Abstract
Recent reports have highlighted a lower-than-expected prevalence of neoplasia in elephants and suggested mechanisms for cancer resistance. But despite infrequent reports in the literature, uterine neoplasia is common in managed Asian elephants (Elephas maximus). This study is an archival review of reproductive tract neoplasia in 80 adult female Asian elephant mortalities in managed care facilities in the United States from 1988 to 2019. Neoplasms occurred in 64/80 (80%) of cases. Most were in the uterus (63/64; 98%) with only a single case of ovarian neoplasia. Myometrial leiomyomas were present in 57/63 (90%) cases with uterine neoplasia. Uterine adenocarcinoma was present in 8/63 (13%) cases. Remaining cases included endometrial adenoma (2), focal carcinoma in situ in endometrial polyps (1), anaplastic carcinoma (1), endometrial hemangioma (1), primitive neuroectodermal tumor (PNET; 1), and angiosarcoma (1). One case with uterine adenocarcinoma had a separate pelvic mass histologically characterized as an anaplastic sarcoma. Distant metastases were documented in 5/8 (63%) cases of uterine adenocarcinoma, and in the uterine anaplastic carcinoma, PNET, and angiosarcoma. Four uterine adenocarcinomas and one carcinoma in situ were examined immunohistochemically for pan-cytokeratin, vimentin, and estrogen receptor. In all, neoplastic cells were pan-cytokeratin positive and vimentin negative, and in 2 cases were immunoreactive for estrogen receptor. Results show that female reproductive tract neoplasia, particularly of the uterus, is common in Asian elephants and is not limited to leiomyomas. Importantly, uterine neoplasms have the potential to impact fecundity and may represent obstacles to conservation in managed care.
Keywords
Current scientific literature suggests that neoplasia in elephants is rare. Few descriptions exist, and the majority are individual case reports. 7,8,16,20,28,31,34,45,46,52,54,56,60,62,67 Elephants have been used as an example of Peto’s paradox, the observation that incidence of cancer at the species level does not correlate with the number of cells in an organism. Studies cite a lower-than-expected rate of neoplasia in elephants given their large size and extended lifespan. 1,21,23,57,58,61 Some reports have proposed that cancer resistance exists in elephants and may be mediated by multiple copies of the tumor suppressor gene, TP53, in the African elephant (Loxodonta africana) genome. According to this hypothesis, a resulting increase in the cellular apoptotic response following DNA damage, as compared with human cells, protects against carcinogenesis. 1,18,61,65 However, confirmatory evidence of a causal link between extra TP53 copies and cancer suppression in elephants has not been documented. 1,10 Furthermore, it is likely that elephant neoplasia is underreported in the scientific literature. Analyses of archival postmortem data from one large zoo-based veterinary pathology program revealed an estimated lifetime prevalence for malignancies of 33.3%, and with inclusion of benign neoplastic lesions, prevalence of neoplasia was 66.7%. These data show that prevalence of neoplasia in captive elephants is similar to humans and other captive zoo mammals. 6,49 Despite the limited literature, the practical experience of veterinarians, pathologists, and caretakers indicates neoplasia may be an important factor in health and welfare.
In mature female Asian elephants (Elephas maximus), neoplasia involving structures of the reproductive tract is common, and myometrial leiomyomas have been recognized most often. 41,51,56 Professionals involved in the managed care of elephants would expect to find myometrial leiomyomas during the postmortem examination of an aged female Asian elephant. However, to date, no comprehensive assessment of reproductive tract neoplasia in Asian elephants has been done. Assimilating information on the morphologic characterization, prevalence, and possible clinical implications of reproductive tract lesions is a high priority for understanding and improving the management of reproductive health in an aging, managed, endangered population for which continued reproduction is a desired goal. Historically, growth and maintenance of managed elephant populations in the United States were facilitated by importation, but such practices are no longer favored. Current management preferences are to foster self-sustaining populations maintained through captive-breeding. Optimal reproductive health and fertility are essential to the fulfillment of such goals. 2,25,68
The objective of this study was to describe reproductive tract neoplasia in adult, female Asian elephant mortalities from managed care facilities in the United States.
Materials and Methods
Archival Review of Postmortem Records
Postmortem reports, including gross and histologic findings, for adult (greater than 10 years of age at time of death) female Asian elephants submitted for examination between 1988 and 2019 were requested from 15 collaborating veterinary diagnostic programs. Reports were evaluated to (1) document any gross and/or histologic neoplastic lesions involving the ovaries, uterus, cervix, or vagina; (2) characterize and categorize specific tumor types and assess malignancy including associated metastatic disease; (3) summarize gross, histologic, and any available immunohistochemical descriptive information for the spectrum of reported neoplasms; and (4) note any concurrent, non-neoplastic reproductive lesions including cystic endometrial hyperplasia, endometrial polyps, and adenomyosis. In addition, age and, where available, information regarding reproductive history were recorded. Gross and histologic descriptive data were derived from evaluation of the archival reports. Histologic sections were not reviewed for the purposes of this study. Similarly, most immunohistochemistry (IHC) data were gleaned from the archival review of the postmortem reports. These immunohistochemical analyses were performed at the time of initial diagnostic evaluation by the receiving pathology service, and protocols were not repeated for the purpose of this study. Protocol information (antibody source, controls) is provided in Supplemental Table S1. Archival immunohistochemical data were reported in this study as described in the submitted postmortem reports. Cases were diagnosed by veterinary pathologists certified by the American College of Veterinary Pathology, many having extensive experience with zoo and wildlife species. Thus, original results and interpretations provided in postmortem reports were considered sufficient for analysis. All available, pertinent information was transferred to a database for summary and analysis. Data were then used to determine prevalence of reproductive tract neoplasia in the study population, organ-specific prevalence, and prevalence of benign and malignant tumors.
Malignant uterine neoplasms in 12 Asian elephants (Elephas maximus) from United States managed care facilities.
Abbreviation: PNET, peripheral neuroectodermal tumor.
This case also had a large pelvic mass histologically categorized as an anaplastic sarcoma.
b This case had a large mass at the pelvic inlet presumed to be of uterine origin. Uterine adenocarcinoma was also present in this case (same case as “a” above).
Immunohistochemistry
For the purpose of the study, additional immunohistochemistry was performed on a subset (n = 4) of uterine adenocarcinomas and one carcinoma in situ arising within an endometrial polyp for which archival materials (formalin-fixed paraffin-embedded [FFPE] tissues) were available. In all 4 cases, immunohistochemistry was done using the Ultravision Quanto Detection System AP (Thermo Fisher Scientific) for pan-cytokeratin (CK) and vimentin, and the MACH 4-AP Universal kit (Biocare Medical) for estrogen receptor (ER). Serial sections incubated with Tris-buffered saline (TBS; Biocare Medical) in place of primary antibody served as negative controls. Protocol positive controls included sections of domestic canine skin (CK and vimentin) and uterus (ER). Additionally, a section of elephant uterus including endometrium, smooth muscle, and vessels was used as a species-specific positive control to confirm antibody cross-reactivity. Five-micrometer-thick sections were cut from FFPE tissues, mounted on charged slides, air-dried overnight, deparaffinized, and rehydrated.
