Abstract
Growth anomalies (GAs) are a morphologically diverse and poorly understood group of lesions affecting corals. The aim of this study was to describe the prevalence and morphology of GAs affecting the faviid corals Diploria labyrinthiformis, Pseudodiploria strigosa, Psudodiploria clivosa, and Colpophyillia natans on St. Kitts. Three gross morphological variants of GAs (exophytic, nodular, and ruminate) were equally prevalent, together affecting 7.8% of corals surveyed across 5 reefs. Prevalence varied by reef and coral species, being highest in C. natans (35.7%). Median colony diameter was larger in corals with GAs relative to those without (Mann-Whitney U test, P < .001). Histopathological examination of exophytic GAs consistently showed corallite and polyp gigantism (n = 7), characterized by polyp enlargement and retained microanatomical structures. In contrast, nodular GAs (n = 9) were consistently hyperplasia of the basal body wall with skeletal dystrophy, composed of micronodular skeletal deposits with abundant hyaline lamellae, bordered by calicoblastic epithelial hyperplasia, interspersed with distorted gastrovascular canals and islands of mesoglea. Endolithic organisms, particularly fungi and algae, were common among GA and apparently healthy biopsies. While pathogenesis of these lesions remains uncertain, a neoplastic basis for GAs on Caribbean faviids could not be established using diagnostic criteria conventionally applied to tumors of vertebrate taxa, in line with other recent observations of coral GAs.
Coral disease is a global threat to coral reef ecosystems, particularly in the Caribbean basin which has been termed a “coral disease hot-spot” due to high disease prevalence and increasing reports of disease. 19,36 Nevertheless, causes of a vast majority of coral diseases remain unknown. 32 Basic descriptions of gross and microscopic lesion morphology are lacking for most coral diseases, 38 presenting an impediment to identifying their cause, as well as establishing case definitions for accurate diagnoses. 43 It is hoped that expanded efforts to comprehensively document and describe the morphology of coral lesions will facilitate an understanding of their pathogenesis, and potentially provide insights needed to devise effective mitigations.
The Federation of Saint Christopher and Nevis, hereto referred to as St. Kitts and Nevis, is a pair of sovereign islands that form part of the Lesser Antilles. St. Kitts is the larger of the 2 islands, with a high population density of 164 people per square kilometer. 2 The nearshore marine environment is home to fringing reefs, formed from volcanic and eroded material colonized by scleractinian coral species. These reefs have become macroalgal dominant, with relatively low coral cover (6% to 13%) resulting from historical incidences of disease and bleaching as well as competition from other benthic organisms. 6 Much of the fringing reef framework was built by members of the genus Orbicella, along with now-dead Acropora palmata colonized by small colonies of fast-growing or brooding corals such as the genus Porites. 6 Members of the family Faviidae are among the largest remnant corals. Faviids have become primary reef building species as other complexes have declined in the region. 7 The faviids Pseudodiploria strigosa, Diploria labyrinthiformis, Pseudodiploria clivosa, and Colpophyllia natans together comprise around 10% of live coral cover on St. Kitts’ reefs. 7 Prevalence of scleractinian coral diseases in the federation was assessed at 1.2% in 2012. 6
Hard corals have a limited repertoire of gross morphological manifestations of disease, and these can be assigned to 1 of 3 lesion categories: tissue loss, pigmentation (ie, discoloration), or growth anomaly. 39,43 Growth anomalies are a distinctive group of coral lesions, presenting as masses involving both tissue and skeleton. The first-described growth anomaly in a scleractinian was in P. strigosa that was thought to have overgrown a Meandrina meandrites. 37 The discovery of a similar lesion in mesophotic Madrepora kauaienis raised the question of whether such lesions were neoplastic. 33 These lesions have been variably referred to as neoplasms, tumors, skeletal malformations, skeletal tissue anomalies, or hyperplasms. 3,11,14,25,31 While there is much to be learned regarding the diverse pathology underlying these lesions, the term “growth anomaly” is the preferred gross morphological diagnosis and brings some clarity to otherwise ambiguous and confusing coral lesion terminology. 16
Growth anomaly morphology appears to have a large degree of variation, although detailed histopathological descriptions are sparse. Work et al described 7 distinct gross morphological types of growth anomalies (GAs) in Indo-Pacific acroporids alone, including exophytic, bosselated, crateriform, nodular, vermiform, fimbriate, and annular variations. 41 Most morphological forms were microscopically composed of gastrovascular canals surrounded by skeleton and lacking other polyp structures. The gastrovascular canals were lined by well-differentiated components of basal body wall, including calicodermis, mesoglea, and gastrodermis, and were thus interpreted as basal body wall hyperplasia. 41 Peters et al had previously described morphologically similar proliferations of the basal body wall in Caribbean A. palmata, but the invasive nature of these GAs led to the conclusion of neoplasia (termed “calicoblastic epithelioma”). 31 GAs on other acroporids of the Gulf of Oman were subsequently diagnosed as calicoblastic epithelioma, although microscopic descriptions were lacking. 11 Montipora capitata GAs were microscopically categorized as basal body wall hyperplasia, although histologic features leading to this interpretation were similarly undescribed. 9 Histopathology of GAs in Platygyra pini, Platygyra sinensis, and Porites compressa revealed enlarged polyps, 14,25 and in Pavona clavus showed no significant morphologic alterations. 17 Histologic assessment of GAs has also identified polyp enlargement, necrosis, or encapsulation responses accompanying algae or metazoan infection. 10,20,43 Microscopic pathology of GAs also inconsistently features reduced dinoflagellate symbionts (ie, Symbiodiniaceae) and gonadogenesis, 9,17,31,41 basal body wall and mesenterial filament necrosis, 41,43 and segments of calicoblastic epithelial hyperplasia. 14,31 Endolithic organisms that reside within the coral’s calcium carbonate aragonite skeleton, including fungi and sponges, are also frequently present within GAs. 11,31,41
Most studies on GAs involve corals of the Indo Pacific, and primarily the families Acroporidae and Poritidae. 17,41,43 Prevalence in Pacific Acroporidae is variable, although it can reach upwards of 20%. 8,41 In the greater Caribbean, GA reports have been limited to A. palmata. 4,31 Various observations, mostly in the form of unpublished data, indicate that GAs also occur in the Caribbean scleractinians Siderastrea radians, Mycetophyllia ferox, Acropora cervicornis, Orbicella annularis, Orbicella faveolata, Montastraea cavernosa, D. labyrinthiformis, P. strigosa, and Pseudodiploria clivosa. 15,16,31,42 GAs are particularly conspicuous in certain members of family Faviidae where associated distortions of the labyrinthine pattern of skeletal ridges are readily apparent. However, there are no reports describing the cellular pathology of faviid GAs, apart from archived observations in D. labyrinthiformis and P. strigosa, summarized as enlarged polyps. 31,42
In order to better understand the pathogenesis of GAs in Caribbean faviids, a pathology survey was undertaken in St. Kitts to describe and classify their morphology at the gross and microscopic level. The objectives of this study were to (1) describe gross morphology of GAs affecting D. labyrinthiformis, P. strigosa, and Colpophyllia natans; (2) estimate the prevalence and distribution of morphological types of GAs among affected species in St. Kitts; and (3) describe cellular pathology of faviid GAs as a comparison for those of other corals.
