Abstract
A 4-year-old, female, spayed Border Collie dog was presented for progressive lethargy, inappetence, and weakness of 4 days duration. The animal had been diagnosed with pemphigus foliaceus 3 months prior and was receiving combination immunosuppressive therapy. Serum biochemistry revealed severely elevated liver enzymes and bilirubin, and humane euthanasia was elected. Gross postmortem examination revealed a diffusely pale tan to slightly yellow, enlarged, markedly friable liver with an enhanced reticular pattern. Histologically, the hepatic changes consisted of multifocal to coalescing areas of severe vacuolar degeneration, numerous coalescing foci of hepatocellular necrosis, and myriad intra- and extracellular protozoa that reacted immunohistochemically with polyclonal antibodies to Neospora caninum, and not Toxoplasma gondii. Neosporosis in the current case is thought to be due to reactivation of latent N. caninum occurring with the administration of glucocorticoid therapy. The severe complication in the present case highlights the importance of early detection and mitigation of common infections in immunosuppressed animals.
A 4-year-old, female, spayed Border Collie dog was presented to urgent care with a 4-day history of progressive lethargy, inappetence, weakness, and collapse. She had previously been diagnosed with pemphigus foliaceus approximately 3 months prior and was currently prescribed oral prednisone (1.8 mg/kg/day) and azathioprine (2 mg/kg/day) for treatment. The dog lived in a suburban area and had indoor and outdoor access. There was no other pertinent medical history.
On physical examination, the patient was recumbent but alert and responsive. She was tachypneic (60 bpm; reference [ref.] range: 18–34 bpm), tachycardic (140 bpm; ref. range: 70–120 bpm), and normothermic (38.5°C; ref. range: 37.5–39.2°C), with weak, but synchronous femoral pulses. Cardiothoracic auscultation revealed increased bronchovesicular lung sounds in all fields. Abdominal palpation revealed hepatomegaly. Blood was observed in the stool on rectal examination. Ophthalmic examination revealed bilateral icterus of the sclera. Alopecic, crusting lesions were multifocally distributed on the nasal planum, periorbital region, and the medial aspect of the forelimbs consistent with the previous diagnosis of pemphigus foliaceus.
Complete blood cell count revealed an inflammatory leukogram (segmented neutrophils, 6.7 × 103 cell/µl; ref. range: 2.6 × 103–11.0 × 103 cells/µl) with a left shift (band neutrophils, 0.6 × 103 cells/µl; ref. range: 0.0 × 103–0.2 × 103 cells/µl), lymphopenia (0.1 × 103 cells/µl; ref. range: 1.0 × 103–4.8 × 103 cells/µl), thrombocytopenia (46.0 × 103 cells/µl; ref. range: 200.0 × 103–500.0 × 103 cells/µl), and icteric serum. Serum biochemistry abnormalities included hyperglycemia (181 mg/dl; ref. range: 70–115 mg/dl), hypocalcemia (8.6 mg/dl; ref. range: 9.0–11.5 mg/dl), hypoalbuminemia (2.7 gm/dl; ref. range: 3.0–4.3 gm/dl), hypercholesterolemia (419 mg/dl; ref. range: 130–300 mg/dl), hyperbilirubinemia (4.9 mg/dl; ref. range: 0.0–0.2 mg/dl), markedly elevated cytokeratin (34,203 IU/l; ref. range: 50–275 IU/l), and a generalized, marked elevation in liver enzymes: alkaline phosphatase (4,468 IU/l; ref. range: 15–140 IU/l), alanine aminotransferase (10,348 IU/l; ref. range: 10–90 IU/l), aspartate aminotransferase (4,283 IU/l; ref. range: 15–45 IU/l), and gamma-glutamyl transferase (737 IU/l; ref. range: 0–9 IU/l). Electrolyte abnormalities included hyponatremia (131 mEq/l; ref. range: 140–150 mEq/l), hypokalemia (3.6 mEq/l; ref. range: 3.7–5.2 mEq/l), hypochloremia (92 mEq/l; ref. range: 108–118 mEq/l), and an elevated anion gap (23 mEq/l; ref. range: 10–21 mEq/l). Urine dipstick analysis demonstrated marked proteinuria (3+; expected value, negative), bilirubinuria (3+; expected value, 0–1+), glucosuria (1+; expected value, negative), and hematuria (3+; expected value, negative). Urine sediment revealed white blood cells (6–10 per high-power field; expected value, none), and occasional red blood cells, transitional and/or renal cells, granular casts, and amorphous and bilirubin crystals.