CK
For antigen retrieval, slides were incubated in HEIR buffer L (Thermo Fisher Scientific) at 90 °C for 5 minutes under 13-15 PSI (Biocare Medical Decloaking Chamber; Biocare Medical), cooled, rinsed, incubated in Carezyme II: Pepsin (Biocare Medical) for 15 minutes, rinsed, buffered in TBS (Biocare Medical), and blocked with UltraV block (Thermo Fisher Scientific). Slides were sequentially incubated in mouse monoclonal Lu-5 pan-cytokeratin antibody (Biocare Medical) for 30 minutes, Primary Antibody Amplifier Quanto (Thermo Fisher Scientific) for 10 minutes, and AP Polymer Quanto (Thermo Fisher Scientific) for 10 minutes.
Vimentin
For antigen retrieval, slides were incubated in HEIR buffer L (Thermo Fisher Scientific) at 100 °C for 10 minutes under 13-15 PSI (Biocare Medical Decloaking Chamber; Biocare Medical), cooled, rinsed, buffered in TBS (Biocare Medical), and blocked with UltraV block (Thermo Fisher Scientific). Slides were sequentially incubated in rabbit monoclonal vimentin SP20 (Biocare Medical) for 45 minutes, Primary Antibody Amplifier Quanto (Thermo Fisher Scientific) for 10 minutes, and AP Polymer Quanto (Thermo Fisher Scientific) for 10 minutes.
ER
For antigen retrieval, slides were incubated in HEIR buffer M (Thermo Fisher Scientific) at pH 8.0, 120 °C for 20 minutes under 13-15 PSI (Biocare Medical Decloaking Chamber; Biocare Medical), cooled, rinsed, and buffered in TBS (Biocare Medical). Slides were sequentially incubated in mouse monoclonal ER 6F11+ SP1 (Biocare Medical) overnight at 4 °C, followed by Mach 4 Universal AP Probe (Biocare Medical) for 30 minutes, and Mach 4 Universal AP Polymer (Biocare Medical) for 30 minutes. All sections were then visualized using Apply Vulcan Fast Chromagen (Biocare Medical) and counterstained with methylene blue (Becton, Dickinson and Company).
Results
Study Population
A total of 80 postmortem reports from adult, female Asian elephants from 26 different holding facilities were reviewed (Suppl. Table S2). Study cases represented 80/94 (85%) of all adult female Asian elephants that died or were euthanized between 1988 and 2019 (Bob Lee, Association of Zoos and Aquariums Elephant Taxon Advisory Group and Species Survival Plan studbook keeper, personal communication). Neoplasia of the reproductive tract was present in 64/80 (80%) of cases. Age of animals with neoplasia ranged from 13 to 71 years (mean = 47 years; standard deviation = 11 years). The age range of animals without neoplasia was 19 to 65 years (mean = 47 years; standard deviation = 14 years). Reproductive history was available for 19 cases; 17 with and 2 without reported neoplasia. All 19 elephants were nulliparous. The majority of neoplasms occurred in the uterus (63/64; 98%); a single case of ovarian neoplasia (carcinoma) was described. One or more ovarian cysts were noted in 11/80 (14%) cases. Hyperplastic, polyploid, or papillomatous mucosal lesions of the vagina/vulva were noted in 10/80 (13%) of cases. No primary cervical neoplasms were reported. No neoplastic lesions involving the epithelium of the distal reproductive tract (vagina, vulva) were reported. A single vaginal leiomyoma was described.
Myometrial leiomyomas were the most frequently identified uterine neoplasms and were present in 57 (71%) of all 80 elephants and 57 (90%) of the 63 elephants with uterine neoplasia. Uterine neoplasia other than leiomyoma was detected in 15/63 (24%) cases; 13 of the neoplasms were malignant (Table 1). Uterine adenocarcinoma was most common and found in 8/63 (13%) of cases. Other neoplasms included endometrial adenoma (2), carcinoma in situ that occurred within endometrial polyps (1), anaplastic carcinoma (1), endometrial hemangioma (1), PNET (1), and angiosarcoma (1). One of the cases with uterine adenocarcinoma also had a separate large pelvic mass that was histologically characterized as an anaplastic sarcoma.
