Abstract
Pemphigus foliaceus (PF) is an autoimmune acantholytic skin disease described in humans, dogs, cats, horses, goats, and sheep. From 2003 to 2016, six Arabian oryx (Oryx leucoryx) at the National Zoological Garden in Pretoria, South Africa, developed progressive, bilaterally symmetrical, hyperkeratotic skin lesions and pustules consistent with PF. Lesions were similar to those observed in domestic animals and primarily affected the pinnae, face and nasal planum, distal legs, and tail tip. Histological evaluation of suspect PF skin lesions in affected animals, evaluation of medical records for treatments received, causative agents in the diet and environment, and special stains for infectious organisms yielded no consistent inciting cause. The Arabian oryx is a species highly adapted to arid environments of the desert and has recently survived from a severe genetic bottleneck; both of these factors may have contributed to the development of PF in these animals.
Keywords
Pemphigus foliaceus (PF) is an autoimmune skin disease that affects the superficial epidermis and has been reported in the human, dog, cat, horse, goat, and sheep. 2,10,13,18 In domestic canids, the disease is caused by autoantibodies produced against desmosomal proteins including desmocollin 1 and to a lesser extent desmoglein 1; these proteins contribute to adhesion between keratinocytes in the superficial epidermis and follicular epithelium. 14 In humans, IgG autoantibodies against desmoglein 1 are causative of PF, and IgA autoantibodies against desmocollin 1 are causative of a disease with similar presentation called subcorneal pustular dermatosis. 12 The pathogenesis of PF has not been fully elucidated in other species but is likely to be similar.
Characteristic lesions of PF in animals include pustules and exfoliative crusts, with often bilaterally symmetric distribution over the pinnae, nasal planum, periorbital skin, and/or pawpads. 13 Histologic lesions include superficial intra-epidermal pustules containing acantholytic keratinocytes. Chronic lesions induce epidermal hyperplasia (acanthosis) with rete ridge formation and hyperkeratosis. Mild to moderate, superficial to perivascular, mixed (ie, lymphocytes, plasma cells, eosinophils, neutrophils) dermal inflammation is most commonly reported with pigmentary incontinence as an inconsistent feature. IgG and IgA immunofluorescence or immunohistochemistry demonstrating antibody deposition between suprabasilar keratinocytes is a feature of PF, though false positive and false negative results can occur. Currently, characteristic histologic lesions and exclusion of infectious disease (dermatophytosis, bacterial infection) are considered diagnostic for PF in animals.
The reintroduction of Arabian oryx (Oryx leucoryx) to its natural range is considered one of the greatest successes of wildlife conservation, as the species was extinct in the wild in the early 1970s and is now listed as vulnerable under the IUCN red list. 8 Successful breeding programs in the United States and Saudi Arabia resulted in the reintroduction of the species into the Arabian peninsula. 8,9,19 Despite concerns of low genetic fitness, given the small number of founder animals, genetic analysis suggests high levels of genetic diversity in some herds. 5,19 However, sporadic reports of chromosomal translocations and pseudo-hermaphroditism are available. 3,15 These few cases stress the importance in monitoring for both congenital disease and susceptibility to acquired diseases when establishing breeding programs for long-term survival of a species.
Between 2003 and 2009, six adult Arabian oryx housed at the National Zoological Gardens of South Africa (NZG) were diagnosed with gross and histological lesions consistent with PF. From 19 pathology submissions recorded in the NZG wildlife disease database (2003–2016), haired skin from 8 adult Arabian oryx was available to investigate this disease in the herd. All skin samples were histologically evaluated, by the same pathologist (JBC), using routine hematoxylin and eosin, Gram, periodic acid-Schiff, and Grocott-Gomori’s methenamine silver stains. Dermatitis was nondiagnostic in 2 adults (O1 and O2). Six animals had neutrophilic pustular acantholytic skin lesions (O3–O8). Animal age group, sex, sample type, and age of disease onset of the 8 animals are shown in Table 1.
Arabian oryx pathology submissions at the National Zoological Gardens of South Africa from 2003 to 2016.
