Abstract
A 13-year-old castrated male Maine coon cat with a 5-year history of atypical mycobacteriosis was euthanized and submitted for necropsy. The cat had been kept in clinical remission since diagnosis using a combination of the antimycobacterial drug clofazimine and additional multimodal antimicrobial therapy. Grossly, tissues were diffusely discolored red-brown to yellow. Histologically, the myocardial interstitum was expanded by numerous, often multinucleated cells, which were distended by uniformly shaped acicular cytoplasmic spaces. These cells were immunopositive for CD18 and immunonegative for desmin, suggesting a histiocytic rather than muscular origin. Macrophages in other tissues contained similar acicular spaces. Ultrastructurally, the spaces were surrounded by 2 lipid membranes, resembling an autophagosome. Based on the clinical history and histologic, immunohistochemical, and ultrastructural data, we diagnosed clofazimine crystal storage. To our knowledge, this is the first report of clofazimine storage in a cat or within myocardial interstitial macrophages.
Clofazimine is a highly lipophilic phenazine dye with antimycobacterial and anti-inflammatory properties. This drug is listed as an essential medicine by the World Health Organization and is most commonly utilized in humans for the treatment of leprosy and multidrug-resistant tuberculosis. 26 In humans, adverse effects are generally self-limiting and include ichthyosis and pink discoloration of the skin in approximately 94% of patients, 19 less frequently, gastrointestinal pain 19 and discoloration of the sclera, 3 and rarely clofazimine storage enteropathy. 23,25 Histologically, clofazimine storage enteropathy in humans is characterized by expansion of the gastrointestinal lamina propria by crystal-laden macrophages following prolonged treatment with high doses of clofazimine. 23,25 This condition can lead to unnecessary laparotomy, either due to clinical signs suggestive of gastrointestinal obstruction 23 or due to its radiologic similarity to neoplastic processes. 24 In humans, storage of clofazimine crystals has also been reported in the macrophages of many tissues, including the lung, 13,20,22 lymphoid organs, 18 liver, 9 and eye. 11
In veterinary medicine, clofazimine is utilized in dogs and cats for the treatment of mycobacteriosis. 4,15,21 Adverse effects are rarely reported but include hepatotoxicity, gastrointestinal signs, photosensitization, discoloration of the skin, and pitting corneal lesions. 4,15 Histologically confirmed cases of clofazimine storage enteropathy associated with treatment for mycobacteriosis have not been reported in domestic animals. However, mice fed approximately 10 mg/kg/day of clofazimine developed pink discoloration of the skin and hair and storage of clofazimine crystals within macrophages in the intestine, liver, spleen, and lungs. 2 Interestingly for the current report, in this murine model, clofazimine did not accumulate within the heart. 2 With transmission electron microscopy, intrahistiocytic clofazimine crystals have been shown to be enveloped in a double lipid membrane, similar to those surrounding autophagosomes. 1
A 13-year-old castrated male Maine coon cat was submitted for necropsy. Eight years prior to necropsy, the cat developed self-limiting lymphadenomegaly, followed by immune-mediated retinal detachment, anemia, and thrombocytopenia. He was treated with multiple immunosuppressive medications over the next 3 years, culminating in hepatotoxicity due to cyclosporine overdosage 4.5 years prior to necropsy. Thereafter, immunosuppression was gradually tapered and discontinued, and the cat developed marked lymphadenomegaly and chemosis. Pyogranulomatous lymphadenitis with intracytoplasmic negative-staining bacilli was identified following cytologic evaluation of an enlarged superficial cervical lymph node. Mycobacterium avium was detected by mycobacterial culture and PCR (polymerase chain reaction) performed on 4 excised enlarged retropharyngeal lymph nodes. It was not possible to determine whether the cat’s mycobacteriosis reflected opportunistic infection secondary to chronic immunosuppression or if it had been the trigger for the cat’s original presentation with immune-mediated retinal detachment, anemia, and thrombocytopenia.
A detailed description of the clinical course of this case and the treatments are presented in the Supplemental Materials. In short, for 8 years, the cat had been treated with various antimycobacterial agents and adjustments had been made as necessary to maintain clinical remission. These medications included enrofloxacin (32.5 mg, q24h), rifampin (75 mg, q24h), clarithromycin (62.5 mg, q12h), amikacin (100 mg, q24h), ethambutol (300 mg, q12h), moxifloxacin (30 mg, q24h), minocycline (50 mg, q24h), azithromycin (50 mg, q24h), pradofloxacin (25 mg, q24h), and clofazimine (50 mg, q24h). Clofazimine therapy was initiated 4 years prior to necropsy but was discontinued 5 months prior to euthanasia due to unavailability for use in veterinary medicine in the United States. Three months following initiation of treatment with clofazimine, the cat’s skin, fur, and sclera developed a pink hue; this discoloration waned after discontinuation of the medication. The cat was presented for euthanasia due to 2 weeks of weakness and rapidly progressive weight loss. At the time of death, the cat was being treated with azithromycin, pradofloxacin, and minocycline.