Myometrial Leiomyoma
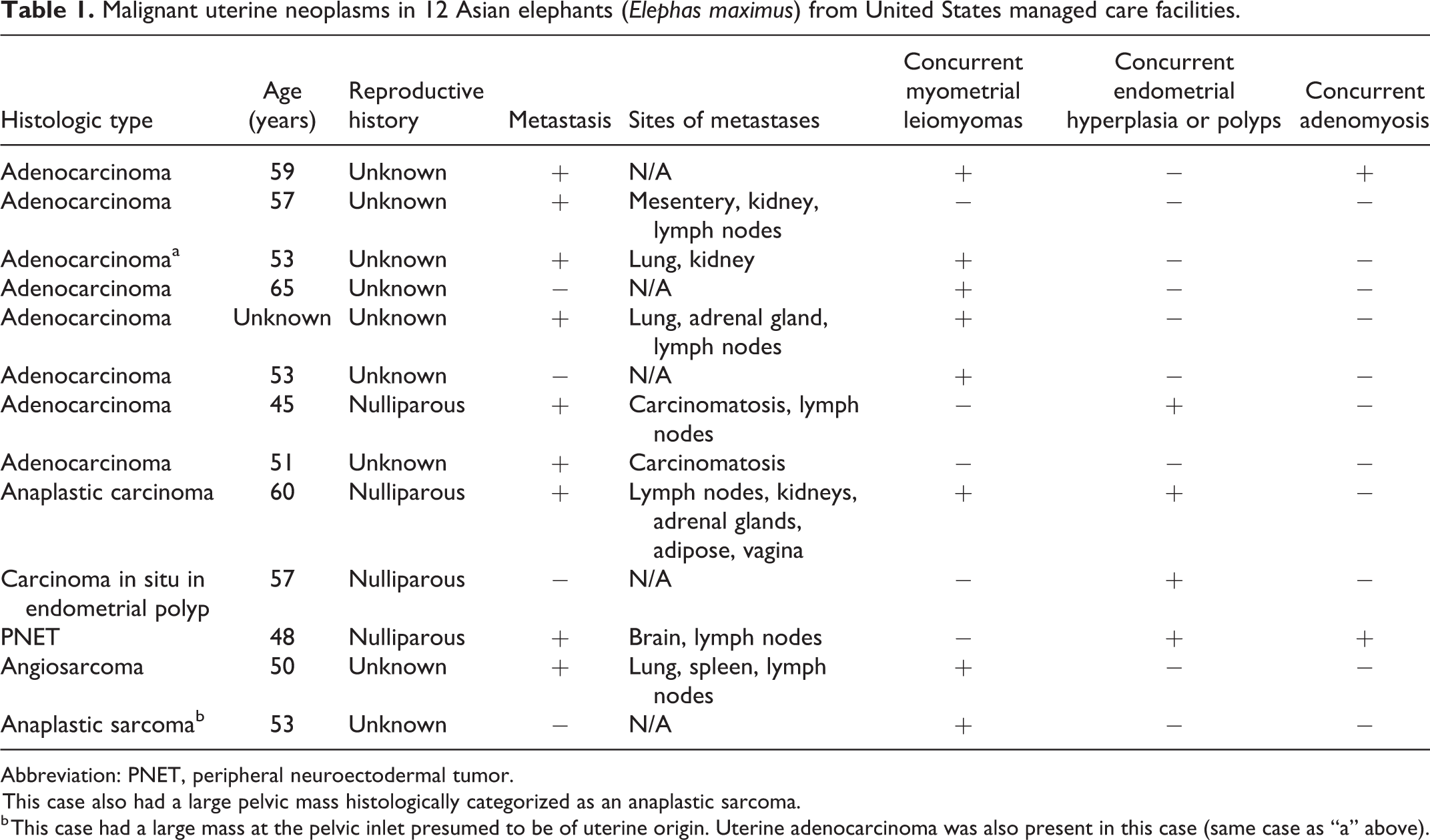
Of the 57 cases with myometrial leiomyoma, the neoplasms were multiple in 51 cases, and solitary masses in 6 cases. Leiomyomas were grossly visible in all but one case, in which the masses were detected histologically. Leiomyomas were present in the uterine horns and body and ranged from microscopic to over 100 cm in diameter, though most were 1 to 10 cm. Grossly, leiomyomas were discrete, unencapsulated, firm, tan-white, nodular, masses that appeared to arise from the myometrium (Figs. 1, 2). In some cases, numerus coalescing masses markedly distorted uterine architecture and impinged upon and obscured the uterine lumen (Fig. 3). Histologic features were typical of benign smooth muscle neoplasms and consisted of long intersecting bundles of large spindyloid mesenchymal cells supported by variable amounts of fibrous stroma (Fig. 4). Cells had moderate amounts of eosinophilic, fibrillar cytoplasm, and indistinct borders. Nuclei were cigar-shaped to fusiform with stippled chromatin and indistinct nucleoli. Anisocytosis and anisokaryosis were minimal to mild. Mitotic figures were either not observed or averaged less than 1 per 400× field. Immunohistochemical characterization of the neoplastic cells was described in the postmortem reports from 4 cases (Suppl. Table S1). Cells were positive for desmin and smooth muscle actin. In one case, immunolabeling for estrogen receptor was performed, and positive staining was detected. No vascular invasion or metastasis to indicate malignancy were reported in any of the cases. In 9 of the 57 cases, a separate, concurrent neoplastic process was also present in the uterus.
Uterine leiomyomas, Asian elephant (Elephas maximus), uterus.
Uterine Carcinoma
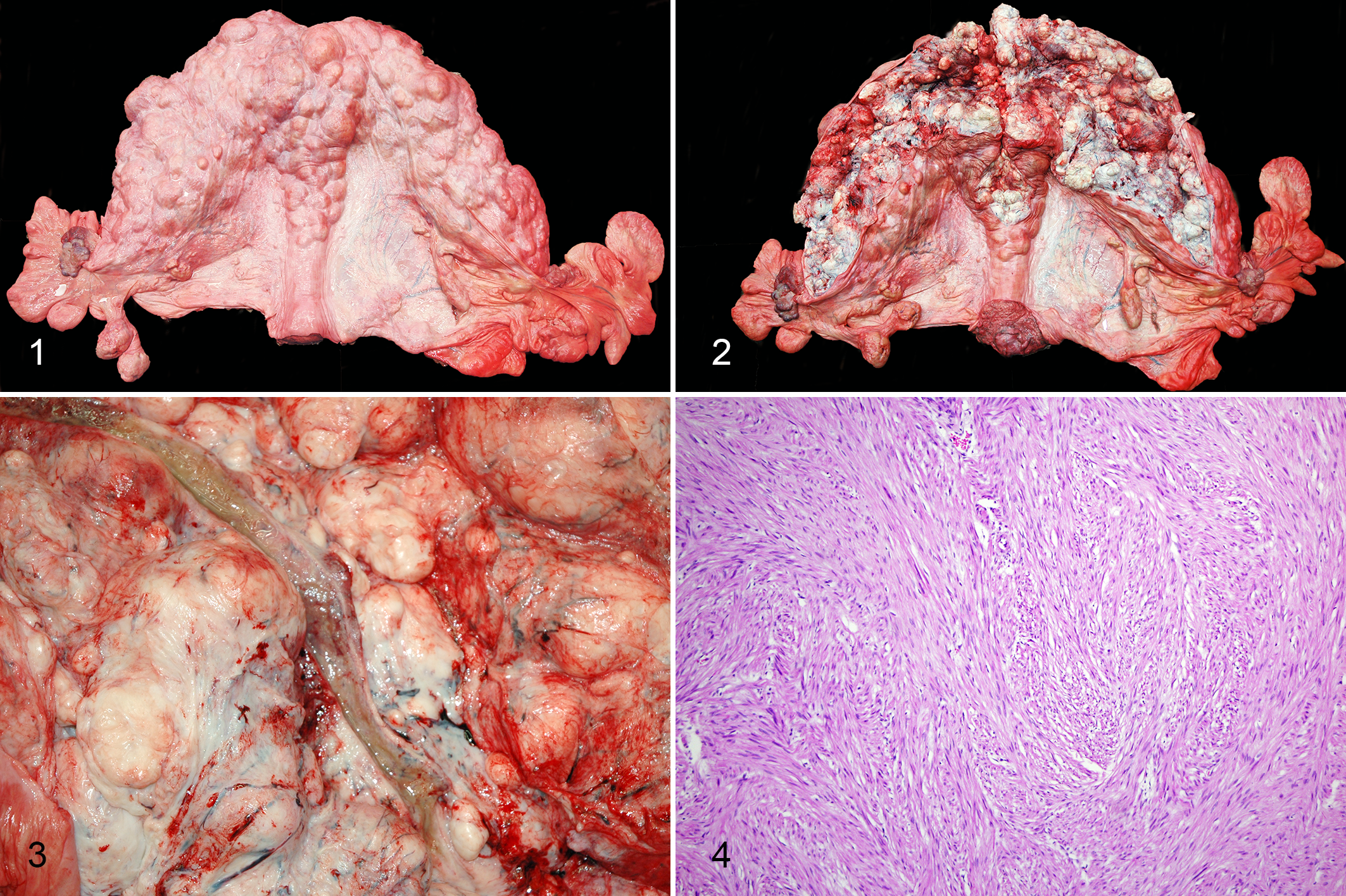
There were 8 cases of uterine adenocarcinoma. All uterine masses were uniform and had gross characteristics compatible with myometrial leiomyomas as described above. Multiple leiomyomas were additionally present in 5 of the 8 cases of uterine adenocarcinoma, and in these cases the adenocarcinoma was uniform and grossly indistinguishable from the leiomyomas. In the remaining 3 cases, adenocarcinomas were poorly demarcated, multinodular masses that ranged from 12 to greater than 30 cm in diameter with distortion of the uterine wall and elevation of the endometrial surface (Fig. 5). These masses were firm with a rough, irregular endometrial surface and, on cut section, often had numerous small cystic cavitations containing yellow-tan, mucoid material, and/or yellow-tan, friable foci consistent with necrosis. Histologically, neoplastic cells were arranged in solid nests, acini or irregular, elongate, tortuous glandular structures, sometimes with luminal papillary projections (Fig. 6). Supporting stroma was typically abundant and densely collagenous. Neoplastic cells ranged from cuboidal or columnar to polygonal and had moderate amounts of eosinophilic granular to wispy cytoplasm and indistinct borders. Nuclei were typically basally oriented with stippled to vesicular chromatin and one or more prominent nucleoli. Of the 8 uterine adenocarcinomas, 6 had histologic features indicative of malignancy (ie, cellular pleomorphism, high mitotic count, stromal invasion, and/or distant metastasis). Distant metastasis was reported in 5 cases (Table 1).