Materials and Methods
GA Prevalence Survey
Five nearshore reefs in St. Kitts were surveyed for the presence of GAs on D. labyrinthiformis, P. strigosa, P. clivosa, and C. natans including Major’s Bay, White House Bay, Timothy Beach, Turtle Beach, and Dieppe Bay (Fig. 1). These sites were selected because of ability to adequately cover the area while snorkeling, known presence of species of interest, and proximity to shore. Surveys were undertaken between September and October 2019, and minimum visibility was at least 5 meters. A roving diver survey approach was used because of low overall coral cover on the island. 6 Each site received equal effort, with a swim time of 60 minutes per site haphazardly following the reef contour. Maximum depth was 7 meters. A single snorkeler led each survey in order to prevent miscounting or counting colonies more than once. Each target species within 5 meters to each side of the surveyor was assessed for presence and type of GA, colony diameter measured, and photographed. A handheld GPS device was used to record the survey track and location of surveyed colonies. Colony diameter was recorded as the maximum diameter in dimension to the nearest 5-cm increment. Lesions were categorized by gross morphologies building on previously established nomenclature. 39,41 GAs in P. clivosa were distinguished from nodular protuberances normally observed in the species by their expansile nature, indicated by the narrowing or closure of valleys in the coral bordering the GA. 25
Location of reefs surveyed for growth anomalies affecting Diploria labyrinthiformis, Pseudodiploria strigosa, Pseudodiploria clivosa, and Colpophyllia natans on St. Kitts, 2019.
Histopathology
Between June and August 2019, histopathology samples were collected from GA affected D. labyrinthiformis, P. strigosa, and C. natans at the same sites assessed with prevalence surveys. Every target coral encountered with a GA within a 2-hour dive was biopsied for this study.
Biopsies were collected using a 3.8 cm hole saw that was hammered into the coral surface and extracted using a chisel. One to 2 lesions per affected coral were biopsied. Biopsies were taken at the lesion margin to capture both apparently healthy and lesioned tissue for histological analysis. An area of apparently healthy tissue was also taken from the base of each colony. Biopsies were placed in individual bags tagged with a colony number and fixed immediately post-dive in a 1 to 4 ratio of zinc-formaldehyde concentrate (Z-Fix, Anatech) and local seawater for a final concentration of 1:5, and with approximately 10 to 1 ratio of fixative to tissue.
Photographs were taken of the entire colony, the biopsied lesion(s) including a scale reference, and the area post-biopsy. These were used in post hoc analysis to help define lesion types as well as aid in trimming tissues for histology.
Samples were fixed for a minimum of 48 hours before being fast-decalcified in formic acid (Formical-2000, StatLab). Fixed tissue was trimmed sagittally near the actinopharynx of the polyp if possible, to span the interface of GA and bordering tissue. In order to facilitate microscopic differentiation of the GA and grossly normal coral, tissue marking dye (Davidson Marking System, Bradley Products) was sometimes applied to the epidermal surface of the GA. Apparently healthy tissues were trimmed in the same manner. Trimmed tissues were placed into histology cassettes containing foam biopsy pads to maintain proper orientation of the tissue, taking care not to stretch the tissue and artefactually widen the space where skeletal ridges had been decalcified. Tissues were decalcified again in formic acid for 20 minutes, then briefly immersed in tap water to remove residual formic acid, and returned to z-fix until processed for histology. Tissues were embedded in paraffin wax, surface decalcified, trimmed in a rotary microtome at 4 μm, mounted to microscope slides, and stained with hematoxylin and eosin using routine methods.
GAs were qualitatively assessed using an Olympus BX51 microscope with an ocular having a standard field number 22 (ie, 40× high-powered field diameter = 0.55 mm), 26 while comparing to tissues from apparently healthy areas. The presence of microscopic lesions were recorded for both GA-affected and apparently healthy biopsies, including necrosis of basal body wall and mesenterial filament, disorientation or distortion of gastrovascular canals, calicoblastic epithelial hyperplasia, presence of gonadogenesis, presence and type of endolithic organisms, and hyaline lamellae deposition.
The severity of certain microscopic lesions, including necrosis, calicoblastic epithelial hyperplasia, hyaline lamellae deposition, and presence of endolithic organisms, were scored based on percent area of coverage in a field area. The field area used for categorizing severity was representative of the most severely affected regions across all sections evaluated. Field diameter was calculated as field number of the ocular/objective magnification, and diameter was used to determine field area. 26 Severities of calicoblastic epithelial hyperplasia, hyaline lamellae deposition, and necrosis were scored as mild if <25%, moderate if ≥25% to 75%, and severe if ≥75% of basal body wall in a field area 0.95 mm2 (20× objective) was affected. Endolithic organisms were scored mild if <25%, moderate if ≥25% to 90%, or severe if ≥90%, of the skeleton was occupied in a field area of 3.8 mm2 (10× objective).
A standard set of microanatomic measurements was collected from each biopsy. These were taken from regions that were considered representative of the predominant features observed across all slides prepared from a single biopsy. Counts were made using imaging software where contours of appropriate length were overlaid on slides (CellSens, Olympus Corporation). Symbiont counts were determined as a mean of 3 counts using 100-µm length of surface body wall gastrodermis. Epidermal cnidocytes were similarly taken as a mean across 3 counts using 100-µm length of epidermis not involving a tentacle. Gonad thickness, as a rough indicator of degree of gonadal maturation, was measured as the thickest width of the gonad observed across all sections examined from the same biopsy.
Microorganisms were categorized to at least the phylum level based on microscopic morphological features similar to those used by others. 41 Sponges were multicellular organisms including choanocytes and a matrix with siliceous spicules. Helminths were vermiform multicellular organisms. Algae were filaments or mosaics of cuboidal to rectangular cells with a distinct cell wall. Fungi were filamentous hyphae, with and without septation or branching. Cyanobacterium were filaments of concatenated trichomes. Crustaceans had a cuticle, segmented appendages, striated muscle, gut, hepatopancreas, and reserve inclusion cells.
Statistical Analysis
GAs were reported as prevalence with 95% confidence intervals constructed using binomial method. Colony diameter data were assessed for normality using a Shapiro-Wilk test. Median colony diameters were compared by presence or absence of GA pathology as well as individual gross GA morphology using a Mann-Whitney U test.
Polyp depth was measured on the formalin-fixed paraffin-embedded tissue block, where depth was defined as the distance between outer epidermal layer and the deepest aspect of basal body wall. A paired Wilcoxon rank sum test was used to compare polyp depth among GA biopsies and apparently healthy biopsies, using significance level of P ≤ .05.
