Abstract
Infectious laryngotracheitis (ILT) is an important upper respiratory disease of chickens. Gross and histologic lesions of ILT in chickens are compared to immunohistochemistry to evaluate the diagnostic test sensitivity. A total of 31 separate ILT-confirmed necropsy submissions (12 commercial meat-type flocks, 13 egg-type producers, and 6 backyard flocks) were arbitrarily selected. Each submission ranged from 1 to 18 birds, for a total of 246 chickens. Cases with available formalin-fixed tissues were selected to include a range of bird production types, ages, clinical histories, and severity of macroscopic and histologic lesions. Macroscopic findings in the respiratory tract varied from increased mucus (55.6%) to fibrinonecrotic exudate (20.3%) and hemorrhages in the larynx and trachea (13.0%). Syncytia with intranuclear inclusion bodies were present in the respiratory tract epithelium with or without hemorrhages. Sections of conjunctiva, sinus, larynx, trachea, lung, and air sac were analyzed by immunohistochemistry (IHC) to detect gallid alphaherpesvirus 1 (GaHV-1) antigen. Positive immunolabeling was detected in the cytoplasm and nuclei of syncytia and epithelial cells in 18/22 conjunctivae (82%), 12/13 sinuses (92%), 18/22 larynxes (82%), 23/25 tracheas (92%), 10/21 lungs (57%), and 3/8 air sacs (37%). Of the 34 tissues with no visible syncytia or inclusion bodies, 8 were positive by IHC. In conclusion, IHC was useful to study the viral antigen tissue distribution and support the diagnosis of ILT when the histopathologic interpretation was doubtful.
Keywords
Infectious laryngotracheitis (ILT) is an important highly infectious viral respiratory disease that mainly affects chickens, although pheasants, peafowl, and turkeys are also susceptible. ILT causes high morbidity, mortality, decreased egg production, and economic losses throughout the world. 2,7,16,22,25 The causative agent of ILT is gallid alphaherpesvirus 1 (GaHV-1), and the disease is characterized by ocular and nasal discharge, gasping, dyspnea, depression, and, in severe cases, expectoration of blood mixed with mucus. 3,7,15 In classic ILT, gross and histological lesions commonly consist of fibrinous, necrotizing and hemorrhagic sinusitis, laryngitis, tracheitis, and conjunctivitis associated with or without syncytia containing eosinophilic intranuclear inclusion bodies. 7,8 Depending on the severity and stage of the disease, syringitis, bronchitis, and bronchopneumonia, with or without syncytial cells, can also occur. Occasionally, ulcerative pharyngitis and esophagitis can also be observed. 21
Although classic ILT is more commonly reported in adult chickens, broilers as young as 16 days of age are also known to be highly susceptible to the disease. 5,9,13,17,22,24 Sometimes in broilers, lesions can be confined to the conjunctiva, nasal passages, and trachea with mild mucoid or catarrhal exudate. This can result in lack of characteristic respiratory signs, termed the “silent form” of ILT. Mortality can vary depending on the age of the birds, vaccination history, production type (broilers or layers), individual susceptibility, virulence of the strain, and occurrence of secondary bacterial infections or concurrent immunosuppressive or respiratory diseases. 3,7,15,16
Currently, histopathology is the screening test of choice for rapid diagnosis of a suspected case of ILT. However, characteristic lesions of GaHV-1 infection, such as syncytia and intranuclear inclusion bodies in the epithelial cells of the respiratory mucosa, are usually observed only from 1 to 5 days after infection. 7,10,20 Rapid and accurate diagnosis of ILT is critical in order to apply adequate corrective measures in the flock.
While histopathology is not conclusive if no syncytia are present, auxiliary tests such as fluorescent antibody (FA) and qPCR (real-time polymerase chain reaction) have also been used to confirm the diagnosis. The efficacy of these tests in diagnosing ILT has been well documented. 1,5,8,10,23 Where FA and qPCR are not available, immunohistochemistry (IHC) is a valuable tool that can be used to detect the antigen in cells, even in the absence of lesions. Although routinely performed in human and veterinary medicine, the use of IHC is often limited in avian diagnostics. 1,5,7 Even though an antibody directed against GaHV-1 is commercially available, studies on the distribution of the GAHV-1 antigen in the respiratory tract of affected chickens are currently lacking. Therefore, the aim of the present study was to review the lesions of ILT and to demonstrate the potential of IHC as an additional diagnostic tool for the investigation of GaHV-1 in chicken tissues, using arbitrarily selected GaHV-1-positive cases that were submitted to the California Animal Health and Food Safety Laboratory System (CAHFS) between 2005 and 2016.
