Abstract
Psittacid herpesvirus 3 (PsHV-3) has recently been implicated as the cause of a severe respiratory disease in Bourke’s parrots (Neopsephotus bourkii) in the United States. In this report, the clinical manifestations and gross and microscopic lesions of PsHV-3 infection in 2 eclectus parrots (Eclectus roratus) in Australia are described. The presence of a PsHV-3 infection was confirmed by polymerase chain reaction amplification and sequencing of PsHV-3 DNA using degenerate and PsHV-3 primers. Electron microscopy of infected cells demonstrated the assembly of herpesvirus virions as well as intranuclear tubular structures. The detection of PsHV-3 in Australia in 2 eclectus parrots broadens the list of known affected species and confirms the presence of this virus in Australia.
Herpesvirus-associated disease is common in avian species and includes significant conditions of domestic poultry such as infectious laryngotracheitis and Marek’s disease. Amongst psittacine birds, 3 distinct species of herpesviridae have been categorized: psittacid herpesvirus (PsHV) 1 and 2, 9 as well as a recently recognized herpesvirus isolated from Bourke’s parrots (Neopsephotus bourkii) in the United States, 8 tentatively called PsHV-3.
PsHV-3 is the only genetically characterized herpesvirus that has been shown to cause respiratory disease in parrots. It is a member of the Iltovirus genus of the Alphaherpesvirus family and was identified in an outbreak of respiratory disease in Bourke’s parrots in 2011. 8 The primary target tissues for this virus were the trachea, bronchi, and air sacs, although in a number of cases, lesions were more systemic (thymus, bursa, inner ear, nasal and conjunctival epithelium, kidney, testes and ovary, thyroid, and spleen). Lesions were characterized by necrosis of the respiratory epithelium and the formation of syncytial cells containing pan-nuclear eosinophilic inclusion bodies. Other psittacine species of varying age groups in this aviary collection (Nymphicus hollandicus, Psittacula spp, Psephotus haematonotus, Forpus coelestis, Ara spp, Amazona spp, Cacatua spp, and members of the Cacatuidae family) did not develop disease, despite close contact. Similar respiratory lesions containing herpesvirus inclusions have been reported in several other species of parrots in North America and Australia. 3,4 With the exception of a recently published article from a North American outbreak, 8 however, identification of the agent has not been reported.
In this report, the clinical manifestations and gross and microscopic postmortem findings in 2 eclectus parrots (Eclectus roratus) purchased from the same commercial source are described. Histologically, viral inclusions were noted, and herpesviral infection was confirmed with polymerase chain reaction (PCR). Further characterization indicated the presence of a virus only recently identified in Bourke’s parrots and identified as PsHV-3. Concurrent infection with psittacine beak and feather disease virus (PBFDV) and an Aspergillus species were present.
The report indicates that PsHV-3 has the potential to infect multiple species and confirms its presence in Australia.
Case Report
A 5-month-old female eclectus parrot (case 1) presented with a 2-week history of delayed crop emptying and gradual weight loss resulting in emaciation. At the time of presentation, a serum biochemical profile was performed. Significant findings included hypoproteinemia (9 g/l; normal range, 32–43 g/l), hypoalbuminemia (<5 g/l; normal range, 11–20 g/l), elevated creatinine kinase (1069 IU/l; normal range, 132–410 IU/l), hypocholesterolemia (1.3 mmol/L; normal, 2.6–6.8 mmol/L), hyperamylasemia (36 200 IU/l; normal, 562–684 IU/l), and hyperuricemia (8.49 mmol/L; normal range, 0.12–0.65 mmol/l). The severity of clinical pathologic findings suggested a guarded prognosis, and the owners elected euthanasia.
During necropsy, variably sized multiple foci of hemorrhage and pallor were noted in the pancreas (Fig. 1). The lung was consolidated, with multiple foci of light yellow, caseous necrosis strongly suggestive of mycotic pneumonia. An extensive range of tissues were selected and either frozen or fixed in 10% neutral buffered formalin and submitted to the State Veterinary Diagnostic Laboratory, Elizabeth Macarthur Agricultural Institute, for analysis.
