Abstract
Melanoma in humans and dogs is considered highly immunogenic; however, the function of tumor-infiltrating lymphocytes (TILs) is often suppressed in the tumor microenvironment. In humans, current immunotherapies target checkpoint molecules (such as PD-L1, expressed by tumor cells), inhibiting their suppressive effect over TILs. The role of PD-L2, an alternative PD-1 ligand also overexpressed in malignant tumors and in patients with anti-PD-L1 resistance, remains poorly understood. In the current study, we evaluated the expression of checkpoint molecule mRNAs in canine melanoma and TILs. Analysis of checkpoint molecule gene expression was performed by RT-qPCR (real-time quantitative polymerase chain reaction) using total RNA isolated from formalin-fixed and paraffin-embedded melanomas (n = 22) and melanocytomas (n = 9) from the Virginia Tech Animal Laboratory Services archives. Analysis of checkpoint molecule expression revealed significantly higher levels of PDCD1 (PD-1) and CD274 (PD-L1) mRNAs and an upward trend in PDCD1LG2 (PD-L2) mRNA in melanomas relative to melanocytomas. Immunohistochemistry revealed markedly increased numbers of CD3+ T cells in the highest PD-1-expressing subgroup of melanomas compared to the lowest PD-1 expressors, whereas densities of IBA1+ cells (macrophages) were similar in both groups. CD79a+ cell numbers were low for both groups. As in human melanoma, overexpression of the PD-1/PD-L1/PD-L2 axis is a common feature of canine melanoma. High expression of PD-1 and PD-L1 correlates with increased numbers of CD3+ cells. Additionally, the high level of IBA1+ cells in melanomas with low PD-1 expression and low CD3+ cells levels suggest that the expression of checkpoint molecules is modulated by interactions between T cells and cancer cells rather than histiocytes.
Keywords
In humans, melanoma is considered the deadliest type of skin cancer. More than 600 000 patients in the United States were recorded as living with melanoma in 2017. 26 Mutations in the RAF and RAS families of oncogenes are detected in high frequency in human melanomas. 5,30 Similarly, canine melanoma is also very aggressive, with median survival time of 22 months after diagnosis for cutaneous melanoma, 5 months for oral melanoma, and <3 months for stage 3 (metastatic) disease. 25 However, in contrast with humans where point mutations in the BRAF family are associated with 50% of the cases, this mutation appears to be rare in canine melanoma 24,38 (Stevenson, Huckle, LeRoith, unpublished data), and few convincing driver mutations have been described for canine melanomas. 10,13,34 Because of this, the dog has been proposed as an attractive animal model to study the oncogenic mechanisms in human triple wild-type (lacking any of the known driver mutations in BRAF, RAS, or NF1) and mucosal melanomas, which comprise almost 40% of the total human melanoma cases. 13,15,35,44
While increases in melanoma diagnoses occur worldwide every year, 2 great progress has been made in the understanding of the mechanisms of the disease, leading to the development of new therapies for human melanoma and other cancers. 12,31 Although melanoma is considered an immunogenic tumor, the function of tumor-infiltrating lymphocytes (TILs) is often suppressed in the tumor microenvironment (TME), failing to control and modulate tumor invasion and leading to escape from immune surveillance. 11,27 Lymphocytes in patients with cancer often experience exhaustion resulting in overexpression of PD-1 co-inhibitory receptor (encoded by PDCD1); moreover, cancer cells frequently overexpress a PD-1 ligand (PD-L1, encoded by CD274), inducing immunosuppression upon their binding to TILs. 23,28,48 The ability of tumor-expressed PD-L1 to inhibit tumor-infiltrating lymphocytes expressing PD-1 is considered a major mechanism by which numerous types of neoplasms achieve immune tolerance. 12,31 Additionally, expression of the PD-1 alternative ligand PD-L2 (encoded by PDCD1LG2) has been detected in numerous tumors, although reports vary as to its correlation with expression of PD-L1. 3,44,48 Interesting, few reports have associated the expression of PD-L2 with cancer infiltration and metastasis. 14,47