Materials and Methods
Specimens and Clinical History
Necropsy submissions for poultry species at the Fresno, Tulare, and Turlock branches of the CAHFS laboratories (University of California, Davis) included from 1 up to 18 bird carcasses, each. Among hundreds of routine poultry necropsy submissions (or “cases”) received, between 2005 and 2016, 31 with available formalin-fixed tissues were arbitrarily selected for this study. Cases were selected to include gross and microscopic findings representative of disease variations of ILT in broilers, layers, and backyard chickens at different ages. Redundant specimens that also qualified for inclusions were omitted from the study.
These 31 cases consisted of a total of 246 chickens belonging to 13 commercial egg-type (119 birds), 12 commercial meat-type (112 birds), and 6 backyard (15 birds) flocks. Chickens were submitted either live (N = 127) or dead (N = 80), or not recorded (N = 39). All live and moribund birds were humanly euthanized by CO2 following the American Veterinary Medical Association guidelines. Commercial meat-type chickens ranged in age from 19 to 42 days for white broilers and up to 10 weeks for the lean brown broilers. Commercial egg-type chickens ranged in age from 5 to 78 weeks. The age of backyard chickens was frequently above 2 years, but this information was not always provided.
The laboratory protocol consisted of obtaining detailed information about each flock including farm locations, total number of birds in the house and in each ranch, clinical history, type of production system such as broilers or layers, sex, age, vaccinations, and treatments administered (Supplemental Table S1). Reported vaccinations included Marek’s disease, infectious bursal disease, Newcastle disease, infectious bronchitis, ILT, avian encephalomyelitis, and fowl pox. No vaccination history was available for 8 of 31 flocks. Diagnostic tests included complete necropsy, gross and histopathological evaluation, GaHV-1 detection by qPCR on tracheal swabs, serology using enzyme-linked immunosorbent assay (ELISA), IHC, FA, virus isolation, and ILT characterization by sequencing and restriction fragment length polymorphism (RFLP).
Necropsy and Histopathology
Complete necropsies were performed on all birds and lesions were recorded for each bird (Supplemental Table S2). Representative tissue samples of the respiratory tract (sinuses, larynx, trachea, syrinx, lungs, air sacs) and conjunctivae, as well as other organs were fixed in 10% neutral buffered formalin, routinely processed, paraffin embedded, sectioned at 4 μm, and stained with hematoxylin and eosin (HE) for histopathology. The presence of syncytia and intranuclear inclusions, as well as the degree of inflammatory and degenerative lesions were subjectively graded as negative (−), mild (+), moderate (++), and severe (+++) (Table 1). From a total of 327 respiratory tissue samples with HE-stained histologic sections, 117 were selected and scored based on their availability and overall quality (integrity of the sections, level of autolysis, presence of artifacts). Samples were often processed as pooled tissues rather than as tissues from individual birds. Therefore, the bird with the representative subjective score per overall case is reported in Table 1 per tissue type. Thus, “individual bird” was considered as the unit of analysis for the HE data.
Scoring of histopathologic lesions and immunohistochemistry for virus antigen in various tissues from 31 cases of infectious laryngotracheitis confirmed by auxiliary tests.