Uterine adenocarcinoma, Asian elephant, uterus.
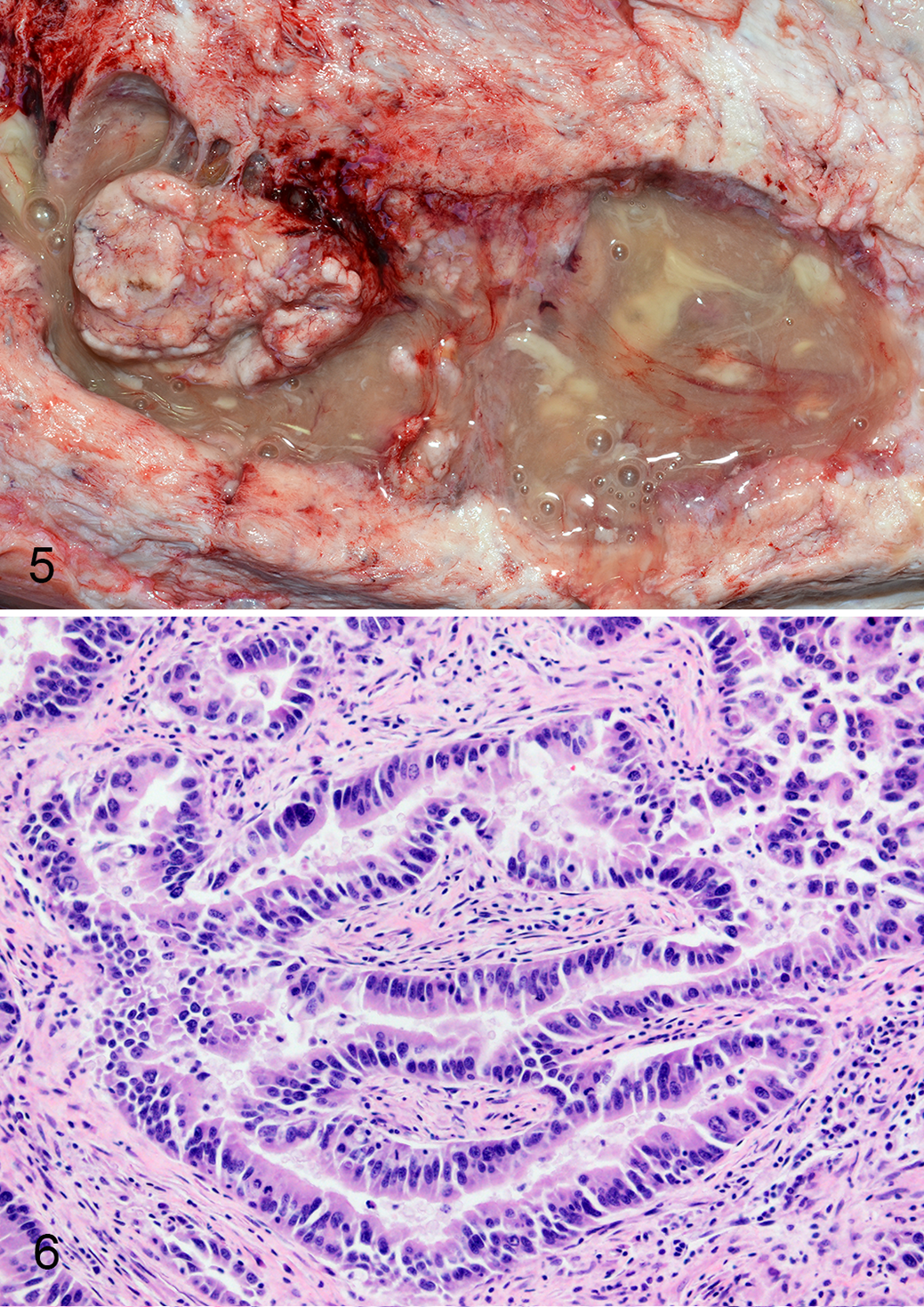
The anaplastic carcinoma was grossly evident as a poorly demarcated, irregular thickening of the endometrium and uterine wall composed of friable, tan-brown tissue, portions of which were sloughed into the uterine lumen. The neoplasm was composed of small nests and dissociated individualized cells supported by fine fibrovascular stroma. Neoplastic cells were pleomorphic and ranged from round or ovoid to polygonal with scant to abundant, eosinophilic, lacy cytoplasm. Nuclei were round with vesicular chromatin and one to multiple prominent nucleoli. Anisocytosis and anisokaryosis were marked and multinucleated cells containing up to 10 nuclei were common. Mitoses were frequent, and many were bizarre (Fig. 7). Lymphatic invasion was described with widespread metastases to lymph nodes, kidney, adrenal gland, mesenteric adipose tissue, and vagina. Neoplastic cells were reported to have strong immunoreactivity to vimentin, and rare cells were positive for pan-cytokeratin. Neoplastic cells lacked immunoreactivity for estrogen receptor, CD3, and CD20 (Suppl. Table S1).
Anaplastic carcinoma, Asian elephant, uterus. A lymphatic vessel is filled with many pleomorphic neoplastic epithelial cells, and some contain multiple nuclei or have bizarre mitoses. Hematoxylin and eosin (HE). Figure 8. Anaplastic sarcoma, Asian elephant, pelvic inlet. The mass contains round to spindyloid neoplastic cells with indistinct borders that are arranged in loose streams supported by moderate amounts of fibrovascular stroma. Binucleated and multinucleated neoplastic cells are present. HE. Figure 9. Primitive neuroectodermal tumor, Asian elephant, uterus. Neoplastic cells separate smooth muscle bundles of the myometrium and are arranged in sheets and trabeculae with frequent rosettes. HE.
