Abstract
A retrospective study of guinea pigs submitted for necropsy revealed intracytoplasmic inclusions in the cardiomyocytes of 26 of 30 animals. The inclusions were found with approximately the same frequency in male and female guinea pigs and were slightly more common in older animals. In most cases, the animals did not have clinical signs or necropsy findings suggestive of heart failure, and the cause of death or reason for euthanasia was attributed to concurrent disease processes. However, the 4 guinea pigs with the highest inclusion body burden all had pulmonary edema, sometimes with intra-alveolar hemosiderin-laden macrophages, suggestive of heart failure. The inclusions were found in both the left and right ventricular myocardium, mainly in the papillary muscles, but were most common in the right ventricular free wall. No inclusions were detected in the atrial myocardium or in skeletal muscle. The inclusions did not stain with Congo red or periodic acid–Schiff. Electron microscopy revealed dense aggregates of disorganized myofilaments and microtubules that displaced and compressed the adjacent organelles. By immunohistochemistry, there was some scattered immunoreactivity for desmin and actin at the periphery of the inclusions and punctate actin reactivity within the aggregates. The inclusions did not react with antibodies to ubiquitin or cardiac myosin, but were variably reactive for alpha B crystallin, a small heat shock chaperone protein. The inclusions were interpreted as evidence of impaired proteostasis.
Keywords
Disorders of protein homeostasis, or proteostasis, have been implicated in a wide range of disorders, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and cystic fibrosis. 18 These disorders are characterized by extra-lysosomal accumulations of misfolded proteins that alter cellular function. Disorders of protein homeostasis affecting skeletal and/or cardiac muscle are known as protein aggregate myopathies. Because of the high metabolic demand, complex cytoskeletal structure, and post-mitotic status of cardiomyocytes, the heart is particularly at risk for disorders of protein homeostasis. Abnormal protein accumulations have been detected in a variety of myocardial disorders, ranging from cardiomyopathies to myocardial ischemia. 12
Protein aggregate skeletal myopathies have rarely been reported in dogs, 6 cats, 3,20 and horses, 35 and there are no previous reports of myocardial protein aggregates in animals other than humans. The purpose of this study was to characterize inclusions commonly found in the myocardium of guinea pigs and compare them to protein aggregates reported in human cardiac diseases.
Materials and Methods
Identification of Cases
The electronic database of necropsy cases at the New York State Animal Health Diagnostic Center was searched for guinea pigs. Gross photographs and necropsy reports for the guinea pigs were reviewed. Slides from all the cases were examined and lesions were scored by a board-certified pathologist blinded to disease status. Guinea pigs were included in the study if a section of heart (transverse or longitudinal) including right and left ventricular free walls and septum was available for histologic analysis.
Image Analysis
The slides were digitized using a Roche iScan HT scanner and the images analyzed using ImageViewer software. The area of each myocardial region (right ventricular free wall [RVFW], interventricular septum [IVS], and left ventricular free wall [LVFW]) was calculated by measuring average wall thickness and length/circumference on a subgross image of the histologic section. Intracytoplasmic inclusions were counted in each region of myocardium, and the burden of inclusions was determined by dividing the inclusion count by the area. Locations of inclusions were annotated on a subgross image to evaluate the distribution in each section. Heart weights were not available for any of the guinea pigs, and image analysis was limited by several variables, including the mechanism of death (barbiturate euthanasia vs natural death) and the sampling method for histology (longitudinal vs cross sections at nonstandard levels).