Gross findings at necropsy included purple-brown to red discoloration of most tissues, including skeletal muscle, kidney, liver, and bone marrow; adipose tissue was discolored yellow to brown. Tissues stained cutting surfaces bright pink, discolored fixation solutions red-orange, and stained histological processing equipment bright pink to dark red. The heart was subjectively enlarged with thickened ventricular walls and weighed 33.8 g, which was 0.78% of body weight (University of Tennessee internal reference range 0.3% to 0.45%) and the liver weighed 240 g, which was 5.5% of body weight (University of Tennessee internal reference range 3% to 3.5%). Cavitary effusions were not present. With the exception of the discoloration and multiple chronic renal infarcts, all organs were grossly unremarkable.
Samples of all major tissues were collected and fixed in 10% buffered neutral formalin, processed routinely, and routinely stained with hematoxylin and eosin for light microscopic examination. For desmin immunohistochemistry Biocare’s Decloaker and Reveal Buffer (Biocare) was used for antigen retrieval, and sections were treated with monoclonal mouse anti-desmin antibodies (Dako, catalogue #M0760; 1:100 dilution, 30 minutes). For CD18 immunohistochemistry, Carezyme I: Trypsin Kit (Biocare) was used for antigen retrieval, then endogenous peroxidase activity was blocked with 3% H2O2 and monoclonal mouse anti-feline CD18 antibody was applied (clone Fe3.9f2, Peter Moore, University of California-Davis, Davis, CA; 1:10 dilution, 30 minutes). 6,8 Diaminobenzidine tetrahydrochloride was utilized as chromogen with hematoxylin counterstain.
For transmission electron microscopy, formalin-fixed samples of myocardium were washed in 0.1 M sodium phosphate buffer, postfixed in buffered 2% osmium tetroxide for 60 minutes, washed in water, and dehydrated in a graded ethanol series with final dehydration in propylene oxide. Samples then were embedded in Embed 812 and semi-thin (1000 nm) and thin sections (100 nm) were prepared on a Leica EM UC7 ultra-microtome and stained with UranyLess stain (Electron Microscopy Sciences) followed by Reynolds lead citrate to increase the contrast. Sections were imaged in a Zeiss Libra 200MC operating at 200 kV.
The most striking histologic feature was expansion of approximately 80% of the myocardial interstitium by many cells with up to 20 nuclei and abundant pale eosinophilic cytoplasm distended by regularly shaped clear acicular spaces (Figs. 1, 2). Similar cells, often multinucleated, were also identified within the interstitium of the skeletal muscle, although in lower numbers. These cells were immunoreactive for CD18 (Fig. 3) and did not label with desmin, indicating a leukocytic origin. Given their multinucleation and similarity to Kupffer cells and pulmonary alveolar macrophages, they were interpreted as histiocytes. Additionally, myocardiocytes were variably sized (up to 3-fold variation) and contained perinuclear brown pigment granules (lipofuscin). Kupffer cells (Fig. 4), and pulmonary alveolar macrophages also contained similar clear, acicular spaces. Kupffer cells also contained abundant brown granular pigment, which stained blue with Prussian blue (data not shown) and was interpreted as hemosiderin. Notably, there was no evidence of these acicular spaces within macrophages in the intestinal lamina propria.
