Abstract
Canine parvovirus type 2 (CPV-2) is among the most important and highly contagious pathogens that cause enteric or systemic infections in domestic and nondomestic carnivores. However, the spillover of CPV-2 to noncarnivores is rarely mentioned. Taiwanese pangolins (Manis pentadactyla pentadactyla) are threatened due to habitat fragmentation and prevalent animal trafficking. Interactions between Taiwanese pangolins, humans, and domestic animals have become more frequent in recent years. However, information about the susceptibility of pangolins to common infectious agents of domestic animals has been lacking. From October 2017 to June 2019, 4 pangolins that were rescued and treated in wildlife rescue centers in central and northern Taiwan presented with gastrointestinal signs. Gross and histopathological examination revealed the main pathologic changes to be necrotic enteritis with involvement of the crypts in all intestinal segments in 2 pangolins. By immunohistochemistry for CPV-2, there was positive labeling of cryptal epithelium throughout the intestine, and immunolabeling was also present in epidermal cells adjacent to a surgical amputation site, and in mononuclear cells in lymphoid tissue. The other 2 pangolins had mild enteritis without crypt involvement, and no immunolabeling was detected. The nucleic acid sequences of polymerase chain reaction (PCR) amplicons from these 4 pangolins were identical to a Chinese CPV-2c strain from domestic dogs. Quantitative PCR revealed a higher ratio of CPV-2 nucleic acid to internal control gene in the 2 pangolins with severe intestinal lesions and positive immunoreactivity. Herein, we present evidence of CPV-2 infections in pangolins.
Keywords
Canine parvovirus type 2 (CPV-2) is now classified under the species carnivore protoparvovirus 1, along with feline parvovirus (FPV), mink enteritis virus, and raccoon parvovirus. 9 Similar to other protoparvoviruses, CPV-2 has tropism for mitotically active S-phase cells for replication, such as intestinal epithelium, bone marrow, and lymphoid tissue. 1,10,13,30 Myocardial susceptibility to CPV in neonates is thought to be related to the initial higher mitotic activity in newborn animals. 1,13,21 Squamous cells in the keratinized epithelium may also be infected transiently following viremia. 12,20 Basophilic intranuclear or intracytoplasmic inclusion bodies with or without inflammation are documented in keratinocytes of the tongue and skin. 12,20 CPV-2 was first described in dogs in 1978 and was presumed to be derived from FPV, although some researchers claimed that FPV and CPV-2 originated independently from common ancestors. 2 Since then, several antigenic variants of CPV-2 have emerged from CPV-2, including CPV-2a, -2b, and -2c, all of which can infect wild carnivores. 2,10 The broad host range of CPV-2 and wide distribution of domestic dogs, who may serve as a reservoir, can cause critical effects on threatened and endangered species. Although phylogenetic studies have indicated that the significant known biodiversity of parvoviruses emerged in nondomestic animals, limited evidence suggests an onward transmission in new host species. 2 In Taiwan, all antigenic variants have been identified in domestic dogs, cats, and wild carnivores such as leopard cats (Prionailurus bengalensis chinensis). 5,6,8,17,34
The Taiwanese pangolin (Manis pentadactyla pentadactyla), endemic to Taiwan, is a subspecies of the Chinese pangolin, and it lives in lowlands and suburban hills below 1000 m asl. 31 All pangolins are threatened by poaching and illegal trade throughout the world, but Taiwanese pangolins are facing more complicated survival problems including trauma caused by gin traps, stray dog attacks, and vehicular collisions. 24 Moreover, toxins from pesticides and herbicides may cause hepatic and respiratory disorders in pangolins in Southeastern Taiwan. 31 Efforts to conserve the Taiwanese pangolin population include rescue and rehabilitation, establishment of the Wildlife Conservation Act by the Taiwanese government, and field investigations, and these are considered successful with a steady increase of the wild pangolin population. 31 However, their contact with domestic animals and humans has increased, thus potentially increasing the likelihood of infection. Nonetheless, infectious diseases have rarely been assessed in this endangered species. In China, parainfluenza virus 5, herpesviruses, and paramyxoviruses have been detected in Malayan pangolins (M. javanica), although gross and histopathological evidence is limited. 18,36 A retrospective study of pathological findings related to Taiwanese pangolins revealed various bacterial and parasitic infections but no viral agents. 15 Here, we provide pathological and molecular evidence of parvoviral infection causing enteritis in recently rescued Taiwanese pangolins critically ill with diarrhea.