Clinical and Histologic Correlations
To determine correlations between inclusions and other changes in the heart and lesions in other organs, inflammatory and degenerative changes were scored on a semiquantitative basis (0 = no lesion; 1 = mild; 2 = moderate; and 3 = severe), while neoplastic conditions were scored as present (1) or absent (0). The scores, along with numeric data such as age and weight, and nonnumeric data such as sex (0 = male; 1 = female) and presence or absence of clinical signs were analyzed in Excel to calculate a Pearson correlation coefficient (
Histopathology, Immunohistochemistry, and Ultrastructural Analysis
The composition of the inclusions in the cardiomyocytes was investigated using polarized light (
Heart tissue from one animal was fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer followed by a postfixation with 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. After 3 washes in distilled water, samples were dehydrated using a 25% to 100% ethyl alcohol gradient. Samples were then infiltrated in EMbed 812 resin (Electron Microscopy Sciences, Hatfield, PA), further embedded, and incubated at 58 °C for 24 hours to polymerize the resin. Embedded samples were sectioned on a Leica UC6 ultramicrotome (Leica Microsystems, Vienna, Austria). Thin sections (70–90 nm) were contrasted with 5% uranyl acetate for 20 minutes and Santos’ lead citrate for 6 minutes. These samples were visualized using a JEOL 1400 transmission electron microscope (JEOL Ltd, Tokyo, Japan). Images were obtained using an AMT Capture Engine Version 7.00 camera and software (Advanced Microscopy Techniques Corp, Woburn, MA).