Other Neoplasms
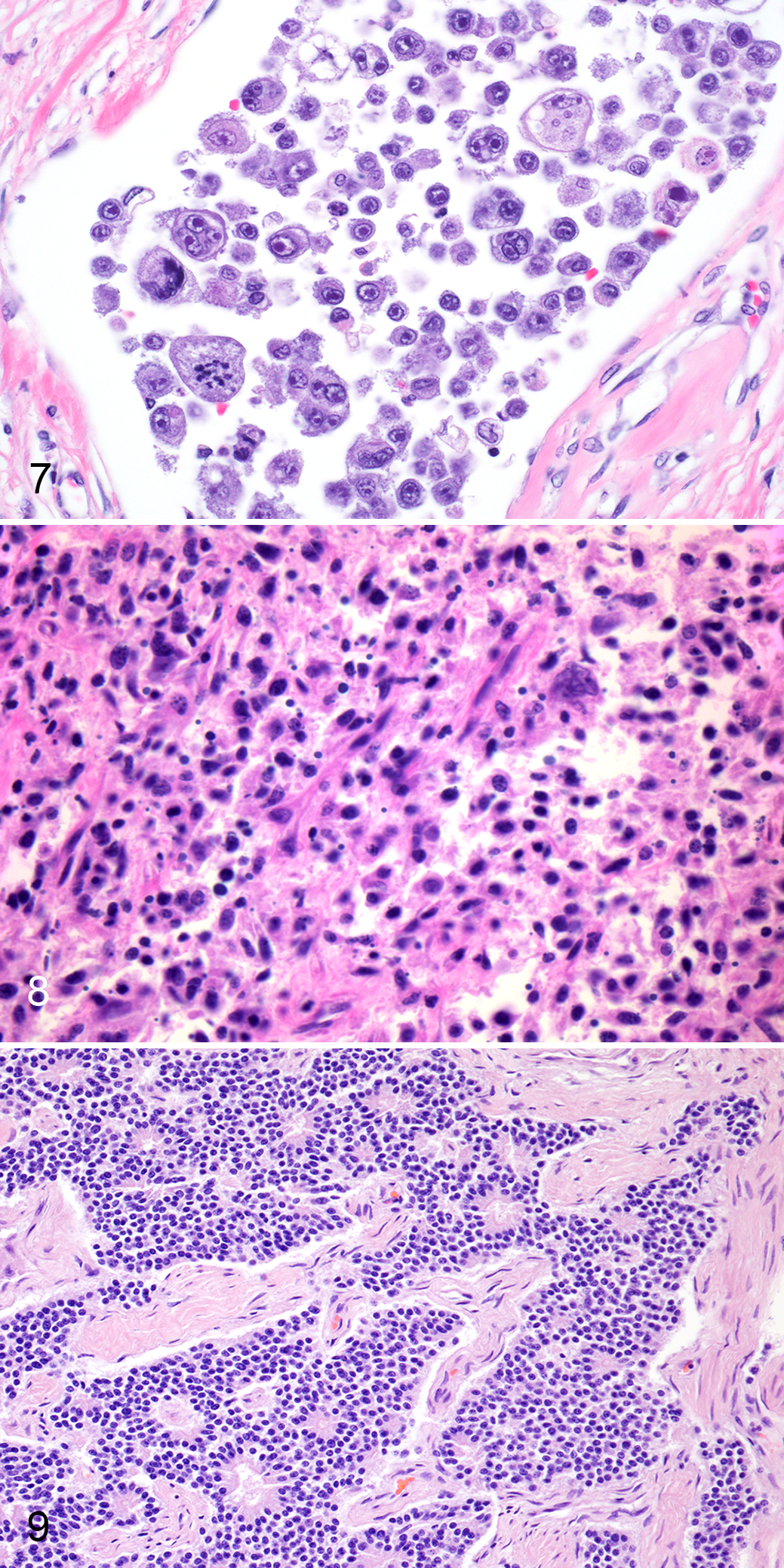
The pelvic anaplastic sarcoma was 59 cm in greatest diameter and weighed 48 kg. The mass was firmly adhered to the inner surface of the pelvic bone with dorsal displacement of the lower urogenital tract. Cut surfaces were lobulated, bulging, and mottled red. Histologically, the neoplasm was densely cellular, nonencapsulated, and infiltrative. Cells were round to spindyloid with indistinct borders and arranged in streams supported by moderate amounts of fibrovascular stroma (Fig. 8). Binucleated and multinucleated neoplastic cells were common. Neoplastic cells lacked immunoreactivity for both pan-cytokeratin and vimentin. Neoplastic cells were also reported to be CD3 negative (Suppl. Table S1). Immunohistochemistry for CD18 and CD79a were described, but the report stated a lack of immunolabeling of positive control elephant tissues.
The PNET had marked infiltration of the right uterine horn, forming a smooth, firm, tan to pink, intramural, luminally occlusive, 44-cm-long mass that blended imperceptibly with the uterine wall. Histologically, neoplastic cells were arranged in sheets and trabeculae with frequent small foci of cells radiating around central accumulations of eosinophilic fibrillar material (rosettes; Fig. 9). Neoplastic cells were polygonal to elongate with small amounts of eosinophilic, fibrillar cytoplasm, and indistinct borders. Nuclei were round to ovoid, occasionally oblong (carrot-shaped), and hyperchromatic with indistinct nucleoli. Neoplastic cells had positive immunolabeling for neuron-specific enolase (NSE), synaptophysin, and chromogranin A. Cells were negative for vimentin, pan-cytokeratin, and GFAP (Suppl. Table S1). Metastasis was manifested as widespread intravascular tumor emboli, effacement of caudal mediastinal lymph nodes, and disseminated neoplastic foci in the brain.
The uterine angiosarcoma was detected histologically within a large (81 × 81 × 101 cm) uterine leiomyoma that was firmly attached at the pelvic inlet, filled half of the peritoneal cavity, and obscured the entire reproductive tract. The leiomyoma contained a second, morphologically distinct population of neoplastic cells (representing the angiosarcoma) that was multifocally mixed with the benign neoplastic mesenchymal population. These cells were round to polyhedral and arranged in sheets and rows within a fibrovascular stroma (Fig. 10). Multifocally, cells formed vague vascular channels containing erythrocytes (Fig. 11). Anisocytosis and anisokaryosis were marked. Mitotic figures averaged 6 per 400× field, and many were bizarre. Masses of neoplastic cells similar to the angiosarcoma were also detected in the lung, spleen, and tracheal and mediastinal lymph nodes. Reported immunohistochemical characterization indicated neoplastic cells were negative for CD3, CD79a, Mac387, CD31, cytokeratin A1/E1, and smooth muscle actin and positive for Factor VIII related antigen (F8RA; Suppl. Table S1).
