Abstract
Pemphigus vegetans is a rare autoimmune blistering acantholytic dermatosis of humans that combines unusually hyperplastic and verrucous pustular skin lesions and mucosal erosions. We report herein the clinical, histopathologic, and immunologic findings in a dog whose lesions resembled, but were not identical to, those of human pemphigus vegetans. A 4-year-old male Greater Swiss Mountain Dog presented with multifocal cutaneous verrucous and crusted papules and pustules, as well as skin and mucosal erosions and ulcers. Microscopic lesions consisted of exophytic papillated epidermal hyperplasia, superficial and deep intraepidermal acantholytic neutrophilic and eosinophilic pustules, and suprabasal epidermal clefts leaving rounded basal keratinocytes at the bottom of the vesicles. Direct and indirect immunofluorescence revealed antikeratinocyte IgG autoantibodies. Immunoprecipitation immunoblotting and immunoabsorption experiments with recombinant canine desmogleins confirmed that autoantibodies recognized desmoglein-1. In this dog, clinical and histopathologic features resembled those of human pemphigus vegetans, while circulating autoantibodies against canine desmoglein-1 were solely identified. This antigen target is different from that of the human disease in which antidesmoglein-3 autoantibodies are detected most commonly.
Keywords
In humans and companion animals, the classification of pemphigus variants relies on the level of cell-cell separation (acantholysis) within cutaneous and/or mucosal epithelia. The main subsets of pemphigus are the deeper vulgaris (PV) and the more superficial foliaceus forms (Pf). Pemphigus vegetans (PVeg) is a rare human acantholytic dermatosis that combines unusual verrucous pustular skin lesions and widespread deep mucosal erosions. Two variants of PVeg have been recognized in human patients: in the Neumann type, cutaneous flaccid bullae and pustules evolve into hyperkeratotic verrucous papules and plaques that are later replaced by large denuded areas. 1 Oral lesions are seen at an early stage, and they resemble those of PV. In the Hallopeau type, pustules are followed by hyperplastic wart-like papules and plaques that do not evolve into widespread erosions. Deep PV-like lesions are usually absent except on mucosae. 1 Histopathologically, lesions of human Neumann- and Hallopeau-type PVeg encompass exophytic epidermal hyperplasia, suprabasal PV-like acantholysis, and neutrophilic and eosinophilic acantholytic pustules scattered throughout all epithelial layers. 1 There are only rare studies on the immunopathology of human PVeg, all demonstrating that the antigen most commonly recognized by autoantibodies is the 130-kDa PV antigen desmoglein-3. 3, 6, 9, 10, 14 Additional epidermal proteins, such as desmoglein-1, 2, 15 plakoglobin, 14 and desmocollins, 2 also have been shown to be identified by circulating autoantibodies in this disease.
Only 2 dogs have been reported previously with a diagnosis of PVeg. In 1977, Scott identified an elderly Fox Terrier with a generalized pustular eruption that evolved into verrucous lesions. 17 Histologically, there were suprabasal to subcorneal eosinophil-rich pustules with acantholytic keratinocytes. Treatment with oral glucocorticoids led to complete remission. 17 In 1980, Schultz reported an adult Chow with a generalized crusted pustular eruption with microscopic suprabasal and intrafollicular eosinophil-rich acantholytic pustules. 16 In this dog, direct immunofluorescence revealed skin-fixed antikeratinocyte autoantibodies. 16 Treatment with prednisolone and antibiotics led to improvement of skin lesions.
Remarkably, in none of these 2 dogs were either mucosal lesions or suprabasal acantholysis observed. These findings are in marked contrast with those of human PVeg, in which PV-like erosions and ulcers are seen in addition to cutaneous verrucous vegetations. For these reasons, the putative diagnosis of PVeg in these 2 dogs must be considered with skepticism. Moreover, the breed and clinical signs of Schultz' dog are more suggestive of the diagnosis of PF rather than PVeg. 11, 12
The purpose of this short communication is to report, in a dog, the clinical, histopathologic, and immunologic characteristics of a mucosal erosive and cutaneous proliferative skin disease resembling human PVeg.