The presence of necrosis, calicoblastic epithelial hyperplasia, and hyaline lamellae were compared among GAs and apparently healthy samples using a Fisher’s exact test, and the severity of these microscopic lesion components was compared using a Mann-Whitney U test. Mean epidermal cnidocytes and symbionts per 100 µm surface body wall were compared among GA types and apparently healthy tissues using a Wilcoxon signed-rank test. To account for multiple histological comparisons among colonies, the significance level was adjusted with a Bonferroni correction to P ≤ .005.
Results
Surveys
Throughout the surveys, 3 gross morphological types of GAs were observed in the target species: exophytic, nodular, or ruminate (Figs. 2–4). Exophytic GAs were solitary raised expansile masses composed of widened skeletal ridges and valleys which followed a meandering pattern similar to the bordering tissue, while retaining polyps and costal detail. Nodular GAs were multifocal papillary to sessile, <2 cm in diameter, solitary or coalescing masses which protruded from ridges, sometimes compressed valleys, had a smooth surface lacking polyps, and were paler in color than bordering tissue. Ruminate GAs were solitary or multifocal discrete slightly raised or depressed areas where skeletal ridges were of uneven width or height being narrowed or subtly nodular, branched to form dead-ends or adjoin with neighboring ridges, and had obscured or collapsed costae and retained polyps.

Growth anomalies, faviid corals.
Of 307 individual corals from the target species surveyed across all sites, 24 individuals were found to have GAs (7.8%; 95% confidence interval [CI]: 4.8% to 10.8%). GA prevalence in the target species was low overall, but varied by reef: 3.4% (95% CI: 0.41% to 11.7%) at Timothy Beach, 4.5% (95% CI: 0.93% to 12.5%) at Turtle Beach, 4.8% (95% CI: 0.58% to 16.2%) at White House Bay, 6.3% (95% CI: 2.1% to 14.2%) at Major’s Bay, and 20.3% (95% CI: 11.0% to 32.8%) at Dieppe Bay. GA prevalence, and distributions of morphological GA type, varied by species (Table 1). Overall GAs were most prevalent in C. natans (35.7%; 95% CI: 21.2% to 50.2%). When target species were pooled, morphological GA types occurred in similar proportions (Table 1). GA colony size was not normally distributed (Shapiro-Wilk test, W = 0.83664, P value < 2.2e-16). GAs tended to be found on larger coral colonies and there was a significant difference in median coral diameter among GA-affected and nonaffected coral colonies (Fig. 5; median diameter of affected and nonaffected colonies was 57.5 and 30 cm, respectively; P = 2.2e-16, Mann-Whitney U test). Median diameter was only higher in nodular GA-affected colonies (median: 64 cm) when compared to nonaffected colonies (median: 30 cm; Mann-Whitney U test, P = .00044).
Prevalence of growth anomalies across 5 reefs surveyed in St. Kitts, by host coral species and gross morphologic category of the growth anomalya.
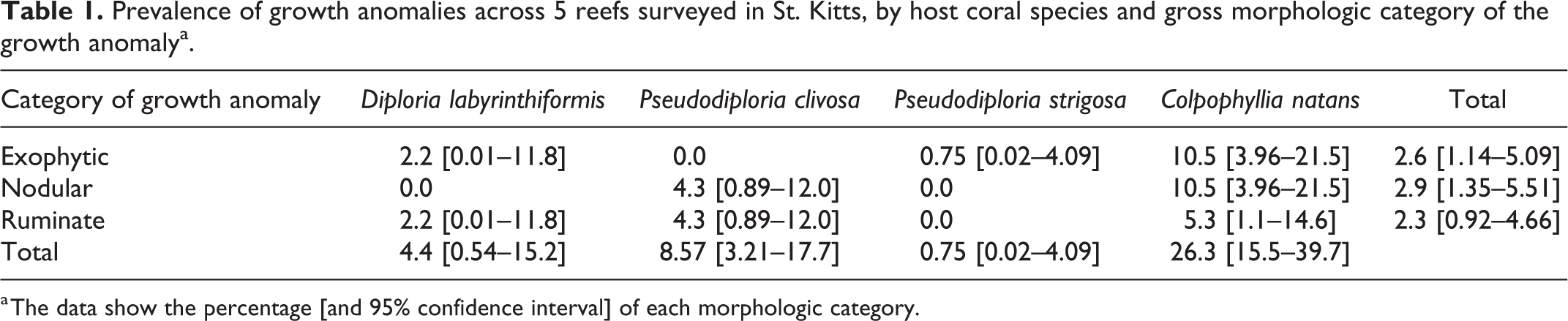
a The data show the percentage [and 95% confidence interval] of each morphologic category.
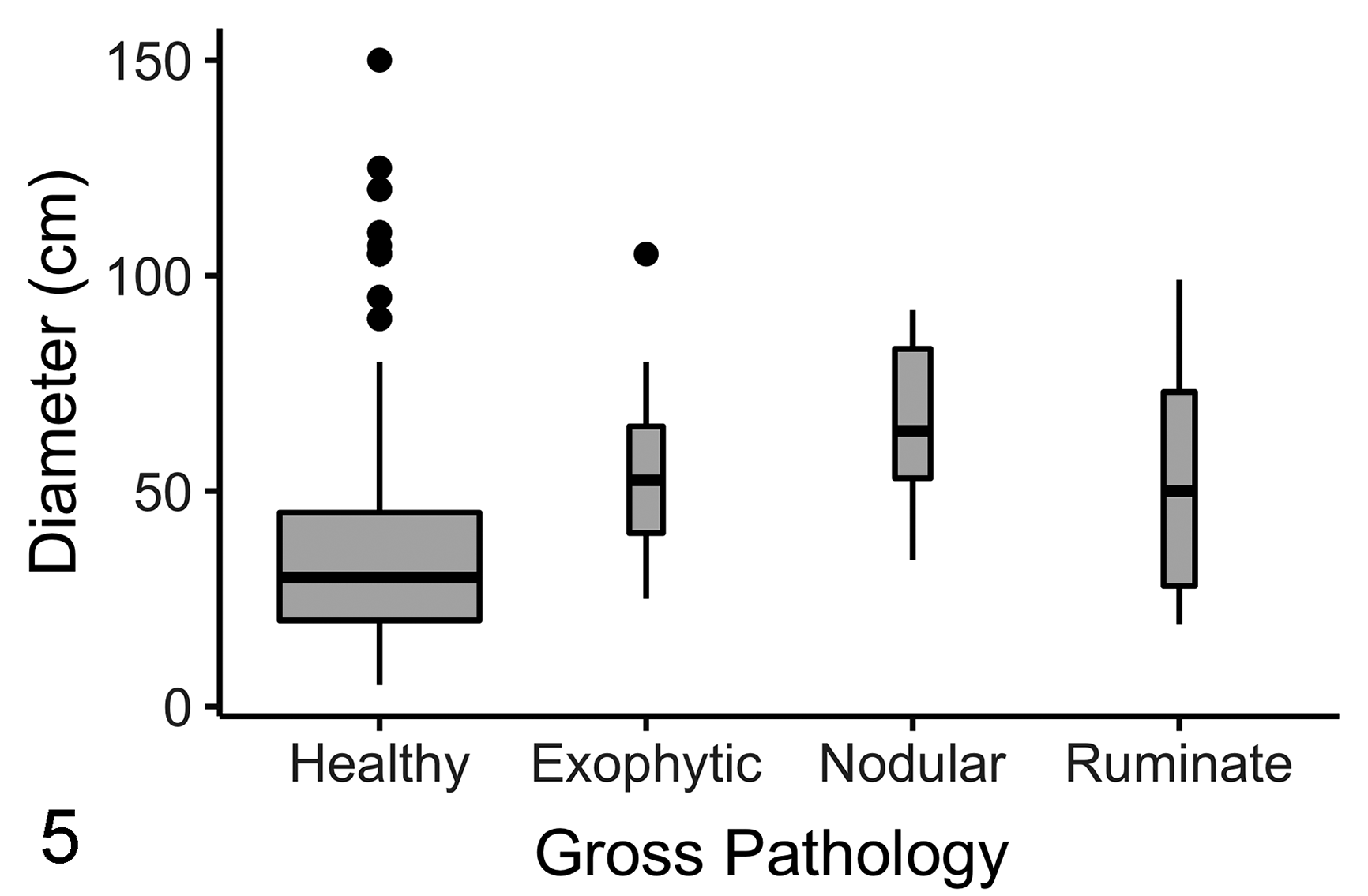
Planar coral colony size compared among apparently healthy faviid corals (n = 282), and faviid corals with ruminate (n = 7), exophytic (n = 8), and nodular (n = 9) growth anomalies surveyed across 5 reefs on St. Kitts, 2019. Horizontal lines indicate median, boxes indicate first to third quartiles, vertical lines indicate the minimum or maximum excluding outliers (dots), and box width indicates relative sample size.
