Abstract
From July 1989 to October 1998, 9/37 (24%) adult captive tiger quolls (
The tiger quoll (
Materials and Methods
Study population
In 1984, a breeding program was established at the CRC (Front Royal, VA) based on two pairs of wild-caught tiger quolls. The majority of tiger quolls were maintained at the CRC, and a small number were kept on exhibit at the National Zoological Park (Washington, DC). Their diet consisted of Toronto Feline Diet (Milliken Wholesale Meat, Toronto, ON, Canada) supplemented with apple biscuits, live and dead mice (2–3 times/week), thawed fish (once weekly), and a multivitamin supplement. Tiger quolls were housed singly except when males were introduced into the females' enclosures for breeding encounters. Animals were housed in large indoor enclosures, either 3.1 × 6.2 × 3.4 m or 6.2 × 6.2 × 3.4 m, with access to outdoor cages of equal size. The flooring substrate was composed of pine bark, solite, and peat moss.
From 1989 to 1998, the collection had a total of 37 adult tiger quolls. During this 9-year period, skin biopsies and necropsies were preformed on nine tiger quolls with similar cutaneous disease. Table 1 contains information on gender, age, and lesion distribution for these nine animals. Six of the nine tiger quolls were born in captivity at the CRC, and three were from the original wild-caught group. The affected population included seven female and two male tiger quolls, ranging from 2.5 to 5.4 years of age at the time of diagnosis (mean = 4.2 years; median = 4.6 years).
Gender, age, and distribution of lesions for nine tiger quolls with subcutaneous atypical mycobacteriosis.
∗ At time of initial diagnosis.
Histopathology
Samples of skin were collected either as biopsies or during postmortem examination. Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5.0 µm, and stained with hematoxylin and eosin (HE). For visualization of mycobacteria in tissue sections, Kinyoun stain was used according to a previously published protocol. 25
Microbiology
For mycobacterial culturing, samples of skin, lymph node, and thoracic abscess were plated onto blood agar and Lowenstein-Jensen medium (Becton Dickinson, Cockeysville, MD) and incubated at 35 C and 7% CO2 for up to 1 week. Mycobacterial isolates were identified by high-performance liquid chromatography and conventional methods (MRL Reference Laboratory, Cypress, CA).
Cytology
Imprints and smears of subcutaneous lesions were air dried and stained with Wright's stain and modified Kinyoun acid-fast stain according to a previously published protocol. 1