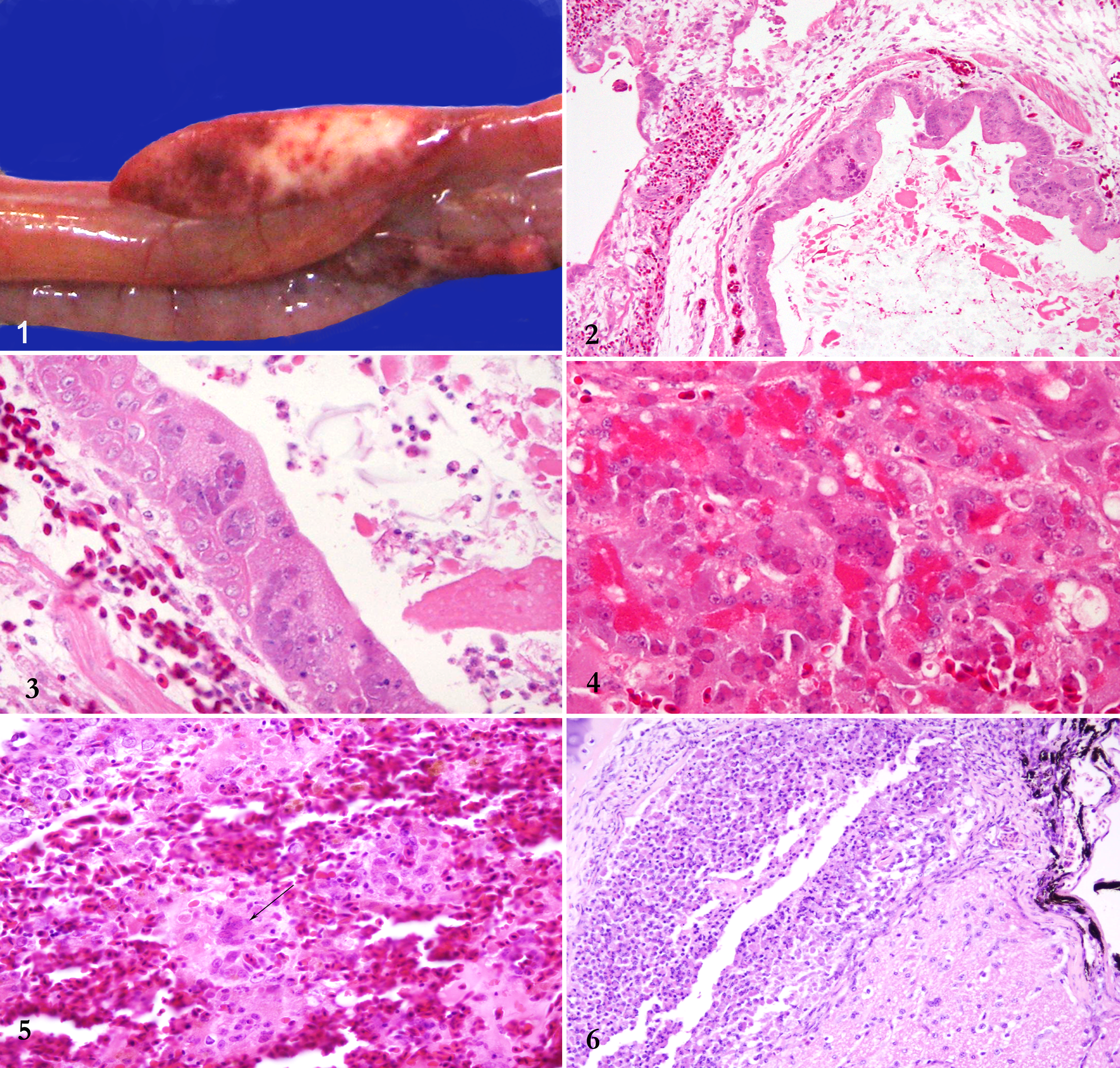
One month later, a second eclectus parrot, 3 months old (case 2), sourced from the same pet shop, presented weak with seizures and upper respiratory signs. Antemortem testing was declined, and the animal was euthanized. Because of the history of its pen mate, tissues were submitted in 10% formalin. Frozen lung was also provided for PCR analysis.