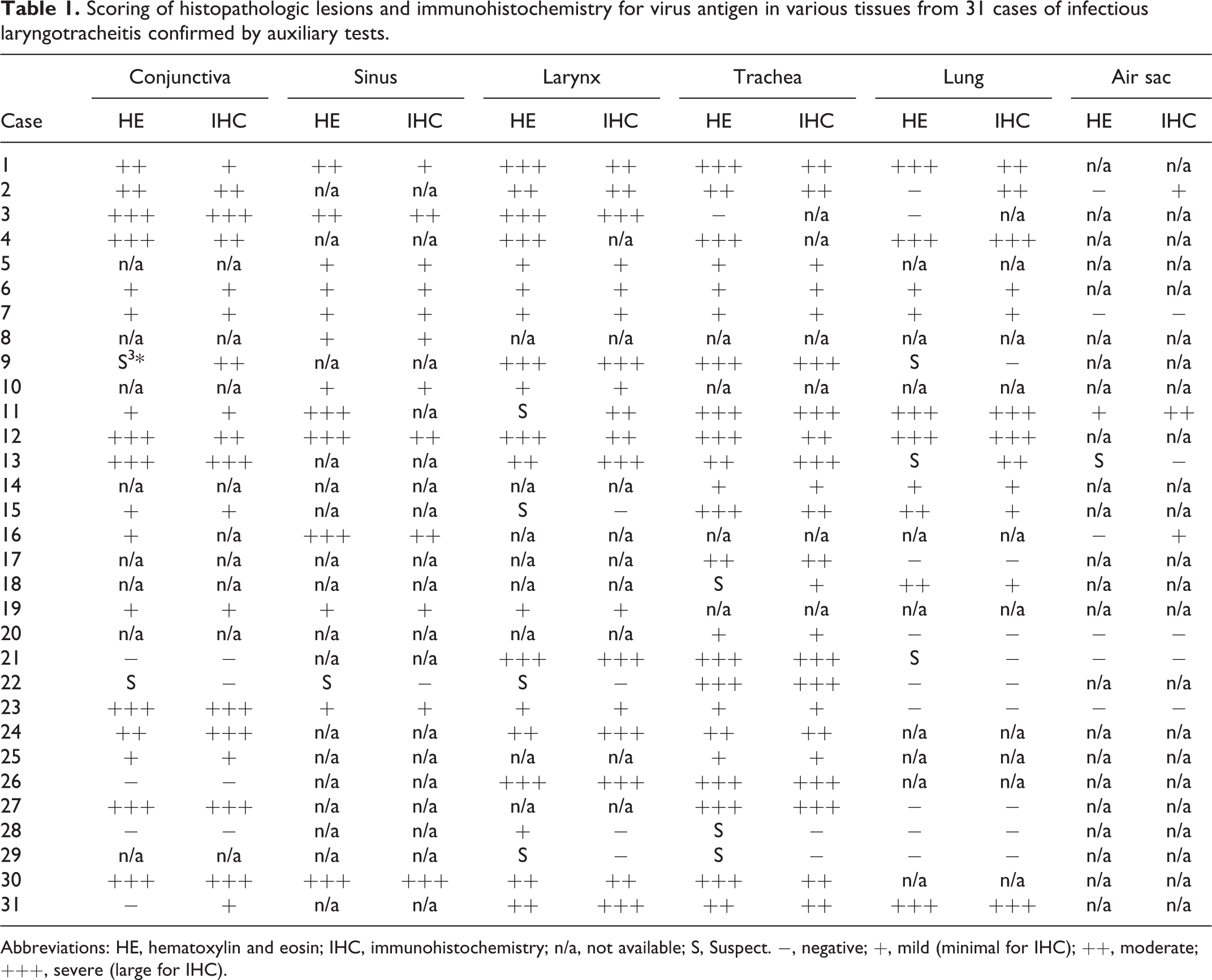
Abbreviations: HE, hematoxylin and eosin; IHC, immunohistochemistry; n/a, not available; S, Suspect. −, negative; +, mild (minimal for IHC); ++, moderate; +++, severe (large for IHC).
Immunohistochemistry
All cases were analyzed by IHC using an immunoperoxidase method (Envision+ System HRP) according to the slightly modified procedure and validated as previously described. 10 Briefly, the estimated sensitivity of the ILT IHC (using qPCR as the reference standard) was 100% with 95% confidence interval (74% to 100%) based on 11/11 samples testing positive on both tests. The estimated specificity of IHC (using PCR as the reference standard) was 100% with 95% confidence interval (44% to 100%) based on 3/3 samples testing negative on both tests (OpenEpi software using the Score [Wilson] method of estimating confidence intervals). 27 Furthermore, no cross-reactivity of ILT IHC was observed in respiratory tissues negative for GaHV-2 and confirmed positive by qPCR for IBV, NDV, IAV, or Pasteurella multocida.
Briefly, 4-μm-thick paraffin sections were mounted on charged slides, deparaffinized, and rehydrated. Following peroxidase inhibition by hydrogen peroxide, antigen retrieval was performed using a pressure cooker and a citrate buffer solution (Diva Decloaker, Biocare Medical) for 10 minutes. The slides were rinsed with TBS-Tween buffer and a casein-based blocking solution (Background Punisher, Biocare Medical) was applied. The tissue sections were incubated with a mouse monoclonal antibody (Mab 11.2 ascites, 1:4000) directed against GaHV-1 for 30 minutes at room temperature. 10 Following incubation with the HRP-labeled anti-mouse polymer for 30 minutes at room temperature, the color was developed with 3-amino-9-ethylcarbazole (AEC Substrate Chromogen, Ready-to-Use, Dako) and sections were counterstained with Mayer’s hematoxylin. Negative and positive controls were included with each IHC run and consisted of PCR-confirmed GaHV-1-negative and -positive tracheas, respectively. Among the 117 respiratory tissues tested by IHC, 6 could not be assessed due to artifacts. The amount of antigen in different tissues, in terms of number of stained cells, was subjectively scored as negative (−, 0%), minimal (+, <10%), moderate (++, 10-50%), and large (+++, >50%) (Table 1). For consistency, IHC results are reported as “individual tissue” and are paired with their respective HE tissue results.
Additional Testing
Portions of superficial trachea with exudate originating from 2 birds were fixed in Karnovsky’s fixative (2.5% glutaraldehyde, 2% paraformaldehyde in phosphate buffered saline) for 10 days, post-fixed in osmium tetroxide, routinely processed and stained with uranyl acetate, lead citrate, and osmium tetroxide, and examined by transmission electron microscopy. 6
Fresh sections of trachea (collected close to the larynx) and conjunctivae, originating from 4 chickens, were trimmed and placed in cryomolds, covered with O.C.T compound (Miles Scientific), frozen at −20 °C until sectioned, and processed using a polyclonal antibody reagent prepared against GaHV-1 in specific pathogen-free chickens (National Veterinary Services Laboratory, Ames, Iowa) and conjugated with fluorescein isothiocyanate diluted 1:40 with sterile 0.01 M, pH 7.2, phosphate-buffered saline. The slides were examined at 495 nm wavelength using a fluorescent microscope.