Results
Clinical findings
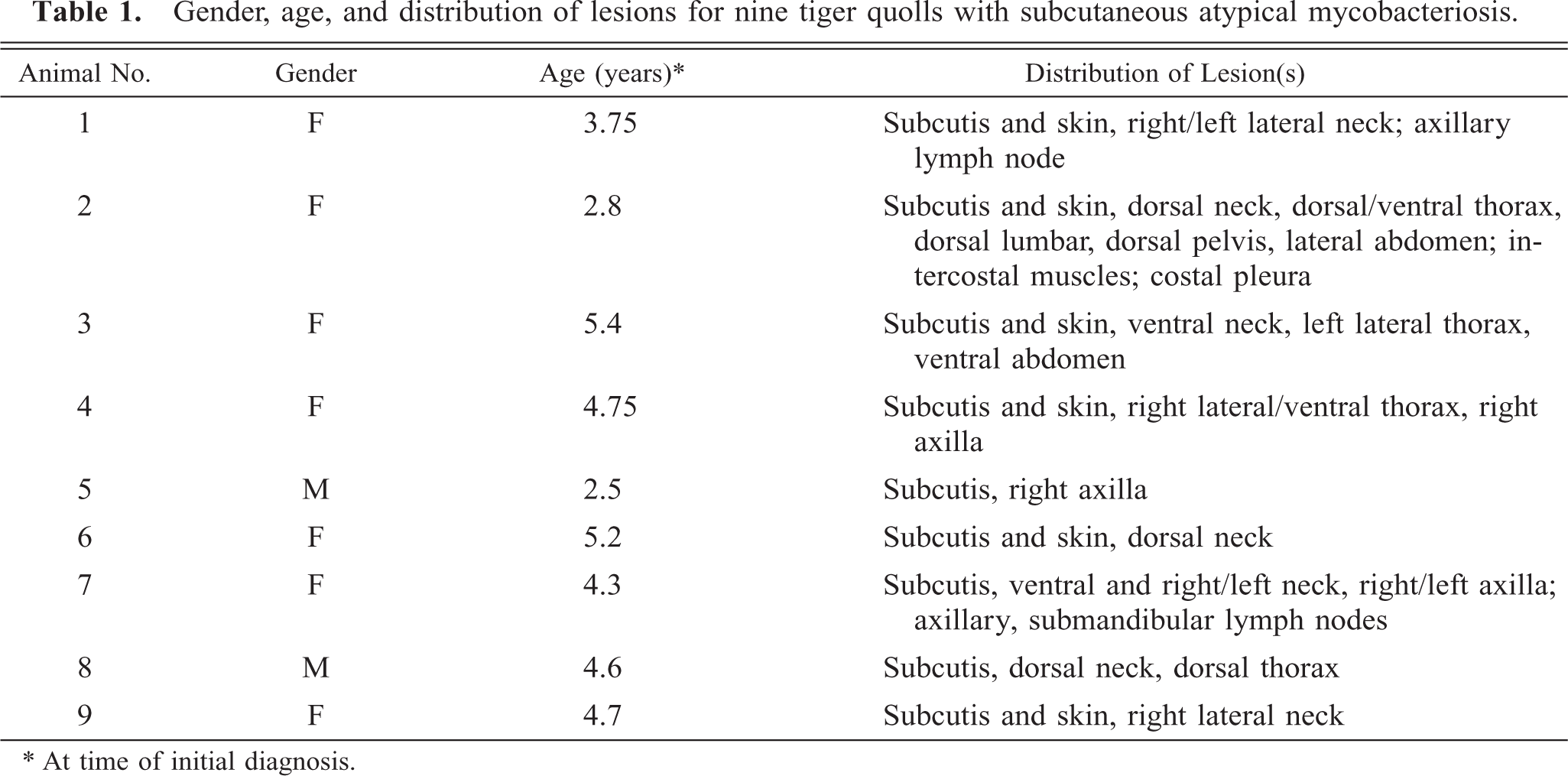
The cutaneous lesions presented as single to multiple thickened areas of skin of various sizes that were sometimes hyperemic and partially alopecic (Fig. 1). The subcutis was multifocally thickened with gray-tan plaques that contained variable amounts of thick, moist to dry, tan-yellow purulent material (Fig. 2). Multifocally, the subcutaneous tissue was edematous and hemorrhagic. The subcutaneous lesions sometimes presented as small draining abscesses or fistulous tracts that communicated with the surface of the skin. The distribution of the lesions was primarily along the dorsal, lateral, and ventral aspects of the neck and thorax (Table 1). Lesions were occasionally evident in the axillary region and rarely in the lumbar, abdominal, or pelvic regions. Two tiger quolls had enlargement of regional lymph nodes (two had enlarged axillary and one had enlarged submandibular lymph nodes). One tiger quoll had suppurative tracts that extended from the dermis and subcutis deep into the underlying intercostal skeletal musculature and communicated with an abscess adhered to the costal pleura.
Skin; tiger quoll. Gross appearance of thickened skin with alopecia from a tiger quoll with subcutaneous atypical mycobacteriosis. Bar = 5.0 cm.

Skin; tiger quoll. Cross section of skin and subcutis from a tiger quoll with subcutaneous atypical mycobacteriosis. Subcutis is edematous and hemorrhagic (arrows) and contains exudative plaques that extend into the dermis (arrowhead). Bar = 5.0 cm.
Two tiger quolls were euthanatized following chronic subcutaneous atypical mycobacteriosis that was unresponsive to treatment. One tiger quoll died during the skin biopsy procedure. One tiger quoll died from spread of subcutaneous mycobacterial infection into the thoracic cavity. The remaining five tiger quolls died or were euthanatized because of other conditions unrelated to the mycobacteriosis.
Cytology
Impression smears of subcutaneous abscesses and plaques were made for all nine tiger quolls. Serial cutaneous smears were examined during the course of the disease for several tiger quolls. In all cases, there was an inflammatory cell population characterized by neutrophils, histiocytes, and few epithelioid macrophages, Langhans giant cells, lymphocytes, and plasma cells. Several extracellular beaded to filamentous acid-fast bacilli (AFB) were noted cytologically in all nine tiger quolls.