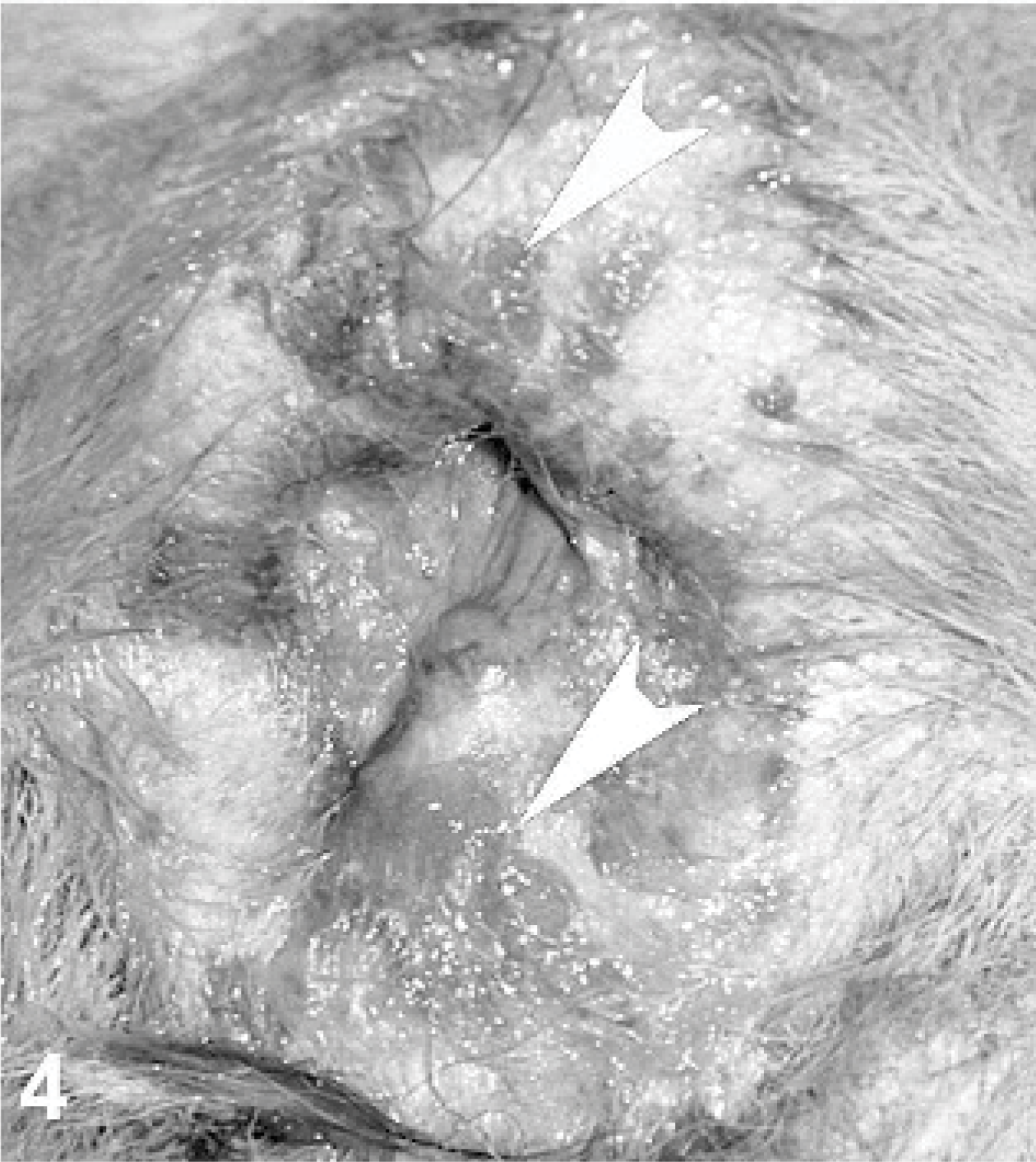
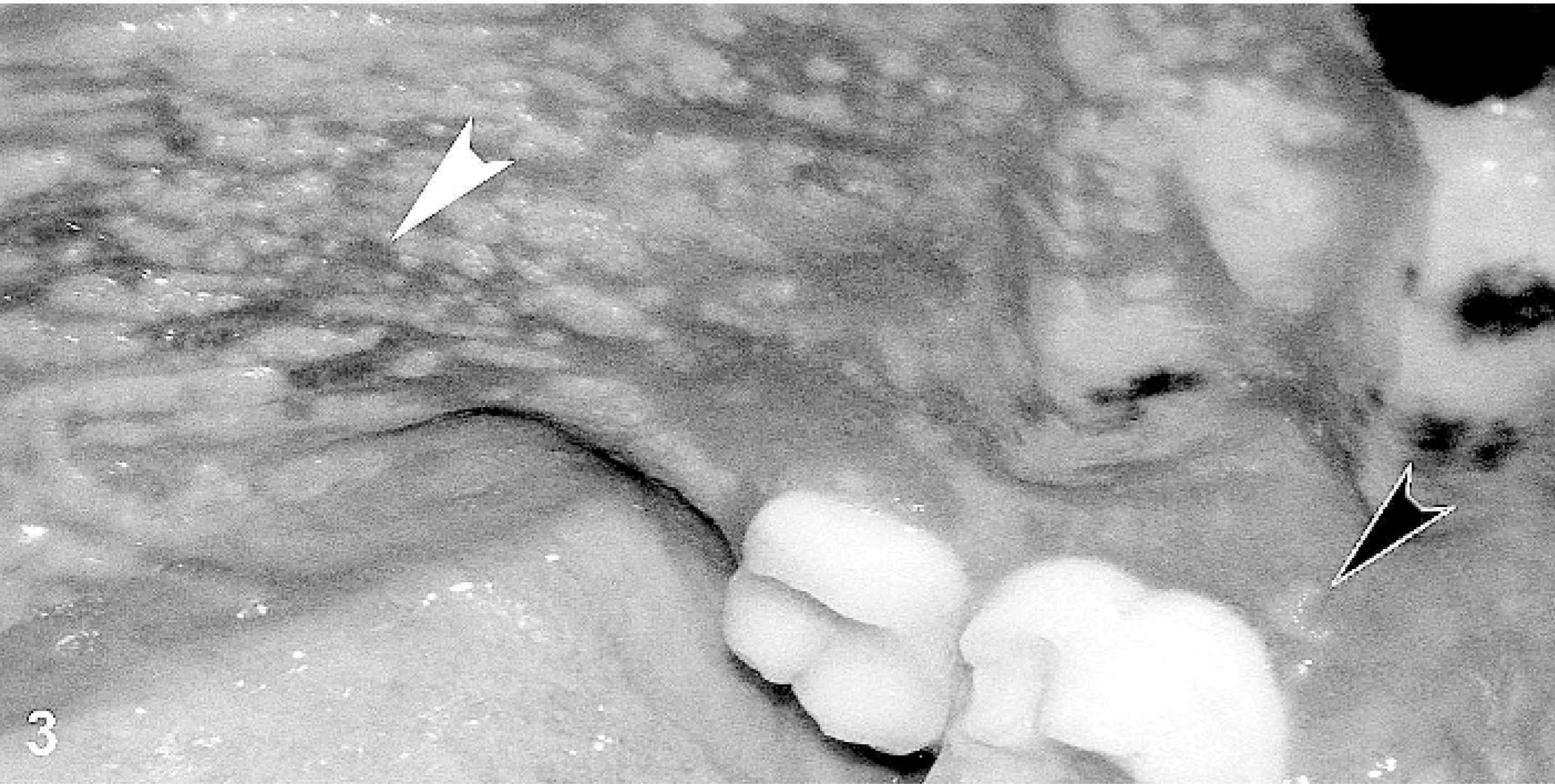
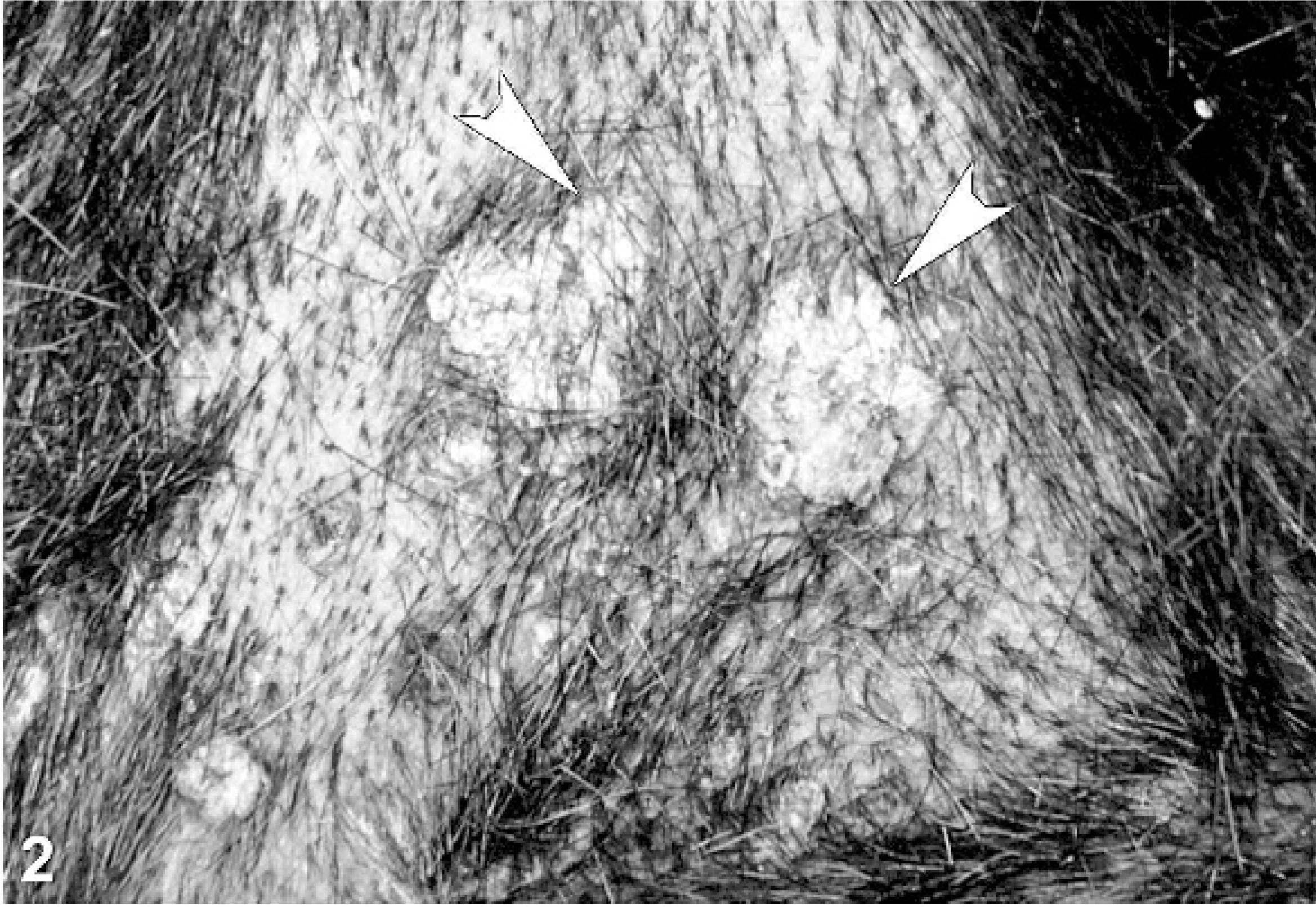
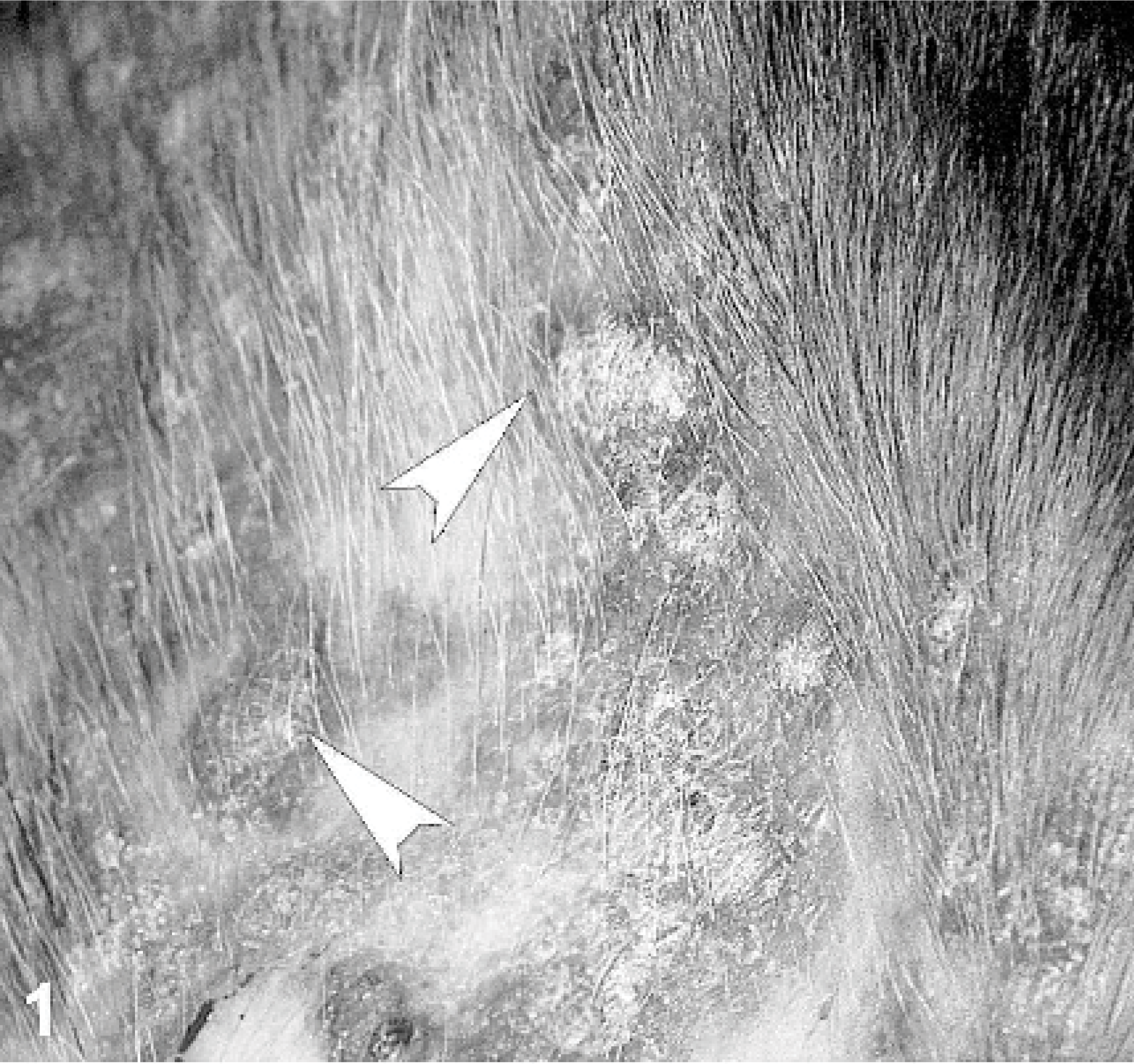
A 4-year-old male Greater Swiss Mountain Dog was first presented to a veterinarian with a 3-month history of mildly pruritic dermatitis. Crusted lesions began over the head and neck before progressively becoming generalized, with later involvement of the oral cavity, lips, and prepuce. Outside of mild hyperthermia (39.7°C) and mucosal and skin lesions, the dog appeared to be in good general condition and there were no clinical signs suggesting systemic involvement. At the time of presentation to the dermatologist, exophytic wart-like crusts overlying erosions were observed on the pinnae (Fig. 1), axillae, and sternum (Fig. 2). On the abdomen, groin, and feet, vesicles and pustules were found also. Bilateral suppurative otitis externa was present. In addition to these skin lesions, poorly defined irregular erosions and ulcers