Pools of tracheal sections and swabs from 11 of the 31 cases were used for total DNA extraction, performed using the QIAamp Mini kit (Qiagen). 5 The qPCR assay for the detection of GaHV-1 and quantification of viral DNA was performed as previously described. 4 Briefly, the primers and probe targeting the viral glycoprotein C gene were synthesized by Integrated DNA Technologies and BioSearch Technologies. Copy numbers of the GaHV-1 genome were estimated per amplification reaction using the standard curve equation generated from the gC plasmid and expressed as log10. RFLP or sequencing were performed in 7 of 31 cases following qPCR results to determine the similarity with the vaccine and field strains. 12,14
Homogenized suspensions of tracheal tissue pools from 18 of the 31 cases were purified and inoculated into the chorioallantoic membrane of 9-day-old specific pathogen-free embryonated chicken eggs and tested for the presence of GaHV-1 as previously described. 5
Serum samples from 4 of 31 cases were tested for GaHV-1 antibodies by ELISA, according to the manufacturer’s instructions (ProFLOCK LT ELISA Kit, Synbiotics Corp).
Results
Clinical History
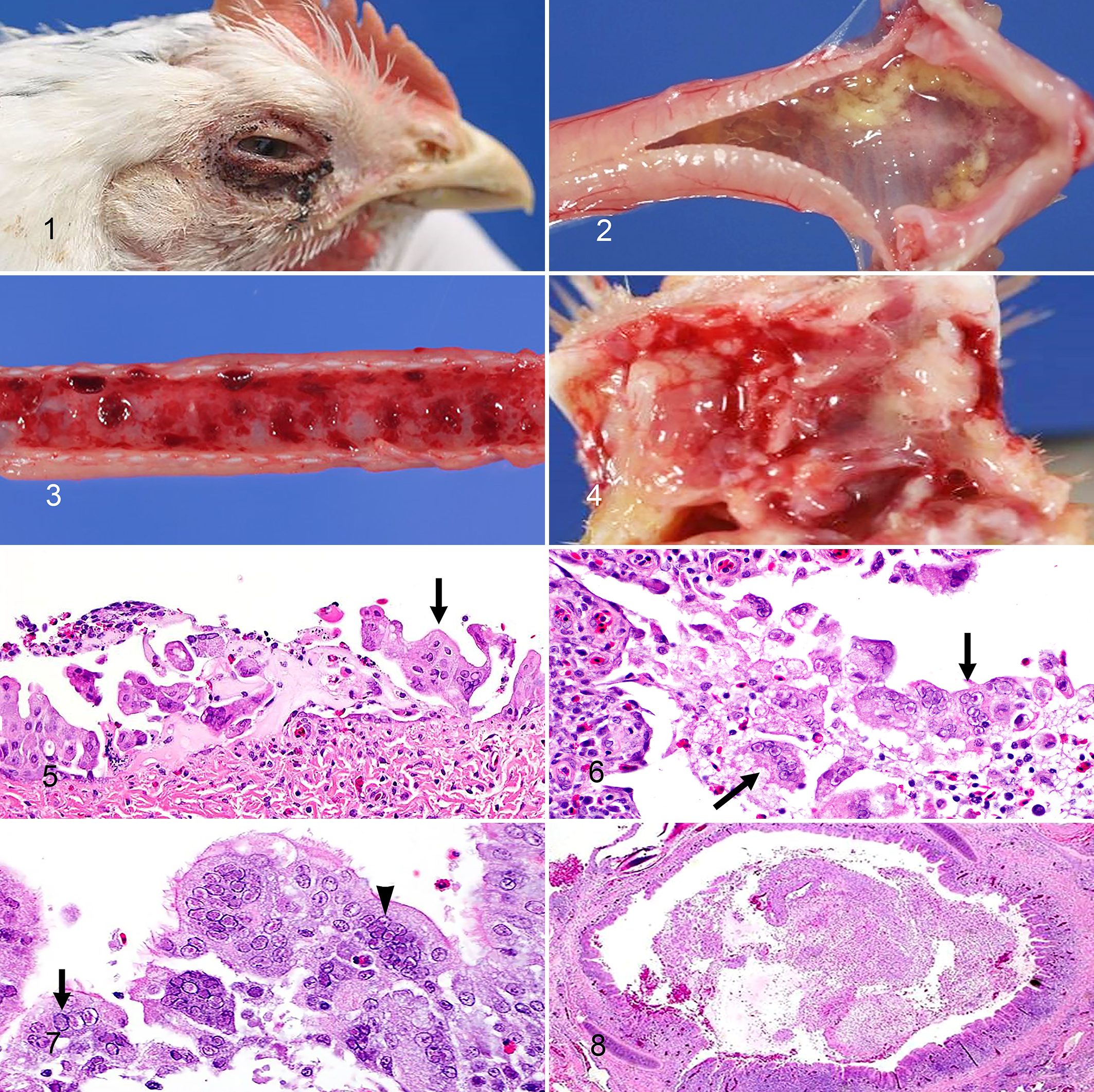
Clinical signs suggestive of GaHV-1 infection were observed in all 31 flocks (100%). Increased mortality (25/31 flocks, 81%) was the most common reason for submission, followed by respiratory signs (22/31 flocks, 71%), which included rales, gasping, sneezing, coughing, and dyspnea. Other reported signs included swelling of the eyelids (14/31 flocks, 45%), watery eyes with occasional conjunctival petechial hemorrhages (4/31 flocks, 12%) (Fig. 1), facial edema or swollen head (2/31 flocks, 6%), nasal discharge (7/31 flocks, 23%), and lethargy, ataxia, head tremors, and anorexia (11/31 flocks, 35%). In laying-type chickens a common history was a drop in egg production (4/31 flocks, 12%). Vaccination history for each flock was also retrieved for most cases (Supplemental Table S1).
Infectious laryngotracheitis, chicken.
Gross Pathology
Macroscopic lesions varied between submissions and between individuals of the same flock (Supplemental Table S2), depending on the age of the birds, the severity and stage of the disease, and the presence of concurrent infections (Supplemental Table S1).
Conjunctival lesions consisted of swelling of the eyelids due to periorbital edema associated with increased lacrimation (63/246 birds, 26%), conjunctival edema, and hemorrhage (from petechiae to diffuse in more severe cases). Mucosae of the nasal turbinates and infraorbital sinuses were also congested, and edematous (21/246 birds, 8%), with mild serous or sanguineous fluid and mucus accumulation within the lumen. Catarrhal to fibrinous exudate in the nasal passages (Fig. 4) were also observed (19/246 birds, 8%). Tracheal submucosal edema with increased mucus in the lumen (137/246 