Ultrasonographic imaging of the abdomen was performed; hepatomegaly with diffusely hyperechoic parenchyma, moderate sedimentation in the gall bladder, and a 1.2-cm hyperechoic lesion with acoustic shadowing located directly distal to the neck of the gall bladder (choledocholithiasis) were documented. Given the diagnosis of severe, acute hepatocellular damage and the need for extensive emergency care and diagnostics, the owner elected for humane euthanasia, and a postmortem examination was performed.
Gross examination of the integument revealed severe, bilateral, multifocal to coalescing, ulcerated skin lesions, located periorbitally, on the nasal planum, and on the medial aspect of both forelimbs. There was also evidence of hyperkeratosis of the epidermis in these same regions. Intra-abdominally, the liver was diffusely pale tan to slightly yellow, moderately enlarged, and markedly friable with an enhanced reticular pattern on the capsular and cut surface (Fig. 1).
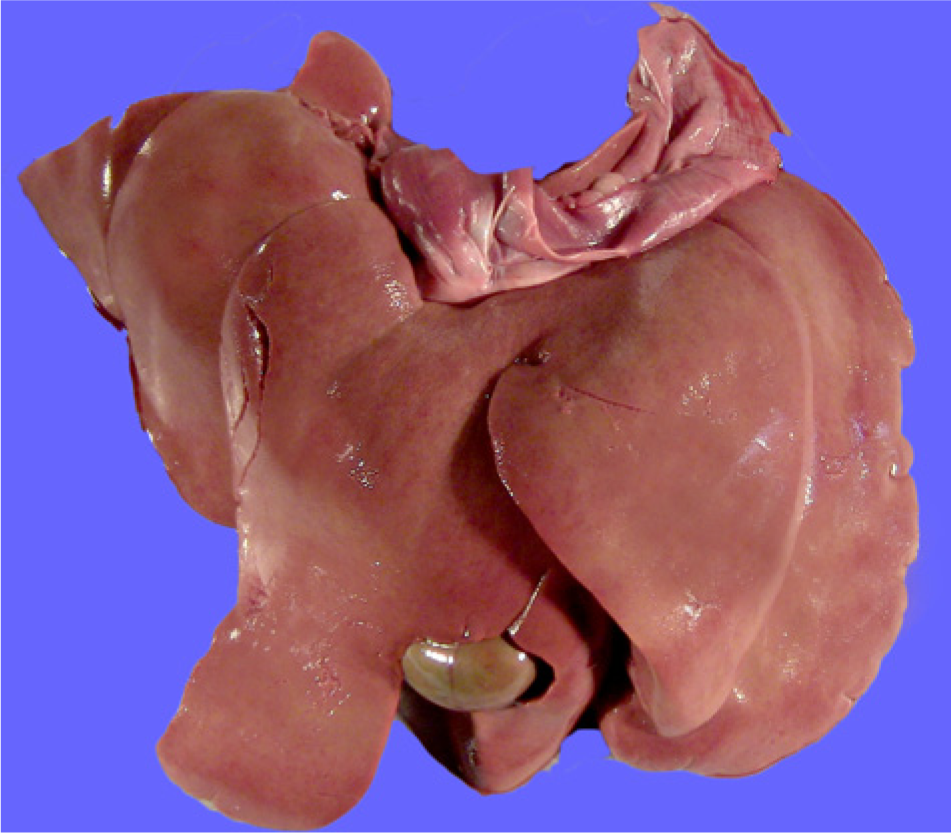
Liver; dog. Gross appearance of liver showing pallor, hepatomegaly, and postmortem fractures due to increased friability.
Histologically, the hepatic changes consisted of multifocal to coalescing areas of severe vacuolar degeneration, characterized by swollen hepatocytes with either indistinct clear cytoplasmic vacuoles (hydropic degeneration), or numerous, small, discrete, clear cytoplasmic vacuoles (microvesicular lipidosis; Fig. 2A). Additionally, scattered randomly throughout the hepatic parenchyma were numerous, multifocal to coalescing foci of lytic hepatocellular necrosis. The necrotic areas were characterized by shrunken, hypereosinophilic hepatocytes with pyknotic to karyolytic nuclei, admixed with abundant amounts of necrotic cellular and karyorrhectic debris, and low to moderate numbers of neutrophils, few macrophages, and lesser amounts of fibrin and hemorrhage. Protozoal tachyzoites, 2–4 µm long, with a centrally located, basophilic nucleus, and thin peripheral rim of clear cytoplasm were found intracellularly within hepatocytes and Kupffer cells as loose or tightly packed groups, as well as scattered extracellularly adjacent to foci of necrosis (Fig. 2B). A 2-step, hemi-nested polymerase chain reaction amplification of the B1 gene for identification of Toxoplasma gondii was performed on formalin-fixed, paraffin-embedded hepatic tissue, and was negative (University of Washington, Department of Laboratory Medicine, Molecular Microbiology Section). Immunohistochemical staining was performed using polyclonal rabbit anti–T. gondii and Neospora caninum as described. 13 The protozoal organisms reacted positively with N. caninum but not T. gondii antibodies (Fig. 2C). Additionally, no periodic acid–Schiff–positive material was identified within the vacuoles of the hepatocytes. Other organs with microscopic changes included severe lymphoid depletion of the spleen, occasional luminal and intramucosal gastric nematodes consistent with Ollulanus sp., and severe, subcorneal, pustular, acantholytic dermatitis with severe acanthosis and orthokeratotic hyperkeratosis consistent with previously diagnosed pemphigus foliaceus. Sections of cerebrum were histologically unremarkable and negative for N. caninum by immunohistochemical staining. Skeletal muscle was not evaluated histologically.