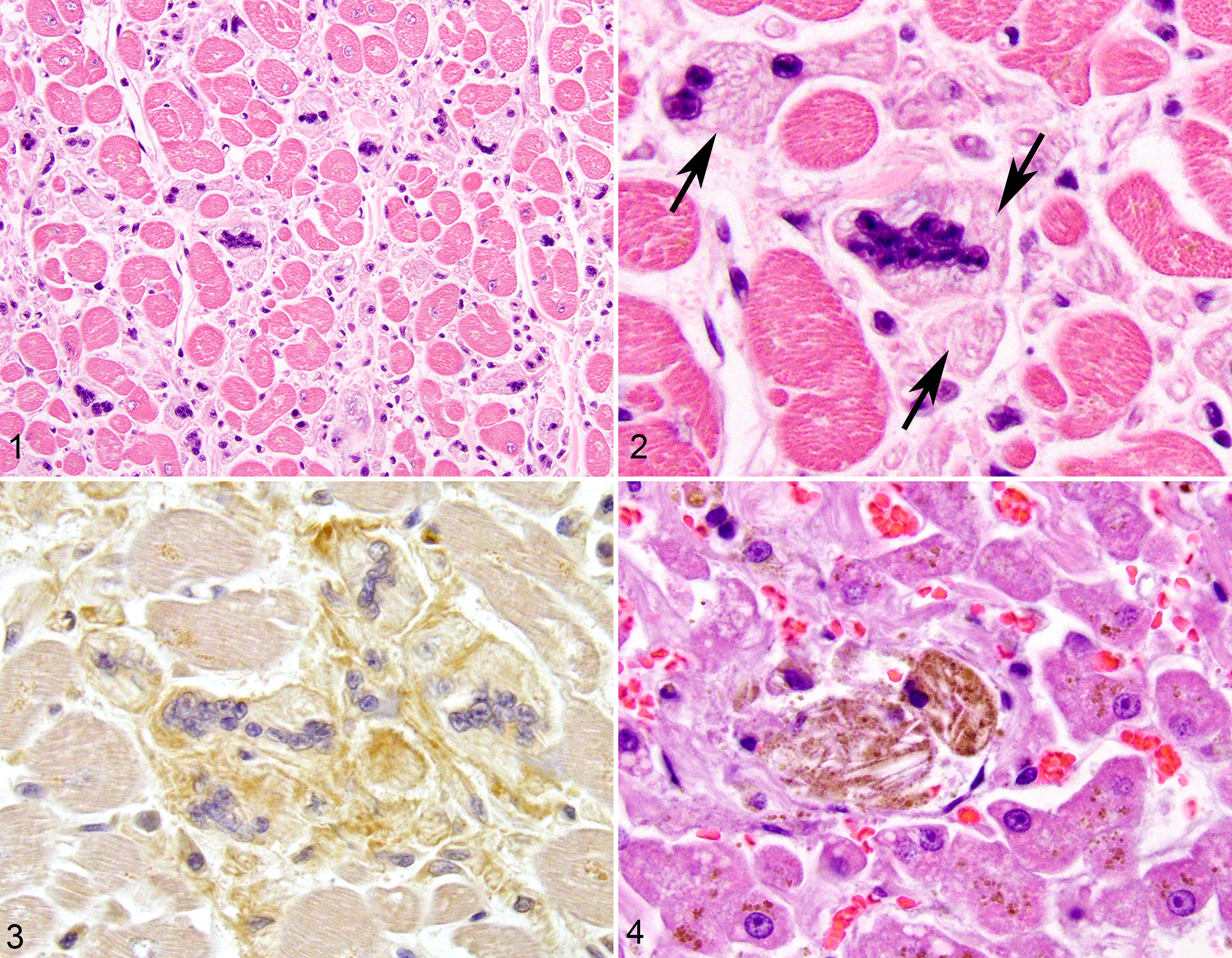
Clofazimine storage, cat.
Ultrastructurally, the spaces in myocardial macrophages were polygonal, electron-lucent, and lined by lipid membranes (Figs. 5, 6), consistent with an autophagosome. Other histologic findings included regionally extensive acute hepatic necrosis and chronic tubulointerstitial nephritis with chronic infarcts. Staining of multiple tissues with Ziehl-Neelsen and Fite-Faraco acid fast stains did not demonstrate acid-fast bacteria. Although considered a significant contributor to clinical decline, a definitive cause of the hepatic necrosis was not identified.
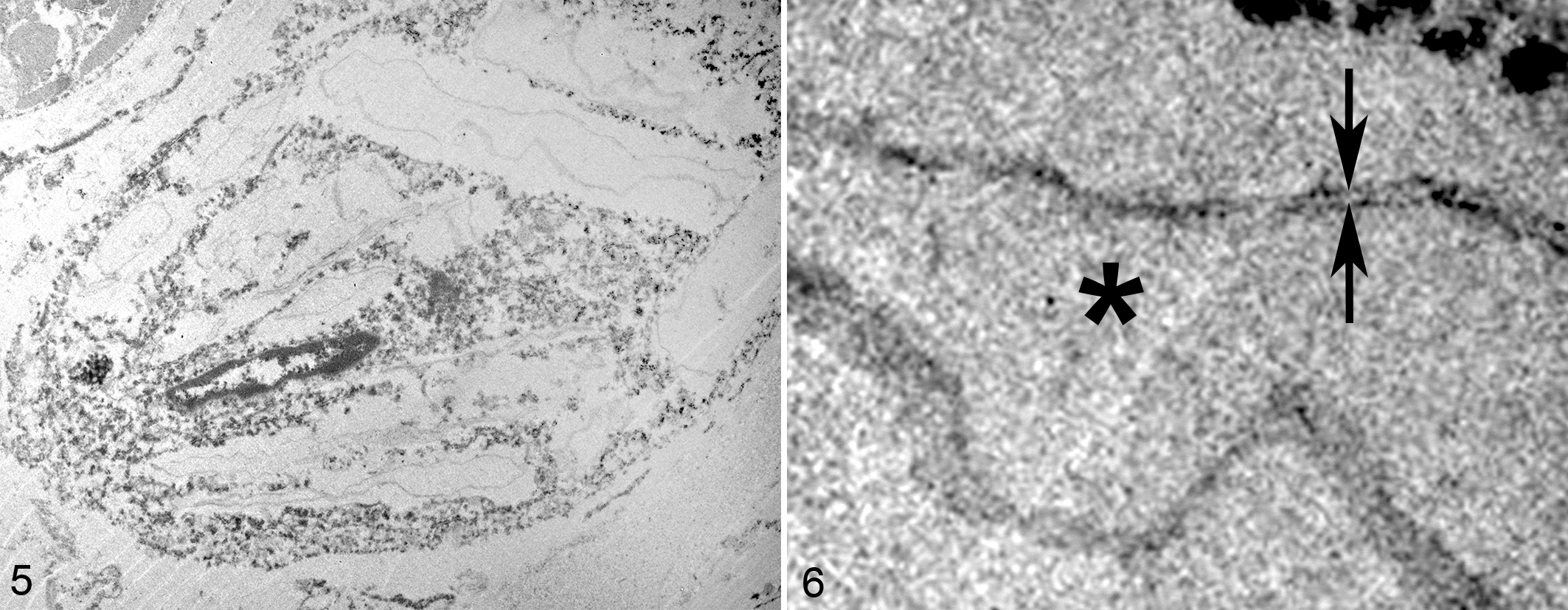
Clofazimine storage, myocardium, cat. Transmission electron microscopy.
Given the historical treatment with clofazimine and the histologic and ultrastructural appearance of the crystalline spaces in macrophages, a diagnosis of clofazimine storage was made. Clofazimine crystals are soluble in organic solvents and alcohols and are therefore lost during routine tissue processing for histology and ultrastructural study, leaving only clear acicular spaces. 24 In order to see crystals histologically, frozen sections must be examined. Frozen samples were not collected in this case.
To our knowledge, this is the first report of clofazimine accumulation within the heart of a cat. Cardiotoxicity resulting in arrhythmias has previously been reported in humans treated with clofazimine for prolonged periods 7,10 and cardiac accumulation of clofazimine has been demonstrated in humans 17 and rats. 16 However, those studies did not include histologic evaluation for comparison to this case. In mice and humans with many forms of chronic heart disease, the number of macrophages within the myocardial interstitium increases. 12 Cats with hypertrophic cardiomyopathy have recently been shown to have increased numbers of macrophages within the myocardial interstitium. 14 Although lacking the classical finding of myocardial disarray, the variably-sized myocardiocytes in this cat, the subjective cardiomegaly with ventricular thickening, and increased cardiac weight (0.78% of body weight) are suggestive of, though not diagnostic for, hypertrophic cardiomyopathy. Alternatively, the increased heart weight may have been the result of infiltration of