Materials and Methods
Animals and Sampling
From January 2013 to June 2019, 32 Taiwanese pangolins from 8 counties were admitted to the local Animal Protection and Health Inspection Office or Wildlife Rescue and Research Center of Endemic Species Research Institute (WRRC-ESRI) for treatment. Among them, 3 Taiwanese pangolins (cases 1–3) from Nantou County and 1 pangolin (case 4) from North Taiwan were admitted to rescue centers due to trauma (cases 1, 2, 4) or from being orphaned (case 3). The onset of clinical signs in cases 1, 2, and 3 occurred within a period of 45 days. Case 4 occurred about 180 days after the previous cases. After being hospitalized for variable periods (all >1 month), these 4 pangolins developed clinical signs including depression, anorexia, and pasty-to-bloody diarrhea, and they eventually died. Clinical information is summarized in Supplemental Table S1. During the period, several carnivorous species, including leopard cats, masked palm civets (Paguma larvata), and ferret-badgers (Melogale moschata), were housed in separate wards of the WRRC-ESRI.
Pathologic examination of the 4 pangolins was performed at the Animal Disease Diagnostic Center, National Chung Hsing University (NCHU-ADDC). The major organs, including the heart, liver, spleen, lungs, kidney, brain, each segment of the gastrointestinal tract, gonads, adrenal glands, and trachea were collected, and fixed in 10% neutral-buffered formalin for histopathology. Frozen tissue samples of the brain, lungs, and liver were stored at −20 °C. The jejunal mucosa of the available carcasses was scraped with a sterile surgical blade and sampled for molecular examination.
Histopathology and Immunohistochemistry
Fixed tissue samples were trimmed, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Semiquantitative histologic grading (− to +++) was applied to histopathological findings to evaluate the intensity and extent of the lesions.
For immunohistochemistry (IHC), tissue sections were placed on silane-coated slides and heated overnight in an oven at 56 °C. After the slides were deparaffinized and rehydrated in graded alcohol, IHC was performed using the EnVision+ Dual Link System-HRP (DAB+) (Dako) according to the manufacturer’s instructions, with mouse anti-parvovirus monoclonal immunoglobulin (Ig) G2a antibody (Abcam). Dual block (0.3% hydrogen peroxide–containing sodium azide and levamisole) treatment for 10 minutes was carried out for endogenous enzyme blocking and nonspecific ionic binding. After being washed twice in Tris-buffered saline Tween-20 (TBST) buffer, the slides were incubated with the primary antibody at 1:1000 dilution at 4 °C overnight, and washed twice. Next, the slides were incubated with the Polymer-HRP reagent for 5 minutes, and then washed twice. Following treatment with DAB+ chromogen for 3 minutes, the slides were rinsed with TBST buffer, counterstained with Mayer’s hematoxylin, rinsed with distilled water, and mounted with Arabic gum. Negative controls (ie, corresponding tissue sections from CPV-negative pangolins and substituting the monoclonal antibody with TBST) were run in parallel in each assay. Samples were considered positive for parvovirus only when distinct red-brown, fine-to-clumped granular signals were detected in the samples and positive controls but not in the negative controls.
In case 1, IHC was conducted on skin sections by incubation with the mouse anti-papillomavirus monoclonal IgG antibody (ab2417, Abcam), to rule out canine papillomavirus infection which could cause a similar dermal lesion at the amputation wound. A skin biopsy specimen diagnosed as pigmented viral plague from a dog was selected as a positive control for canine papillomavirus IHC.