Seventeen GAs from 15 corals were examined histologically across 3 different species over the course of the study, representing the 3 gross morphological types of GA (Table 2). Gonadogenesis was evident in 11 of these corals including 7 C. natans (of which 6 were histologically female, and 2 were male and female), and 3 P. strigosa (of which 2 were histologically male and female, and 1 was female).
Pathologic characterization of 17 growth anomalies (gas) in 3 species of coral from St Kitts.
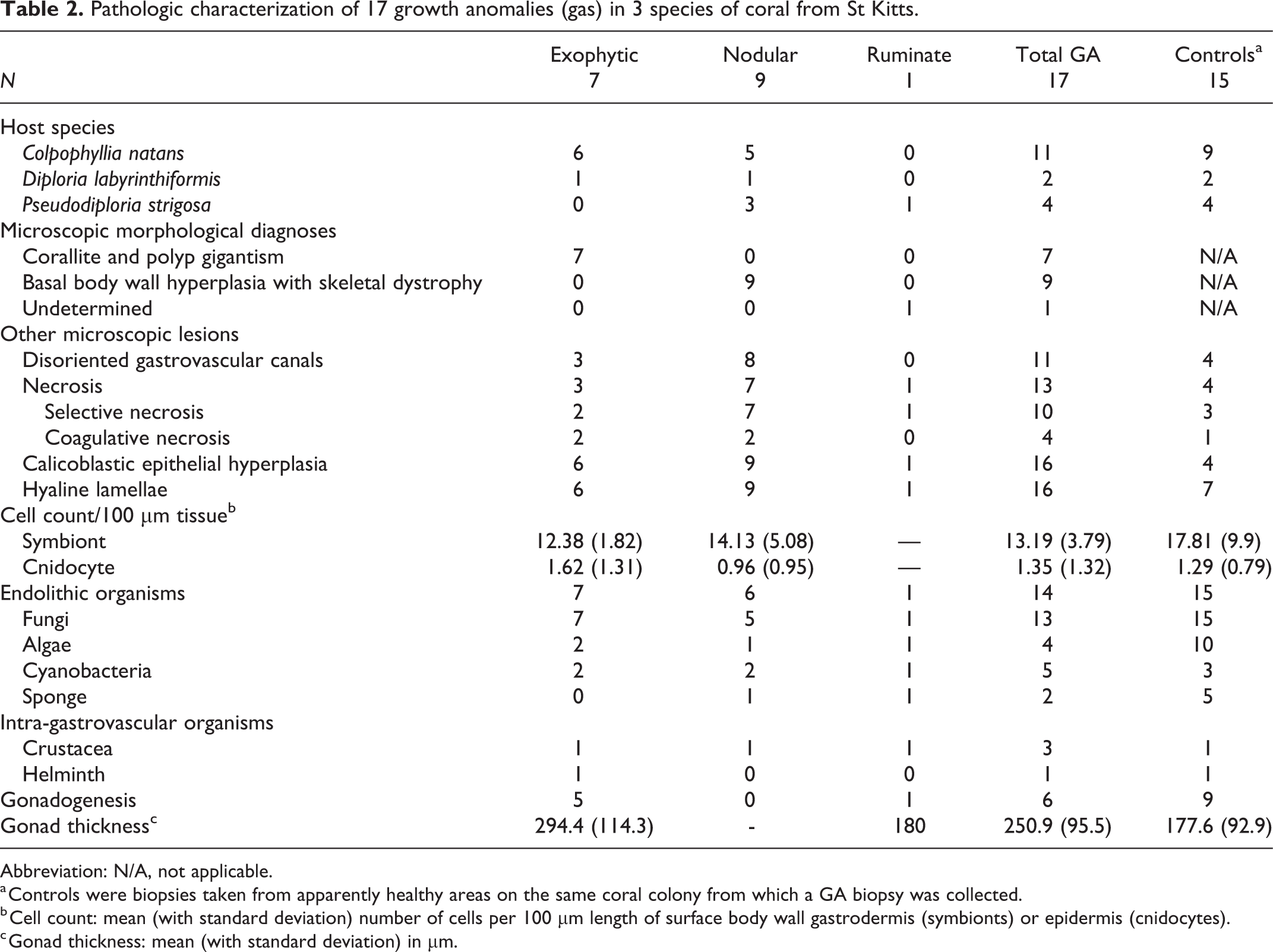
Abbreviation: N/A, not applicable.
a Controls were biopsies taken from apparently healthy areas on the same coral colony from which a GA biopsy was collected.
b Cell count: mean (with standard deviation) number of cells per 100 µm length of surface body wall gastrodermis (symbionts) or epidermis (cnidocytes).
c Gonad thickness: mean (with standard deviation) in μm.
Exophytic Growth Anomalies
Microscopically, all 7 exophytic GAs showed corallite and polyp gigantism, characterized by overall enlargement of the coral organism (Figs. 6–9). Polyp architecture and microanatomic structures were retained (including tentacles, actinopharynx, mesenteries, mesenterial filaments, and gonad). The cells comprising each tissue type of corallite and polyp gigantism were microscopically similar to those of apparently healthy biopsies. Gastrovascular canals were sometimes distorted, losing vertical orientation or seeming somewhat dilated, but this change was also often present in polyps from apparently healthy biopsies, suggesting artefactual distortion of tissues related to processing.
