Abstract
Broiler chickens on several farms from a single poultry company experienced neurological signs and mortality in chicks between 3 days and 10 days of age over a 3-week period after use of a fowlpox-vectored infectious laryngotracheitis virus vaccine in ovo. At necropsy the lungs contained numerous tan or gray, opaque to translucent, 0.5- to 2.0-mm nodules in the parenchyma. Microscopic lesions were a multifocal severe lymphohistiocytic and heterophilic bronchopneumonia. Immunohistochemistry was positive for fowlpox virus in macrophages and lymphocytes, and polymerase chain reaction on paraffin-embedded lung tissues was positive for a fowlpox vector virus commonly used as a vaccine. The cause of the neurological signs was not determined.
Several farms in a commercial broiler company experienced elevated mortality and neurological clinical signs of tremors, recumbency, torticollis, and wry necks in chicks ranging from 3 days to 10 days of age over a 3-week period in the spring of 2008. No management changes had been instituted at the farm level. Affected farms had a change in vaccination at the hatchery. Chicks on these farms received in ovo a vectored vaccine that contains fowlpox virus (FPV) with infectious laryngotracheitis virus gene inserts. Ages of vaccination ranged from 17.5 to 19.0 days of embryonation. The company veterinarian noted that flocks vaccinated at 17.5 to 18.0 days were more severely affected than those vaccinated at 18.5 or 19.0 days of embryonation. Gross lesions noted in the field included multiple tan to gray, opaque to translucent, 0.5- to 2.0-mm nodules within the lung parenchyma. Samples collected on the farms included formalin-fixed tissues (brains and lungs) and swabs for bacteriology of the brains. Additional farms submitted dead chicks, formalin-fixed and fresh tissue, or live chicks to the Poultry Diagnostic and Research Center laboratory at the University of Georgia for additional testing, including virus isolation and mycology culture. A total of 6 submissions for different farms were analyzed.
Brain swabs were negative for bacteria, and virus isolation was negative for avian encephalomyelitis virus in the brains. Lung cultures were negative for bacteria and fungi. Brains and lungs were routinely processed, embedded, and sectioned at 4 μm with routine hematoxylin and eosin (HE) staining. All submissions (cases Nos. 1–6) had similar changes in the lungs. Microscopic changes in the lungs included multiple areas of numerous histiocytes and lymphocytes centered on the parabronchi. The inflammatory cells filled the airways of several parabronchi and extended from secondary bronchi (Figs. 1, 2). There were occasional areas of acute necrosis of the inflammatory cells with mild numbers of heterophils migrating through the histiocytes to the foci of necrotic debris. Approximately one half of the lung parenchyma was affected in several sections. Gomori methenamine–silver staining was negative for fungal hyphae. Values based on central nervous system tissues were within normal limits.
