Abstract
This study describes the clinical and pathological characteristics of cutaneous spindle cell squamous cell carcinoma (SCSCC) in 18 cats. The average age of the cats was 11.8 ± 2.7 years, and all tumors were located in the facial skin, mainly affecting the pinna (13/18, 72%), followed by the periorbital area (4/18, 22%) and the dorsal muzzle (1/18, 6%). Tumors were composed of fusiform neoplastic cells with moderate atypia arranged in solid sheets or fascicles with foci of squamous differentiation. A panel of antibodies against cytokeratins, vimentin, S-100 protein, NSE, GFAP, Melan A, SMA, desmin, CD18, CD31, and p63 was used to help differentiate SCSCC from other spindle cell malignancies. SCSCCs expressed CK5/6 (17/18, 94%), AE1/AE3 (15/18, 83%), and p63 protein (18/18, 100%), but there was no immunolabeling for CK8/18. A role for sunlight exposure in the pathogenesis of the tumors was suggested by changes indicative of actinic keratosis, the location of the tumors in dorsal areas, and the absence of histomorphologic features of papillomavirus infection. Recurrence was not recorded in 14/18 cases (78%) during a follow-up period of 7 to 25 months. Three of 18 (17%) tumors recurred or led to humane euthanasia due to local progression, and one case (5%) had regional lymph node metastasis. Clinical outcome varied with cutaneous location, mitotic count, and invasion of surgical margins; thus, SCSCCs with a more aggressive behavior were located in the periorbital area (4/4 cases), had ≥14 mitoses in 10 high-power fields (2.37 mm2) (4/4 cases), and showed invasion of surgical margins (3/4 cases).
Keywords
Skin tumors are the most frequent neoplasms in cats after those occurring in the lymphoid system. 4,9 Basal cell tumor, mast cell tumor, squamous cell carcinoma, and fibrosarcoma are the most prevalent feline cutaneous tumors. 13,14,17 Squamous cell carcinoma (SCC) is a malignant neoplasm of epithelial cells with differentiation to keratinocytes. Several factors have been associated with the development of SCC, including chronic exposure to ultraviolet light, lack of pigment within the epidermis at the sites of tumor development, and absent or sparse hair coat. 5,8 In addition, papillomaviruses are oncogenic viruses recently associated with SCC as well as other related precursors, such as papillomas, viral plaques, and Bowenoid in situ carcinomas. 3,5,11 Tumors are invasive and locally destructive with metastasis to regional lymph nodes or more distant metastasis rarely described. 8,9,13 SCC in domestic animals is usually classified into well and poorly differentiated categories. 4,6,13 Uncommon subtypes include acantholytic, clear cell, spindle cell (SCSCC), and carcinoma arising in Bowen’s disease. 3 –5,13
In humans, SCSCC is reported to be more aggressive than conventional squamous cell carcinoma (CSCC). 2,12,19 Data in relation to different behavior of nonconventional squamous cell carcinoma in cats are not available.
The present study aimed to improve our knowledge of SCSCC in cats by morphological and immunohistological analyses, and to compare it with CSCC.
Materials and Methods
The study population was 18 cats from which biopsies were submitted to the Veterinary School of Las Palmas de Gran Canaria, Spain. The samples constituted all cases with a diagnosis of SCSCC received between 2014 and 2019. This number represented 2.3% (18/768) of the total cases of SCC in the same period. SCC of all histological types (n = 768) constituted 49% of cutaneous tumors in cats (n = 1567) in our reports, with the facial skin being the principal area affected (515/768, 67%). Cases were retrospectively selected from the database, and the diagnosis was confirmed by histological review of hematoxylin and eosin–stained sections. Inclusion criteria included a morphological diagnosis of SCSCC, characterized by proliferation of atypical spindle and polygonal cells arranged in solid sheets and/or fascicles with ≥70% spindle cell areas along with cytokeratin positivity.