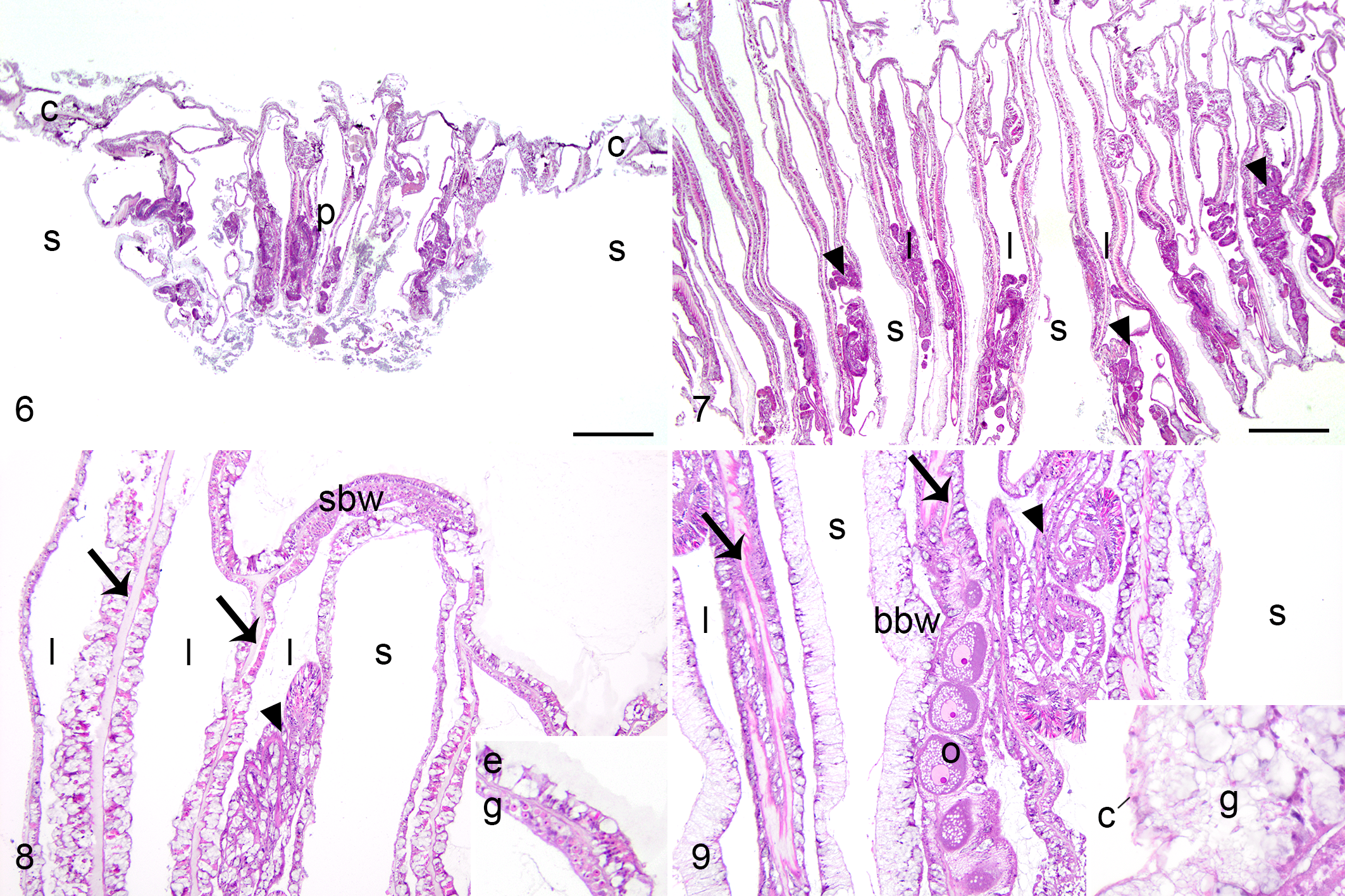
Normal, faviid coral (Colpophyllia natans). The polyp (p) is of normal size. Coenenchyme (c) and clear space representing decalcified skeleton (s) border the polyp. Bar = 1 mm.
Polyp depth was significantly greater in exophytic GAs (median: 5.5 mm) relative to apparently healthy areas (median: 3.5 mm; paired Wilcoxon rank sum test, P = .028).
Nodular Growth Anomalies
All 9 nodular GAs microscopically comprised basal body wall hyperplasia with skeletal dystrophy. These lesions were similar among coral species and consistently comprised nodular masses (Fig. 10), typically involving the edge of a polyp or coenenchyme, lined exteriorly by surface body wall, and consisting of jumbled micronodular deposits of skeleton, interspersed with calicoblastic epithelial hyperplasia, islands of mesoglea, and distorted gastrovascular canals of varying diameter and without vertical orientation (Figs. 11–13). Micronodular deposits of skeleton were unique to these GAs, and were sometimes apparent during the decalcification step of histology processing (n = 5), where tiny nodules of skeleton dissociated from the tissue. The calicodermis lining skeletal spaces was expanded by tall columnar calicoblastic epithelial cells (ie, calicoblastic epithelial hyperplasia; Figs. 11, 12), sometimes with fine eosinophilic apical cytoplasmic granules, and bordered amphophilic lamellar material (ie, hyaline lamellae). The surface of the nodules sometimes had segments of epidermis which invaginated and transitioned to gastrodermis (Fig. 14). Gastrodermis was distinguished from epidermis by apically located nuclei and the presence of intracellular Symbiodiniaceae. Spaces within in the mass were sometimes lined by segments of calicoblastic epithelial hyperplasia transitioning to gastrodermis (Fig. 15), resulting in apparent confluence of gastrovascular canal and skeletal space. Transitional gastrodermis was composed of tall cuboidal to columnar cells lacking symbionts. Small portions of mesenterial filaments were rarely within gastrovascular canals of these GAs, but otherwise they were consistently devoid of other polyp structures including gonad.