Results
Demographics
Thirty guinea pigs met the criteria for inclusion in the study. The age range was 4 months to 8 years (mean = 3.8 years), and the group included 14 males and 16 females. In 26/30 (86%) guinea pigs, the sarcoplasm of cardiomyocytes contained 10 to 50 µm diameter, ovoid to amorphous, eosinophilic inclusions (Fig. 1).
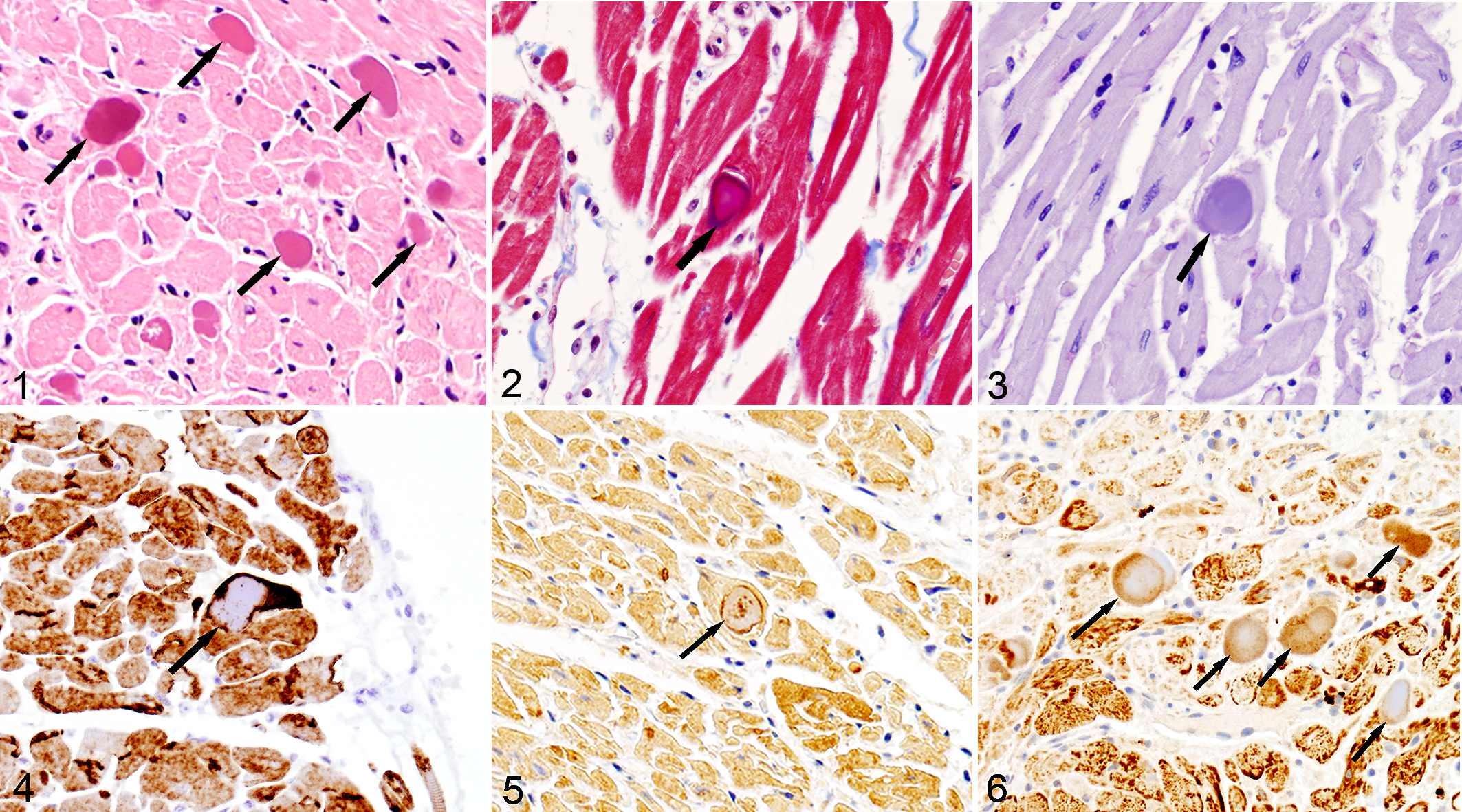
Protein aggregates, myocardium, guinea pig.
Histopathology, Immunohistochemistry, and Ultrastructural Analysis
The inclusions were not birefringent under polarized light, stained red to purple with Masson’s trichrome (Fig. 3), and did not stain with Congo red or periodic acid–Schiff (Fig. 3). With immunohistochemistry, the inclusions did not label for ubiquitin or cardiac myosin. Immunohistochemistry for desmin (Fig. 4), sarcomeric actin (Fig. 5), and muscle actin had variable peripheral immunoreactivity with some scattered punctate reactivity toward the center of the inclusion. Inclusions had variable peripheral, punctate to diffuse reactivity with antibodies to alpha B crystallin (Fig. 6).
Using electron microscopy, the inclusions were composed of large sarcoplasmic aggregates of disorganized myofilaments and microtubules surrounded by aggregates of loose intermediate filaments (Figs. 7, 8). The inclusions compressed and displaced adjacent myofibrils, endoplasmic reticulum, and mitochondria. Autophagic vesicles were not detected in any of the examined cardiomyocytes (with or without inclusions). One cardiomyocyte contained a single concentric lamellar body. No other ultrastructural abnormalities of the myocardium were detected.