were located in the oral cavity, especially on the palate (Fig. 3) and gingiva, as well as on the prepuce and anus (Fig. 4). Small usually flaccid vesicles also were seen on the gingiva.
Anus; dog. Erosions and ulcers are observed on the margin of the anus (white arrowheads).
Oral cavity; dog. Irregular erosions (white arrowhead) are present on the palate. A recently ruptured vesicle can be seen next to the molar (black arrowhead).
Skin; dog. Wart-like crusted papules (arrowheads) are visible on the sternum.
Skin; dog. Hyperplastic crusted papules (arrowheads) cover the concave aspect of the pinnae.
Microscopic examination of a smear made from the content of a pustule revealed nondegenerated neutrophils and acantholytic keratinocytes. Punch skin biopsies were obtained from oral, abdomen, and pinnae lesions. Microscopically, sections exhibited a combination of acantholytic pustules and suprabasal clefts. The pustules were superficial and deep within the epidermis and contained nondegenerated neutrophils, eosinophils, and isolated or clustered acantholytic keratinocytes (Fig. 5). Suprabasal clefts were lined by rounded basal keratinocytes, and they contained few neutrophils, mononuclear cells, red blood cells, and rare acantholytic keratinocytes (Fig. 6). Histopathology of oral cavity lesions also revealed suprabasal acantholysis and cleft formation. In all sections of haired skin, the epidermis was acanthotic and exhibited orthokeratotic hyperkeratosis, both lesions being more severe in fully developed lesions where the hyperplastic epidermis had a papillated pattern and often exophytic projections (Fig. 7). Even in these chronic lesions, deep pustules were seen. Remarkably, both patterns of epithelial separation were observed also within hair follicle infundibuli. Dermal inflammation was of moderate severity, with a superficial perivascular to interstitial pattern composed of neutrophils, lymphocytes, histiocytes/dendritic cells, mast cells, and occasional eosinophils.