Excisional biopsy was the only treatment in all cases. Tumor samples had been fixed in neutral buffered formalin and embedded in paraffin wax. Sections (4 μm) were cut and stained with hematoxylin and eosin, and also used for immunohistochemical analysis. Mitotic figures were counted in 10 high-power fields (HPF), equivalent to 2.37 mm2 (diameter of the field of view = 0.55 mm; 40× objective and 10× ocular; Olympus BX40 microscope), and the results expressed as median per 10 HPF ± SD.
Immunohistochemistry (Suppl. Table S1) was performed on deparaffinized sections using the avidin-biotin-peroxidase complex technique for cytokeratins (AE1/AE3, CK5/6, and CK8/18), vimentin, S-100 protein, neuron-specific enolase (NSE), glial fibrillary acidic protein (GFAP), Melan A, smooth muscle actin (SMA), desmin, CD31 (Dako), CD18 (Bio-Rad), epithelial/myoepithelial p63 marker, and major-capsid protein of papillomavirus (Abcam). All antibodies used in the study showed cross-reactivity with feline tissues. A biotinylated rabbit anti-mouse or swine anti-rabbit IgG (Vector Laboratories), diluted 1:200, were applied as secondary reagents for 30 minutes at room temperature. An ABC complex (Vector) diluted 1:50 was applied as the third reagent. Sections were incubated for 5 minutes with 3,3′-diaminobenzidine tetrahydrocloride (Sigma) 0.035% in TBS containing H2O2 0.1%. After rinsing in tap water, slides were lightly counterstained with Harris’s hematoxylin and mounted under DPX mountant (BDH Laboratory Supplies) for microscopy. Eighteen CSCC with similar facial locations (Suppl. Table S2) were included in the study. Additionally, tissues from feline cutaneous tumors composed of fusiform cells that can resemble the morphology of SCSCC, which included hemangiosarcoma, amelanotic melanoma, fibrosarcoma, peripheral nerve sheath tumor, leiomyosarcoma, and progressive histiocytosis, were run alongside in the immunohistochemical procedures.
Clinical information was obtained from accompanying biopsy submission forms, and follow-up data were retrieved from the clinical files and/or obtained by phone calls to the referring veterinarians or owners. Age, breed, sex, and clinical outcome are summarized in Table 1. Surgery was performed for both diagnosis and therapy, and no additional sampling or postmortem examination was carried out for this study. Date of death (when applicable) and recurrence and/or metastasis, determined by clinical examination, aspiration cytology, and radiographic evaluation, were recorded. Data were analyzed using IBM SPSS 26.0 for Windows (IBM Corp), and differences were considered statistically significant at P < .05. Analysis of variance was used to compare immunohistochemical results, mitotic count, tumor location, and clinical outcome. Correlations between variables was determined with the Student test for independent samples to access the differences of means. Mann-Whitney test was used when normality was not verified. Associations between pathological findings and clinical variables were evaluated by means of the Fisher χ2 test.
Clinical and Histological Data of Spindle Cell Squamous Cell Carcinoma in 18 Cats.