birds, 56%) was more frequently encountered in birds younger than 10 weeks. Only birds over 7 weeks of age had the classic diffuse tracheal hemorrhages (32/246 birds, 13%; Supplemental Table S2). Subacute to chronic lesions were characterized by formation of grey, firm, friable, caseous casts obstructing the lumen of the larynx and trachea (50/246 birds, 20%). Other gross findings included diffuse pulmonary congestion and caseous casts within the bronchi (24/246 birds, 10%), mild fibrinous exudate over the air sacs (20/246 birds, 8%), and bursal atrophy (56/246 birds, 23%). No remarkable differences were observed based on the bird’s production type, but this comparison was limited due to individual variations and complex clinical histories.
Histopathology
Since the number of tissues varied per case, the tissue with the representative subjective score per overall case and tissue type is reported as “individual bird” (Table 1). Therefore, pathognomonic syncytia and intranuclear inclusion bodies were present in the conjunctiva (17 of 23 birds, 74%; Fig. 5), respiratory epithelium of the nasal passages (13 of 14 birds, 93%), larynx (19 of 23 birds, 83%), trachea (23 of 27 birds, 85%; Figs. 6, 7), lung (10 of 22 birds, 45%), and air sacs (1 of 8 birds, 13%) in association with variable lesion severity. Overall, 83 out of 117 total respiratory tissues presented characteristic syncytia and intranuclear inclusions. In 14 of 117 tissues examined by HE, lesions were present but typical syncytia and inclusions were lacking or inconclusive due to sloughing of the surface epithelium or advanced necrosis.
Associated microscopic findings ranged from mild to severe and included segmental to diffuse loss of cilia, goblet cell hyperplasia, single-cell to diffuse coagulative necrosis, and sloughing of the respiratory epithelium. Acute cases were characterized by marked heterophilic inflammation, fibrin exudation, formation of fibrinous and heterophilic pseudomembrane-like exudate in the trachea (Fig. 8), and effacement of the mucosa. When present, caseous casts partially or completely obstructed the lumen of the airways and were characterized by necrotic cellular debris admixed with fibrin, erythrocytes, and viable or degenerating mixed inflammatory cells. Bacterial colonies were occasionally observed on the mucosal surface, within the intraluminal exudate or penetrating the ulcerated mucosa. In the nasal turbinates, larynx, trachea, and less commonly bronchi, the lamina propria was often expanded by variable numbers of inflammatory cells that were mostly lymphocytes and plasma cells and to a lesser extent, heterophils, macrophages, and multinucleated giant cells. Widespread hemorrhages and edema, occasionally extending beneath the cartilage septa or rings, were also present. In the most severe cases, marked hyperemia of vessels also accompanied the hemorrhages.