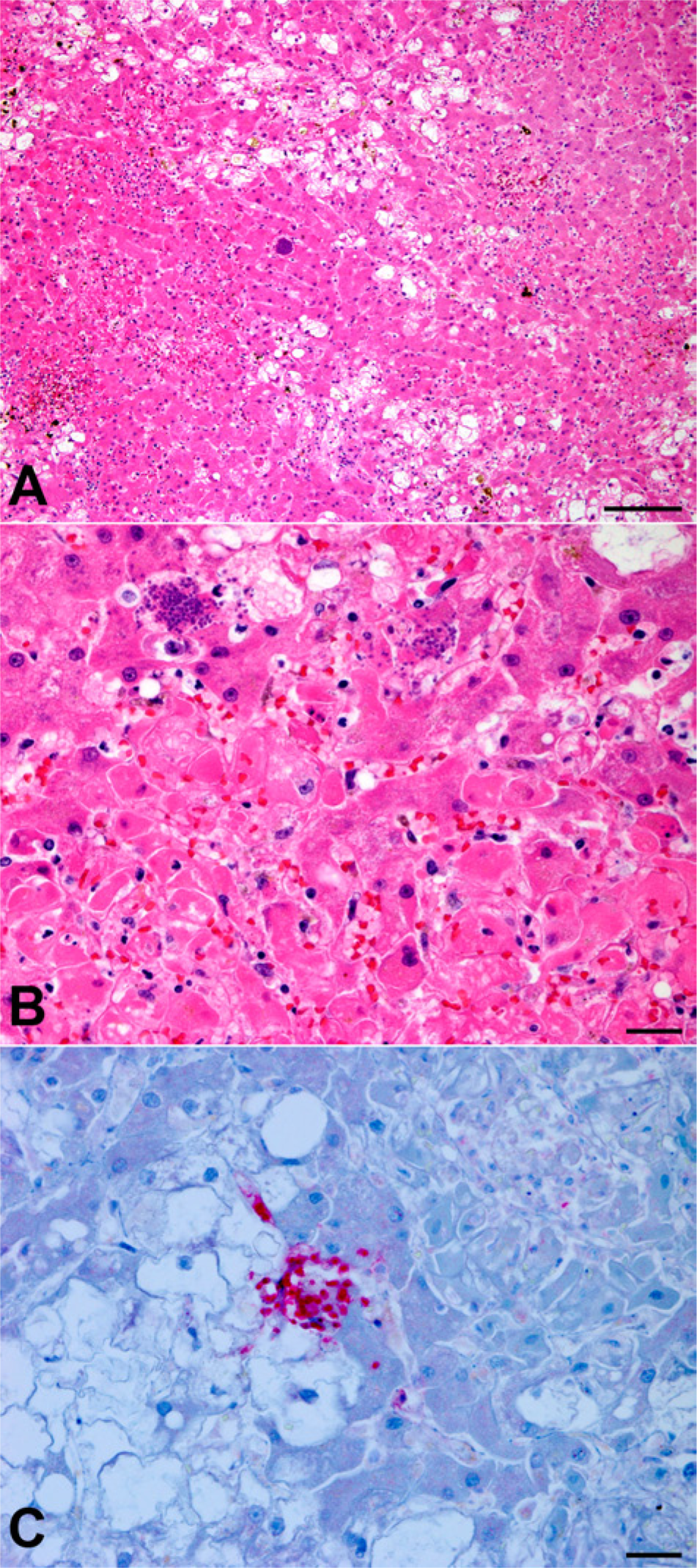
Liver; dog.