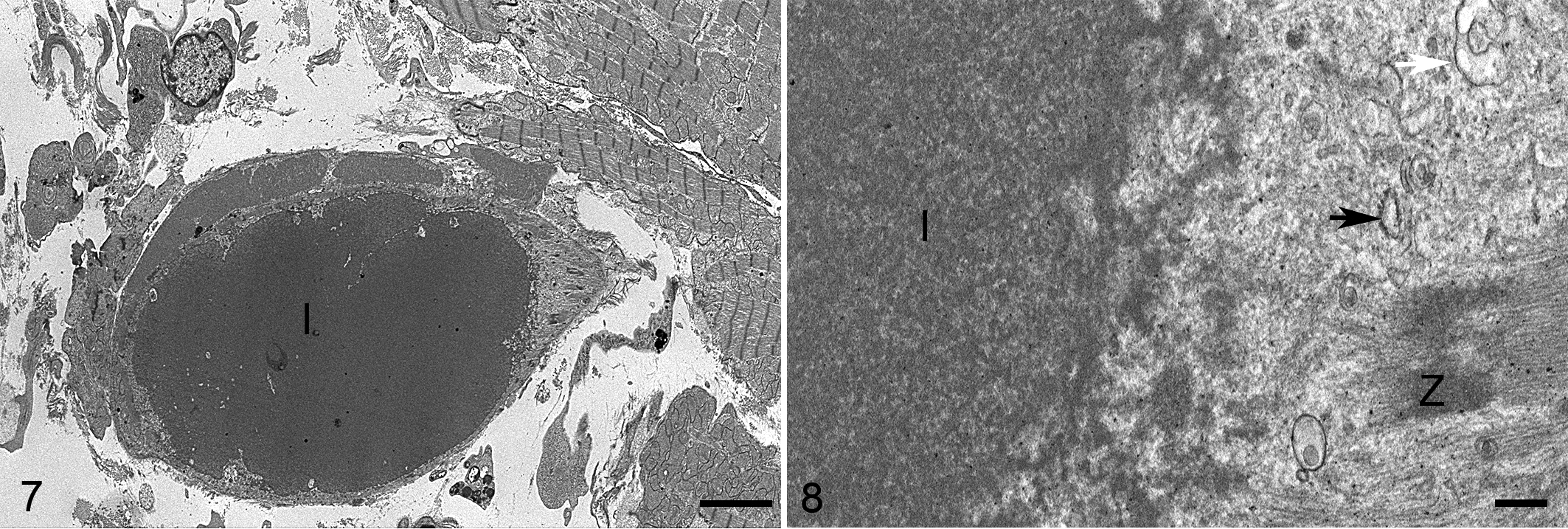
Protein aggregates, myocardium, guinea pig, case 28. Transmission electron microscopy.
Image Analysis
The inclusion burden in the ventricular myocardium ranged from 0.01 to 16 per mm2 (average 0.25 inclusions/mm2). The inclusion burden was highest in the right ventricular free wall (average 4.8/mm2, range 0–47/mm2), followed by the left ventricular free wall (0.7/mm2, range 0–9/mm2), and the septum (0.5/mm2, range 0–3/mm2). In all regions of the myocardium, the inclusions were most numerous in the papillary muscles and subendocardial myocytes (Fig. 9), and were often found in clusters, sometimes with multiple inclusions in a single cardiomyocyte.
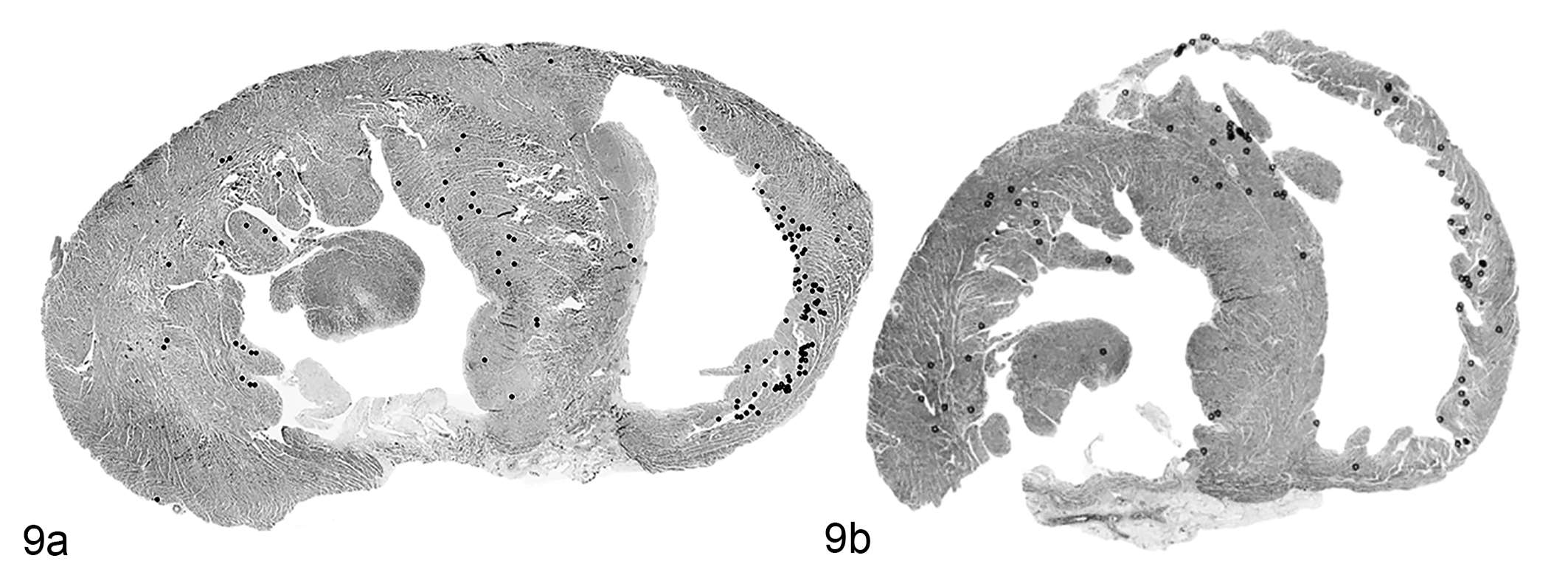
Distribution of protein aggregates, heart, guinea pig, cases 28 (Fig. 9a) and 29 (Fig. 9b). Annotation on subgross sections. Each black dot represents a microscopically detected protein aggregate. The aggregates are present in the right ventricular free wall, interventricular septum, and left ventricular free wall and are most concentrated in the subendocardial myocardium.
Statistical Analysis and Clinical Correlations
Inclusion burden did not appear to vary based on sex (
No inclusions were found in the atrial myocardium or skeletal muscle; however, skeletal muscle was only examined histologically in 5 of the guinea pigs.