Polymerase Chain Reaction (PCR) for CPV-2 Detection
After being homogenized with Biomasher II homogenizer (Nippi), DNA was extracted from fresh tissue samples using the DNeasy blood and tissue kit according to the manufacturer’s instructions (Qiagen). For cases when fresh intestinal mucosa was unavailable, 10 consecutive 10-μm-thick sections of FFPE blocks were deparaffinized with xylene, washed with 99.5% alcohol, and used for DNA extraction as described above. Nested polymerase chain reaction (PCR) was conducted using CPV-2-specific primers targeting the VP1/VP2 gene fragment as previously described. 3,16 Bidirectional sequencing with the original primers (for nested-PCR, inner primer set pCPV-2N(F) and pCPV-2N(R) were used) was conducted using the fluorescent BigDye-Terminator v3.1 Cycle Sequencing Kit (Thermo Fisher) following the manufacturer’s instructions. Alignment and editing of the sequences were conducted using the Lasergene package of SeqMan software 7.1.0. 4 The sample obtained from a confirmed case of CPV-2a infection in a dog was used as positive control and nuclease-free water (Qiagen) was used as a negative control in parallel in each PCR run.
Relative Quantification of CPV-2 Nucleic Acid
Real-time PCR was performed on the DNA samples of CPV-2-positive cases to compare the amount of CPV-2 DNA between each sample. The internal control primers 3F (5′-TCAAGCCTATCACCCGCAAG-3′) and 3R (5′-AGGTCCAGGGGTTTTGGTTG-3′) were designed to amplify 230-bp gene fragments of the mitochondrial ribosomal protein L16 (MRPL16) according to the Chinese pangolin sequence (GU990982.1). The primer pair M1/M2 was selected to detect the gene of CPV-2. 29 SYBR Green-based real-time PCR assays were performed using CFX 384 quantitative PCR instrument (BioRad) with SYBR Green Master Mix (BioRad). Each 10-µL of reaction mixture contained 0.2 μM forward and reverse primers and 3 µL of DNA extract. The thermocycling conditions were as follows: 2 minutes at 50 °C, 2 minutes at 95 °C, and 40 cycles each at of 15 seconds at 95 °C, 1 minute at 61 °C and 1 minute at 68 °C. All samples were run in triplicate. The positive and negative controls as mentioned previously were included. The relative quantification of the gene of CPV-2 was calculated using the delta-delta Ct (2–ΔΔCt) method, where Ct is the mean threshold cycle. 19 The liver sample of case 1 was used as the calibrator of gene expression.
Detection of Concurrent Infections
Molecular screening for canine morbillivirus and canine coronavirus was conducted as described elsewhere. 14,26 Brain smears (eg, the cerebrum, cerebellum, and brainstem) were prepared for rabies virus antigen detection following the standard fluorescent antibody test procedure. 7 Bacterial culture was performed on the mesenteric lymph node of case 1 and liver of case 2. Parasite identification was performed through direct wet mount examination of eggs or adult parasites. 15,28 In cases in which parasites were obtained and identified by morphological features, PCR was conducted using NC1 and NC2 primer sets for rDNA of helminths. 33 Bidirectional sequencing with the original primers was conducted as mentioned earlier.
Results
Gross Lesions
Gross lesions are summarized in Table 1. Overall, all 4 pangolins had poor body condition. They had white-tan pasty to bloody feces (Fig. 1), and swollen, tan yellow-to-brown mesenteric lymph nodes (Fig. 2). In the jejunum and ileum, lesions varied from large amounts of pasty content within the lumen to thickened and reddened mucosa (Figs. 3, 4). There were several translucent nematodes (Necator americanus), approximately 7 mm in length anchored in the jejunal mucosa of cases 1 and 4. In the heart of case 1, the epicardium of the right ventricle was locally extensively mineralized, and multiple areas of gastric mucosa showed crateriform lesions. In case 2, there were petechiae and/or ecchymoses along the coronary sulcus of the heart. Cases 1 and 2 had surgical amputation wounds of the right and left hindlimbs, respectively, that were the reason for entry to the facility; these lesions were swollen, edematous, and covered with fibrinous exudate. Pulmonary and interlobular edema and congestion in multiple organs were present in all 4 pangolins and considered nonspecific.
Gross Lesions in the Digestive tract of 4 Pangolins Infected With Canine Parvovirus.

Abbreviations: MLN, mesenteric lymph node; −, none; +, mild; ++, moderate; +++, severe gross lesions.