Skin (sternum); dog. Hyperkeratotic exophytic lesions exhibit papillated epidermal hyperplasia. Inset: a neutrophilic pustule develops in a juxtabasal location. HE. Bar = 200 μm
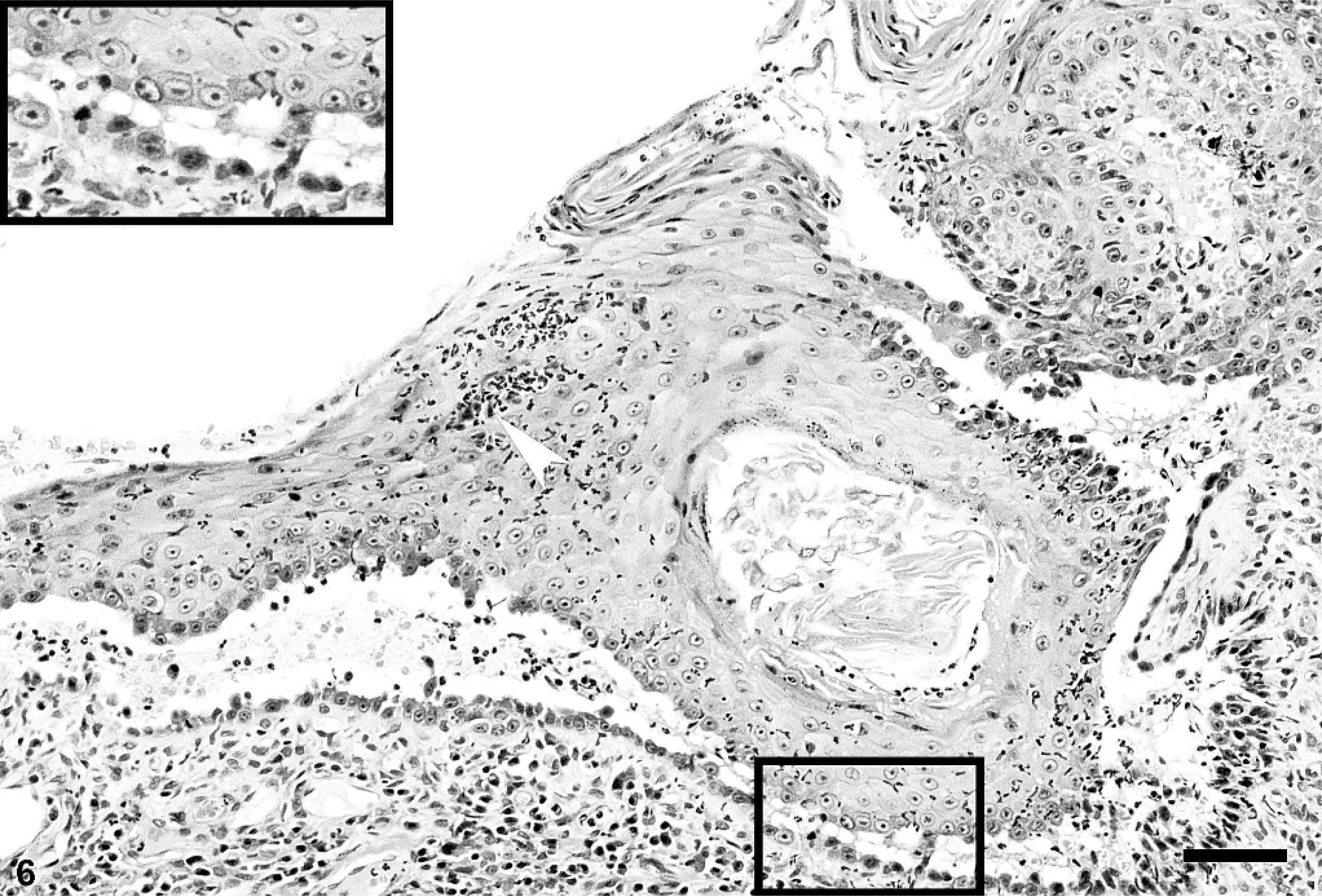
Skin (prepuce); dog. Suprabasal acantholytic clefts leave stratum basale keratinocytes at the bottom of the splits. Neutrophilic pustules occur in the upper stratum spinosum (arrowhead). Inset: suprabasal acantholysis is evident. HE. Bar = 75 μm.