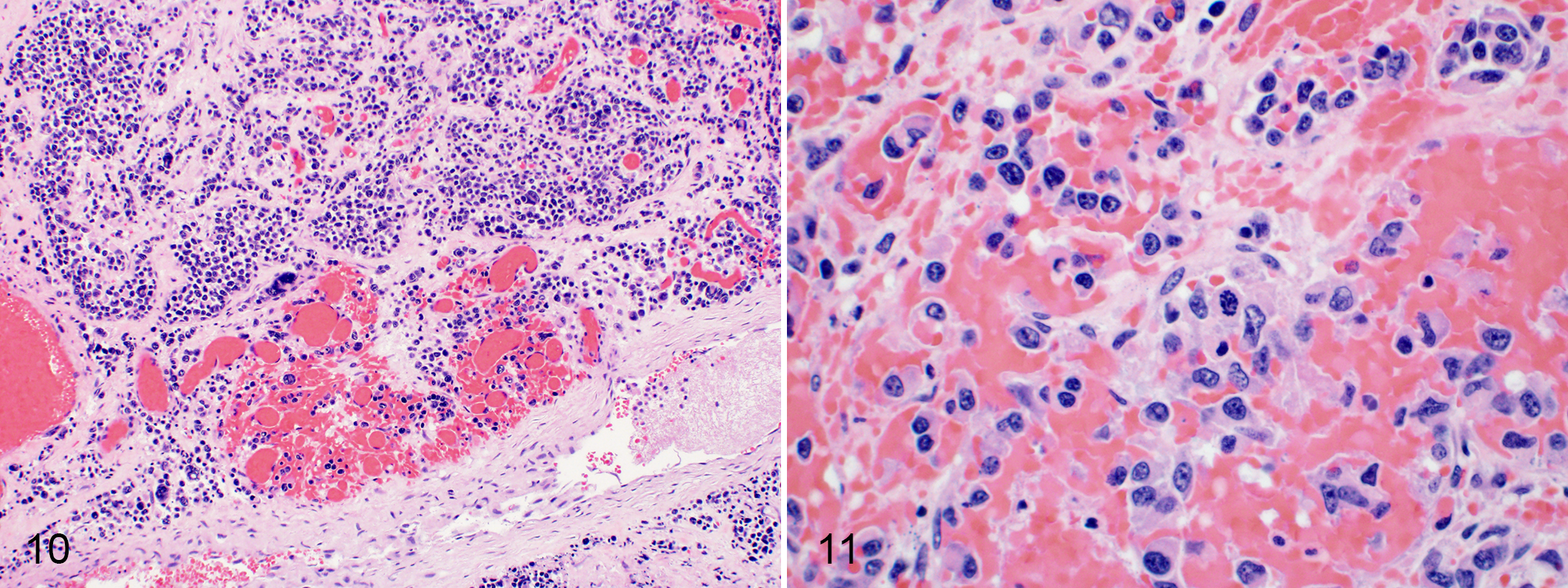
Uterine angiosarcoma, Asian elephant, uterus.
Non-Neoplastic Lesions
In 29/63 (46%) of the uterine neoplasia cases, cystic and/or polyploid endometrial hyperplastic lesions were also described. Most cases with endometrial hyperplasia also had leiomyomas (24/28; 86%). In one case, 2 separate endometrial polyps had evidence of carcinoma in situ. Lesions of carcinoma in situ were densely cellular and nonencapsulated with cellular atypia, formation of glands and papillary projections and regions of necrosis as described for invasive uterine adenocarcinomas. Another case had concurrent leiomyoma and endometrial adenoma. A single case of uterine adenocarcinoma reported concurrent endometrial hyperplasia. Uterine neoplasia in the remaining 4 cases with endometrial hyperplasia included an endometrial adenoma, anaplastic carcinoma, and PNET. Adenomyosis was only reported in 4 cases that had concurrent uterine neoplasia: 2 cases with leiomyomas, 1 case with adenocarcinoma and leiomyoma, and 1 case with PNET.
In the 16 cases in which no reproductive tract neoplasia was documented grossly, histologic evaluation of reproductive tissues was performed in 7 cases. Five of these 7 cases reported endometrial hyperplastic lesions. Adenomyosis was not described.
Immunohistochemistry
The positive control elephant uterus with cystic endometrial hyperplasia had positive immunolabeling for pan-cytokeratin and estrogen receptor in endometrial gland epithelium and positive immunolabeling for vimentin in mesenchymal components of the endometrium and myometrium. In all 4 examined adenocarcinoma cases, neoplastic cells were positive for pan-cytokeratin and negative for vimentin (Figs. 12–14). In 2 cases, neoplastic cells had positive intranuclear immunoreactivity for estrogen receptor (Fig. 15). The single case of carcinoma in situ was positive for pan-cytokeratin, vimentin, and estrogen receptor.
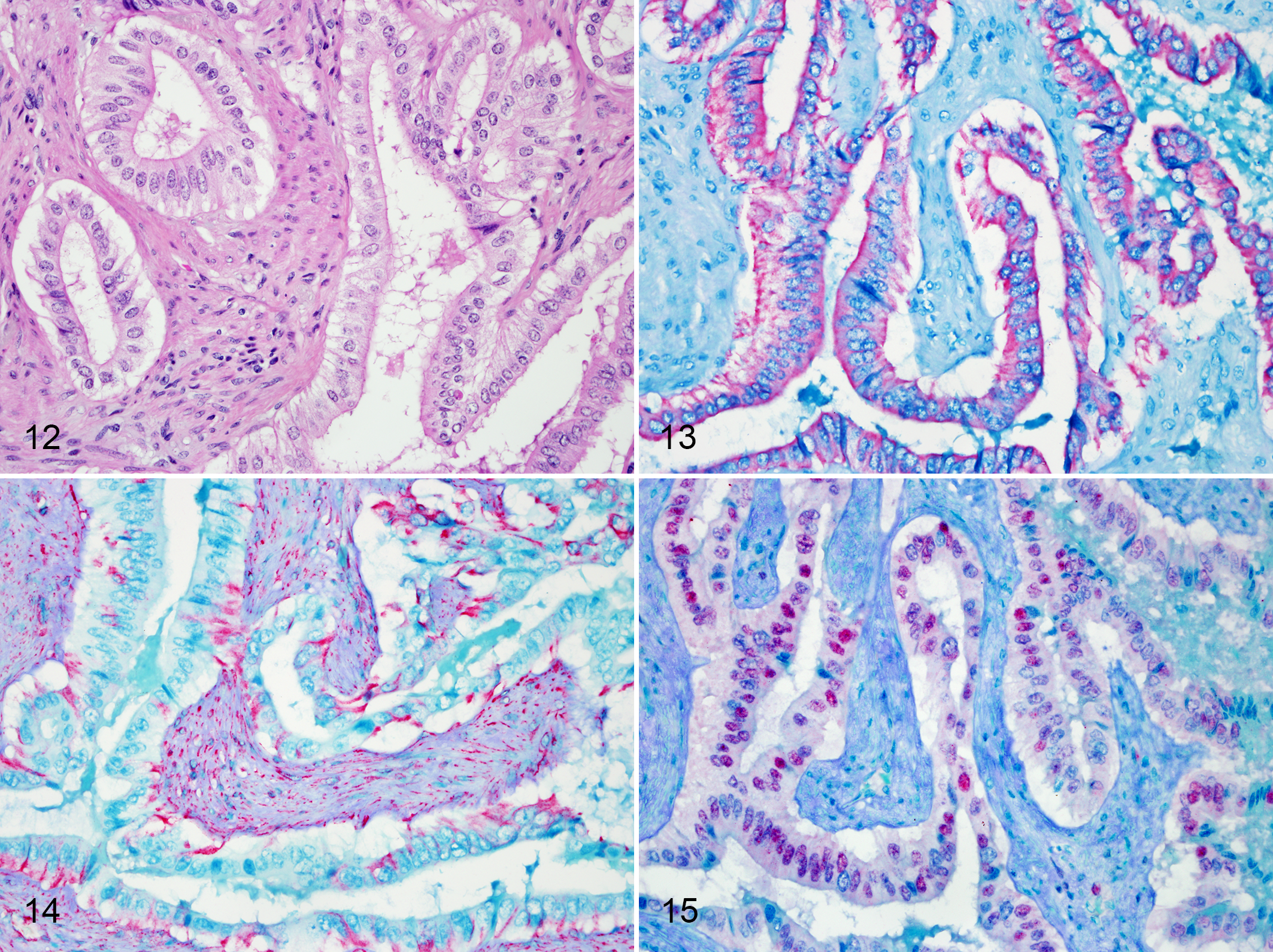
Uterine adenocarcinoma, Asian elephant, uterus.
