet been established, but infection, inflammation, and other effects may result in epithelial cell proliferation and increased rates of apoptosis in antral cells. 8 It is possible that M. ornithogaster induces similar effects. Infection with this organism in budgerigars is more often associated with inflammation than in other birds, and it seems most likely that chronic irritation may be driving the transformation event rather than some form of oncogenic by-product of the fungus. Further studies such as transmission studies are necessary to further elucidate any association between infection and gastric neoplasia in budgerigars and the role if any that different strains of the organism may play in the pathogenesis of the disease.
Conclusion
In this retrospective study of archived necropsy data collected from 1998 to 2013, there was a significant and positive correlation between simultaneous infection with M. ornithogaster and the presence of proventricular adenocarcinoma at the time of necropsy in budgerigars submitted for pathologic evaluation. This correlation provides further evidence to suggest that M. ornithogaster is a potential cause of proventricular adenocarcinoma in budgerigars, although an alternate possibility is that M. ornithogaster selectively colonizes neoplastic gastric tissue. Further studies are warranted to elucidate the causality of this association.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818823773 - Macrorhabdus ornithogaster Infection and Spontaneous Proventricular Adenocarcinoma in Budgerigars (Melopsittacus undulatus)
Supplemental Material, DS1_VET_10.1177_0300985818823773 for Macrorhabdus ornithogaster Infection and Spontaneous Proventricular Adenocarcinoma in Budgerigars (Melopsittacus undulatus) by Lauren V. Powers, Mark A. Mitchell and Michael M. Garner in Veterinary Pathology
Footnotes
Acknowledgements
We thank Christie Buie of Northwest ZooPath for image layout, Cathy Minogue of Northwest ZooPath for data retrieval, and LeRoy Brown of Histology Consultation Services for slide preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