Histologic lesions in affected lungs ranged from mild multifocal to severe diffuse necrohemorrhagic bronchitis characterized by sloughing of the epithelium into the lumen and marked lymphoplasmacytic inflammation expanding the lamina propria. Lymphocytes and plasma cells often extended past the cartilaginous rings to the peribronchial interstitium and adjacent air sinusoids and septa. Air sacs were multifocally expanded mainly by lymphocytes and plasma cells, and lesser heterophils and macrophages, edema, fibrin, and hemorrhage. Syncytia with intranuclear inclusion bodies originated exclusively from epithelial cells and were most common in the respiratory epithelium of the conjunctiva, sinuses, larynx, trachea, bronchi, and air sacs in acute cases when the epithelium was still intact. In more advanced stages, syncytia with intranuclear inclusion bodies were mainly within intraluminal caseous casts and necrotic debris. Intranuclear inclusion bodies were present in syncytial cells or less often in individual epithelial cells. The inclusions were round to oval and often induced margination of chromatin to the periphery of the nuclei. Nuclear shape and size were minimally affected by the inclusions. Two different types of inclusions were identified, even within the same syncytium: one type was amphophilic to basophilic, completely filled the nucleus, and had a “glass-like” appearance; the second type was small, eosinophilic or “target-like” because it was surrounded by a halo, and did not entirely fill the nucleus (“eye-shaped”—Cowdry type A; Fig. 7). No correlation was found between birds’ age or production type and the presence of syncytia or intranuclear inclusion bodies.
Immunohistochemistry
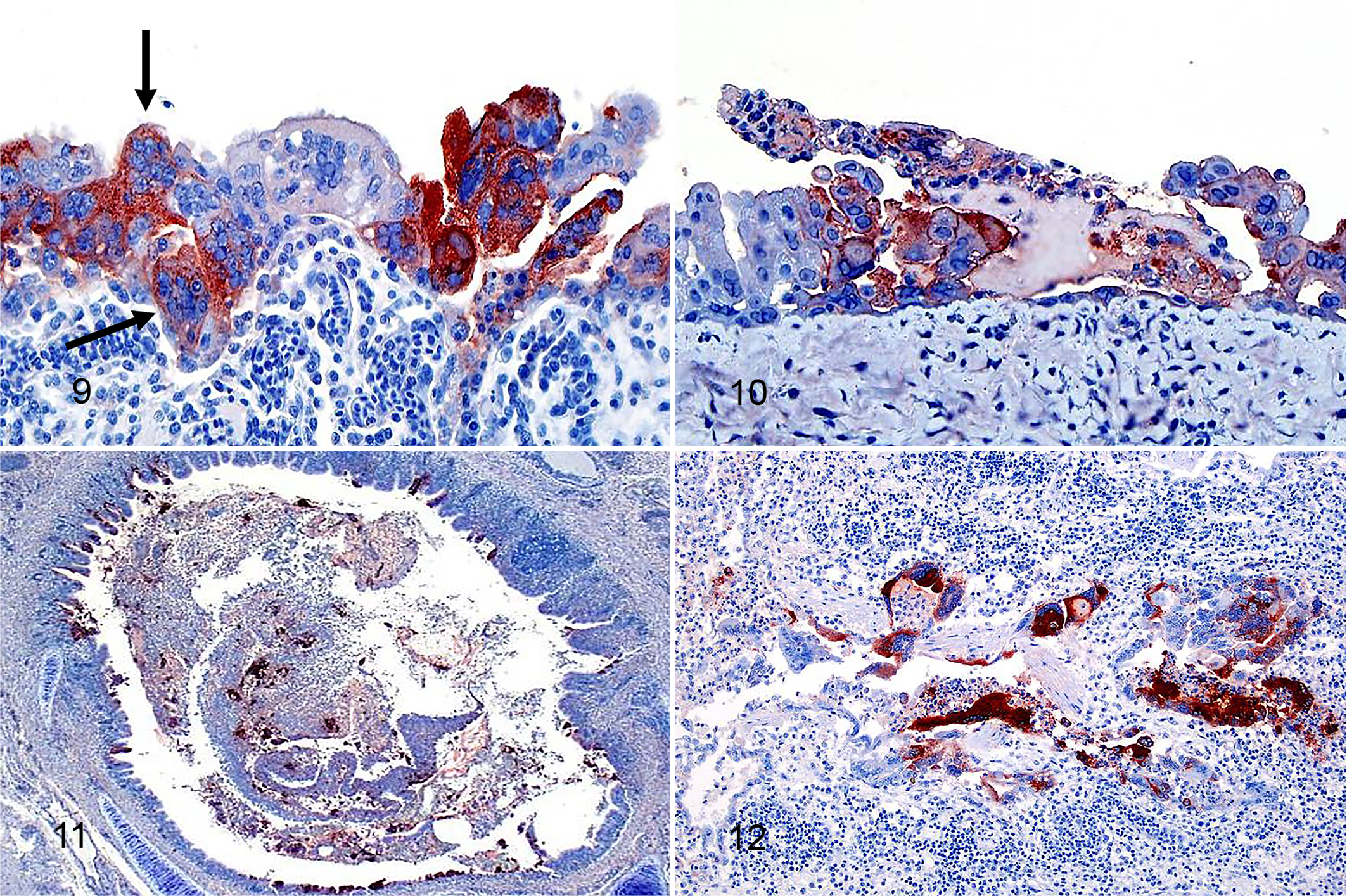
There was strong intracytoplasmic and intranuclear immunolabeling for viral antigen in GaHV-1-positive respiratory tissues (Table 1). The viral antigen was detected in the trachea (23 of 25 birds, 92%; Fig. 9), conjunctiva (18 of 22, 82%; Fig. 10), larynx (18 of 22, 82%), lung (12 of 21, 57%; Figs. 11, 12), sinuses (12 of 13, 92%), and air sacs (3 of 8, 37%). Of the 117 total tissue samples evaluated by histopathology (syncytia and/or inclusions) and 111 by IHC, 78 were positive by both tests, 24 were negative by both tests, 8 were positive by IHC but negative by histopathology, and 1 was negative by IHC but positive by histopathology. The majority of the negative tissues were lung and air sac, and occasionally conjunctiva (Table 1). The IHC test was positive for viral antigen in 8 tissues out of 34 that lacked characteristic syncytia and intranuclear inclusions, including 2 sections of conjunctiva, 1 of larynx, 1 of trachea, 2 of lung, and 2 of air sac (Table 1). Among these, 4 tissues were suspicious due to the presence of inflammation and hemorrhage. The remaining 10 ILT-suspicious tissues were negative by IHC. Concomitant respiratory infections were generally associated with higher lesion scores and stronger IHC positivity, with rare exceptions (Table 1, Supplemental Tables S1 and S2).