Discussion
Results of this retrospective study identified that uterine neoplasia is common in adult Asian elephants. Study findings challenge assertions of several recent reports focusing on the scarcity of neoplasia in elephants. According to these studies, enhanced apoptotic activity facilitated by multiple copies of the TP53 gene in the African elephant genome provides for cancer resistance. 1,21,23,57,61 However, carcinogenesis is a complex process in which evolutionary, genetic, and epigenetic factors contribute. In the reproductive tract, hormonal influence may have profound effects and lead to varying degrees of proliferative disease. Additionally, it is important to note that the multiple TP53 gene copies identified in the elephant genome are not functional alleles. The extra TP53 copies are retropseudogenes that are incapable of encoding a complete p53 protein. Experimental evidence indicates some of the retropseudogenes are transcribed and may be translated, and in situ investigations have demonstrated an effect on apoptotic responses of elephant cells. 1,18,61,65 Still, the role and functional importance of the multiple TP53 retropseudogenes in the elephant genome and their role in influencing carcinogenesis in vivo remain unclear.
Previous studies have described a high prevalence of reproductive tract disease in aged Asian and, to a lesser extent, African elephants. 2,25 Cystic and polyploid endometrial hyperplasia (CEH) is common and may be a factor that impacts fertility. In elephants, CEH is linked with nulliparity and is associated with prolonged phases of both estrogen and progesterone influence, similar to carnivores. 2 The current case series indicates that myometrial leiomyoma is another common geriatric female reproductive tract lesion in Asian elephants. The study population of Asian elephants was considered representative of the managed population in the United States as a whole (reviewed cases comprised 85% of the total adult female Asian elephant mortalities for the time period), which is largely aged and nulliparous. In the study population, reproductive history was only known and documented in a small proportion of reports. Some cases were wild-caught individuals for which complete reproductive history was unavailable. In other cases, no reproduction was documented during tenure at the final holding facility, but such information was not available from all previous holding facilities. Despite incomplete histories for some cases, the majority were likely nulliparous given the dynamics of the Asian elephant population in the United States (Kay Backues, personal communication). Historic (prior to ∼1980) management practices in the United States that relied on importation for elephant population maintenance put less emphasis on elephant breeding and resulted in widespread nulliparity. 68 Possibly, nulliparity and the accompanying hormonal milieu play a role in leiomyoma pathogenesis similar to mechanisms recognized in women. 11,53,66
Most elephant uterine tumors identified in this review were leiomyomas. Leiomyomas are benign neoplasms of smooth myocytes. Myometrial leiomyomas, typically referred to as uterine fibroids, are common neoplasms in women. They occur in 77% of women, and though histologically benign, are the primary indication for hysterectomy in the United States due to resultant excessive menstrual bleeding and pelvic discomfort. 59,66 In women, several specific risk factors are associated with the occurrence of uterine fibroids. Prevalence peaks in the perimenopausal years and declines after menopause. Obesity and early age at menarche are recognized risk factors. Parity also plays a role in pathogenesis; age at birth of the last child is inversely associated with risk. These factors all contribute to a women’s overall lifetime exposure to estrogen. 47,64,66 In women, fibroids are steroid hormone-dependent and hyperresponsive to estrogen with elevated levels of estrogen and progesterone receptors. 11,53,66
With the exception of miniature pig (Sus scrofa) breeds, reproductive tract leiomyomas are uncommon in domestic ungulates. 13,30,44 Genital leiomyomas have been documented sporadically in horses (Equus callabus). 5,12,36 Though leiomyomas are also seen in cattle (Bos taurus), uterine adenocarcinoma is more frequent in this species. 12,14,17,38,48 Leiomyomas in miniature pigs have been proposed as a possible animal model for fibroids in women. 43 Additionally, a retrospective study of reproductive tract lesions in a variety of exotic/wild Suidae and Tayassuidae species in managed care found a high prevalence of uterine neoplasia including leiomyoma (29.6%) and endometrial carcinoma (12%). It was postulated that long-term, lifetime exposure to estradiol and progesterone through many estrous cycles may contribute to carcinogenesis in suid species, as is recognized in women. 19 Among other exotic ungulates, rhinoceroses are notable for a high prevalence of reproductive tract leiomyomas, and similar to elephants, represent another charismatic megavertebrate for which reproduction in the managed care setting is a conservation goal. 15,24,26,55 In Indian rhinoceros (Rhinoceros unicornis) particularly, age-related development of reproductive tract leiomyomas has been implicated in reduced fecundity of the species in managed care. Analyses of international studbook data for the Indian rhinoceros has confirmed a reduced reproductive lifespan in managed populations that correlates to onset and development of reproductive tract leiomyomas. It is typical for female rhinoceroses greater than 12 years of age to have reproductive tract leiomyomas, and number and size of masses increases over time. All portions of the genital tract are affected, though a higher prevalence has been noted in the cervix and vagina. 15,24 This contrasts with the current study findings in elephants indicating the uterus as the primary site for leiomyoma.
Though most elephant uterine tumors were leiomyomas, malignant neoplasms accounted for 19% of reported uterine neoplasms. Importantly, uterine malignancies had metastasized in 67% of cases. Most uterine malignancies were adenocarcinomas analogous to type I endometrioid carcinomas in women. In humans, endometrial cancer is a disease of the affluent, developed world; incidence rates are up to 10 times higher in Western, industrialized countries than in Asia or rural Africa. Environmental risk factors including low level of physical activity and obesity are associated with >40% of disease incidence in women. 32 Endogenous hormones also play an important role in endometrial carcinogenesis, and increased risk has been associated with early menarche and late menopause. Parity decreases risk, and exogenous estrogen administration increases risk. Type I carcinomas are associated with mutations in the ras proto-oncogene and PTEN tumor suppressor gene, but usually do not have p53 mutations. 32,37
Hormones have regulatory effects on the cellular processes of proliferation, differentiation, and apoptosis. These regulatory effects provide the principal mechanisms by which hormones may influence uterine carcinogenesis. Estrogen stimulates endometrial proliferation via increasing production of insulin-like growth factor-1; progesterone counterbalances these mitogenic effects by stimulating production of insulin-like growth factor binding protein-1. Uterine carcinoma risk is increased with high plasma levels of bioavailable estrogens and/or low plasma progesterones. Ovarian and adrenal-origin androgens can also contribute to elevated estrogen levels following metabolism by aromatase enzymes in adipose tissue. 32,40 Body condition may have profound impacts on hormone metabolism. Obesity can lead to insulin resistance and hyperinsulinemia, increased bioavailability of steroid hormones and localized inflammation, all of which potentially contribute to carcinogenesis. Insulin stimulates ovarian and adrenal cortical production of androgens, reduces liver production of sex hormone-binding globulin, and functions as a direct endometrial growth factor, similar to insulin-like growth factor-1. Obesity also facilitates increased peripheral aromatization of androgens leading to elevation in bioavailable estrogen levels. 9,22,27
All cases of elephant uterine adenocarcinoma occurred in geriatric individuals (range = 45–65 years; mean = 55 years) confirmed or presumed to be nulliparous. Though it would seem likely that similar hormone-mediated mechanisms contribute to endometrial carcinogenesis in elephants as have been recognized in women, information regarding plasma estrogen or progesterone levels at time of death or previously during the course of carcinogenesis were not available in any cases to provide objective evidence. Ovarian atrophy/senescence was described in a single case, but no ovarian lesions were noted in the other 6 cases in which ovaries were examined histologically. All elephants with uterine adenocarcinoma were reported to have been in good to reduced body condition evidenced by adequate or decreased adipose stores, respectively. Though obesity was not described in any of the cases, body condition at the time of death may not have been reflective of body condition prior to or during carcinogenesis. A high percentage of elephants housed in North American zoos have an elevated body condition score. 42 Due to the physical constraints of the managed care environment, relative reduction in physical activity compared with free-ranging individuals likely contributes to overconditioning. 29 Possibly, body condition and activity level could be factors impacting hormonal homeostasis and metabolism in elephants and are recognized contributors to cancer risk in women.