Therapeutic use of monoclonal antibodies against PD-1 and PD-L1 for humans has been remarkably effective at reversing immune escape by tumors and increasing patients’ median overall survival time. 1,8 Interestingly, patients that developed resistance to anti-PD-L1 therapy had overexpression of PD-L2, 39,41 and at the same time the presence of TILs has been associated with a better outcome and prolonged survival time. 8
As yet, no effective treatments have been developed for canine melanoma. A DNA vaccine, Oncept, approved by the Food and Drug Administration for canine oral melanoma, has shown mixed results in survival times 4,32,42 and is unable to maintain antitumor efficacy, perhaps owing to the expression of immunosuppressing checkpoint molecules on vaccine-activated lymphocytes. 4,16,32 As in humans, detection of overexpression of PD-1 by TILs and PD-L1 from malignant tumors representing different types of canine cancers has been reported. 19,21 However, only one small pilot clinical study has demonstrated objective antitumor response for oral melanoma targeting PD-L1. 22
A rationale for extension of advances in human melanoma immunotherapy to dogs requires a deeper comparative understanding of disease mechanisms across species, including characterization of the role of tumor-expressed checkpoint molecules in suppressing host immune response. Accordingly, the objectives of the research reported here are to determine the expression of PD-1, PD-L1, and PD-L2 mRNAs in canine melanoma and to evaluate the association of TILs with the expression of these immune modulators.
Materials and Methods
Sample Identification
Thirty-one formalin-fixed and paraffin-embedded specimens from biopsies with the diagnosis of nondigital cutaneous (6 cases) or oral melanoma (16 cases) were retrieved from the Virginia Tech Animal Laboratory Services (ViTALS) archives. Melanocytomas (9 cutaneous cases) were used as a control group. These tumor biopsies were from different privately owned dogs, submitted for routine histopathological evaluation. Cases were dated from 2017 to 2019. Diagnostic criteria for melanomas and melanocytomas included mitotic rate and tissue infiltration, as previously described. 37 Briefly, those tumors with a mitotic index ≥3 for cutaneous neoplasms and ≥4 for oral neoplasms, cytological features such as large nucleoli and atypical mitoses, and the presence of vascular or lymphatic invasion in hematoxylin and eosin–stained slides were considered to be melanomas. 36 Two amelanotic cases were confirmed subsequently by the presence of melanin in biopsies of recurrent tumors. Lymphovascular invasion was assessed morphologically in tissue sections and by clinical staging where provided. No oral melanocytomas were identified.
Real-Time Quantitative Polymerase Chain Reaction (RT-qPCR)
Four to 6 scrolls of 10 µm thickness each were cut serially from paraffin blocks of all cases. RNA was extracted using the QIAamp RNA FFPE tissue kit (Qiagen), and the RNA concentration and purity were estimated by UV spectrophotometry. Only isolates with RNA concentrations greater than 100 ng/µl were used for this study. Random-primed cDNA was synthesized from 1 µg total RNA using the High Capacity cDNA kit (Life Technologies) and stored at −20 °C.
RT-qPCR primers and TaqMan exon junction-spanning probe sets targeting canine PD-L1 (NM_001291972; sense primer: GGTGCTGACTACAAGCGGATTA; antisense primer: GTGACAGGATCCACAGAAATTCTTT; probe: FAM-TGAAAGTTCATGCCCCGTAC-MGBNFQ) and PD-L2 (XM_847012; sense primer: CGCCTGGGACTACAAATATCTGA; antisense primer: GATCCTGAGGAAATGAGTGTTTATTTT; probe: FAM-TGAAAGTCAAAGCTTCCTACAA-MGBNFQ) were designed using PrimerExpress software (Applied Biosystems) and validated for amplification efficiencies of >95%. Prevalidated primers and probe sets for canine PD-1 and the internal reference marker 18S rRNA were obtained from Thermo Fisher (cat. nos. Cf04947484_m1 and 4333760F, respectively).