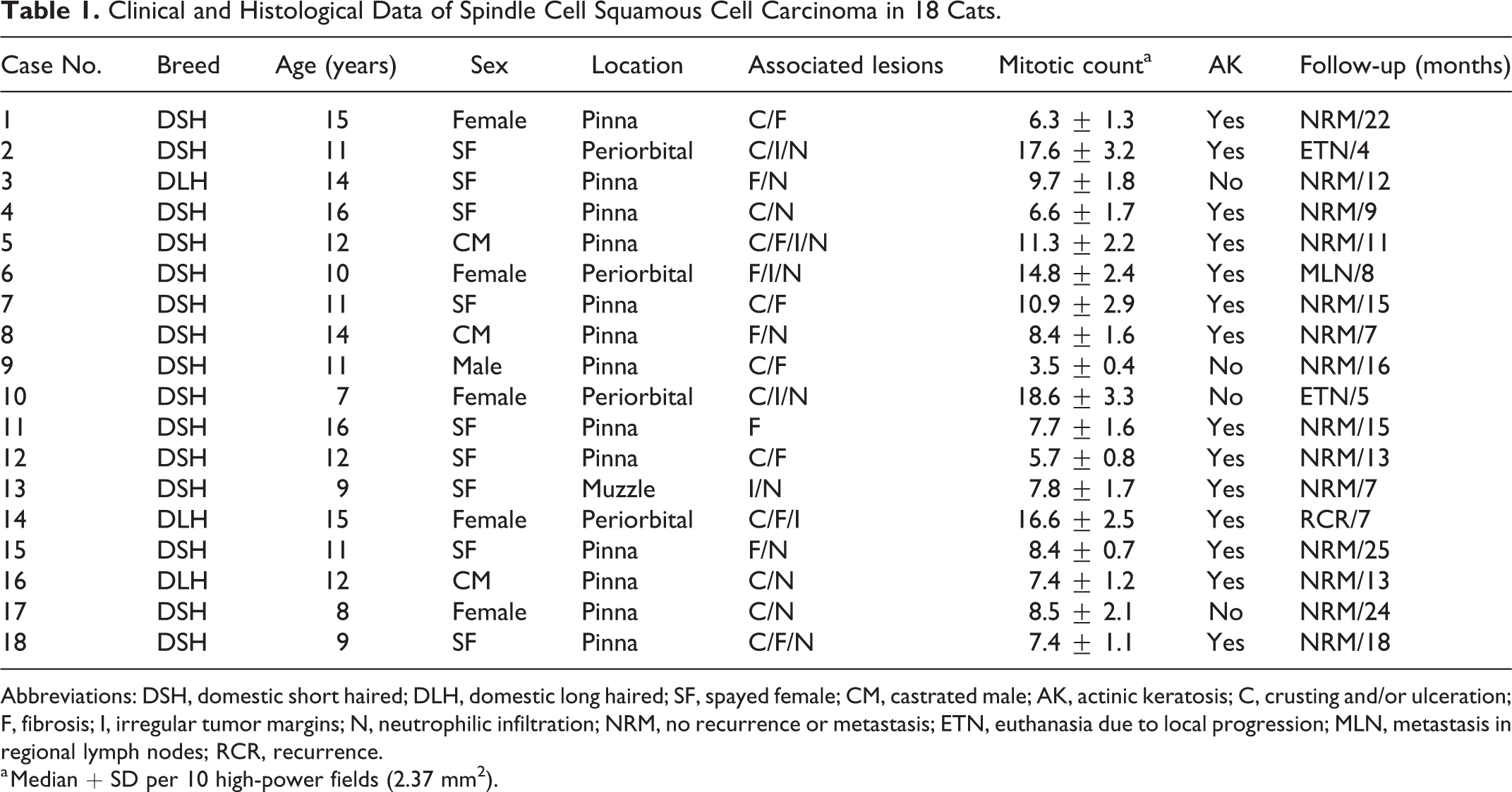
Abbreviations: DSH, domestic short haired; DLH, domestic long haired; SF, spayed female; CM, castrated male; AK, actinic keratosis; C, crusting and/or ulceration; F, fibrosis; I, irregular tumor margins; N, neutrophilic infiltration; NRM, no recurrence or metastasis; ETN, euthanasia due to local progression; MLN, metastasis in regional lymph nodes; RCR, recurrence.
a Median + SD per 10 high-power fields (2.37 mm2).