Four guinea pigs had notably high inclusion burdens (>1 inclusion/mm2; 3 males and 1 female, age 2–6 years). Of note, all 4 of these guinea pigs had pulmonary edema, and hemosiderin-laden macrophages (“heart failure cells”) were detected in the lungs of 2 of these animals. Two of these 4 guinea pigs were euthanized (one was recumbent and one was having seizure-like episodes) and 2 died. In addition to pulmonary edema, 1 of these 4 guinea pigs had bacterial pneumonia, 1 had hepatic necrosis, 1 had meningitis and renal disease, and the cause of death in one was not determined but suspected to be secondary to hypoxia associated with air transportation.
Causes of death in other guinea pigs with inclusions included chronic renal disease (3/22 animals), amyloidosis (4/22), and lymphoma (3/22). Hepatic lipidosis (13/30), glomerulonephropathy (11/30), cystic ovaries (7/16 females), and adrenal cortical hyperplasia (14/30) were common histopathologic findings regardless of inclusion burden. The 4 guinea pigs with no detected inclusions ranged in age from 4 months to 1.5 years and included 3 males and 1 female. Two of these guinea pigs were euthanized and 2 died. Causes of death/reason for euthanasia in these 4 guinea pigs were meningitis, suppurative rhinitis, protein-losing nephropathy, and gastric perforation. The clinical and pathologic data for all guinea pigs are included in Supplemental Table S2.
None of the guinea pigs in this study were, to our knowledge, closely related (siblings, parent-offspring). All but one of the guinea pigs lived in upstate New York at the time of death. Information about the home of the other guinea pig was not available.