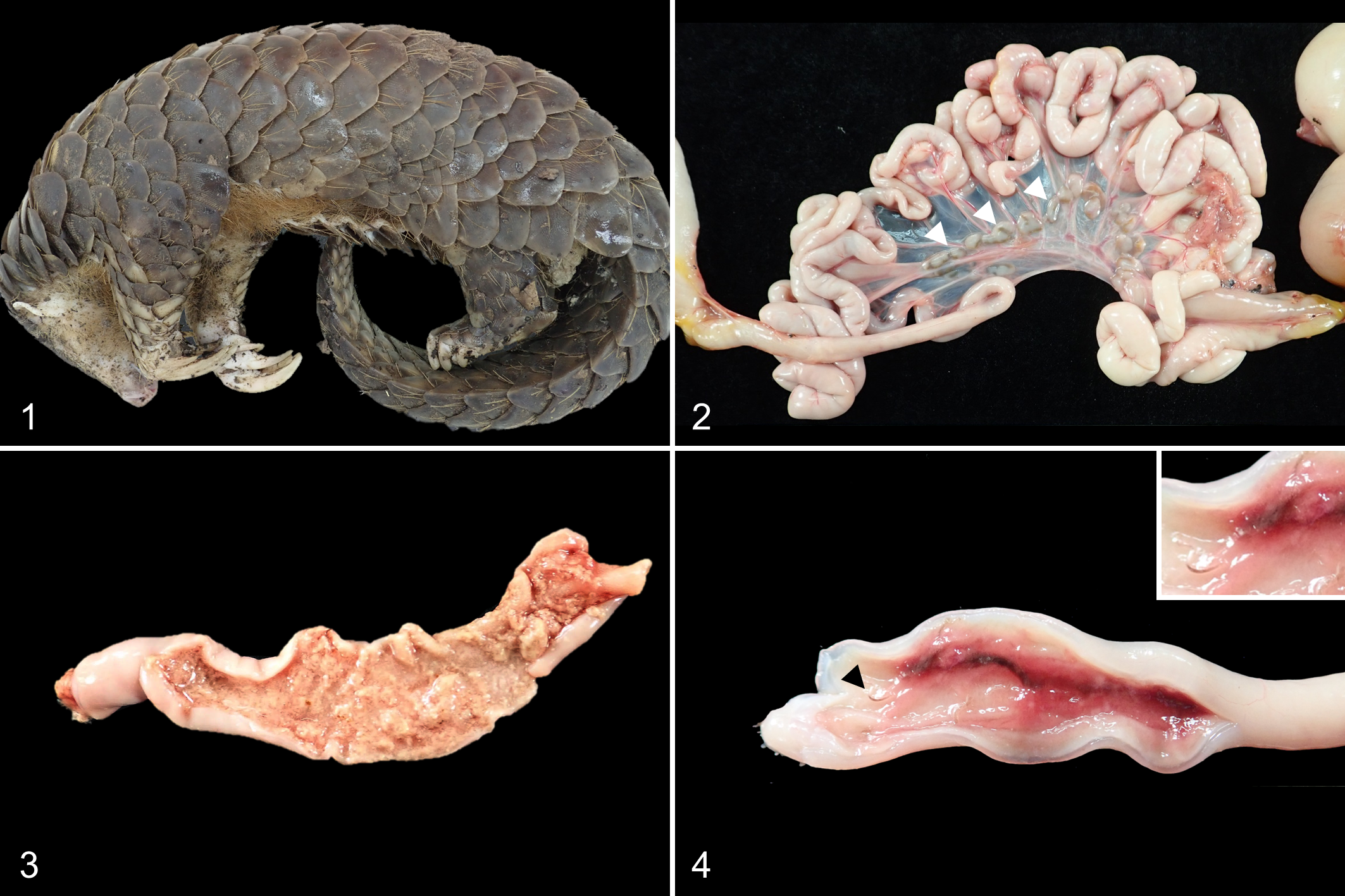
Canine parvovirus infection, pangolin, case 1.