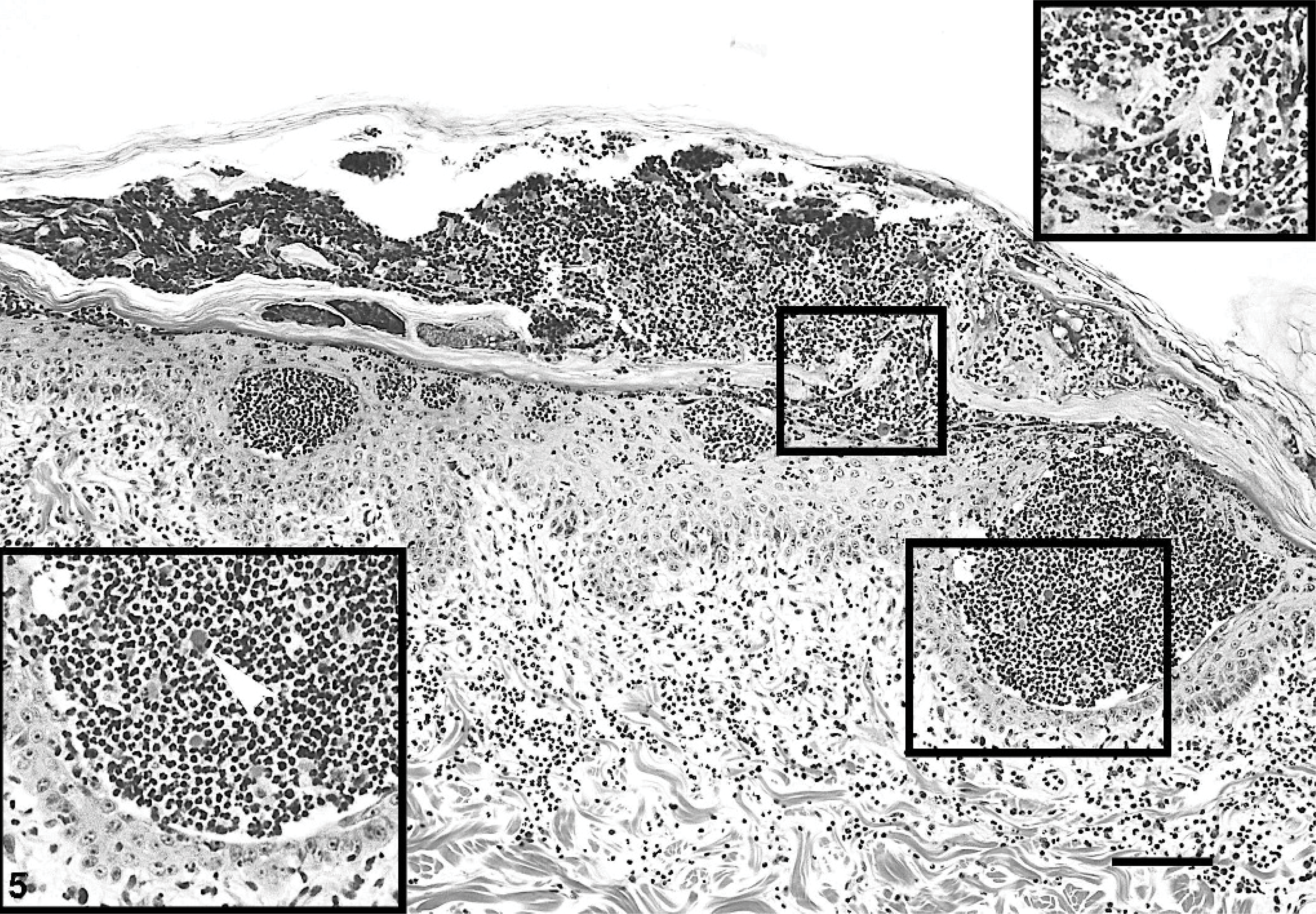
Skin (abdomen); dog. Neutrophil-predominant pustules also containing eosinophils and acantholytic keratinocytes (arrowheads) are located at various epithelial depths, from subcorneal to juxtabasal. Insets: deep (thin-walled inset) and superficial (thick-walled inset) epidermal neutrophilic pustules with acantholytic keratinocytes. HE. Bar = 150 μm.
On direct immunofluorescence performed on paraffin-embedded sections, the most consistent finding was an intercellular IgG deposition at all epidermal layers, with the strongest fluorescence seen in the stratum spinosum. Skin-fixed IgA, IgM, and C3 deposits were not evident. Indirect immunofluorescence, performed with the dog's serum and various epithelial substrates, revealed a strong antikeratinocyte membrane-staining pattern at all levels of stratified epithelial substrates with the strongest fluorescence in the stratum spinosum and weakest in the stratum basale and uppermost layers (Fig. 8). This pattern resembled that seen in rare dogs with PF that have antidesmoglein-1 autoantibodies. 5 Circulating antikeratinocyte autoantibody titers varied from 1 : 2,500 (canine gingival, neonatal mouse skin) to 1 : 10,000 (canine esophagus). Indirect immunofluorescence was unremarkable when canine bladder was used as substrate. Immunoprecipitation immunoblotting was done using baculovirus-expressed recombinant extracellular domains of canine desmogleins 1 and 3. 7, 8 This method confirmed that this dog's IgG autoantibodies recognized recombinant canine desmoglein-1. In contrast, IgG directed against desmoglein-3 was not detected (Fig. 9). Remarkably, immunoabsorption of this patient's serum with recombinant canine desmoglein-1 completely abolished previously positive indirect immunofluorescence on living MCA-B1 canine keratinocytes, a cell line that expresses both desmogleins 1 and 3. 4 These results suggest that this dog's circulating autoantibodies were directed against the extracellular segment of desmoglein-1.