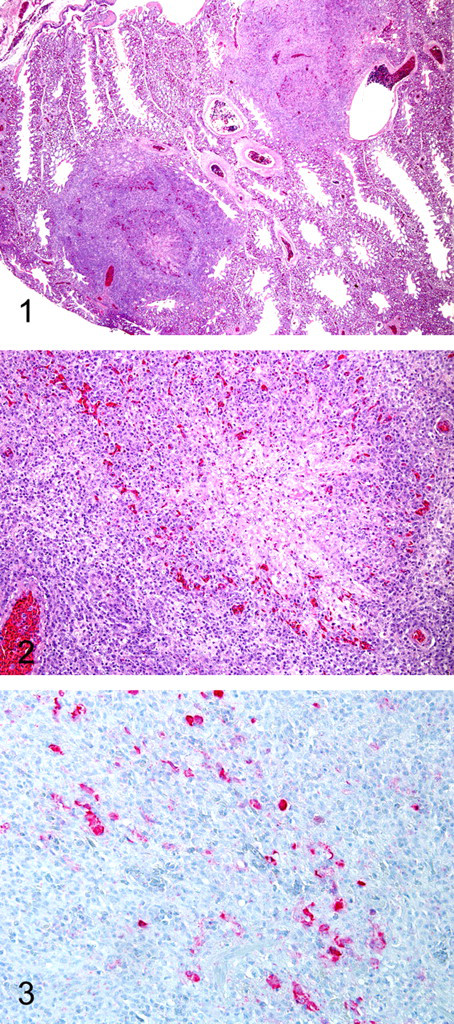
Immunohistochemical analyses were performed on lung sections (case Nos. 1 and 2) and stained for lysozyme and pan-cytokeratin to confirm the presence of histiocytes and absence of extraepithelial cells. Briefly, slides were heat treated at 60°C for 6 minutes in a baking oven. The slides were deparaffinized by a series of steps in xylene and graded alcohol baths. Next, the slides for lysozyme detection were heat treated with citrate buffer (pH 6.0; Biogenex, San Ramon, CA) for antigen retrieval. Slides were barcode labeled and placed on the Dako Autostainer (Dako, Carpenteria, CA). All slides were treated with 3% H2O2 for 5 minutes. Slides for pan-cytokeratin detection were treated with proteinase K for 5 minutes for antigen retrieval. All slides were treated with Power Block (Biogenex) for 5 minutes. The primary antibodies for pan-cytokeratin (1:100) (Lu-5 clone, Biogenex) or Lysozyme (1:1000) (Dako) were applied for 45 or 30 minutes, respectively. Biotinylated antimouse (Vector, Burlingame, CA) or biotinylated antirabbit (Vector, Burlingame, CA) antibody was, respectively, applied for 10 minutes. Horseradish peroxidase–labeled StreptAvidin (Dako) was applied for 10 minutes. Finally, DAB (Dako) was applied for 12 minutes. Slides were then counterstained with Gill II hematoxylin (Fisher Scientific, Suwanee, GA) and bluing reagent, dehydrated, cleared, and mounted. The infiltrating cells stained positively for lysozyme and negatively for pan-cytokeratin. Unstained slides (case Nos. 1 and 2) were sent to Michigan State University, Diagnostic Center for Population and Animal Health, for FPV immunohistochemical analysis. 10 An Enhanced V Red Detection System (alkaline phosphatase red) (Ventana Medical Systems, Inc., Tucson, AZ), as well as bulk buffers specifically designed for use on the BenchMark Automated Staining System (Ventana Medical Systems, Inc.) were used for immunolabeling and visualization. Briefly, slides were baked in a drying oven at 60°C for 30 minutes. The slides were then barcode labeled and placed in the BenchMark for deparaffinization and heat-induced epitope retrieval. Antigen retrieval was performed using retrieval solution CC1 (Medical Systems, Inc., Tucson, AZ) with an 8-minute-heating and 8-minute-cooling cycle. A mouse monoclonal anti-FPV antibody (U.S. Department of Agriculture, Avian Diseases and Oncology Laboratory, East Lansing, MI), at a concentration of 1:1,000,000 for 32 minutes, was used as primary antibody. The slides were counterstained using Ventana hematoxylin (Ventana Medical Systems, Inc.) and bluing reagent for 2 minutes each, then dehydrated, cleared, and mounted. Formalin-fixed, paraffin-embedded sections of chorioallantoic membrane inoculated with FPV were used as positive controls. For negative controls, the primary antibody was replaced with homologous nonimmune serum. Positive staining for FPV was observed within the lymphohistiocytic inflammatory cells (Fig. 3).