Histologically, in both cases, there was extensive expansion of secondary bronchioles and parabronchi with heterophils, fibrin, cellular and nuclear debris, and numerous branching fungal elements. Similar inflammatory infiltrates extended into adjacent airs sacs. Fungal elements consisted of regularly septate hyphae 3 to 6 μm in diameter with thin, parallel walls and acute-angle dichotomous branching, consistent with an Aspergillus sp. Lining epithelial cells were frequently degenerate and contained expansile intranuclear eosinophilic inclusion bodies (Figs. 2 and 3), often associated with marginated chromatin. Numerous syncytial cells were noted affecting both respiratory and air sac epithelium and extending into the trachea (case 2). There was moderate (case 2) to extensive (case 1), multifocal to coalescing disruption of the pancreas with multiple foci of necrosis and multifocal acinar hemorrhage. Numerous degenerate acinar cells contained intranuclear inclusion bodies (Fig. 4), identical to those in the lung. Syncytia were frequently noted. Peripherally, there was acinar atrophy and ductular proliferation. Within the spleen, there were multiple foci of necrosis, with lakes of fibrin, karyorrhectic debris, and numerous syncytial cells (Fig. 5) with intranuclear inclusion bodies. Within the duodenal mucosa and the renal parenchyma, scattered similar syncytial cells were present. A focal accumulation of heterophils and histiocytes with rare intranuclear inclusion bodies was noted at the base of the optic nerve, expanding the infraorbital sinus in case 2 (Fig. 6), with extension of similar but minimal inflammatory infiltrates into the meninges of the cerebral cortex and cerebellum. Additional findings included mild lymphoplasmacytic nephritis with multifocal necrosis and intranuclear inclusion bodies in tubular epithelial cells (case 2) and mild ventricular koilin disruption with occasional associated fungal hyphae, consistent with Aspergillus sp. Examination of the liver in both birds did not reveal significant changes.
Fungal culture of lung and air sac from case 2 was carried out on Sabouraud media for 5 days at 30°C and confirmed a heavy growth of an Aspergillus sp.
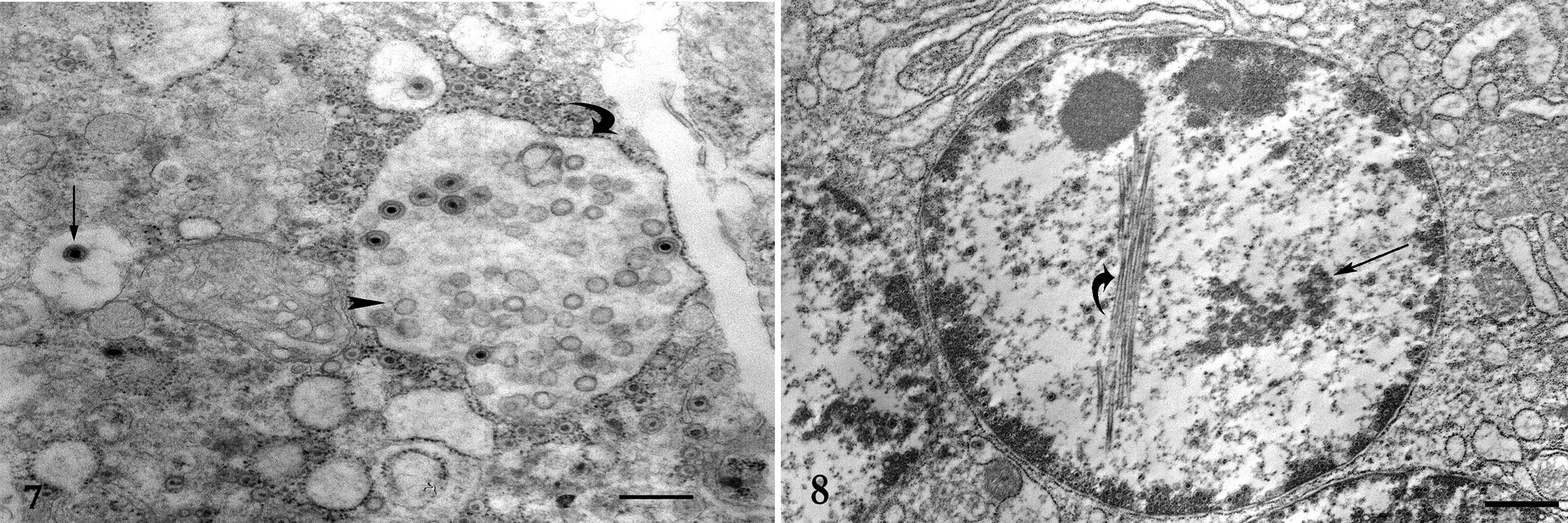
Pulmonary, pancreatic, and renal tissue samples from extant paraffin blocks of cases 1 and 2 were selected and processed for ultrastructural examination. Ultrathin sections were mounted on copper grids, stained with uranyl acetate and lead citrate, and examined and photographed using a transmission electron microscope. In the pancreas and the lung, numerous syncytial cells were noted containing myriad nucleocapsids and empty viral particles within the nucleus or free within phagocytic vacuoles (Fig. 7). In the kidney, similar changes were seen but with fewer syncytia. There was extensive nuclear proteolysis. The outer diameter of the virions was 90 to 120 nm in diameter. The presence of virion cores was variable. Frequently, intranuclear tubular structures were present that contained a clear core (Fig. 8). Viral particles were confirmed in lung, pancreas (case 1), and kidney (case 2).