Histopathology
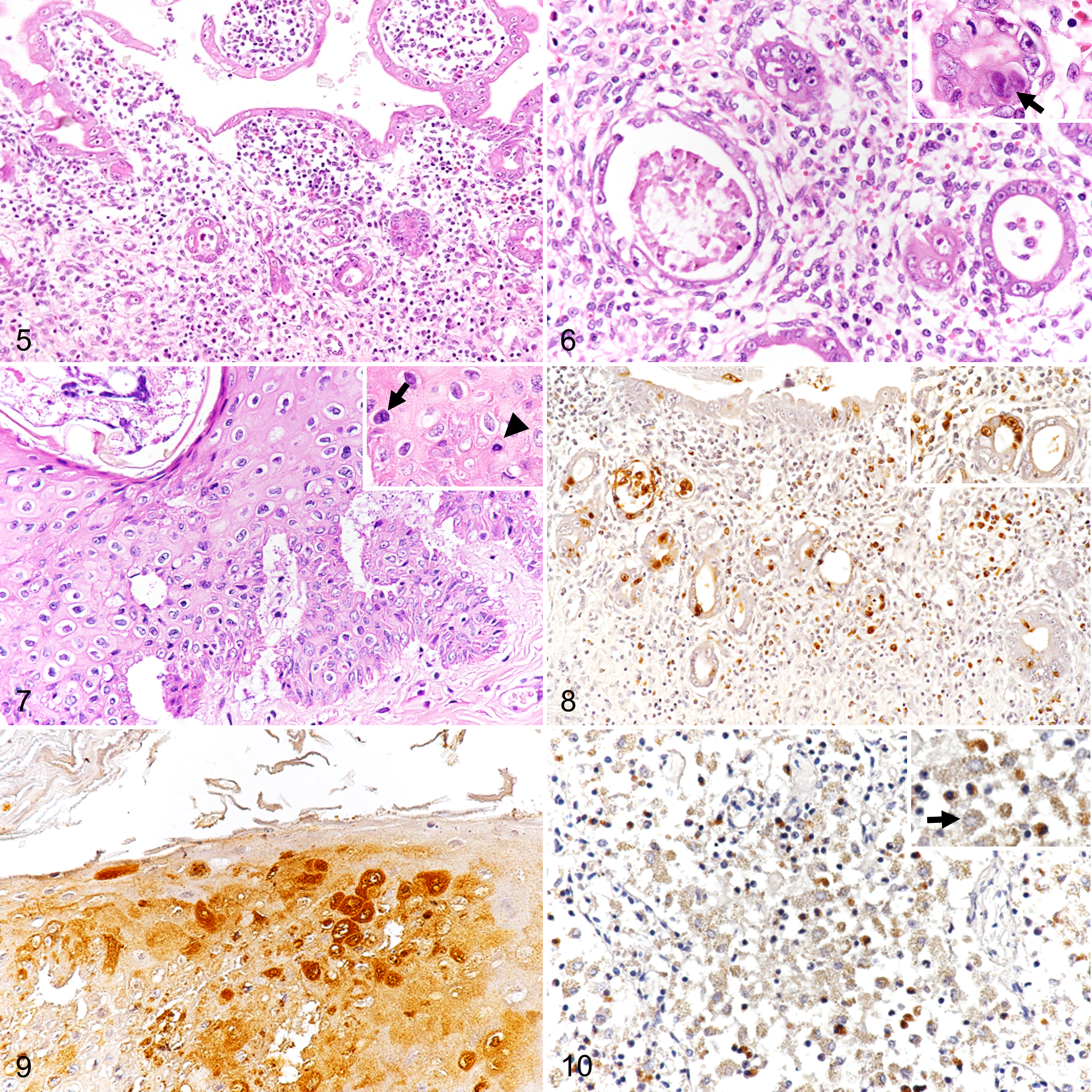
Table 2 summarizes the histopathological findings. The most severe lesions were in the digestive system. In cases 1 and 3, crypt necrosis, villous atrophy, and fusion affected 75% to 100% in each intestinal section (Fig. 5). There were occasional round-to-oval, basophilic to amphophilic inclusion bodies with marked chromatin marginalization in the nuclei of cryptal epithelial cells (Fig. 6). The lamina propria was expanded with proteinaceous fluid and diffusely infiltrated with moderate to large number of lymphocytes, plasma cells, and neutrophils (Fig. 6). Peyer’s patches in the ileum were hyperplastic and populated by lymphocytes. Cases 2 and 4 had relatively mild villous atrophy in the ileum with moderate numbers of plasma cells and neutrophils in the ileal lamina propria, but neither crypt hyperplasia nor intranuclear basophilic inclusion bodies were identified in these 2 cases. Several sections of the jejunum and ileum of cases 1 and 4 also had neutrophils and eosinophils in the lamina propria. In the distal ileum, the lumen contained degenerate inflammatory cells, cell debris, and strongyle eggs that had morulae and a surrounding annular clear space (Supplemental Fig. S1).