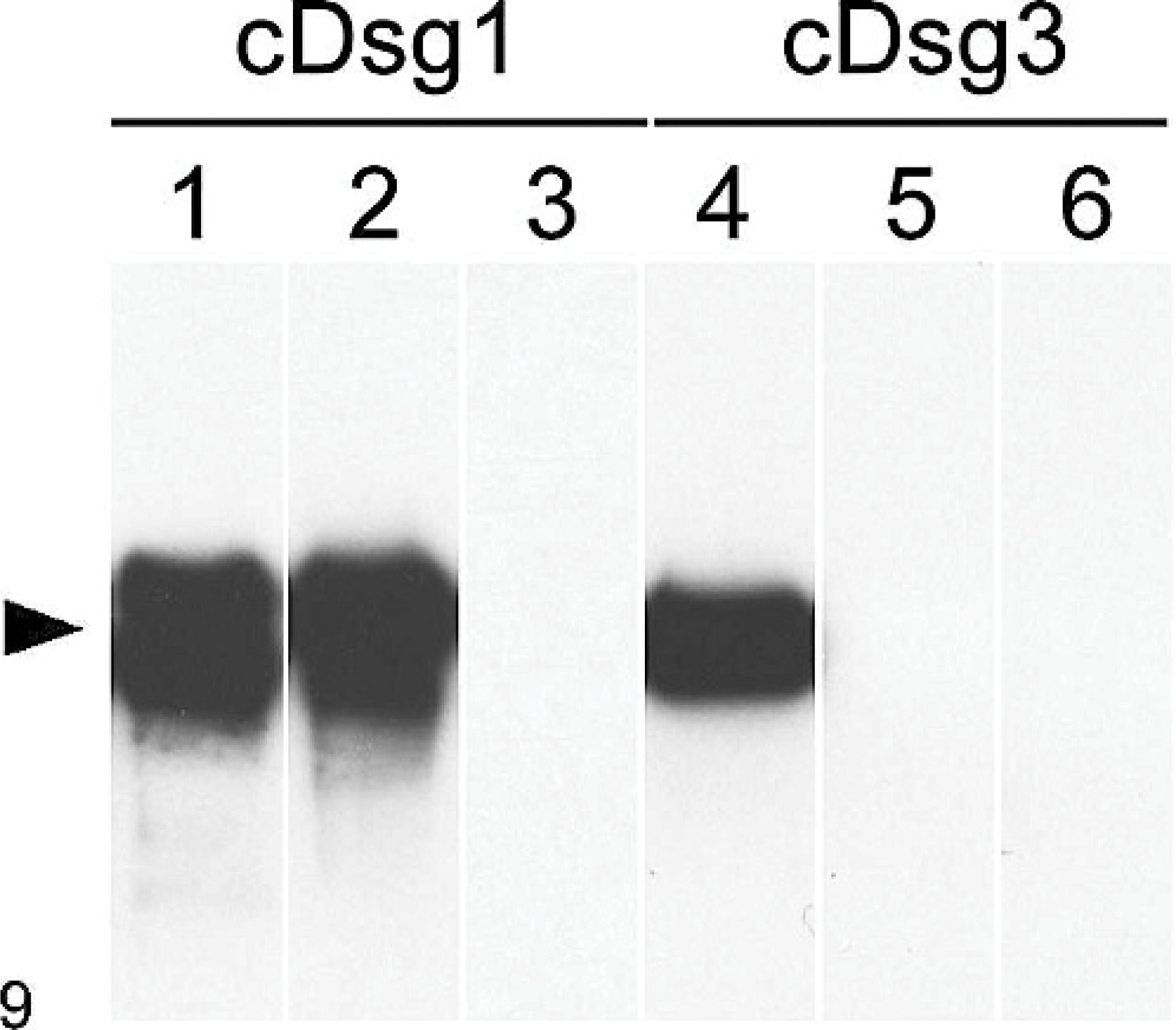
In this dog with pemphigus vegetans (PVeg) (lanes 2 and 5), circulating IgG autoantibodies recognize canine desmoglein-1 but not canine desmoglein-3. Immunoprecipitation immunoblotting; immunoprecipitation of recombinant E-tagged canine desmoglein-1 (lanes 1−3) and desmoglein-3 (lanes 4−6) with serum from a human with mucocutaneous PV (lanes 1 and 4), this dog with PVeg (lanes 2 and 5), and a normal dog (lanes 3 and 6) followed by immunoblotting with anti-E−tag monoclonal antibodies.
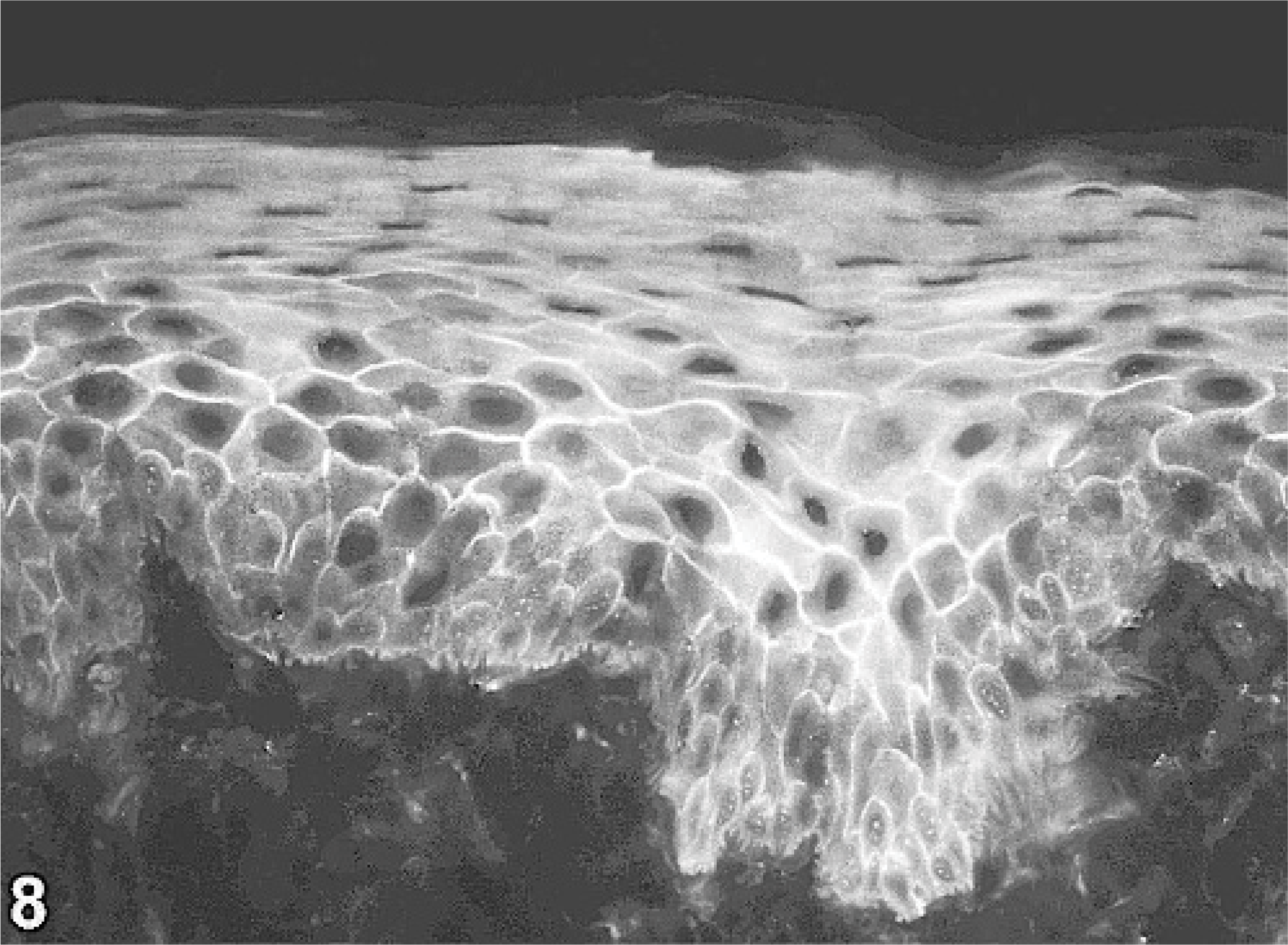
gingival substrate; normal dog. Serum IgG autoantibodies bind to keratinocytes of gingival epithelium, especially in the stratum spinosum. Indirect immunofluorescence, anticanine IgG-fluorescein.
In this dog, immunosuppression was induced with prednisolone at 2 mg/kg twice daily and azathioprine at 2.5 mg/kg once daily. Clinical signs improved rapidly, and the dosage of both drugs was reduced progressively to reach maintenance dosages of prednisolone of 1 mg/kg every other day alternated with azathioprine at 1.5 mg/kg every other day. During a follow-up of 3 years, this dog experienced occasional minor recurrences of crusted lesions over the pinnae and prepuce, all controlled with adjustment of drug dosages. The dog eventually died of a car accident.
This dog exhibited a unique combination of macroscopic and microscopic lesions. Clinically, cutaneous exophytic wart-like crusted skin lesions coexisted with oral and cutaneous vesicles evolving into deep erosions. Histologically, there were neutrophilic and eosinophilic acantholytic pustules at all levels of the epidermis and follicular infundibuli, lesions found to be more variable than those of typical canine and human PF. 12 In addition, both mucosal and skin specimens exhibited suprabasal acantholytic cleavage resembling that seen in canine and human PV. Finally, papillated epidermal hyperplasia was present in chronic lesions. Whereas it could be argued that the presence of acantholytic skin lesions at all levels of the epidermis warrants the diagnosis of “panepidermal pustular pemphigus” 18 instead of PVeg, the dog described herein did have skin and mucosal lesions, whereas those affected with “panepidermal pustular pemphigus” only exhibit skin lesions. Moreover, as discussed in details in a recent review, there is little evidence supporting the separation of canine “panepidermal pustular pemphigus” from the main superficial pemphigus variant, PF. 11