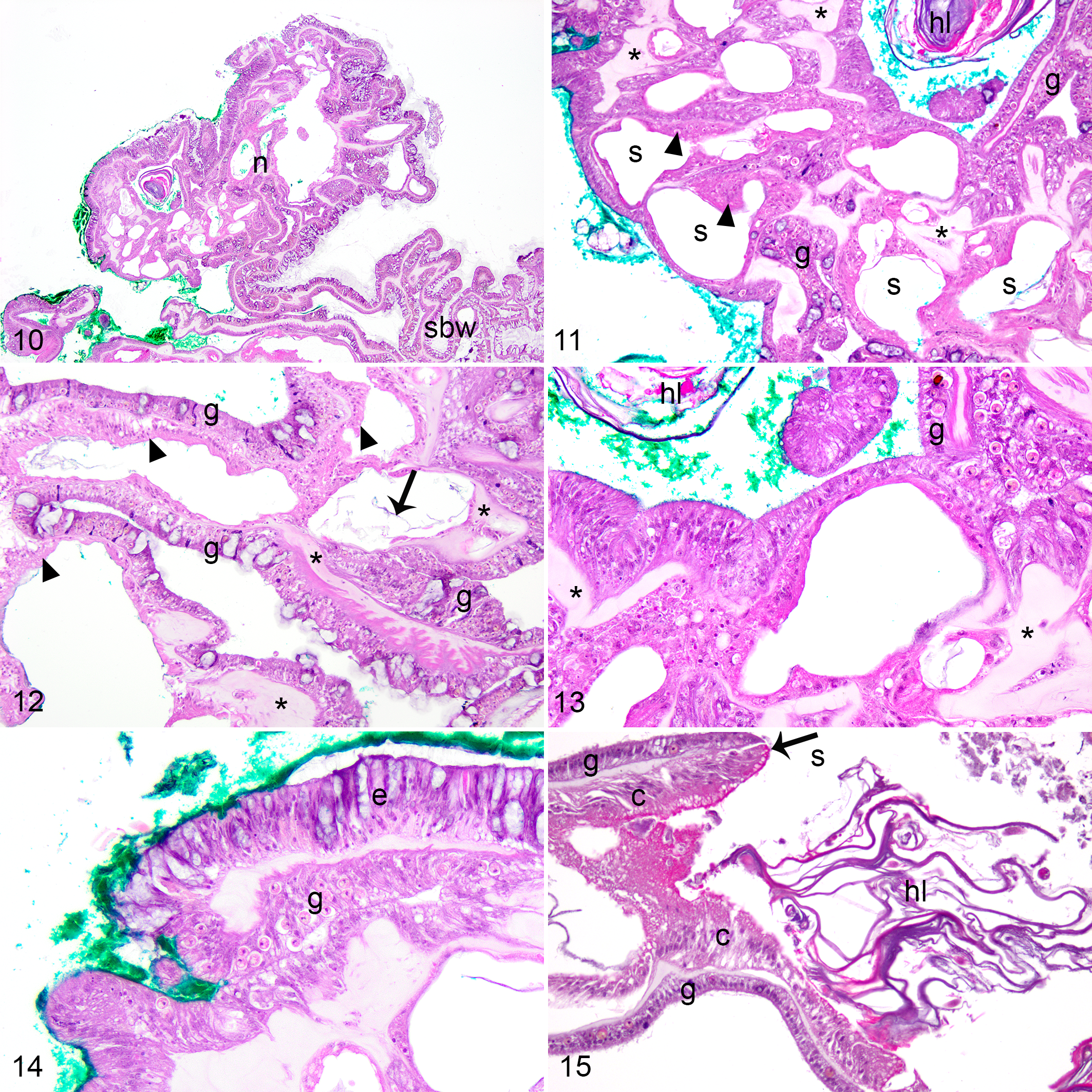
Nodular growth anomalies with basal body wall hyperplasia and skeletal dystrophy, faviid corals. Colpophyllia natans, Turtle Beach (Figs. 11–14); Pseudodiploria strigosa, Dieppe Bay (Fig 15). Green surgical ink was used to microscopically distinguish the growth anomaly. Hematoxylin and eosin.
Ruminate Growth Anomalies
Sampling of ruminate GAs was problematic due to their delicate nature where biopsies crumbled into numerous <5 mm fragments immediately after collection. Sampling was abandoned after multiple attempts failed to produce decent cores. Histology was performed on one sample, and extensive tissue fragmentation precluded a meaningful assessment of tissue architecture and provision of a microscopic morphological diagnosis. However, most anatomic structures were present in this sample, including tentacles, mesenteries, mesenterial filaments, and gonad. Gastrovascular canals were distended with mucus and selective necrosis (described below) of gastrodermis was severe.
Quantitative Microscopic Assessment
Necrosis of gastrodermis of gastrovascular canals (n = 11) and mesenterial filaments (n = 6) was commonly observed among biopsies (Table 2). Two morphological types of necrosis were observed. In coagulative necrosis (n = 5), regional areas of basal body wall and mesenterial filaments had loss of cell detail, cytoplasmic hypereosinophilia, and nuclear pyknosis (Figs. 16, 17). In selective necrosis (n = 13), clusters of gastrodermal epithelial cells and associated Symbiodiniaceae were detached and in rounded clusters within gastrovascular canals, with loss of distinction of cell borders, scattered karryorhectic or pyknotic debris, and cytoplasmic vacuolation (Figs. 18, 19). Both types of necrosis were seen in apparently healthy and GA tissues, and neither necrosis type was more commonly observed in GAs than in apparently healthy samples (Fisher’s exact test, P = .042), nor in nodular relative to exophytic GAs (Fisher’s exact test, P = .45). Severity of necrosis was not different among GAs (mild = 5, moderate = 5, severe = 1) relative to apparently healthy samples (mild = 4, moderate = 0, severe = 0; Mann-Whitney U test, W = 34, P = .083).
Calicoblastic epithelial hyperplasia consisted of segments of calicodermis lined by calicoblasts that were columnar rather than squamous, and pseudostratified, with eosinophilic granular apical cytoplasm, and often indistinctly vacuolated basal cytoplasm (Fig. 11). While this was a consistent and predominant component of nodular GAs, it was also observed in other GAs (exophytic n = 6; ruminate n = 1) and in apparently healthy samples (n = 4; Table 2). Hyaline lamellae (ie, eosinophilic to amphophilic laminar proteinaceous deposit at the skeletal interface) accompanied this change in all instances (Figs. 12, 13), although this change was also in one exophytic GA sample which did not show calicoblastic epithelial hyperplasia. Calicoblastic epithelial hyperplasia and hyaline lamellae were more commonly observed in GAs than apparently healthy samples (Fisher’s exact test, P = .0001 and P = .005, respectively). Calicoblastic epithelial hyperplasia was more severe in nodular (mild = 0, moderate = 0, severe = 8) than exophytic GAs (mild = 3, moderate = 2, severe = 1; Mann-Whitney U test, W = 52, P = .002). The severity of hyaline lamellae did not significantly differ among exophytic (mild = 3, moderate = 2, severe = 1) and nodular GAs (mild = 2, moderate = 3, severe = 3; Mann-Whitney U test, W = 16.5, P = .182).
Mean epidermal cnidocytes and Symbiodiniaceae per 100 µm surface body wall in exophytic (means = 1.62, 12.4, respectively) or nodular of GAs (means = 0.96, 14.1, respectively) were not significantly different than those in the paired apparently healthy areas (mean = 1.29 and 17.8, respectively; Wilcoxon signed-rank test, V = 53, P = 1, and V = 24, P = 0.08, respectively; Table 2).