Discussion
Over 86% of the pet guinea pigs in this study had protein aggregates in the cytoplasm of cardiomyocytes, suggestive of abnormal protein homeostasis. Protein function depends on molecular shape, which is in turn determined by 4 levels of structure: (1) primary structure, or the sequence of amino acids; (2) secondary structure, or folding/coiling of the polypeptide backbone (forming alpha helices and beta pleated sheets); (3) tertiary structure, or 3-dimensional folding determined by interactions between hydrophobic domains in the amino acids; and (4) quaternary structure, or 3-dimensional interactions between individual polypeptide chains. The sequence of nucleotides in a specific region of DNA, which is transcribed to mRNA and translated by the ribosome into a chain of covalently linked amino acids, specifies the primary structure. Protein folding, which determines the secondary, tertiary, and quaternary structure of the molecule, begins during protein synthesis, while the growing polypeptide is still attached to the ribosome. The way a polypeptide chain is folded is a function of the sequence of amino acids, the cytoplasmic environment (pH, redox status, ion concentrations), and interactions with cofactors and chaperone proteins. Genetic mutations, alterations in cytoplasmic pH or redox status, and deficient or abnormal cofactors/chaperones can all result in protein misfolding. 28 Misfolded proteins are detected by heat shock proteins, resulting in upregulation of chaperones and cofactors to prevent aggregation and either fix the folding error or degrade the abnormal protein via the ubiquitin/proteasome or autophagy/lysosome pathway. Misfolding of proteins causes exposure of hydrophobic domains, which, if not promptly segregated, repaired, or degraded, leads to extra-lysosomal, cytoplasmic aggregation into insoluble complexes. Misfolded proteins can also persist in the cell as soluble oligomers, which in diseases such as Alzheimer’s are believed to be the principle cause of adverse effects on the cell (proteotoxicity). 26 The soluble oligomers can disrupt normal protein function, interact with membranes and lipids, and sequester chaperone, signaling, or trafficking proteins. Sequestration of these proteins into insoluble aggregates may represent a defense mechanism against the harmful effects of the soluble oligomers. 1 Aging and disease-associated stress, such as ischemia and decreased ATP production, can increase the incidence of protein misfolding and decrease repair and degradation capabilities. 31
Protein aggregate myopathies (PAMs) are characterized by extra-lysosomal accumulations of abnormal proteins which can affect skeletal and/or cardiac muscle. Causes include a wide variety of sporadic and familial genetic mutations, as well as aging and other processes which can affect normal protein folding or degradation. The complex cytoskeleton of muscle cells is composed of a vast array of proteins (Suppl. Fig. 1), 22 including sarcomeric components (actin, tropomysin, troponins, myosin, myosin-binding proteins, titin, nebulin/nebulette, and Z-disc proteins such as ZASP, FHL1, myotilin, and BAG3) and extrasarcomeric components (desmin, actin microfilaments, plectin, synemin, alpha B crystallin, and microtubules). Protein aggregate myopathies can involve any of these proteins and are broadly categorized based on desmin immunoreactivity of inclusions. Myofibrillar myopathies have desmin-positive protein aggregates. PAMs with desmin-negative inclusions include actin filament aggregate myopathies (actinopathies), and myosin storage myopathies (myosinopathies). 13
Myofibrillar myopathies, including desminopathy and desmin-associated myopathies, are the largest group of PAMs. 30 Although the clinical presentation is variable, these disorders are most commonly associated with adult-onset skeletal muscle weakness in the lower extremities, atrioventricular nodal conduction block, other arrhythmias, and cardiomyopathy. 30 Desmin is an intermediate filament protein that forms a 3-dimensional scaffold linking Z-disks (and thus the myofibrillar apparatus) to other cellular structures. Molecular alteration in the desmin gene causes abnormal structural or functional organization of this extrasarcomeric cytoskeleton, resulting in desmin accumulations near the Z-disc (desminopathies). 14 Similarly, molecular alterations in desmin-associated sarcomere-cytoskeletal linker proteins (plectin, 29 ZASP, 15 FHL1, 38 and myotilin 34 ) result in desmin-positive protein aggregates. Alpha B crystallin (CryAB; also known as heat shock protein B5) is a small chaperone protein abundantly expressed in the lens of the eye, skeletal muscle, and cardiac muscle. In muscle tissue, CryAB chaperones desmin and is therefore essential to cytoskeletal organization and function. 8 Mutations in the gene for CryAB result in aggregation of CryAB and desmin in the cytoplasm (crystallinopathies). 10 Other mutated proteins associated with myofibrillar myopathies include selenoprotein, 11 filamin, 37 lamin, 4 and valosin. 13
Actin filament aggregate myopathy, which is caused by mutations in the
In myosin storage myopathy or myosinopathy, aggregates of slow/beta-cardiac myosin heavy chain accumulate in type 1 myocytes. Myosin heavy chain is the molecular motor of the sarcomere and mutations in the tail of the slow/beta-cardiac myosin heavy chain gene (
In the heart, protein accumulation has occasionally been associated with hypertrophic cardiomyopathy 25 and dilated cardiomyopathy. 19 Pressure overload, 39 ischemic stress, 2 and aging 31 have been shown to impair cardiac proteostasis, and protein accumulation can be both a cause and a consequence of heart disease. Because of the abundance of specialized proteins for conduction and contraction, high metabolic demand, and post-mitotic state, myocardial cells are particularly vulnerable to disorders of proteostasis. 17 In mitotically active cells, accumulations of abnormal proteins are often sequestered in 1 of the 2 daughter cells, which subsequently is lost through apoptosis. 27 Mitotically inactive cells, such as cardiomyocytes, have no mechanism to dispatch the abnormal proteins and no means of replacing cells that die 5 as a result of proteotoxicity.