Immunologically, skin-fixed and circulating antikeratinocyte IgG were detected by immunofluorescence, and these autoantibodies were found to recognize desmoglein-1, the major human but minor canine PF antigen. 13 The outcome of our dog's disease was favorable after implementation of combined immunosuppressive therapy with prednisolone and azathioprine. This very rare coexistence of hyperplastic and wart-like crusted skin lesions and mucosal erosions respectively stemming from superficial and deep epithelial pustules and suprabasal clefts (both with acantholytic keratinocytes), along with antikeratinocyte IgG recognizing the desmosomal protein desmoglein-1, altogether support the diagnosis of PVeg for this dog. The absence of widespread cutaneous erosions may be more suggestive of the Hallopeau than the Neumann subtype of human PVeg, keeping in mind that both forms of this human disease are believed to represent a continuum of the same entity. 6 Of interest was the discovery that IgG autoantibodies in this dog identified canine desmoglein-1 instead of desmoglein-3. Indeed, most human patients with PVeg have detectable desmoglein-3−specific circulating autoantibodies, 3, 6, 9, 10, 14 whereas rare individuals also have antidesmoglein-1 IgG. 2, 15 The reason for this discrepancy is unknown, but it could be due to either stage of the disease or to a low titer of circulating antidesmoglein-3 IgG undetectable with our immunoprecipitation immunoblotting and immunoadsorption experiments.
In conclusion, the disease described herein has most, but not all, features used for diagnosing PVeg in humans: 1) presence of oral erosions and cutaneous pustules that evolve with hyperplastic wart-like crust formation; 2) microscopic exophytic epidermal hyperplasia, suprabasal clefts, and intraepidermal neutrophilic and eosinophilic pustules with acantholysis; and 3) detectable skin-fixed and circulating IgG autoantibodies that recognize desmosomal antigens. The main difference between the canine and human diseases resides in the antigen recognized by circulating autoantibodies. Whereas in human PVeg, autoantibodies principally recognize desmoglein-3, antikeratinocyte antibodies in this dog identified desmoglein-1, the major human and minor canine PF antigen.
Footnotes
Acknowledgements
The authors are grateful to Dr. Vincent Rademaeker for his referral of this dog to the veterinary dermatologist (LB) and to the owners of this dog for their compliance with treatment and follow-up.