Histologic Lesions in 4 Pangolins Infected With Canine Parvovirus.
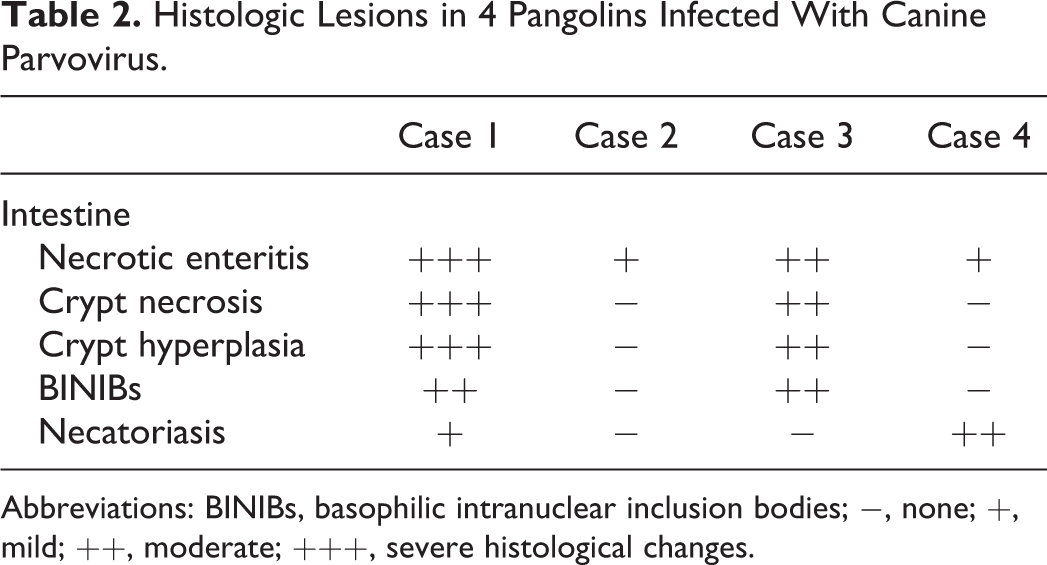
Abbreviations: BINIBs, basophilic intranuclear inclusion bodies; −, none; +, mild; ++, moderate; +++, severe histological changes.
Canine parvovirus infection, pangolin, case 1, Hematoxylin and eosin.
In the surgical amputation wound of case 1, the epidermis contained oval arthropod (mite) burrows, accompanied by acanthosis and focal parakeratotic hyperkeratosis (Fig. 7). There were multifocal clefts containing small amounts of proteinaceous fluid separating the dermis and epidermis. There were individualized hypereosinophilic keratinocytes with pyknotic nuclei, anisocytosis, and binucleated cells in the stratum spinosum. Keratohyalin granules in the stratum granulosum of the hyperplastic region were more prominent than in the unaffected region. In the subcutaneous tissue, there was marked infiltration of degenerative neutrophils with fibrinous exudates and erythrocytes, which was thought an action related to the ongoing tissue repair reaction in the amputation wound.
In all pangolins, mesenteric lymph nodes were depleted of lymphocytes, and there was moderate to severe hemosiderosis. In case 1, a locally extensive subepicardial area was mineralized with multinucleated giant cells, fibroblast proliferation, marked angiogenesis, and degeneration of surrounding cardiomyocytes. In all 4 cases, there were pulmonary edema and emphysema with variable amounts of eosinophilic proteinaceous fluid, variable number of pulmonary alveolar macrophages, and fusion of the alveolar spaces without prominent inflammatory cell infiltration.