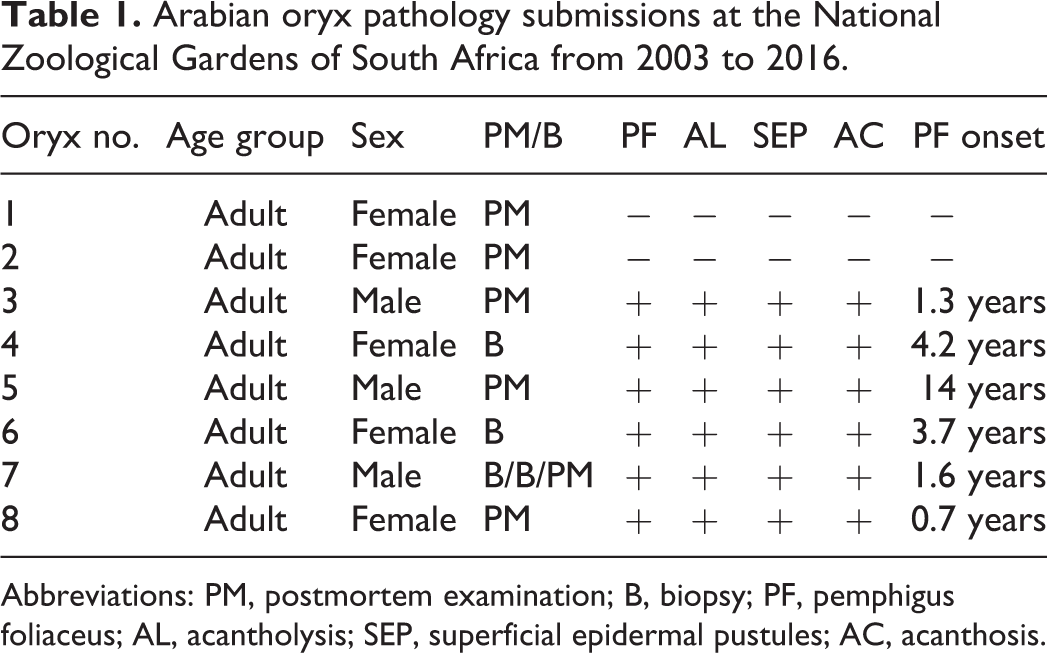
Abbreviations: PM, postmortem examination; B, biopsy; PF, pemphigus foliaceus; AL, acantholysis; SEP, superficial epidermal pustules; AC, acanthosis.
Four of the 6 affected PF animals (O3, O5, O7, O8) had chronic skin disease listed as a primary cause or comorbidity resulting in euthanasia. Lesion onset was as early as 8 months of age to as late as 14 years of age. Cases consisted of 3 males (O3, O5, O7) and 3 females (O4, O6, O8). Case O4 was a heavily pregnant female. Pruritus was noted in 2 cases (O6, O7). Macroscopically, affected oryx developed progressive, bilaterally symmetrical, rough, scaly to raised skin lesions with variable alopecia and skin depigmentation. Lesions were most commonly on the pinnae, but also over the face and nasal planum, coronary bands and distal legs, and tail tip (Figs. 1, 2). No causative bacteria, fungi, arthropods, or viral inclusions or cytopathies were observed in any of the PF cases on hematoxylin and eosin or special stains.
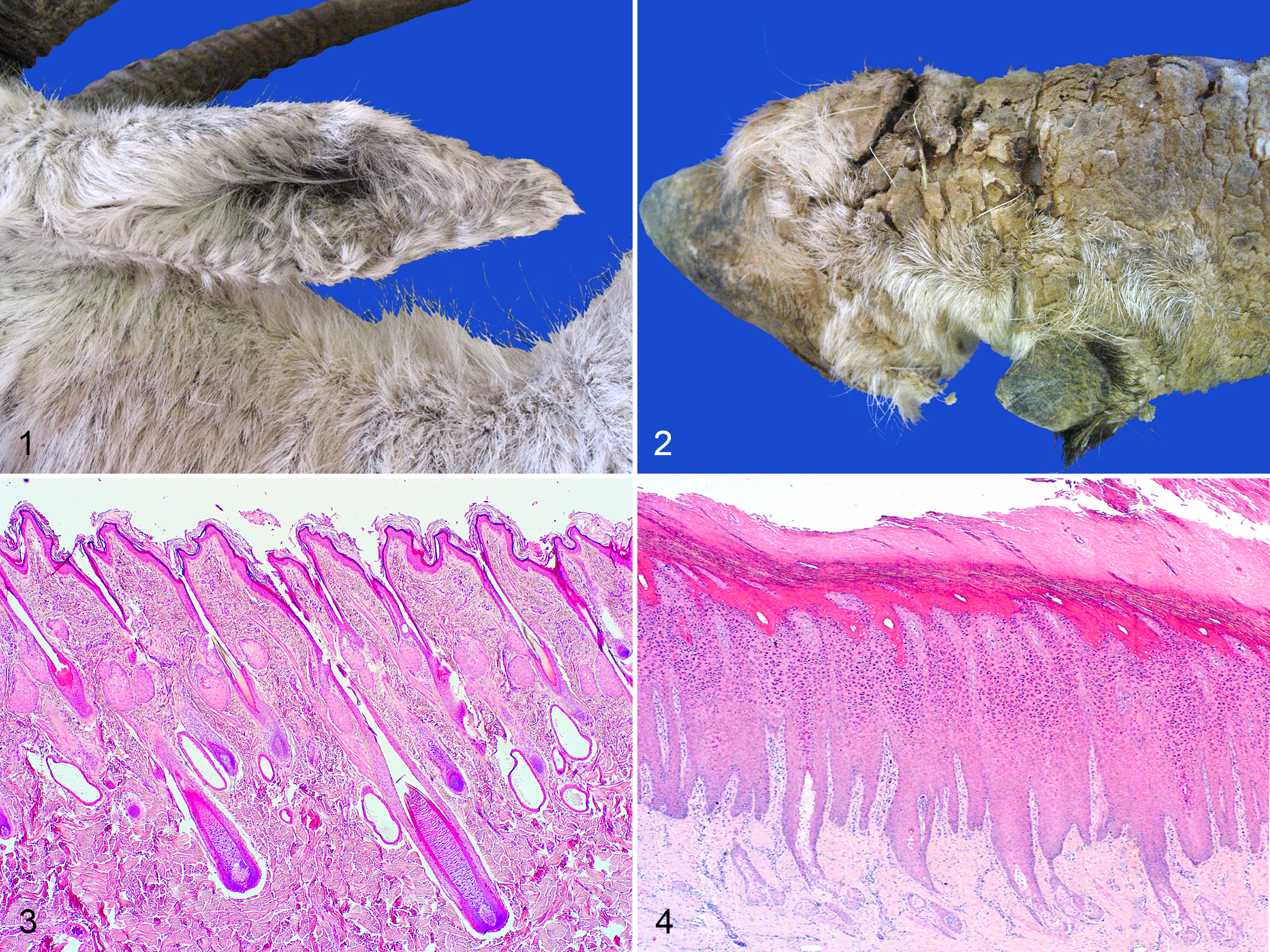
Pemphigus foliaceus (PF), skin, Arabian oryx.