Results
Cats with SCSCC were >10 years old in 13/18 cases (72%), ranging from 7 to 16 years (mean 11.8 ± 2.7). Significant differences in relation to breed, age, and sex were not detected between SCSCC and CSCC. The tumors affected sun-exposed areas, especially pinnae (13/18, 72%), followed by the periorbital area (4/18, 22%) and dorsal muzzle (1/18, 6%). In the pinna, margins (7/13, 54%) were more commonly affected than the base of the ear (6/13, 46%). Tumors were bilateral in 4/13 cases (31%) and affected nonpigmented areas in 17/18 cases (94%). Growths consisted of elevated plaques or exophytic masses, frequently (12/18, 67%) with crusted or ulcerated surfaces and a solid, white to gray appearance when sectioned.
Diagnosis of SCSCC was made when a biphasic tumor was composed of ≥70% malignant spindle-shaped cell component of epithelial origin, confirmed by immunohistochemistry, and had areas with recognizable SCC histomorphology. Tumors consisted of a proliferation of pleomorphic elongated cells (Figs. 1–3), with variable cellular density, arranged in solid sheets and/or fascicles. Nuclei were large and vesicular, containing finely stippled or marginated chromatin, and prominent single or multiple nucleoli. There was moderate anisokaryosis and anisocytosis. The mitotic count was highly variable (Table 1), with a median of 9.8, and it was >7 in 14/18 cases (78%).
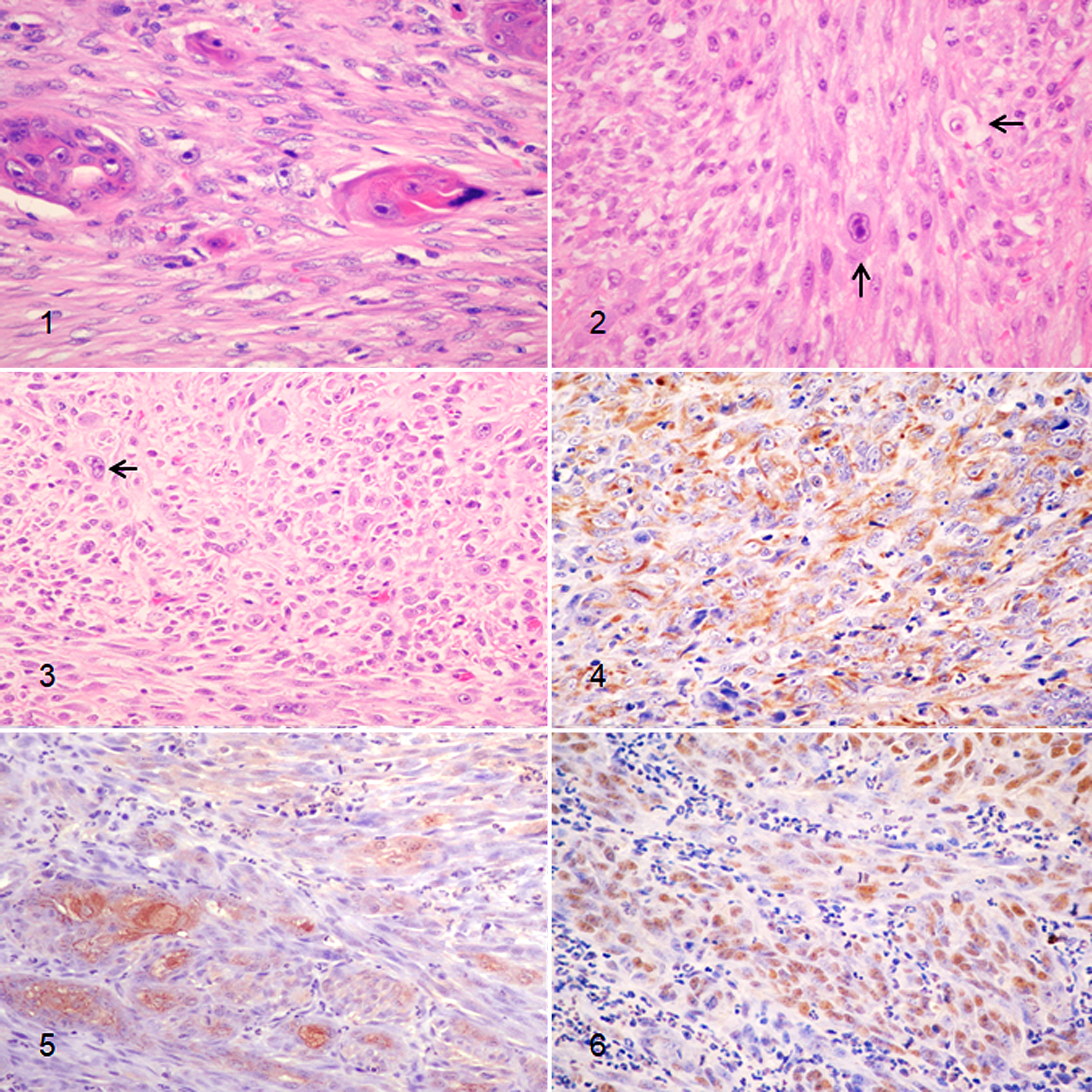
Spindle cell squamous cell carcinoma, skin, cat.