All reactions were run in triplicate on an Applied Biosystems 7500 thermocycler using the Fast Real-Time PCR program, consisting of an initial incubation of 20 seconds at 95 °C, followed by 40 cycles of denaturation (3 seconds at 95 °C) and annealing/extension (30 seconds at 60 °C). Triplicate Ct values were averaged and used to determine the relative gene expression of PD-1, PD-L1, and PD-L2 by the comparative Ct method. 18 For each sample, mean Ct values for each target were first normalized internally to 18S rRNA expression (yielding ▵Ct), then compared to the same target’s mean normalized expression in melanocytoma controls (yielding ▵▵Ct). For display, results were calculated as a fold change relative to mean expression in melanocytoma (2-▵▵Ct).
Immunohistochemistry
Immunohistochemistry was performed on 10 melanoma cases using antibodies against CD79a, CD3, and IBA1. Cases were selected in 2 groups according to the relative expression of PD-1 detected by RT-qPCR analysis: the 5 cases with the lowest and the 5 cases with the highest relative expression of PD-1. Each of these IHC groups contained 3 cases of oral melanoma and 2 cases of cutaneous melanoma. Sections of 5-µm thickness from each formalin-fixed paraffin-embedded tissue block were deparaffinized and stained for each cell marker. Monoclonal antibodies recognizing CD79a (Biocare Medical clone HM47/A9; 1:250) and CD3 (Dako clone A0452; 1:100) and polyclonal antibodies recognizing IBA1 (FUJIFILM Wako #019-19741; 1:1000) were used at the dilutions indicated. Universal Alkaline Phosphatase Red Detection kit (Ventana UltraView) was used as secondary antibody, and visualization of antibody was obtained with Fast Red chromogen incubation. Slides were processed on an automated Ventana Benchmark XT using standard and previously-validated protocols at Virginia Tech Animal Laboratory Services, an AAVLD-accredited veterinary diagnostic laboratory. Positive controls for each cell marker were tissues from colon and lymph nodes. Sections of melanoma tissue stained without addition of primary antibody served as negative controls. Hematoxylin was used as a counterstain.
Blinded assessment and characterization of the 10 cases were performed by a single board-certified veterinary pathologist estimating the percentage of positive cells infiltrating the tumor averaged over 10 high-power field (HPF; 2.37 mm2).
Statistical Analysis
Normalized expression of individual checkpoint molecules (as ▵Ct) in melanocytoma and melanoma samples were compared by 2-tailed Student’s t test using SAS 9.4, after confirming normality of distribution. The relationships between expressions levels of paired checkpoint molecules (eg, PD-L1 vs PD-1) within each tumor type were examined by least-squares linear regression analysis of normalized (▵Ct) values and analysis of covariance of curve slopes. Mitotic indices and the densities of CD3+, IBA1+, and CD79a+ cells in stained sections of low versus high PD-1-expressing cases were compared by 2-tailed Student’s t test. In all cases, P values <.05 were considered statistically significant. The data analyzed in this study are available upon request to the corresponding author.
Results
Checkpoint Molecule mRNA Expression in Melanoma Relative to Melanocytoma
RT-qPCR analysis was performed targeting PD-1, PD-L1, and PD-L2 using total RNA extracted from 31 formalin-fixed, paraffin embedded melanoma (n = 22) and melanocytoma (n = 9) tissue blocks. Results for each specimen are expressed as checkpoint molecule mRNA normalized internally to 18S rRNA and relative to mean normalized expression in melanocytoma (2-ΔΔCt; Figs. 1 –3). Measured expression of all 3 checkpoint molecules was higher in melanomas relative to melanocytomas; based on 18S rRNA-normalized (ΔCt) values for each marker, the expression differences reached statistical significance for PD-1 (P = .0012) and PD-L1 (P = .0043) but not for PD-L2 (P = .0633, 2-tailed Student’s t test).