the myocardium by clofazimine-laden macrophages. A similar phenomenon of myocardial histiocytosis has also been demonstrated in humans and mice with chronic kidney disease 5 but not, to our knowledge, in cats. The accumulation of clofazimine crystals within Kupffer cells 2 and pulmonary alveolar macrophages 13 20 22 has been previously documented in experimental and human literature and closely mirrors this case. Interestingly in this case, and in contrast to humans 23,25 and experimental models, 2 macrophages in the intestinal lamina propria did not contain clofazimine crystals. The accumulation of crystal-laden macrophages within the skeletal muscle intersititum, to the authors’ knowledge, has not been previously reported and may suggest an unusual distribution of clofazimine crystal storage in cats.
Although clofazimine is a mainstay of treatment of mycobacteriosis in cats, 15 21 adverse effects rarely have been reported. Previously reported adverse effects are limited to gastrointestinal upset and photosensitization, both of which typically resolve following cessation of the medication. This report demonstrates clofazimine crystal storage within myocardial and skeletal muscle interstitial macrophages, Kupffer cells, and pulmonary alveolar macrophages. Myocardial accumulation of clofazimine in this case could represent an adverse effect of clofazimine, or could potentially be a consequence of underlying cardiac pathological changes. In this case, the multiple adverse effects associated with administration of other medications may reflect an individual or species-specific sensitivity to various medications. Unfortunately, the role of clofazimine in the clinical decline in this case is not determined.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Helgert_et_al - Intrahistiocytic Storage of Clofazimine Crystals in a Cat
Supplemental Material, Combined_supplemental_materials-Helgert_et_al for Intrahistiocytic Storage of Clofazimine Crystals in a Cat by Nathan D. Helgert, Debra L. Miller, Jacqueline C. Whittemore and Mee-Ja M. Sula in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to acknowledge the University of Tennessee Advanced Microscopy and Imaging Center for instrument use, as well as scientific and technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