Histopathologic findings
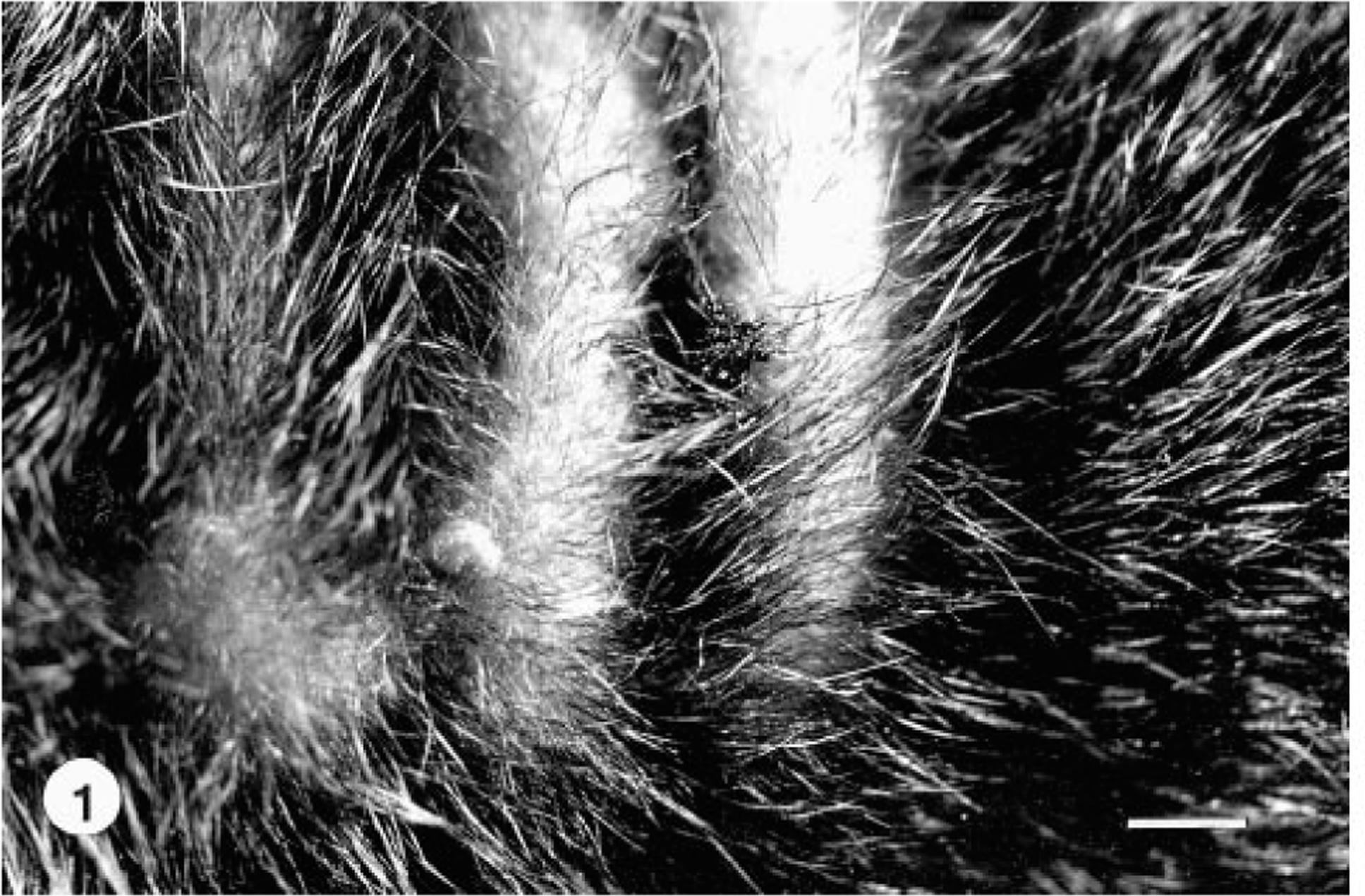
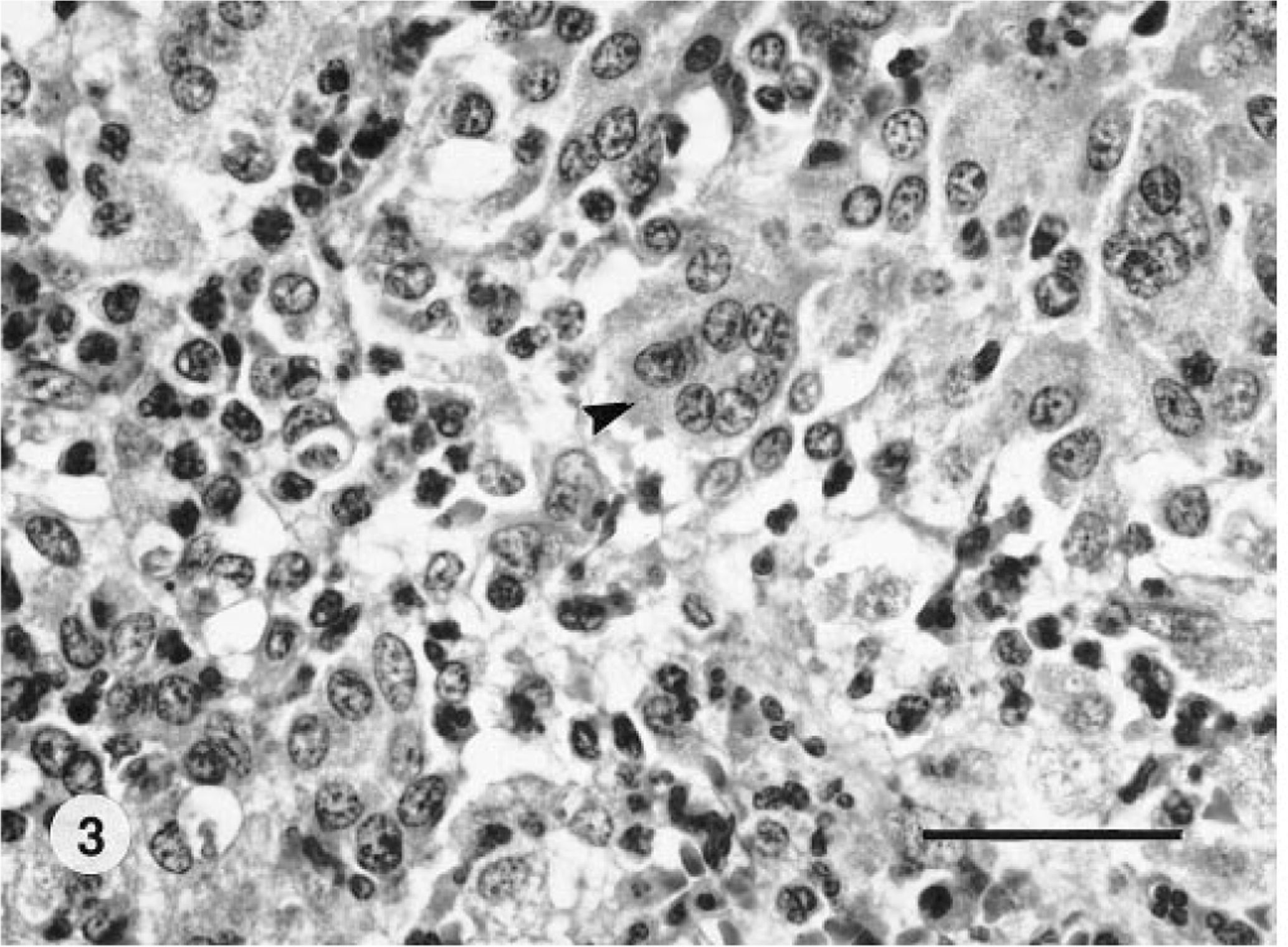
Histologic lesions in the subcutis were similar in all nine tiger quolls. The predominant lesion was nodular to diffuse pyogranulomatous panniculitis and cellulitis. The inflammatory infiltrate was characterized by histiocytes, neutrophils, epithelioid macrophages, lymphocytes, plasma cells, and few Langhans giant cells and mast cells combined with fibrous connective tissue (Fig. 3). Multifocally within the pyogranulomatous inflammation were regions of necrosis, mineralization, and hemorrhage of various sizes. The inflammatory infiltrate expanded and effaced normal subcutaneous adipose tissue. In six animals, the inflammation extended into the dermis, forming fistulous tracts. The overlying epidermis was ulcerated in four animals. One tiger quoll (No. 2) had an extension of the pyogranulomatous inflammation into the underlying intercostal skeletal musculature and costal pleura, and another tiger quoll had involvement of dermal sebaceous glands. In two tiger quolls (Nos. 1, 7), there were neutrophils, histiocytes, and few epithelioid macrophages within subcapsular and medullary sinusoids of regional axillary (two animals) and submandibular (one animal) lymph nodes. In 7/9 (78%) tiger quolls, few aggregates of extracellular beaded to filamentous AFB were evident within large, clear lipid vacuoles embedded within the pyogranulomatous inflammation (Fig. 4). In two tiger quolls (Nos. 1, 9), AFB were not demonstrated histologically but were demonstrated by cytology of abscess exudate, and atypical mycobacteria were cultured from respective skin samples.