Infectious laryngotracheitis, chicken. Immunohistochemistry for gallid alphaherpesvirus 1.
Additional Testing
With transmission electron microscopy, syncytia and intranuclear viral particles were present in tracheal epithelium collected from 2 infected birds. Viral particles were round and approximately 200 nm diameter with an outer envelope, a protein tegument layer, and an inner capsid with icosahedral symmetry.
Of the 27 cases tested, 24 (89%) were positive for GaHV-1 antigen by direct immunofluorescence testing of trachea. The qPCR test was positive for GaHV-1 DNA in all 11 tested trachea samples. All 7 cases tested by RFLP were compatible with the CEO-like vaccine strains. GaHV-1 was successfully isolated and confirmed by FA testing of the chorioallantoic membrane in only 6 of the 18 cases tested (33%). Only 6 of 23 tested serum samples tested positive for GaHV-1 antibodies (26%) and the titers were lower than 1:256 (1:20 is the limit of detection).
Discussion
This study describes the gross and histologic lesions of ILT in chickens of different ages and production types, and compares the diagnostic test sensitivity of histopathology and IHC in birds with ILT. GaHV-1 causes an acute respiratory disease in chickens in which both severe and mild forms have been observed. Macroscopically, birds presented swelling of the eyelids due to periorbital edema, lacrimation, conjunctival petechiae, and congestion and edema of nasal turbinates and infraorbital sinuses with mucoid to fibrinonecrotic hemorrhagic exudate accumulation within the lumen. Submucosal tracheal edema in younger birds (<7-weeks) contrasted with the classic tracheal hemorrhages observed in older birds. Reasons for this difference is unknown, but it is described in the literature as “silent ILT,” a synonym for the milder form of the disease. 7,22 Laryngeal and tracheal caseous cast formation was also variable but rarely observed in younger birds.
Airsacculitis and bronchopneumonia associated with ILT have not been well documented and the incidence in our study was also low compared to lesions in other organs. The reason for this finding is not exactly known. Our clinical observations (HLS, unpublished data) suggest that airsacculitis and bronchopneumonia occur in the early stage or in mild forms of the disease. In addition, birds that die of ILT tend to have minimal lesions in the lower respiratory tract (HLS, unpublished data) perhaps because those with acute severe upper respiratory lesions die before the lesions extend into the lungs and air sacs. Thus, airsacculitis and bronchopneumonia might be more frequent in birds submitted live than in birds that die of ILT.
Depletion of lymphoid organs was a common finding especially in younger birds. However, thymus and cloacal bursa are difficult to assess in adult birds because of physiologic involution.