Adenocarcinomas in this study were cytokeratin-positive and vimentin-negative. Appropriate labeling of negative and positive control tissues provided confidence in the immunohistochemistry results, and labeling of non-neoplastic elephant tissues gave evidence of antibody cross-reactivity in the species. Immunohistochemical labeling in the elephant uterine adenocarcinomas was similar to expression described in the few studies of domestic feline and canine uterine carcinomas 39,50 In contrast, as observed in the carcinoma in situ in one elephant, human endometrial carcinomas most often have both cytokeratin and vimentin immunoreactivity. Vimentin immunoreactivity is a feature that can aid in distinguishing human endometrial and endocervical carcinomas. 33,35 In 2 elephant adenocarcinomas and the carcinoma in situ, immunoreactivity for estrogen receptor was also demonstrated. Estrogen receptor expression is a prominent feature in both human and domestic animal endometrial carcinomas. Expression in the elephant cases could provide support for hormone-mediated mechanisms of uterine carcinogenesis as noted in women and presumed in domestic animal species, but analysis of additional cases is required to confirm findings. 4,39,50 Immunohistochemical characterization of the elephant adenocarcinomas was hindered by the archival/retrospective nature of the study. The time tissues spent in formalin prior to processing was generally unknown. Additionally, given the size of elephant specimens, tissue sections collected at the time of necropsy may have been large and ill-suited to timely and uniform fixation. Thus, formalin fixation and processing likely impacted antigen retrieval and overall IHC performance, which may limit interpretation of the results.
Adenocarcinoma was not the only uterine malignancy documented in Asian elephants during the study period. Single cases of anaplastic carcinoma, anaplastic sarcoma, angiosarcoma and PNET also occurred. Multicentric metastasis was reported for the anaplastic carcinoma, angiosarcoma, and PNET. The anaplastic carcinoma resembled the human malignancy referred to as undifferentiated/dedifferentiated endometrial carcinoma, in which cytokeratin immunoreactivity may only be focal and vimentin immunoreactivity is described. 3,69 The anaplastic sarcoma was a large pelvic mass that obscured and displaced the lower urogenital tract. Uterine origin was presumed, though size and tissue distortion precluded definitive determination. The angiosarcoma was only diagnosed histologically within sections of the large uterine leiomyoma, mediastinal lymph node, spleen, and lung. Endothelial origin was determined by positive immunolabeling for factor VIII–related antigen of neoplastic cells. Immunohistochemical results combined with neoplastic cellular morphology and arrangement were most consistent with epithelioid angiosarcoma. Uterine origin of the angiosarcoma was presumed, and other sites of neoplasia were interpreted as metastases. However, marked distortion of the uterus and surrounding pelvic tissues by the massive uterine leiomyoma prevented confirmation. Interestingly, epithelioid angiosarcomas arising within uterine leiomyomas are described in women. 63 Possibly, this elephant case represented a similar presentation. Though immunoreactivity to factor VIII–related antigen was supportive of epithelioid angiosarcoma, neoplastic cells lacked immunoreactivity for other markers described in human epithelioid angiosarcoma (eg, CD31, cytokeratin). Inconsistencies may reflect a lack of validated antibodies for use in elephants, although there was labeling of internal positive controls. Alternatively, lack of expression by neoplastic cells due to poor differentiation was considered.
A single case of ovarian neoplasia (carcinoma) was documented. The few ovarian follicular cysts were interpreted as incidental findings. No other important ovarian lesions were reported. No primary cervical neoplasms were noted. In at least one uterine adenocarcinoma case, neoplasia was localized adjacent to the cervix; however, histologic features suggestive of cervical origin were lacking. Hyperplastic, polyploid, or papillomatous mucosal lesions of the vagina/vulva, similar to those previously reported in Asian elephants, were noted in a small proportion (9/80; 11%) of cases. 62 No neoplastic lesions involving the epithelium of the distal reproductive tract (vagina, vulva) were described. A single case had a vaginal leiomyoma. Study findings indicate ovarian, cervical and vaginal/vulvar neoplasms are uncommon in the species.
The retrospective nature of this study was a known limitation. Analysis was achieved through review of archival postmortem records, and thus was reliant on the observations and interpretations of various veterinary clinicians and pathologists. Due to the retrospective nature of this study, confirmation of gross abnormalities by the authors was not possible. Histologic findings were also assessed via evaluation of reports; it was not feasible to obtain and review all slides for histopathology. Similarly, most IHC data were gleaned from the archival review of the postmortem reports, and protocols were not repeated for the purpose of this study. Given that all cases were originally diagnosed by American College of Veterinary Pathology board-certified pathologists, many of which with extensive experience with zoo and wildlife species, original results, and interpretations were considered sufficient for analysis.
Conclusions
Results of this study indicate that female reproductive tract neoplasia, particularly of the uterus, is common in Asian elephants, and that neoplasia is not limited to benign myometrial tumors. A variety of uterine malignancies occurred in elephants, and most were associated with distant metastases. Importantly, benign and malignant neoplasms affecting the uterus have the potential to impact fecundity, and thus may represent obstacles to conservation in managed care. 25,41 Additional research is necessary to better characterize these neoplasms, elucidate their pathogenesis, and determine the role of hormonal influences in their development and progression.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211031843 - Reproductive tract neoplasia in adult female Asian elephants (Elephas maximus)
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211031843 for Reproductive tract neoplasia in adult female Asian elephants (Elephas maximus) by Jennifer A. Landolfi, Patricia M. Gaffney, Rita McManamon, Nicole L. Gottdenker, Angela E. Ellis, Raquel R. Rech, Sushan Han, Linda J. Lowenstine, Dalen Agnew, Michael M. Garner, Denise McAloose, Charlotte Hollinger, Judy St. Leger, Scott P. Terrell, Mary Duncan and Allan P. Pessier in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank all contributing elephant holding facilities for granting permission to release archival postmortem reports for this review. Yvonne Cates of the San Diego Zoo Global Disease Investigations Histopathology Laboratory provided immunohistochemistry support. Lisa Farina and Jackie Gai assisted in identification and acquisition of case materials. The authors are also grateful to the America Zoo and Aquarium Association (AZA) Elephant Taxon Advisory Group and Species Survival plan (TAG-SSP) for endorsing the study and to Martha Fisher, Bob Lee, and Kay Backues for providing population data vital to interpretation of study results.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