Immunohistochemistry
By IHC, cases 1 and 3 had positive immunoreactivity for CPV antigen (Table 2) that was almost exclusively in the nuclei and sometimes in the cytoplasm of the cryptal and villous epithelium in all intestinal segments (Fig. 8). In the surgical amputation wound of case 1, immunoreactivity in keratinocytes was limited to the hyperplastic region and was distributed from the stratum basale to the stratum spinosum (Fig. 9). Scant but positive immunoreactivity was present in the cytoplasm of lymphocytes and macrophages of lymphoid organs, including the spleen and mesenteric lymph nodes (Fig. 10). Other organs had no immunoreactivity, even when lesions were present. No immunolabeling for CPV antigen was present in case 2 and 4. Papillomaviral antigen was not detected in the epidermis of the amputation wound of case 1.
PCR and Sequence Analysis CPV-2
CPV nucleic acid was detected by PCR in the intestinal mucosa and liver samples of all 4 pangolins (Table 3). The sequences of the PCR amplicons of these 4 pangolins (GenBank accession numbers MN900866-MN900869) shared 100% identity with the partial VP1/VP2 gene sequence of a Chinese CPV-2c strain (accession number: MK518005.1).
Detection of Canine Parvovirus by Immunohistochemistry and Polymerase Chain Reaction in 4 Pangolins.

Abbreviations: CPV-2c, canine parvovirus type 2c; IHC, immunohistochemistry; MLN, mesentery lymph nodes; PCR, polymerase chain reaction; +, occasional signal; ++, moderate signal; +++, abundant signal; −, not detected; N/D, not done.
a Mixed formalin-fixed, paraffin-embedded tissue.
Quantification of CPV-2 nucleic acid relative to an internal control gene was analyzed by real-time PCR (Supplemental Table S2). The levels of CPV-2 DNA in pangolins with typical CPV-2 related lesions (cases 1 and 3) were similar (232.32 in case 1 FFPE-I and 229.13 in case 3, relative to the calibrator). By contrast, the levels in pangolins without obvious lesions was low (0.07 in case 2 and 1.42 in case 4, relative to the calibrator).
Concurrent Infection
None of the 4 pangolins were positive for rabies virus, canine morbillivirus, or canine coronavirus. Mixed bacterial populations were isolated from the intestinal lumen and mesenteric lymph nodes of case 1 and liver of case 3.
The intestinal parasites in cases 1 and 4 were identified as the American hookworm (Necator americanus) based on the presence of dorsal cutting plates in the buccal cavity and elliptical eggs containing clusters of cells (morulae; Supplemental Figs. S2, S3). Helminth nucleic acid was detected using PCR in mucosal samples of cases 1 and 4.
Discussion
This study presents evidence of CPV-2c infection in rescued Taiwanese pangolins. Recently, CPV-2c infection was reported in 1 Taiwanese pangolin rescued in northern Taiwan. 35 Pathological changes including blunt intestinal villi, regeneration of the intestinal crypts, and infiltration of mixed inflammatory cells are typical findings related to CPPV-1 infection. In addition, multifocal ulceration and vesicle formation were present in the mucosa of the tongue and esophagus, and no other causative pathogens were detected. 35 In our study, lesions in cases 1 and 3 were consistent with subacute canine parvoviral enteritis occurring in other susceptible wildlife species. 30,32 While the intestine had severe lesions, neither vesicles nor ulcers were present in the upper gastrointestinal tract. 35 Positive immunoreactivity was also noted in mesenteric lymph nodes and spleen consistent with a previous study. 35 Thus, CPV infection in pangolins may have a similar pathogenesis as for other host species, with the virus amplifying in the lymphoid organs and a tropism for cells in S phase. 22,25
The pangolins also had skin lesions associated with trauma, surgical wounds, and mite infestation. Mite infestation is considered the primary cause of epidermal hyperplasia among pangolins. 15 The hyperplastic epidermal lesion associated with mite infection at the surgical amputation site of case 1 co-localized with IHC immunoreactivity against CPV-2, whereas the relatively normal epidermis had no immunoreactivity. This suggests that focal transient infections of CPV-2 in the skin might result from the higher mitotic rate of epidermal keratinocytes due to mite infestation and wound repair.
In case 1, locally extensive myocardial fibrosis and mineralization were identified but was restricted to the right ventricle. There was no positive immunoreactivity for CPV in these lesions, so it is uncertain if this focal circumscribed lesion was a sequel to CPV-induced myocarditis or an infarct.