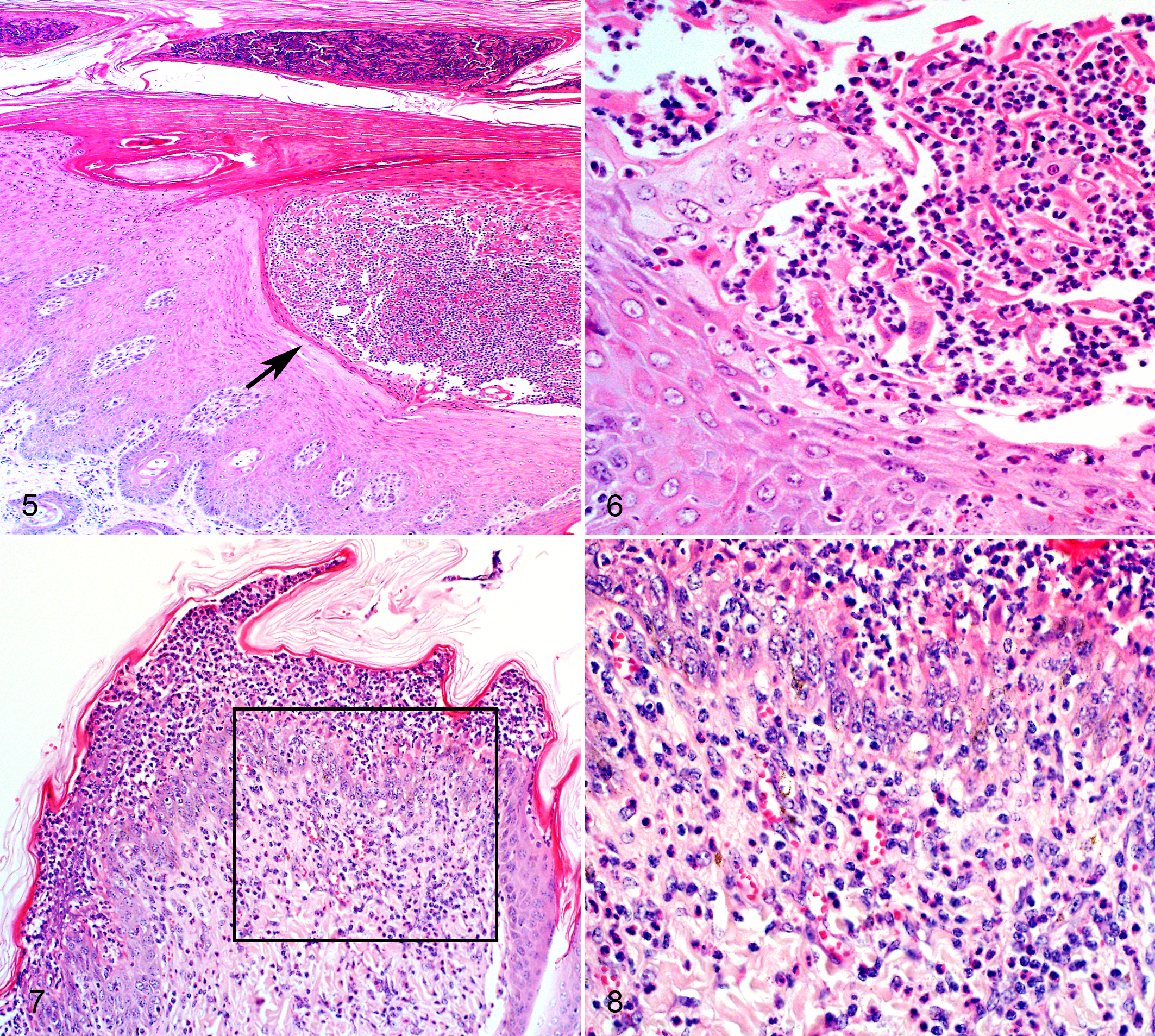
All affected haired skin sections, compared to normal sections (Fig. 3), had acanthosis with basket-weave and/or compact orthokeratotic hyperkeratosis of varying severity and superficial intra-epidermal pustules containing neutrophils, eosinophils, acantholytic keratinocytes, and sloughed cellular debris (Figs. 4–8). Pustules occurred in the stratum granulosum or stratum corneum layers. Cases O4, O7, O8 had pustules in multiple outer stratum layers, sometimes in alternating layers with orthokeratosis, indicative of “waves” of pustule formation and consequent exfoliation (Fig. 5). Vesicle formation was described macroscopically in O3 though no vesicles were noted histologically. Dermal inflammation included mild to moderate, superficial, scattered to perivascular infiltrates of lymphocytes, plasma cells, eosinophils and neutrophils with eosinophilic and neutrophilic exocytosis (Figs. 7, 8). In all 6 cases, mild scattered pigmentary incontinence, superficial dermal capillaries lined by plump activated endothelial cells, and mild to moderate apocrine gland ectasia were also present. Ectatic apocrine glands were either empty or filled with small amounts of pale eosinophilic granular to wispy material. Scattered leukocytoclasia and mild superficial dermal fibrosis were noted in the majority of cases. Case O7 included 2 skin biopsies taken approximately 5 and 1.5 months prior to euthanasia. The earliest biopsy was of the pinna and included histologic lesions classic for PF. However, the biopsy taken 1.5 months prior to death showed only moderate to severe orthokeratotic hyperkeratosis (Fig. 4). At postmortem examination, histologic sections of skin above the coronary band (Figs. 5, 6) had severe, yet classic PF lesions. In a chronic case (O5), histological lesions in the lumbar skin had severe dermal fibrosis and adnexal atrophy, moderate to severe acanthosis and hyperkeratosis, minimal superficial dermal inflammation, and moderately dilated apocrine glands. Acantholytic cells were only noted in superficial serocellular crusts, a finding commonly reported in chronic PF lesions.
Pemphigus foliaceus (PF), skin, Arabian oryx. Hematoxylin and eosin.