Endolithic organisms were common among both GA and apparently healthy samples, including algae, fungus (Fig. 20), cyanobacteria and sponges (Table 2). Endolithic organisms were not more commonly present in GAs than in apparently healthy samples (Fisher’s exact test, P = .23), nor were they more severe (GAs: mild = 2, moderate = 6, severe = 6; apparently healthy: mild = 4, moderate = 6, severe = 4; Mann-Whitney U test, W = 131, P = .234). Microscopically, endolithic organisms were observed within foci of hyaline lamellae (21/24, 88%), calicoblastic epithelial hyperplasia (15/20, 75%), and necrosis (6/15, 40%). Crustacea, potentially endosymbiont copepods of the Family Asterocheridae, 34 were in the actinopharynx and tightly embedded within the gastrovascular canals of both apparently healthy tissues and GAs of a P. strigosa and within a GA of a C. natans from Major’s Bay (Fig. 21).
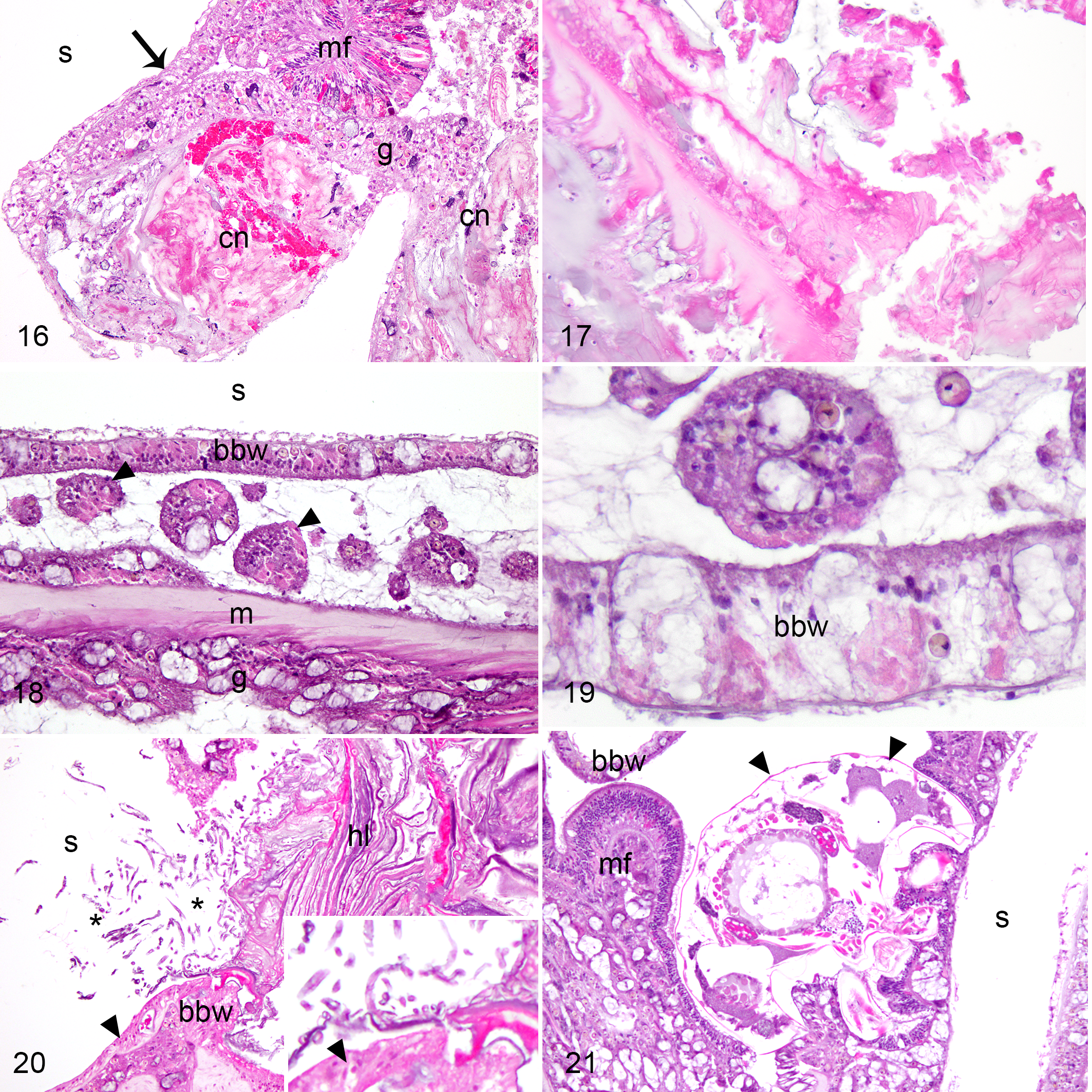
Growth anomalies, faviid corals. Hematoxylin and eosin.
Discussion
This study was the first to comprehensively investigate the pathology of growth anomalies in several species of Caribbean corals from the family Faviidae. Two distinct microscopic lesions were identified and could be predicted by gross lesion morphology: exophytic GAs were a reflection of corallite and polyp gigantism and nodular GAs were basal body wall hyperplasia with skeletal dystrophy. In no instance was the cause of a GA microscopically apparent, suggesting that an infectious cause is unlikely unless involving agents not apparent with light microscopy, such as viruses. However, the fact that such diverse microscopic lesions underlie GAs suggests divergent pathogenesis and emphasizes the need for research investigating GA etiology and epidemiology to accurately categorize lesions according to microscopic pathology.
Exophytic GAs affected around 2.6% (Table 1) of surveyed corals, including C. natans, D. labyrinthiformis, and P. clivosa. Microscopically, gigantism consisted of enlargement of the coral organism, without hyperplastic or neoplastic proliferation of the coral’s cells. Enlargement was evident from microscopy and measurements of polyp depth compared among GA and control biopsies. The only other histological descriptions of GAs in D. labyrinthiformis and P. strigosa similarly indicate enlarged corallites and polyps, 31,42 and earlier gross descriptions of GAs in these species appear synonymous with the exophytic morphological type of GA. 4,37 Together these findings suggest gigantism and corresponding exophytic GAs are not limited to faviids on St. Kitts. Moreover, GAs of Agaricia lamarcki have also been attributed to gigantism, 31 and those of Porites compressa and Platygyra spp. were similarly described as enlarged, hypertrophic, and hyperplastic polyps, 14,25 suggesting that gigantism may be a microscopic basis for GAs shared among several coral species. Exophytic GAs of acroporids microscopically do not consist of corallite and polyp gigantism, 31,41,43 indicating that the gross appearance of a GA may only be predictive of microscopic abnormalities on a species-specific basis.