Based on histochemical staining, the cytoplasmic aggregates in cardiomyocytes of these guinea pigs are non-congophilic protein. Electron microscopy showed a heterogeneous mixture of myofilaments and microtubules. The aggregates had variable immunoreactivity to desmin, sarcoplasmic and muscle actin, and alpha B crystallin, also suggesting a mixture of proteins. These immunohistochemistry and ultrastructural features are most consistent with a myofibrillar myopathy.
The high prevalence of the aggregates in these pet guinea pigs suggests a species predisposition to disrupted cardiomyocyte proteostasis. Interestingly, similar inclusions have been detected in 3 porcupines and 2 chinchillas at Cornell University (Suppl. Figs. 2, 3). Guinea pigs, chinchillas, and porcupines are closely related, all belonging to the rodent infraorder Caviomorpha.
The involvement of CryAB, a protein that functions to prevent protein aggregation, may suggest decreased or abnormal functionality of this small heat shock protein in the development of myocardial protein aggregates in guinea pigs.
In humans, mutations in CryAB are associated with a variety of central nervous system, cardiac and skeletal muscle disease known as crystallinopathies. 7 The ultrastructural appearance of the inclusions in desmin-related myopathy associated with a missense mutation in CryAB in humans is very similar to what was seen in the guinea pigs. 36 Alternatively, or in addition to a genetic predisposition to protein aggregation, almost all of the guinea pigs with cardiomyocyte inclusions in this study had significant extra-cardiac disease, and this might have contributed to alterations in redox status or cytoplasmic pH, conditions known to increase protein misfolding. The only guinea pig with a high inclusion burden that did not have significant extracardiac lesions was one that died during air transport, likely due to hypoxia, which could also alter pH or redox status.
Although guinea pigs are frequently used as research animals, and even as models of cardiac function and disease, 16 these myocardial inclusions have not been previously described. Spontaneous heart disease is uncommon in guinea pigs. 9,23 Protein aggregate myopathies are rarely reported in other animals. Nemaline rod myopathy, an inherited disorder involving the structure of the sarcomere in skeletal muscle, has been reported in dogs 6 and cats. 3,20 Myofibrillar myopathy, with myofibrillar degeneration and aggregates of desmin in skeletal muscles, has been suspected in Warmblood horses. 31 None of the previous veterinary reports describe myocardial protein aggregates.
Conclusion
This report describes novel protein aggregates in the cardiomyocytes of pet guinea pigs. Guinea pigs with the highest burden of protein aggregates had changes in other organs suggestive of heart failure. This is, to our knowledge, the first report of protein aggregates in the myocardium of animals other than humans.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211042586 - Myocardial protein aggregates in pet guinea pigs
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211042586 for Myocardial protein aggregates in pet guinea pigs by Teresa Southard, Kathleen Kelly and Anibal G. Armien in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