Subcutis; tiger quoll. The subcutis is infiltrated by histiocytes, lymphocytes, epithelioid macrophages, neutrophils, and multinucleated giant cells (arrowhead). HE. Bar = 60 µm.
Subcutis; tiger quoll. Multifocal, clear, lipid vacuoles are surrounded by pyogranulomatous inflammation within the subcutis. A few small filamentous, extracellular bacteria (arrow) are within vacuoles. HE. Bar = 30 µm.
Microbiologic findings
Seven tiger quolls were positive for group IV mycobacteria by aerobic culture. Table 2 contains a list of the species of mycobacterium isolated.
Mycobacterium species isolated from seven tiger quolls with subcutaneous atypical mycobacteriosis.

Treatment
Eight tiger quolls were treated empirically for atypical mycobacterial infection of the subcutis and skin. The duration of treatment varied from several weeks to >4 months. Antimicrobial agents used were amikacin (Fort Dodge Laboratories, Fort Dodge, IA), enrofloxacin (Bayer Corp., Shawnee Mission, KS), cephalexin (Teva Pharmaceuticals, Sellersville, PA), ciprofloxacin (Bayer Corp.), chloramphenicol (Sanofi, Overland Park, KS), rifabutin (Pharmacia and Upjohn Corp., Kalamazoo, MI), and rifampin (Hoechst Marion Roussel, Kansas City, MO) at appropriate dosages. In a majority of cases, affected areas were surgically debrided prior to antibiotic therapy. Some animals responded temporarily to therapy, but most were refractory to treatment. In addition, several tiger quolls had recurrence of atypical mycobacterial infection in new cutaneous locations. Only two tiger quolls (Nos. 6, 9) that responded to treatment had apparent resolution of skin lesions until their death; however, skin from previously affected areas from one of these tiger quolls (No. 9) was lost to follow-up examination.
Discussion
From 1989 to 1998, 24% of captive adult tiger quolls in the National Zoological Park collection were diagnosed with atypical mycobacterial infection that involved primarily the subcutis and overlying dermis. This condition was the most commonly diagnosed disease in the collection. The disease had a similar clinical presentation in all nine tiger quolls, with thickened areas of skin and subcutis primarily along the neck and thorax. Histologic lesions were similar in all tiger quolls and consisted of pyogranulomatous panniculitis and cellulitis. One or more different species of Runyon group IV mycobacteria were cultured from the skin of seven tiger quolls. The disease in these tiger quolls is similar to subcutaneous atypical mycobacteriosis (SAM) in other animals and humans. 3 , 38
In humans, cutaneous infection with rapidly growing (atypical) mycobacteria is usually an opportunistic disease, particularly in patients that are immunocompromised.
3
Humans cases of chronic cutaneous abscessation due to atypical mycobacteria have been associated with previous trauma to the skin.
33
Cutaneous infections with atypical mycobacteria in humans can also be nosocomial and have been associated with contaminated water in medical devices.
3
SAM is most commonly reported in domestic felids and rarely in dogs and other animals.
8
,
9
,
13
,
17
,
19
,
29
,
34–37
It is caused by infection with one or more different Runyon group IV mycobacteria.