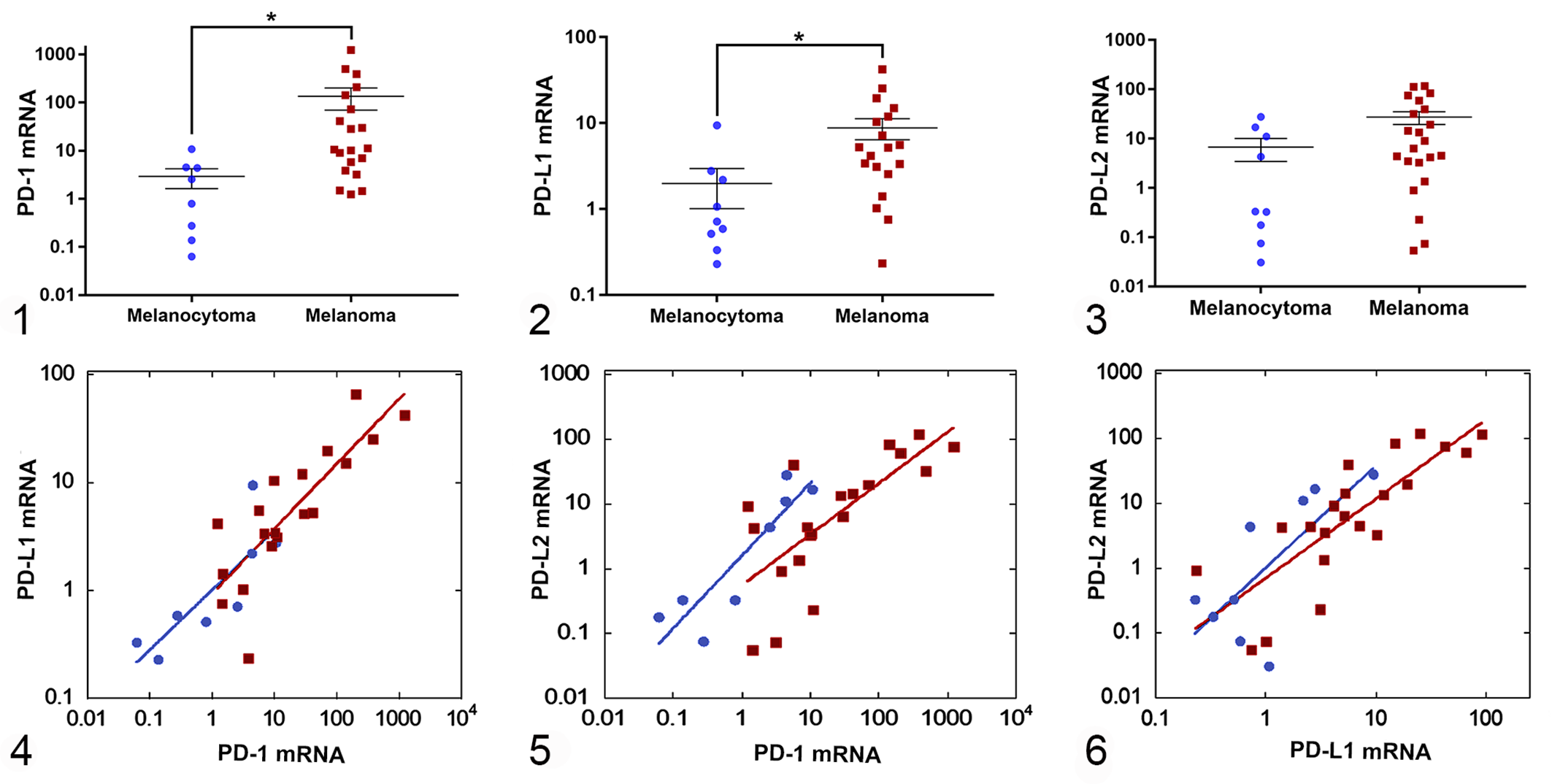
Expression of PD-1, PD-L1, and PD-L2 mRNAs in canine melanoma and melanocytoma. Each symbol represents checkpoint molecule mRNA expression in an individual melanoma or melanocytoma case, shown as a ratio (2-▵▵Ct) to the mean normalized expression in melanocytoma.