DNA extraction was performed and herpesvirus DNA was amplified according to the method described in the online supplemental files. Herpesvirus sequences were aligned, creating a sequence of 1050 base pairs, and this sequence was then compared with the 850 base pairs of the original PsHV-3 sequence. 8
Because of the ubiquitous nature of PBFDV in Australian psittacine populations, PBFDV extraction and amplification were carried out, and both birds were positive for PBFDV, despite the lack of histologic evidence. For additional information and for the nature of the primer sets used to amplify both PsHV-3 and PBFDV, please see the online supplemental files.
Discussion
This article describes the first identification of PsHV-3 infection in any species other than the Bourke’s parrot and is only the second instance of isolation of this virus globally. The virus has only recently been identified 8 and was the cause of an outbreak of respiratory disease in captive Bourke’s parrots housed in a collection in the United States. As was the case in the Bourke’s parrots, both eclectus parrots in this report were captive animals, but only 1 showed respiratory signs similar to those reported in the Bourke’s parrots. Antemortem and postmortem examination demonstrated widespread, multiorgan involvement. Both parrots shared the same microscopic lesions of the bronchi and air sacs, trachea, spleen, and kidney as those reported in PsHV-3 infections of Bourke’s parrots. Lesions of the pancreas (1 bird), small bowel, meninges, and infraorbital sinuses were unique to the eclectus parrots in this report. In neither the eclectus nor the Bourke’s parrots were lesions noted in the liver, a preferential site of replication and associated necrosis for PsHV-1, the causative agent of Pacheco disease (and, at first consideration, a reasonable differential diagnosis). In addition to this lack of target specificity between PsHV-1 and PsHV-3, tissues in the current cases were rich in syncytial cells, a histopathologic characteristic that is rarely seen in birds infected with PsHV-1. 7
Of note in the current cases was the finding of intranuclear tubular structures (Fig. 8). This finding is infrequently reported, but it has been noted in association with other herpesvirus infection of birds, 3,4,11,12 mice, 5 and cats. 1 These tubular structures are thought to represent aberrant assembly of viral structures or may have a role in the intranuclear transportation of viral particles. 2
The viruses identified in the Bourke’s and the eclectus parrots varied genetically by less than 0.1% in the region sequenced and thus would be considered essentially identical. The variation in the tissues involved between outbreaks may have been caused by differences in the host, differences in the stage of the infection, or possibly genetic differences in the viruses not detected in this relatively conserved portion of the genome. It is also highly probable that concurrent infection with the PBFDV and Aspergillus sp could have led to more widespread distribution of the virus. The presence of both of these agents is not unusual in psittacine birds. The PBFDV is a common infection in both wild and captive species of parrots in Australia, and secondary or tertiary fungal infections are common in many species. The PBFDV targets multiple cells of the immune system, and the eponymous clinical characteristics do not necessarily manifest in all cases. These 2 eclectus parrots were at an age where a cloacal bursa was still present, but PBFDV inclusions were not identified in the bursae. Therefore, infection would not have been detected without PCR analysis of tissue (in this case, lung).
Given that this is the first time that PsHV-3 has been confirmed to be present in Australia and the first time it has been detected outside of the United States, a more widespread distribution is implicit. Similar lesions were reported in Bourke’s parrots 6 in Australia in the 1980s (although no agent was confirmed)—it is possible, therefore, that this virus evolved in Australian parrots and was exported to the United States, either directly or via other transit points. Alternatively, it is equally possible that the virus was introduced into Australia—for many years, there has been both legal and illegal movement of budgerigars, lovebirds, and finches into Australia from other markets, which could have been a source for this virus. Similar lesions have been reported in parrots (no agent identified) in Japan and the United States. 3,4,6,10 The presence of these cases raises the possibility that PsHV-3 is more widely spread and suggests that it should be added to the list of potential pathogenic agents of significance in psittacine populations. Since birds have most likely been transported to destinations worldwide, with the possibility of viral dissemination, examination of archival materials may be prudent to further elucidate the potential global distribution of this virus.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