Gigantism in corals appears to comprise a developmental anomaly because it occurs in a localized cluster of polyps, presumptively reflecting a somatic genetic alteration passed on to progeny polyps via asexual reproduction (polyp budding). While anatomy was not convincingly altered in histological sections (limited to disoriented gastrovascular canals), it may be that other anatomic malformations accompany gigantism and additional techniques such as macroscopic skeletal examination, corallite scanning electron microscopy, or skeletal radiography, 3,14,17,25,44,45 would provide a more accurate assessment for anomalous 3-dimensional architecture. The fact that GAs with enlarged polyps in P. compressa have reduced gonad development supports a pathological state. 14 Reproductive effects were not obvious in corallite and polyp gigantism on St. Kitts as gonadogenesis was evident within most instances of gigantism (5/7, 71%). Polyploid gigantism occurs in certain marine invertebrates, best known for its advantages in aquaculture of bivalves. 21 To the authors’ knowledge, no studies have yet cytogenetically assessed gigantism in corals, including evaluation of chromosome number.
Nodular GAs affected around 2.9% of corals (Table 1), observed in C. natans, D. labyrinthiformis, P. clivosa, and P. strigosa. These lesions microscopically were composed of a basal body wall proliferation, similar to other histological descriptions of GAs. 9,11,31,41,43 In contrast to basal body wall proliferations in acroporids, the architecture of gastrovascular canals was substantially distorted. We interpreted this lesion as a cellular adaptation (ie, controlled proliferation) rather than neoplasia. Thus, we used the morphological diagnosis of basal body wall hyperplasia with skeletal dystrophy, to reflect the abnormal growth and micronodular deposits of skeleton within these lesions. The transitions observed among epidermis, gastrodermis, and calicodermis (suspicious for metaplasia) also are in contrast to basal body wall proliferations of acroporids. Similar transition from calicodermis to epidermis occurs in wound repair in Montipora capitata, suggesting that nodular GAs in faviids may represent an aberrant healing attempt. 40 The consistent presence and predominance of necrosis in nodular GAs, and its association with hyaline lamellae (protein deposition from calicoblasts), further supports a response to injury. Nodular GAs are grossly consistent with what have been termed “gemmae” in D. labyrinthiformis, P. clivosa, and P. strigosa. 35 While thought to initially form at sites of injury, these nodules may separate from the parent colony and reattach to benthic substrate, and have been considered an unusual form of asexual reproduction albeit with low overall survival.
Neoplasia was discounted as a morphological diagnosis in nodular GAs because the size was restricted to <2 cm, there were multiple tissue types comprising the lesion (calicodermis, gastrodermis, and epidermis), the well-differentiated nature of each tissue type, and the lack of a single monomorphic “transformed” cell type, cellular anaplasia, and mitotic activity. These absent features have not been convincingly demonstrated in any GA study to date. However, it is possible that neoplasia in invertebrate taxa may not conform to the diagnostic criteria used in vertebrates. 29,31 Apart from disseminated neoplasia of presumed hemic origin in bivalves, malignant neoplasia has not been well documented in invertebrates, 30 thus it is unclear if cellular criteria of malignancy used for vertebrates are applicable in these taxa. The concept of “collateral hyperplasia” has been used to explain the presence of more than one tissue lineage within coral GAs. 31 Mitosis in invertebrates may be controlled hormonally or diurnally, with peak activity in corals occurring at midnight. 18 Thus, mitotic figures may be infrequent in biopsies collected during the daytime. From this regard, methods other than morphological examination could be beneficial for substantiating neoplasia among coral GAs, including longitudinal observations of GA growth and spread, cytogenetic analyses, or molecular clonality assessment of tumor cells. 22
Surveys showed GAs to be an uncommon lesion (7.8%) of St. Kitts faviids, similar to observations in other coral species. 1,8,41 This observed prevalence contrasted greatly to an estimated overall prevalence of 0% taken from a meta-study of Caribbean GA reports, 42 and were perhaps underreported in the past due to their morphological plasticity or cryptic nature. Large colony size is associated with the presence of GA in Pacific corals, 8,25 and median colony diameter was larger in GA-affected relative to nonaffected faviids on St. Kitts. Association with size may reflect coral age, where larger corals are more likely to have accrued somatic genetic alterations or be exposed to etiologies or environmental factors that may influence GA formation over time. GAs were most common at Dieppe Bay (20.3%) relative to other study sites, a finding which is likely biased by the predominance of C. natans and large colony size on that reef (data not shown).
Necrosis seems to be a common feature of GAs. 41 –43 We observed 2 types of necrosis in St. Kitts’ GAs: coagulative necrosis and a morphologic form that did not conform to conventional classifications of necrosis for vertebrate animals, 12 which we designated as selective necrosis based on the prior use of this term for a similar form of necrosis observed in acroporid GAs. 41 Selective necrosis was distinguished by the sloughing of cohesive sheets of cells, which were rounded up and showing features of degeneration, whereas tissue organization was largely retained in coagulative necrosis. A morphologically similar pattern, referred to as necrotic cell spherules, also occurs at tissue loss margins in A. cervicornis and A. palmata (E. Peters, personal communication). 27,28 The patterns of necrosis and the corresponding categories of causal mechanisms have not been well studied in invertebrates, 13 and observations of unconventional morphologies of necrosis in corals emphasize the need for basic research that addresses their fundamental and morphological responses to injury. The rounded up rafts of cells resemble those observed in collagenase-dissociated tissue preparations of Pocillopora damicornis. 24 Those “tissue balls” were metabolically active and showed autophagy; therefore, the potential role of this selective form of necrosis in facilitating tissue regeneration deserves further investigation.
Endolithic organisms are also a common feature of GAs, 11,14,31,41,43 and it is tempting to consider them as a potential cause of GAs because some fungal compounds are known to influence cell growth. 42 Endolithic organisms were abundant within all biopsies on St. Kitts, regardless of GA status. However, their presence in control samples may have been biased by sampling at the colony’s margin where benthic organisms may easily access the coral skeleton. Moreover, endolithic fungi are common among hard corals and their pathogenicity is debated. 5,23 Evaluation of a greater number of GAs and control biopsies sampled from nonmarginal areas of the colony, using a less subjective approach than histology for characterizing endolithic community composition and quantity, is needed to clarify the potential etiological role of endolithic organisms.
In conclusion, this study helps fill a knowledge deficit regarding the microscopic morphology of coral GAs. Future studies are required to confirm whether the morphological trends observed in GAs of St. Kitts’ faviids hold true across a larger geographical area and broader representation of each faviid species. However, 2 contrasting themes of pathology that underlie GAs are increasingly apparent: enlargement of the polyp and corallite (ie, gigantism), and various proliferations of basal body wall. Whether those GAs of the latter category represent a cellular adaptation or neoplasia is yet to be unequivocally determined, but the present study could not confirm a neoplastic basis for GAs.
Footnotes
Acknowledgements
Coral biopsies were collected under permit from St. Kitts Department of Marine Resources. Kathleen Gilbert and Nicole Atherley assisted with field surveys and coral sampling. David Hilchie assisted with histological processing. This work was supported by an intramural grant from Ross University School of Veterinary Medicine, Center for Conservation Medicine and Ecosystem Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Center for Conservation Medicine and Ecosystem Health, Ross University School of Veterinary Medicine, St. Kitts, West Indies.