To ascertain whether elevation of expression of multiple checkpoint molecules occurred in the same samples, plots of PD-L1 versus PD-1, PD-L2 versus PD-1, and PD-L2 versus PD-L1 normalized expression were prepared (Figs. 4 –6). The slopes of all curves were nonzero for both melanocytoma and melanoma (analysis of covariance P < .05), indicative of positive relationships between expression of the paired checkpoint molecules. For example, relatively higher expression of PD-1 was associated with relatively higher expression of PD-L1 in the same sample. While this relationship was observed in all of the comparisons for both the melanocytoma and melanoma groups, slopes of the regression lines did not differ between the 2 tumor types.
Immunohistochemistry
In order to examine the potential for contribution by infiltrating lymphocytes to the observed PD-1 expression in melanoma, immunohistochemistry was performed to identify T cells (CD3+), macrophages and monocytes (IBA1+) and B cells (CD79a+; Figs. 7, 8). This analysis focused on 2 subgroups of the melanoma samples: the 5 with the highest observed relative PD-1 expression and the 5 with the lowest expression (Fig. 1). Although these groups differed markedly in PD-1 expression, they were similar in diagnostic features, with the low PD-1 expressors showing a numerically higher mean mitotic index that was not statistically significant (Fig. 9). Each group consisted of 3 oral and 2 cutaneous melanomas.
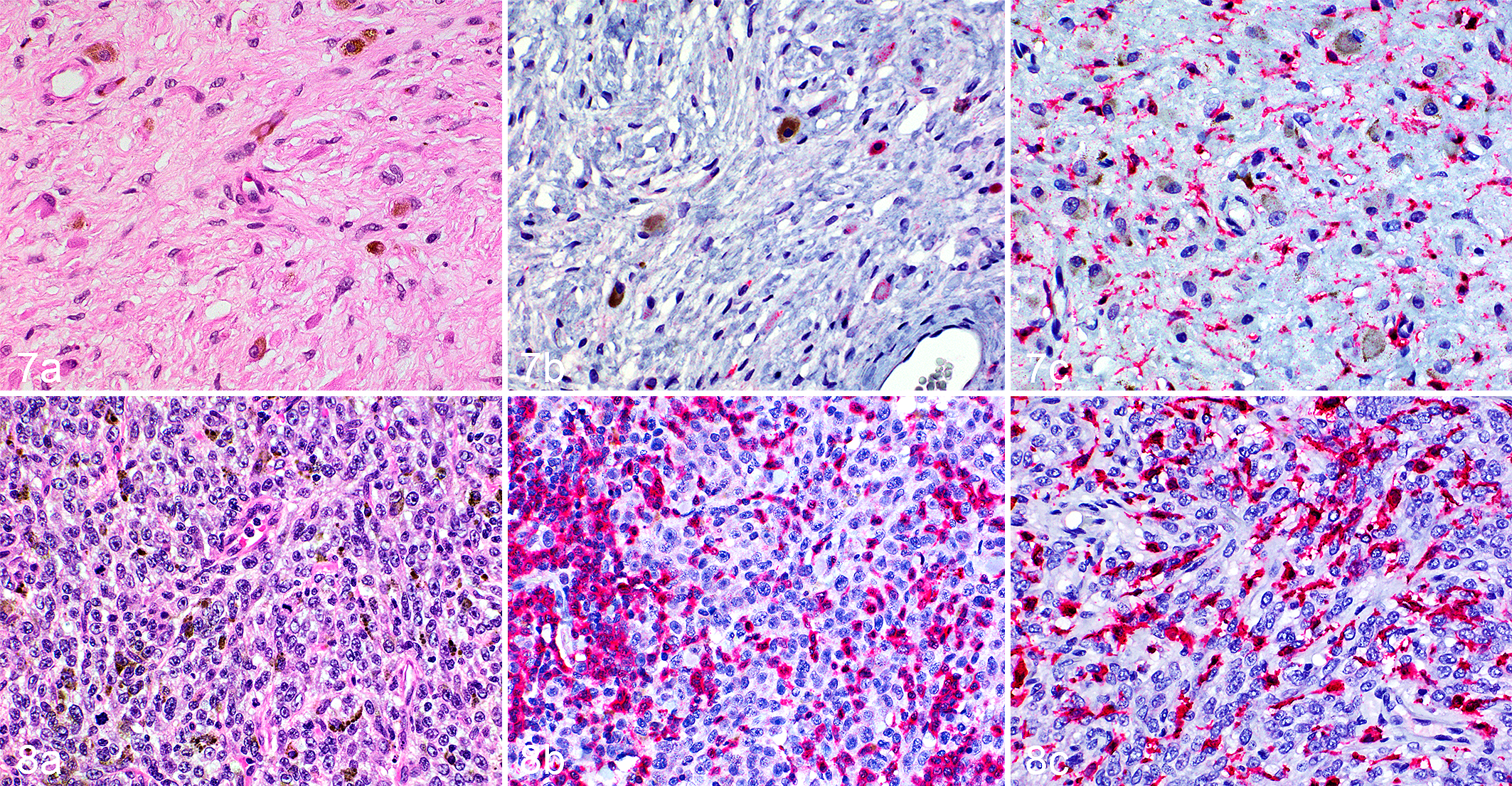