Additional postmortem findings in the oryx with PF included a few cestode cysts on the rumen serosa (O3) and peritoneum (O5); moderate intestinal infestation with Trichostrongylus spp. (O7); mild atrioventricular valvular endocardiosis (O3, O5); mild myocardial fibrosis (O8); mild colloid goiter (O3, O7, O8); moderate chronic renal infarcts (O5); mild (O7) to marked (O8) lymph node lymphoid hyperplasia; mild splenic follicular lymphoid hyperplasia (O8); moderate bone marrow and neutrophilic myelopoiesis (O7); mild adrenocortical hyperplasia (O8); mild ruminal parakeratosis with microabscesses (O7); and mild glomerulonephritis (O7) and enteritis (O7, O8).
The nutritional background, environmental, genetic, epidemiological, and enclosure features were recorded to evaluate potential etiologic factors. Onset of lesions was mainly in the dry winter season. Other than periodic oral anti-helminthic treatment, the oryx received no regular drug therapy and were not exposed to significant levels of phenols or tannins. No clinical response to supplementation (multivitamin, mineral, and essential fatty acids) was observed but prolonged glucocorticoid treatment resulted in remission in one case (O6).
While PF has been reported in humans and domestic animals including ruminants, this is, to the authors’ knowledge, the first report of PF in oryx. 2,3,9,10,13,18 However, skin disease is often reported in Arabian oryx under different managed-care situations (personal communication with Dr Jane Budd, Breeding Centre for Endangered Arabian Wildlife (BCEAW), United Arab Emirates) and similar macroscopic and histological lesions have been described in partially free-ranging and captive oryx. 1
The factors that trigger acantholysis and the production of antibodies to epidermal epithelial cell adhesion molecules are not fully understood though likely involve loss of desmosomal attachments. 13,14 Sunlight exposure has been implicated as a trigger in dogs and genetic predispositions have been recorded in some cases, with possible breed predispositions in horses. 13 Certain infections such as dermatophytosis and superficial bacterial infection, ingestion of Echinacea purpurea, chronic tannin ingestion, and antibiotic therapy have also been described as causes for pustular acantholytic disease. 4,13 The possibility of immune-dysregulation is supported by the resolution of some cases (including O6 in our series) with long-term glucocorticoid therapy. 2,13
The Arabian Oryx came from a small group of founder animals, so a genetic predisposition to this disease, similar to endemic pemphigus foliaceus (Fogo selvagem) in humans from Brazil, could be implicated in Arabian oryx. 20 Prior exposure to antigens from simulid insect bites and other hematophagous insects have also been implicated as risk factors for development of lesions. 7,11 A winter seasonal predilection has been recorded in some horse cases, though possibly as a result from prior sensitization from biting insects. 18 In this case series, no antibiotic or anthelmintic treatments, diet changes, pathogens, or exposure to biting insects could be correlated with onset of the disease.
The Arabian oryx has special adaptations to life in the desert, including a white coat, large hooves, selective brain cooling, and adaptive heterothermy. Like other desert-dwelling artiodactyls, this species is reported to have decreased evaporative water loss during the heat of the day as a means to maintain hydration and thermoregulation in other portions of the day. 17 A case of PF in a Barbary sheep (Ammotragus lervia), a ruminant which is native to arid and semi-arid climates, is recorded. 2 Interestingly, transient acantholytic dermatosis (Grover’s disease) in humans has been linked to suspected impaired expression of cutaneous cholinergic receptors which influence sweat secretion. 16 In hot climates, cases are linked to heat, hospital confinement, sweating, and solar damage, whereas in cold climates cases are linked to dry skin. 6 Winter in the region of the NZG of South Africa is a dry sunny season, so a combination of environmental and genetic or adaptive factors may at least partly explain the development of PF in these Arabian oryx.
The stage of disease, location of sampling, and care to preserve superficial crusts in tissue processing are all strong factors in aiding the histologic diagnosis of PF. Furthermore, PF should be included on any differential diagnosis list where an animal has periodic hyperkeratotic skin disease and lesion distribution fits the clinical picture. Research into the levels of genetic relatedness, possible inciting factors in the food or environment, and correlation to climate patterns is still needed to determine causative factors involved in PF in the Arabian oryx.
Footnotes
Acknowledgements
Histology staff at the Faculty of Veterinary Science and National Health Laboratories in Pretoria; Nozipho Khumalo and Dr Ian Espie for practical assistance; and Drs Antoinette Kotze and Karen Terio for financial and logistical support. Technical graphical support by Ms Estelle Mayhew.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