Polymerase chain reaction (PCR) amplification was performed on nucleic acid extracted from Vectormune FP-LT vaccine strains (Ceva/Biomune, Lenxa, KS; hereafter, FPV-LT [fowlpox virus–laryngotracheitis]) and a live-attenuated fowlpox vaccine, Fowl Pox Vaccine (Merial, Gainesville, GA; hereafter, CEO-FPV [chicken embryo origin–fowlpox virus]) and from paraffin-embedded lung tissues from FPV-LT-vaccinated birds (cases Nos. 1–6). Both vaccine viruses were extracted directly from the vial using the Qiamp Mini Kit (Qiagen, Valencia, CA) with modifications from the manufacturer’s recommendations, and the paraffin-embedded lung tissues were deparaffinized using CitroSolv (Fisher Scientific) and hydrated before DNA extraction. DNA extraction was performed with the Qiamp Mini Kit (Qiagen). Nucleic acid was eluted with 100 μl of elution buffer provided in the kit. Two sets of primers were utilized for PCR amplifications. Both sets of primers are located in reticuloendotheliosis virus–long terminal repeat (REV-LTR) sequences retained in the FPV genome. These remnants of integrated reticuloendotheliosis virus presumably arose during homologous recombination at one of the two regions common to both long terminal repeats or during retroviral excision from the FPV genome. 9 As a result, different portions of the integrated reticuloendotheliosis virus 5′ and 3′ long terminal repeats are found among FPV isolates. One set of primers was designed to amplify REV-LTR conserved sequences found among most FPV isolates and vaccine strains from the Unites States 5 (F-CTCTTCCTGTCGCCGTTAAG; R-GCCGATTCCGAGAAATGATA), and a second set of primers was designed to amplify FPV strains that have retained truncated REV-LTR sequences 5 (F-GGCTCGCTAACTGCCATATT; R-CTGCCCCACCTAGTGTGCAAG). All amplifications were performed using a PTC-200 thermocycler (BioRad, Hercules, CA). All reactions were conducted in a 50-μL reaction mixture containing 10 mM Tris-HCl (pH 9.0), 50 mM KCl, 1% triton X-100, 1.5 mM of MgCl2, 0.2 mM of each deoxynucleoside triphosphate, 5 μM of each primer, and 2.5 U of Taq DNA polymerase (Promega, Madison, WI). The amplification program consisted of a denaturation step at 94°C for 2 minutes, followed by 35 cycles of 94°C for 2 minutes, 57°C for 2 minutes, and extension at 72°C for 1 minute. A final extension cycle was performed at 72°C for 5 minutes. DNA from uninfected chick embryo fibroblast cells was routinely included in each assay as negative controls. Amplification reactions were analyzed by electrophoresis in 1X Tris Borate EDTA buffer at 100 V on 1% agarose gel with ethidium bromide (0.5 μg/ml). A PCR product of 195 base pairs was generated with primers homologous to the conserved REV-LTR region present in the CEO-FPV and the FPV-LT vaccines (Fig. 4, lanes 17 and 18) as well as in the lung tissues vaccinated with the FPV-LT vaccine (Figure 4, lanes 11 to 16). A 182–base pair fragment was amplified with primers homologous to the truncated REV-LTR region. Amplification was observed for the FPV-LT vaccine (Fig. 4, lane 9) and from lung tissue of FPV-LT-vaccinated birds (Figure 4, lanes 2 to 7). No amplification was observed for the CEO-FPV vaccine with this set of primers (Figure 4, lane 8). Based on these results, viral DNA extracted from the lungs of FPV-LT-vaccinated chickens does share REV-LTR sequences similar to the FPV-LT vaccine.
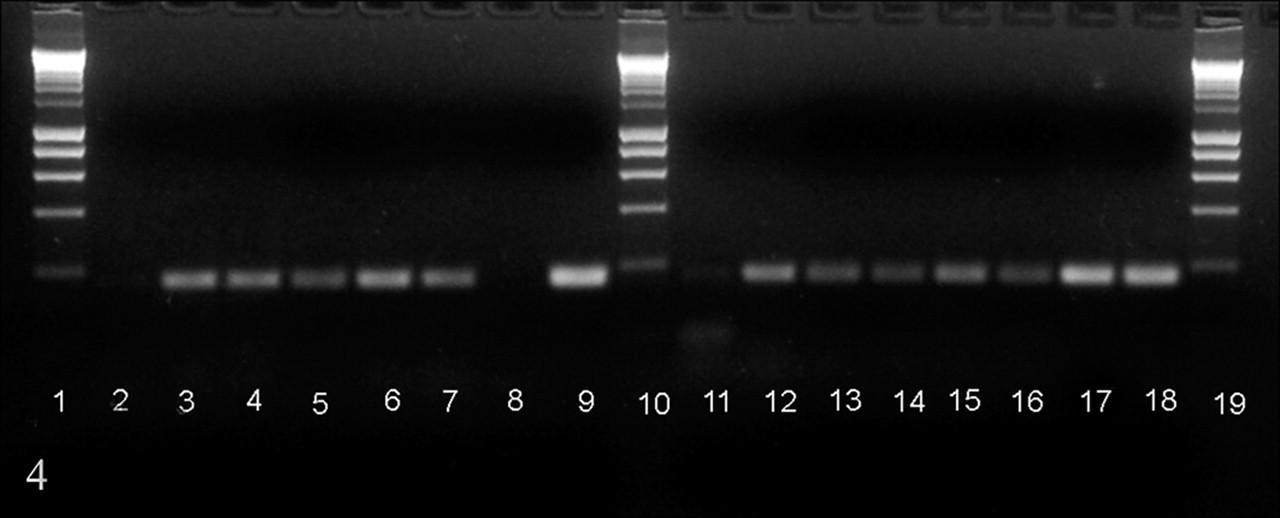
Polymerase chain reaction amplification on lung sections from chickens vaccinated for fowlpox virus–laryngotracheitis (FPV-LT). Lanes 1, 10, 19: molecular weight marker (Bioline, Tauton, MA); lanes 2 to 9, amplification with primers specific for fowlpox virus strains that retained truncated reticuloendotheliosis virus–long terminal repeat sequences; lanes 11 to 18, amplification with primers for conserved reticuloendotheliosis virus–long terminal repeat sequences retained among a variety of fowlpox virus strains. Lanes 8 and 17, FPV-LT vaccine; lanes 9 and 18, chick embryo origin–fowlpox virus vaccine; lanes 2 to 7 and 11 to 16, lung tissues from FPV-LT-vaccinated chickens.