Cats presenting with tumors that had ≥14 mitoses per 10 HPF (median 16.9 ± 1.6) had significantly worse clinical outcome in relation to those with <14 mitoses per 10 HPF (median 9.8 ± 4.3).
Cytoplasm was abundant with distinct cell borders. Dispersed foci of individual cell keratinization, affecting polygonal-epithelioid cells, observed in 17/18 cases (94%), helped distinguish SCSCC from other spindle cell malignancies on routine hematoxylin and eosin–stained sections. Transition of pleomorphic spindle cells to hyperplastic and dysplastic overlying epidermis was observed in 15/18 cases (83%).
Vascular and surgical margin invasion was detected in 3 of 18 cases (17%; cases 2, 6, and 14), which significantly influenced the clinical outcome.
Changes indicative of actinic keratosis, including prominent irregular hyperplasia of the epidermis, pleomorphism of keratinocytes, alterations in nuclear polarity, nuclear enlargement, and increased mitotic activity in basal and suprabasal epidermal cells, were detected in the proximity of 14/18 tumors (78%).
During the period of study, no recurrence or metastasis was recorded in 14/18 cases (78%). In contrast, 3 of 18 tumors (17%) recurred and/or the cats were humanely euthanized due to local progression, and one case (case 6) developed regional lymph node metastasis confirmed by fine-needle aspiration.
Immunohistochemical results (Table 2) showed that neoplastic cells had cytoplasmic expression of CK5/6 in 17/18 (94%) SCSCC (Fig. 4), and expression of AE1/AE3 in 15/18 (83%; Fig. 5). Similarly, 17/18 (94%) and 18/18 (100%) CSCCs expressed CK5/6 and AE1/AE3, respectively. Immunoreactivity with CK8/18 was not detected in any SCSCC or CSCC. Nuclear positivity for p63 protein was detected in 18/18 (100%) of both SCSCCs and CSCCs (Fig. 6). The remaining 28 spindle cell tumors included in the study, which consisted of hemangiosarcomas, melanomas, fibrosarcomas, peripheral nerve sheath tumors, leiomyosarcomas, and progressive histiocytosis, were negative for cytokeratins and p63. Finally, SCSCCs were not labeled with the antibody against major-capsid protein of papillomavirus or with the other cellular markers evaluated, which included vimentin, S-100, NSE, GFAP, Melan A, SMA, desmin, CD31, and CD18.
Number (%) of Tumors Immunolabeled With Cytokeratins and p63 in Spindle Cell and Conventional Squamous Cell Carcinomas, and Other Cutaneous Spindle Cell Malignancies in Cats.