Syncytia with intranuclear inclusion bodies were key to histologic diagnosis of ILT. These were most frequently present in epithelium of the trachea, nasal turbinates and sinuses, conjunctiva, larynx, and primary bronchi. With the progression of the disease, the laryngeal and proximal tracheal epithelium degenerates and sloughs into the lumen forming the exudate that accumulates in and obstructs the syrinx. At the beginning of the infection, the first permissive tissues to develop intranuclear inclusions and syncytia are also those most affected by the inflammatory response and associated cytolysis, and thus the inclusion bodies might be progressively lost from these tissues. 7,19,20,24,26 In experimental infections of chicken embryo kidney cells, syncytia and intranuclear inclusions developed as early as 12 hours post infection. 20 However, these usually disappear by day 5 after infection, following necrosis and desquamation of the superficial epithelial cells. 7,20,22 The inclusion bodies that are the basis for histopathologic diagnosis could be obscured by the host response to the infection and may be absent by day 5 after infection. Thus, to increase the likelihood of identifying syncytia with inclusion bodies, different portions of trachea, sections of syrinx with caseous casts, and cranial areas of the lungs with primary bronchi and respiratory epithelium should be included among examined tissues.
Under experimental conditions, the age, breed/strain of bird, and viral dose are known factors that affect disease severity and susceptibility. 18 From our set of diagnostic cases, the numbers of syncytia and intranuclear inclusion bodies in the tissues did not seem influenced by either the age of the birds or the production type but appeared directly correlated to the disease severity. This can be explained as age and breed/strain variations are overcome by high virus challenge. 18,22 Concomitant respiratory infections were correlated with higher lesion scores, increase in numbers of syncytia with viral inclusions, and stronger immunoreactivity for the GaHV-1 nucleoprotein (Table 1, Supplemental Tables S1 and S2), with rare exceptions. Prior vaccination history did not correlate with disease severity, although limited information on the type of vaccines, quality and timing of the administration was provided.
In summary, the disease severity, as well as the number and distribution of inclusions and syncytia, depend on multiple factors, including the route of infection, repetitive exposures, stage of the disease, virus strain and tissue tropism of the virus, host age, bird breed/strain, concomitant infections, vaccination history, and individual differences in sensitivity and in response to the infection. 3
Currently, the most frequently used confirmatory diagnostic test for ILT is histopathology. Histopathology is relatively rapid (24 hours or less) and specific, although it relies on adequate sampling procedures and processing techniques. In addition, the temporal development of the intranuclear inclusions and their persistence in tissue samples must be carefully considered when analyzing histologic sections. 3,7,19,20,26 Compared to histopathology, we confirmed that IHC is useful to demonstrate infection when classical histologic lesions were absent or inconclusive. In particular, 8/34 tissue sections that were negative or ambiguous for syncytia and inclusion bodies were positive by IHC. Therefore, IHC identifies the tissue distribution of viral antigen and its localization in infected cells and necrotic debris, regardless of the presence of pathognomonic gross and histologic lesions. As for other IHC tests, limitations may include false negatives from prolonged exposure of tissues to formalin or from background staining, and the technique requires adequate laboratory capabilities. 11 Furthermore, neither IHC nor histopathology are able to differentiate ILT lesions caused by field strains from those caused by CEO-like vaccinal strains that reverted to virulence. This requires molecular characterization.
Beyond doubt, the most efficient and sensitive detection method is qPCR (11 out of 11 positive samples in this study). However, qPCR results need to be correlated with presence of characteristic lesions in order to exclude detection of inert viral DNA, or spontaneous intermittent shedding of nonpathogenic vaccine virus. 7 The least efficient methods for diagnosis of ILT were virus isolation (6 out of 18 positive cases) and serology (6 out of 23 positive cases). Virus isolation is traditionally associated with low diagnostic sensitivity, higher costs, and slow turn-around time, and is considered valuable only if further genetic characterization of the virus is required. 5,7 Serologic tests can be used to confirm exposure of a flock during the late stage of the infection and proof of immunization post-vaccination but provides limited diagnostic advantages early in the course of the disease.
The present study confirms the reliability and consistency of IHC for the diagnosis of ILT in chickens and its usefulness as a supplementary diagnostic test in subclinical cases and/or in research settings. While qPCR is probably the method of choice for quick turnaround times and diagnostic sensitivity, IHC is a reliable tool to study pathogenesis and distribution of virus antigen in tissues.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211035388 - Infectious laryngotracheitis of chickens: Pathologic and immunohistochemistry findings
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211035388 for Infectious laryngotracheitis of chickens: Pathologic and immunohistochemistry findings by Silvia Carnaccini, Chiara Palmieri, Simone Stoute, Manuela Crispo and H. L. Shivaprasad in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank the staff from the California Animal Health and Food Safety System including Karen Sverlow for developing and validating the immunohistochemistry for the ILT virus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