The obligate intracellular coccidian parasite, N. caninum, can infect a wide variety of hosts, with dogs and cattle being the primary domestic species affected.4,9 Currently, domestic and wild canids are the only known definitive hosts, acquiring the disease through ingestion of infected tissues or transplacentally from infected dams. 9 Clinical manifestation of the disease in canids is uncommon, but typically presents as a progressive, ascending neuromuscular disorder characterized by polyradiculitis, polymyositis, and meningoencephalitis.1,6 Clinical signs vary depending on the degree of tissue invasion and the amount of cellular necrosis caused by the pathogen’s intracellular replication (tachyzoite stage). 2 Subclinical neosporosis occurs when these tachyzoites localize as tissue cysts (bradyzoites); the cysts can persist within the tissue and may reserve the potential for reactivation later in life.2,6 The most common sites for tissue cyst localization are the central nervous system and muscle.1,4
Although the disease does not appear to have any age, sex, or breed predilections overall, it is important to note that N. caninum has been associated with concomitant cytotoxic and glucocorticoid therapy, as well as age-related immunodeficiency.7,11,12,16 At the time of presentation, the patient was being prescribed immunosuppressive doses of prednisone and azathioprine (1–2 mg/kg/day and 2 mg/kg/day, respectively 17 ) for treatment of pemphigus foliaceus, an autoimmune disease characterized by autoantibodies targeting the desmosomal proteins present between keratinocytes of the superficial epidermis. 19 The deficiency in cellular immunity that resulted from treatment most likely facilitated the establishment of clinical neosporosis. It is unknown whether the current case occurred due to reactivation of congenital infection or recent acquisition, but clinical and experimental evidence has shown recrudescence of latent N. caninum with the administration of glucocorticoids,6,11 suggesting the use of immunosuppressive therapy in this patient contributed to the activation and clinical manifestation of disease.
The most salient feature of the present case is hepatic neosporosis, which has only rarely been documented as the most severe lesion. A similar example to the present case was described in a 4-year-old Poodle in New Zealand that likely developed Neospora-associated hepatitis as a result of reactivated infection following drug-induced immunosupression. 11 In that case, however, the extent of liver involvement was unknown because the diagnosis was based on liver biopsy and other organs were not examined. Additionally, hepatitis was the most prominent feature in an 8-month-old female Basset Hound diagnosed with generalized neosporosis in the United States. 4 Most (>75%) of the Basset Hound’s liver was necrotic, and masses of tachyzoites were present within lesions (case 6). 4 The dog also had widely disseminated Neospora infection and, unlike the present report, there was no history of immunosuppressive treatment, suggesting an alternate pathology than exogenous immunosuppression.
Infectious diseases are relatively common sequelae of human beings and animals undergoing immunosuppressive therapy for neoplastic and immune-mediated conditions, highlighting the importance of monitoring and early detection. Antemortem N. caninum methods include serology, cytology, histology, and polymerase chain reaction.1,5,18 For serology, the indirect fluorescent antibody test has been most commonly used, and titers are almost always greater than 1:800 in clinical animals, but titers as low as 1:25 have been documented in confirmed cases of neosporosis.1,5,8 Clinical neosporosis is uncommon, but if positive titers are discovered in immunosuppressed patients, such individuals benefit from immediate treatment in order to mitigate reactivation and prevent the manifestation of a potentially fatal disease. Clindamycin, trimethoprim–sulfonamide, and pyrimethamine—often used in combination—are the treatments most commonly utilized in dogs.1,5 Prognosis is more favorable in cases with slow onset of disease, less severe clinical signs, and rapid initiation of treatment. 1
In addition to the extensive areas of hepatocellular necrosis, multifocal vacuolar degeneration not associated with N. caninum replication was also present. Steroid hepatopathy, a common side effect of long-term glucocorticoid administration resulting in glycogen accumulation within hepatocytes, is normally characterized by vacuolar changes diffusely or zonally distributed throughout the hepatic parenchyma. 10 The histologic changes present in the current case, however, are atypical for steroid hepatopathy given the random distribution and lack of periodic acid–Schiff–positive histochemical staining. Alternatively, azathioprine hepatotoxicity has been well documented in human beings, and is understood to occur in dogs, but is not currently described in the veterinary literature. In human beings, this often manifests as cholestasis, endothelial and vascular injury, nodular hyperplasia, or acute hepatocellular necrosis3,14,15,20; however, the histologic presentation in dogs is yet to be documented. Thus, azathioprine may have contributed, or co-contributed, to the pathologic changes noted in the liver as an atypical manifestation not discussed previously.
Immunocompromised animals, whether etiologically congenital, acquired, or iatrogenic, are at risk for a multitude of secondary infections. In the present case, fulminant hepatic disease due to N. caninum was a severe complication of immunosuppression. To the authors’ knowledge, hepatic neosporosis has not previously been reported in an animal being treated for immune-mediated skin disease, making this case a unique presentation for the protozoal infection and highlighting the importance of early detection and mitigation of common infections in immunocompromised animals.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