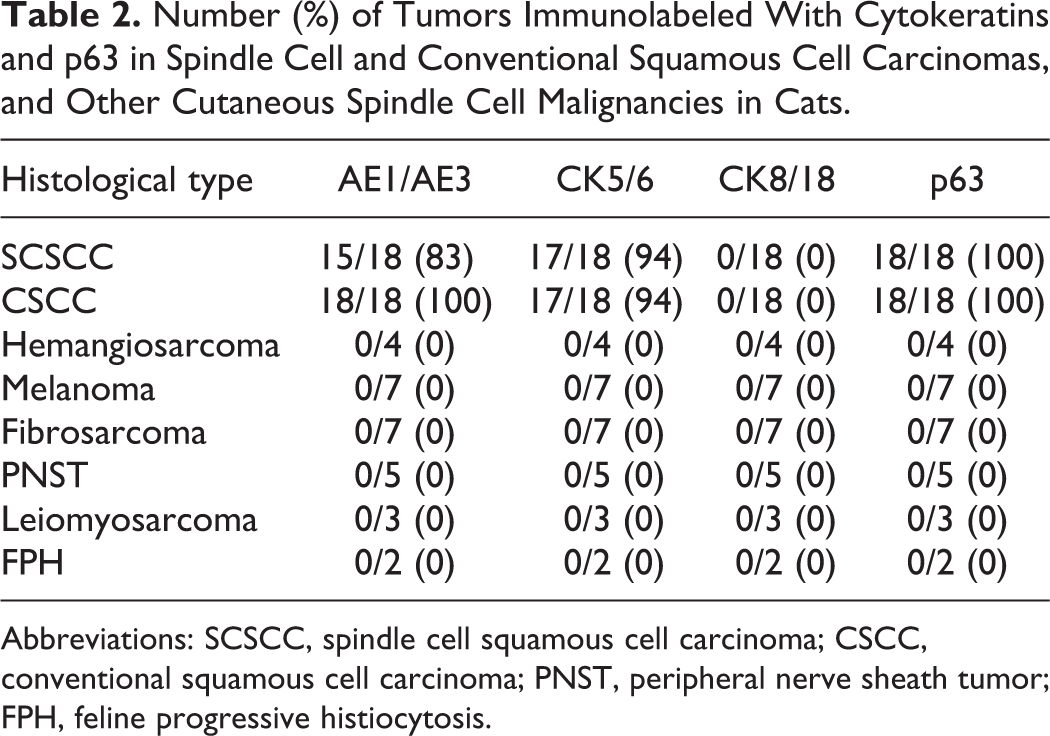
Abbreviations: SCSCC, spindle cell squamous cell carcinoma; CSCC, conventional squamous cell carcinoma; PNST, peripheral nerve sheath tumor; FPH, feline progressive histiocytosis.
Discussion
SCSCC occurred in older cats, similar to the age of animals with CSCC in the current study (10.7 ± 2.5 years) and previous reports, 3,9,13 the facial skin being the most common site. 4,5 In humans, this neoplastic pattern is mainly described in the upper respiratory and digestive tracts, skin, and breast. 10,12,18 The indications of actinic keratosis, the location of the tumors in dorsal nonpigmented areas, and the high level of solar radiation in the geographical location of the animals (Canary Islands) suggest that ultraviolet radiation played a significant role in the pathogenesis. Ultraviolet exposure, among other factors, can induce mutations of p53 tumor suppressor gene, which is critical in inducing apoptosis. 4,5,20 The altered suppressor gene can undergo impaired degradation and lead to abnormal accumulation of proteins, causing positive immunostaining. 3,20 p63, a member of the p53 protein family, has been used as epithelial/myoepithelial marker in human and feline carcinomas 2,15,16 as demonstrated in the current study, in which there was no significant differences of p63 staining in relation to the clinical outcome or the histological type.