The concept of in ovo vaccination has been well documented in the literature. 3,6-8 In ovo vaccination results in exposure to antigens between 17 and 19 days of embryonation and allows the developing chick to begin mounting an immune response before hatching, thus providing some protection when placed in the poultry house at 1 or 2 days of age. 4,5,7 Most poultry respiratory diseases are controlled by spray vaccination at day of age in the hatchery and with boosting, either by spray vaccination or in the drinking water, at 14 to 18 days of age in the field. 3
The severe vaccination reaction occurred in chicks vaccinated at 17.5 to 18.0 days of embryonation, whereas birds vaccinated at 18.5 or 19.0 days of embryonation experienced few clinical signs. The site of injection with in ovo vaccination varies with the age of embryo, needle gauge and length, and other factors. 1,2 Vaccination at earlier ages is associated with a higher percentage of injections into the amnionic fluid, and as the embryo ages, the percentage of injections into the embryo proper increases. Injection at earlier ages is also associated with a greater (though still small) percentage of injections into unintended sites, such as the air cell, allantois, or yolk sac. Billions of eggs are vaccinated in ovo each year in the USA. The reason for the adverse reaction in this case is unknown, but this reaction has been anecdotally reported from other operations using fowlpox vaccines in ovo (JA Smith, personal communication). The association of more frequent and severe reactions with administration at the earlier embryo ages suggests that either the injection of the unintended sites (air cell, allantois, or yolk sac) or the amnion is associated with the problem. However, even though the amniotic route becomes less frequent with advancing embryo age, it still accounts for a high percentage (approximately 30%) of injections at 19 days of embryonation. The percentage of injections into the unintended sites approached 6% in embryos at the earliest developmental stages in one study, and this figure is consistent with the incidence of problems noted on the most severely affected farms with chicks vaccinated at 17.5 to 18.0 days of embryonation (3 to 5% affected), suggesting a possible association. The reaction is also anecdotally reported to be dose dependent and is reportedly worse with CEO-FPV vaccines and less with tissue culture–derived fowlpox vaccines (C Williams, personal communication).
The cause of the neurological signs was not determined. Owing to the combination of neurological signs and the gross lesions in the lungs, as well as the initial association with chicks produced on one day at one hatchery, a systemic fungal infection with Aspergillus fumigatus or Onchroconis gallopava was initially considered. These were ruled out by fungal culture and histopathology. Other likely differential diagnoses—including avian encephalomyelitis virus, Newcastle disease virus, Marek disease virus, bacterial brain abscesses (Enterococcus durans or other bacteria), and nutritional encephalomalacia (vitamin E or vitamin A deficiencies)—were not supported by bacteriology, mycology, and virus isolation and particularly by the lack of characteristic histological lesions in the central nervous system.
The use of this particular vaccine in ovo is an off-label use, and the product was not licensed for this type of administration at the time. It is licensed for wing web injection at 8 weeks of age or older for adequate protection against infectious laryngotracheitis and fowlpox. After the outbreak of vaccine-induced pulmonary lesions, adjustments were made at the hatchery to vaccinate only at 19 days of embryonation, and the problem discontinued.
Footnotes
Acknowledgements
We would like to thank Gwin Kerce and Abbie Bulter for excellent histology and immunohistochemistry expertise.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