In cats, lesions due to infection with atypical mycobacteria are usually within the dorsal lumbosacral or ventral abdominal and inguinal regions. 19 , 29 , 34 , 36 Typically, the clinical presentation is firm, plaquelike, warm, and sometimes painful swellings within the subcutis and skin. Draining fistulous tracts frequently are associated with the masses. 34 The distribution of the skin infection in the tiger quolls was different from that in domestic cats in that it was concentrated primarily in the dorsolateral cervical and thoracic regions. Only one tiger quoll had lesions along the ventral abdomen. However, the clinical presentation and gross lesions of infection in tiger quolls were similar to that seen in domestic cats.
Histologically, SAM in cats manifests as nodular to diffuse pyogranulomatous panniculitis and dermatitis. The inflammation can extend into the superficial dermis and subjacent skeletal muscle. Usually, within the pyogranulomatous inflammation there are smooth-bordered extracellular lipid vacuoles that contain fine filamentous to beaded extracellular AFB. 21 The histologic lesions in the tiger quolls of this study were identical to those described in cats diagnosed with SAM. AFB were not observed histologically in two animals, but all tiger quolls were positive for AFB by cytology. It appears that cytology is a more sensitive method than histology for detecting AFB in tiger quolls with SAM.
An apparent association exists between cutaneous trauma and SAM in domestic cats. 19 In one study, all cats infected with atypical mycobacteria had previously sustained trauma to the skin via bite wounds or road accidents. 36 Similar cutaneous injuries have been described in dogs with atypical mycobacterial infections of the skin. 13 A delay of months to years can occur between the initial skin injury and clinical presentation. Tiger quolls reportedly have protracted breeding that sometimes involves the infliction of bite wounds by males into the females' necks. 7 , 10 , 28 Approximately 3 months after copulation, females dig nests in the substrate, which they frequent until parturition. Because group IV mycobacteria are saprophytes found typically as soil and water contaminants, 11 the substrate used in the tiger quoll enclosures was suspected as a source of infection. Open skin wounds acquired during mating may have become infected via contaminated substrate. However, repeated attempts to culture atypical mycobacteria from the substrate were unsuccessful. Also, not all cases could be easily described by this scenario. Long intervals from the time of bite wounds to clinical presentation were observed in tiger quolls with SAM; some of these intervals were >2 years. One female and both male tiger quolls had no recorded history of cutaneous injury; however, unnoticed skin trauma cannot be discounted in these cases.
Marsupials are more susceptible to mycobacterial infections than are eutherian mammals.
12
,
24
Most case reports of mycobacteriosis in marsupials are from captive populations. The prevalence and susceptibility of wild brushtail possums (
Antibiotic and surgical treatment of cats infected subcutaneously with atypical mycobacteria is usually unsuccessful. 34 Surgical resection of mycobacterial skin lesions usually results in wound dehiscence and possible occurrence of new lesions. 17 However, there are a few reported cases of remission in cats with or without treatment. 18 , 27 , 29 Most (75%) tiger quolls treated for SAM failed to respond to any type of antimicrobial or surgical therapy. Two of eight tiger quolls treated for atypical mycobacteriosis had apparent clinical resolution of infection.
Tiger quolls are particularly susceptible to atypical mycobacterial infections, which manifest primarily as pyogranulomatous panniculitis. Diagnosis is best made by histopathology and cytology combined with mycobacterial culture of skin lesions. The substrate likely served as a source of infection, and mycobacteria may well have entered the skin through cutaneous wounds acquired during mating interactions. The delay in onset of clinical signs can sometimes be very long, and not all tiger quolls had historic evidence of cutaneous trauma. Atypical mycobacterial infection is an important differential diagnosis for pyogranulomatous skin disease in captive tiger quolls.
Footnotes
Acknowledgements
We thank D. Fischer, A. Bratthauer, and V. Bonshock (NZP, Smithsonian) for laboratory assistance, Dr. Phillips (George Washington University) for lymphocyte proliferative assay results, R.-A. V. Ferris (Armed Forces Institute of Pathology) for photography, and Dr. Collins, Dr. Citino, and the tiger quoll keeper staff at CRC. The project was supported by FONZ SR. Fellowship 98-3545-A.