There were no histomorphologic changes compatible with papillomavirus infection, such as large and irregular granules of keratohyalin, presence of koilocytes, expanded blue-gray cytoplasm, or intranuclear inclusion bodies. 4,5,11,13 Cutaneous SCSCC in humans has been associated with sun-damaged skin, burn scars, and radiation exposure but, in agreement with the current study, no association has been found with papillomavirus infection. 10,12
Although SCSCC is considered a poorly differentiated variant with an aggressive clinical course and poor response to treatments in humans, 18,19 there was no association between the histological type and the clinical behavior in the current study. Despite the fact that SCC located in the pinnae has occasionally been reported to invade the meninges, lymph node, eye, and internal ear, 14 neoplasms in this location had a significantly better prognosis, probably due to the possibility of complete surgical resection and the low metastatic potential previously described at this site. 8,9
In addition to the histopathological features that are similar to those of various types of sarcoma, the heterogeneous profile of cytokeratin expression, with a wide range of positivity for pan-cytokeratin AE1/AE3, CK1, and CK5/6, the immunoreaction with vimentin, and the occasional expression of S-100 and smooth muscle actin in some cases of SCSCC in humans, make it a diagnostic challenge. 10,18,19 In the current study, positive immunostaining for cytokeratins and p63 was the most accurate way of differentiating SCSCC from other spindle cell malignancies and also from the desmoplastic response frequently seen in SCC. 4,19
Epithelial-mesenchymal transition (EMT) is considered a feature of carcinoma progression because invasive neoplastic cells often lose epithelial characteristics and acquire mesenchymal phenotype, which confer metastatic properties by enhancing mobility, invasion, and resistance to apoptosis. 1 EMT is also regulated by alterations in the expression of structural cytoplasmic proteins, specifically repression of cytokeratin and activation of vimentin, which affect cell shape, motility, and disaggregation of cell-to-cell adhesion. 1,18 In our series, although 3/18 (17%) of the SCSCCs were not immunolabeled with cytokeratin AE1/AE3, all SCCs were negative for vimentin, which could indicate lack of association of SCSCC with EMT, a different immunophenotype profile, or less invasiveness of cutaneous SCSCC in cats in relation to CSCC of the feline oral cavity 7 and SCSCC in humans. 18
Analysis of subtypes of cytokeratins in neoplastic cells are used to help determine the histogenesis of epithelial neoplasms. 5,10,19 Cytokeratin expression confirmed a diagnosis of SCSCC in all but one case (case 16), in which the positivity with p63 was required to confirm the epithelial nature. 2,15,16 In agreement with reports in humans, 2,19 CK5/6 and p63 antibodies were the most sensitive for SCSCC. None of the SCC included in the study immunoreacted with CK8/18, which is detected in carcinomas of glandular origin. 6 Negative labeling with vimentin, S-100, NSE, GFAP, Melan A, SMA, desmin, and CD31 allowed differentiation of SCSCC from other spindle cell neoplasms frequently seen in cats, such as fibrosarcoma, malignant peripheral nerve sheath tumors, leiomyosarcoma, spindle cell amelanotic melanoma, and hemangiosarcoma. Finally, the leukocyte marker CD18 served to exclude feline progressive histiocytosis 5,6 that can mimic poorly differentiated SCC.
In conclusion, SCSCC is an uncommon neoplasm in cats, involving the facial skin, associated with changes indicative of prolonged sunlight exposure. Clinical outcome varied with cutaneous location, mitotic count, and invasion of surgical margins, but there were no significant differences between SCSCC and CSCC. Immunohistological analysis may be required to accurately differentiate it from other spindle cell malignancies.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820985126 - Cutaneous Spindle Cell Squamous Cell Carcinoma in Cats: Clinical, Histological, and Immunohistochemical Study
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820985126 for Cutaneous Spindle Cell Squamous Cell Carcinoma in Cats: Clinical, Histological, and Immunohistochemical Study by Francisco Rodríguez Guisado, Alejandro Suárez-Bonnet and Gustavo A. Ramírez in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