Oral melanoma, dog. Compared to a tumor with low PD-1 expression (Fig. 7), a tumor with high PD-1 expression (Fig. 8) has greater cellularity (a, hematoxylin and eosin), greater abundance of immunoreactivity for CD3 (b), and comparable immunoreactivity for IBA1 (c).
Infiltration of the respective immune cell types in high and low PD-1 expressors was estimated by calculating the number of cells positive for each marker as a percentage of total cell numbers, averaged over 10 HPFs (Figs. 10 –12). Markedly higher relative abundance of CD3+ cells was observed in the group with higher PD-1 expression when compared with the group with lower expression (38.0 ± 11.5% vs 6.2 ± 2.3%, mean ± SEM; P = .026; Fig. 10), whereas infiltration of IBA1+ cells showed no evident relationship with PD-1 expression (45.0 ± 8.1% vs 35.0 ± 3.2%; P = .28; Fig. 11). Although there was a small numerical difference observed between in mean abundance of CD79a+ cells in high- versus low PD-1 expressors (4.8 ± 1.5% vs 1.8 ± 0.2%), these cells were sparse in both groups and comparison of their means did not indicate a statistically significant difference (P = .077; Fig. 12).
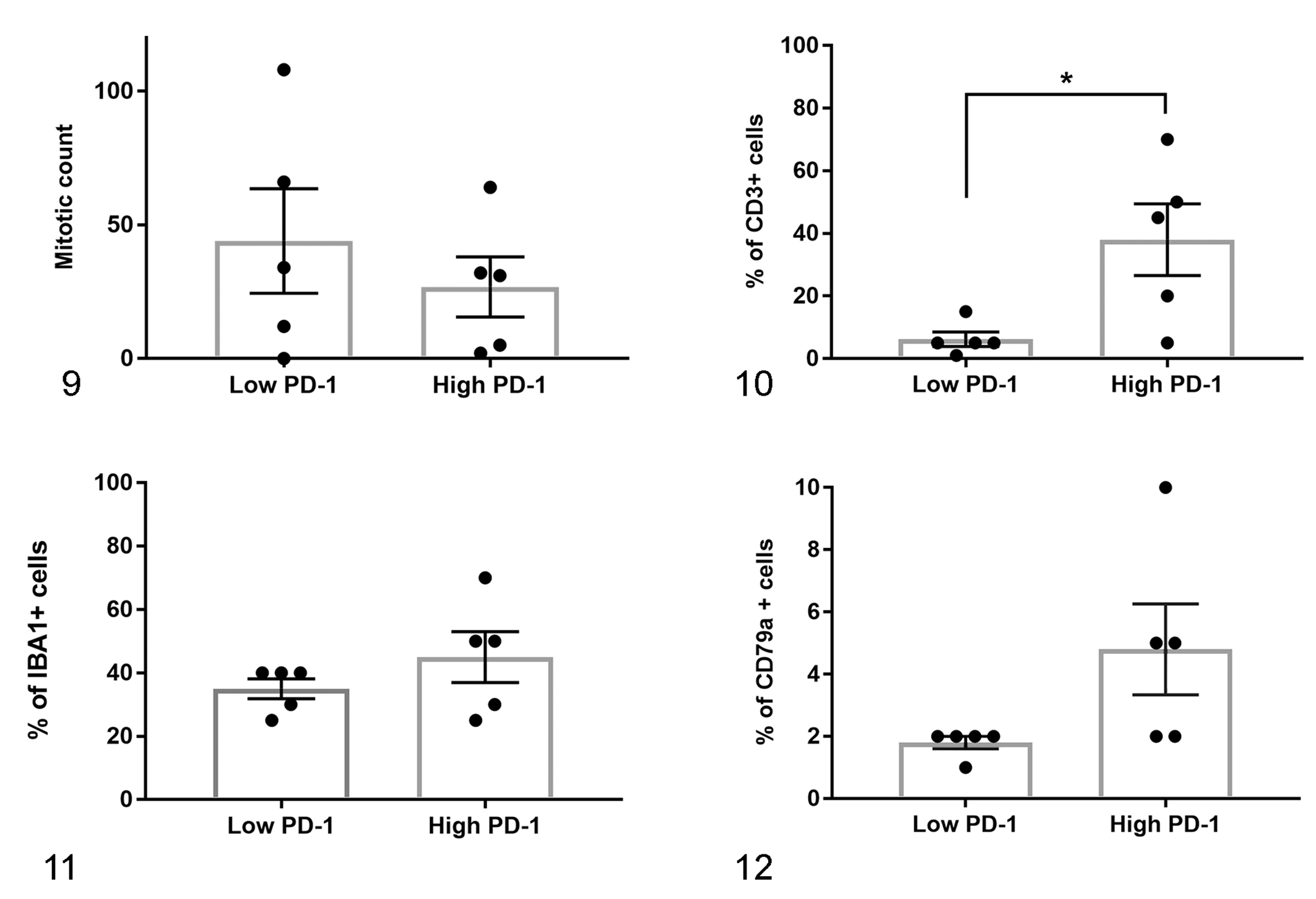
Quantification of mitotic figures and immunolabeling for CD3 (T cell marker), IBA1 (macrophage marker), and CD79a (B cell marker) in melanomas with high and low expression of PD-1 mRNA (“Low PD-1” and “High PD-1”) (means ± SEM). A significantly higher percentage of CD3+ cells infiltrating melanomas was observed in the high PD-1 group (P = .026; Fig. 10), whereas percentages of infiltrating IBA1+ cells were similar in the low and high PD-1 melanoma groups (P = .28; Fig. 11). Percentages of CD79a+ cells were low in both PD-1 high and low groups (P = .077; Fig. 12).
Discussion
In this study, we evaluated the expression of mRNAs encoding molecules of the PD axis, specifically PD-1, PD-L1, and PD-L2, in canine melanoma. Expression of these markers in melanoma was compared to that in melanocytoma, a benign neoplasm derived from melanocytes. Our study of checkpoint molecule expression was limited to analysis of mRNAs extracted from FFPE tissues for several reasons, the principal one being our ability to devise suitably sensitive and quantitative assays based on knowledge of the relevant mRNA sequences. While it would be desirable to confirm expression changes at the protein or cell-type level, approaches such as anti-checkpoint molecule IHC or in situ hybridization were outside the scope of this study. Moreover, validated anti-canine checkpoint molecule antibodies are not available that would allow us to adequately interpret an IHC-based analysis of FFPE tissues. However, other investigators have reported a correspondence between transcript and protein for these targets in tumor tissue. 9,17 We chose melanocytoma as a control tissue for comparison of checkpoint molecule expression owing to its shared melanocytic origin, but markedly different disease outcome, relative to melanoma. Since we wish to examine the relationship between checkpoint molecule expression and disease progression, we maintain that melanocytoma is a more appropriate control tissue than normal skin for our study.