CPV was detected in cases 2 and 4 by PCR and confirmed through sequencing, but these 2 pangolins had relatively low levels of CPV-2 based on real-time PCR testing. Consistent with the low levels of virus detected, no specific intestinal lesions were present in these cases and IHC was negative. It is reported that human and carnivore parvoviruses can be detected in both subclinical infection and during recovery from previous infection. 2,25 The different patterns of pathological findings may be due to age of the host, maternal antibody (MAb) titer, immune status, and differences in the pathogenicity between viral strains. 10 In these pangolins, the major difference was age: although it could not be estimated precisely, cases 1 and 3 were likely younger than cases 2 and 4 based on the body length and weight. In dogs, young animals are considered more susceptible than adults. Our data indicate that this is the case in pangolins as well. Antibody titers to CPV were not tested. The sequences of a short fragment of VP2 generated from all 4 pangolins were identical at the nucleotide level, although this does not rule out differences in virulence. Thus, CPV-2 can be associated with either severe or mild lesions in Taiwanese pangolins.
The partial VP2 gene sequences obtained from pangolins in our studies were identical to CPV-2c strains from dogs in China and CPV-2c has become more prevalent than CPV-2a and -2b in dogs and wildlife in Taiwan. 5,6,35 This finding suggests that the CPV-2c strains are circulating in Taiwanese wildlife population, and the host range of these strains include not only carnivores but also pangolins.
Bacterial, protozoal, and metazoal pathogens have frequently been identified in pangolins, yet their implications for pangolin health are rarely discussed. 23,24,37 Several gastrointestinal parasites including nematodes, cestodes, and protozoa have been found at necropsy and on fecal assessment. 23,37 Necator americanus hookworms were identified in the jejunum and ileum of cases 1 and 4 in the present study and were associated with intestinal wall thickening and bloody content. Similar to other Ancylostomatidae species, Necator americanus is blood-sucking, which may cause anemia and growth retardation in susceptible hosts. 27 In our cases, the hookworm infection together with CPV-2 may synergistically damage the intestinal epithelium. 11 Necatoriasis has also been reported in rescued Taiwanese pangolins in Southeastern Taiwan. 15 Thus, hookworm and parvoviral infections are differential diagnoses for melena or hemorrhagic enteritis in pangolins.
From a clinical perspective, veterinary clinicians and keepers in wildlife rescue facilities should consider CPV-2 as a causative agent when pangolins show signs of anorexia, emaciation, vomiting, and diarrhea. Strict isolation procedures (disinfection of fomites, workwear, and environment) should be followed when handling suspected or confirmed CPV-2-infected animals to prevent the introduction of CPV-2 into an uninfected population. Routine molecular screening may help identify subclinically infected individuals, thus guiding prevention strategies. Humans are the principal host for Necator americanus, and strict biosafety strategies with routine fecal assessments can help avoid this neglected zoonosis.
In summary, we provided data on CPV-2 infection with involvement of the intestinal tract and immune organs in 4 Taiwanese pangolins by using pathological, immunohistological, and molecular methods. Severe systemic CPV2-induced injury was probably the major contributor to the death of these pangolins. Although the relationship between CPV-2 genotypes and host range alteration within carnivores is well-studied, there has been limited evidence of CPV-2 spillover to phylogenetically close species. In our study, CPV-2 should be considered an issue for pangolin preservation because of the uncertainty of the sources of these viral strains and the high environmental resistance of parvoviruses. Elucidation of the origin of these viral strains, their dynamic interaction in the field, and whether modified live vaccines protect against this novel strain are required to inform future conservation strategies and policies.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211002198 - Canine Parvovirus Infections in Taiwanese Pangolins (Manis pentadactyla pentadactyla)
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211002198 for Canine Parvovirus Infections in Taiwanese Pangolins (Manis pentadactyla pentadactyla) by Yen Chi Chang, Zhi Yi Lin, Yan Xiu Lin, Kuei Hsien Lin, Fang Tse Chan, Shun Ting Hsiao, Jiunn Wang Liao and Hue Ying Chiou in Veterinary Pathology
Footnotes
Acknowledgements
We would like to express our gratitude to the staff and faculty of the NCHU-ADDC and ESRI-WRRC for helping with this study. All authors read and approved the final manuscript. The authors would also like to express appreciation to Wallace Academic Editing service.
Author Contributions
All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