Higher mean levels of PD-1, PD-L1, and PD-L2 mRNAs were detected in the melanoma group compared to melanocytomas, reaching statistical significance for PD-1 and PD-L1. Since PD-L1 and PD-L2 can be expressed by antigen presenting cells as well as neoplastic cells, immunohistochemistry was performed to evaluate the potential contribution of lymphocytes and macrophages to checkpoint molecule expression in the tumor microenvironment. Our results revealed a significantly higher proportion of cells with positive cytoplasmic immunoreactivity using antibodies against the T cell marker CD3 in the group of melanomas with higher expression of PD-1, whereas the number of cells with positive cytoplasmic immunoreactivity using antibodies against IBA1 (macrophages) was similar in PD-1 High and PD-1 Low melanoma groups. Additionally, large numbers of IBA1+ cells were seen in both high and low PD-1 expressing melanomas, including those with a relatively low number of CD3+ cells. The elevation of PD-L1 and PD-L2 in these same high PD-1-expressing tumors suggests a modulated co-expression of the PD axis molecules in melanoma involving the neoplastic cells and T lymphocytes, rather than macrophages.
Our results are consistent with the expectation that the higher PD-1 expression in the study samples is contributed by the more abundant T cell population; confirmation of this conclusion awaits cellular localization when appropriate antibodies are available for IHC. Our findings of TILs in all 10 melanoma cases examined are consistent with those of other investigators 29 who reported the presence of TILs (as CD3+ and CD20+ cells) in a high percentage of melanomas (78.1% of oral cases and 64.7% of cutaneous cases). It should be noted that our melanoma group included both oral (16 cases) and cutaneous (6 cases) melanomas. While these forms of melanoma may differ in disease progression and likely oncogenic origin, we did not observe pronounced differences with regard to checkpoint molecule expression or T cell infiltration (data not shown). However, our samples sizes did not allow their rigorous comparison.
The lack of effective therapies for canine melanoma emphasizes the importance of comparative oncology to understand common mechanisms between the species and the potential value of immunotherapy development. Increased PD-1 and PD-L1 expression has been previously associated with shorter survival times and lymph node metastasis in human lung adenocarcinoma 46 and urothelial carcinoma. 43 Increased PD-L2 expression has been linked to increased cell invasion in human patients with colorectal cancer, 14 and increased PD-1, PD-L1, and PD-L2 expression was found to be associated with metastasis when compared with primary tumors in renal carcinoma. 3 These results also correlate with studies in canine cancers, where PD-L1 was overexpressed in melanoma, osteosarcoma, histiocytic sarcoma, mast cell tumor, mammary carcinoma, and prostatic cancer. 21 Additionally, a single study demonstrated overexpression of PD-L2 in dogs with B cell lymphoma. 40 Since the expression of checkpoint molecules has been related to an increase in tumor infiltrating lymphocytes 8 and a better response to immunotherapy in humans, 6,45 our findings suggest that immunotherapies targeting the PD-1 axis have potential for efficacy in canine melanoma and other cancers, as has been demonstrated. 22
The majority of studies evaluating checkpoint molecules have compared their expression between malignant neoplasms and healthy control tissue. However, only a few studies performed in human cancers have compared expression between malignant and benign neoplasms of the same cellular origin. 7,33 In the current study, we showed that checkpoint molecule mRNA expression is higher in malignant tumors when compared to the benign counterpart. To our knowledge this is the only study in dogs evaluating the expression of checkpoint molecules using such a control, and brings interesting new insight into the mechanisms of tumor development and antitumor immunity.
While this manuscript was in final review, Maekawa et al. 20 published findings from their further studies of chimeric anti-PD-L1 monoclonal antibody therapy of canine melanoma.
Footnotes
Acknowledgements
We thank Dr Stephen Werre for statistical analysis and Dr Sheryl Coutermarsh-Ott for guidance in RNA extraction from formalin-fixed tissues.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work was provided by the Research & Graduate Studies Office of the Virginia-Maryland College of Veterinary Medicine.
