Abstract
In disease, blood vessel proliferation has many salient roles including in inflammation, when granulation tissue fills superficial defects, or in the recanalization of an occluded blood vessel. Sometimes angiogenesis goes awry—granulation can be exuberant, and plexiform proliferation of vascular components can contribute to pulmonary hypertension. This review focuses on the diverse manifestations of pathologic vascular overgrowth that occur in the brain, spinal cord, and meninges of animals from birth until old age. Entities discussed include systemic reactive angioendotheliomatosis in which glomeruloid vascular proliferations are encountered in various organs including the central nervous system (CNS). The triad of CNS vascular malformations, hamartomas, and benign vascular proliferations are an especially fraught category in which terminology overlap and the microscopic similarity of various disorders makes diagnostic classification incredibly challenging. Pathologists commonly take refuge in “CNS vascular hamartoma” despite the lack of any unique histopathologic features and we recommend that this diagnostic category be abandoned. Malformative lesions that are often confusing and have similar features; the conditions include arteriovenous malformation, cavernous angioma, venous angioma, and capillary telangiectases. Meningioangiomatosis, a benign meningovascular proliferation with dual components, is a unique entity seen most commonly in young dogs. Last, accepted neoplastic conditions range from lower-grade locally acquired growths like hemangioblastoma (a tumor of mysterious interstitial stromal cells encountered in the setting of abundant capillary vasculature proliferation), the rare hemangioendothelioma, and the highly malignant and invariably multifocal metastatic hemangiosarcoma. Additionally, this review draws on the comparative medical literature for further insights into this problematic topic in pathology.
Keywords
Proliferative vascular disorders include exuberant disturbances of angioarchitecture (vascular malformations), reactive vascular proliferations, as well as borderline and neoplastic vascular lesions. At one end of the spectrum, the developmental disturbances are characterized by an architectural disarray of blood vessels formed by mature, well-differentiated cells. At the other end sits the vascular endothelial sarcomas featuring high cellularity, anaplasia, and pleomorphism, which in areas may lack any semblance of normal vascular structure.
In total, this group constitutes a heterogeneous mixture of nonneoplastic and neoplastic conditions in human and veterinary pathology. Many are idiopathic with little known of their pathogenesis. Two members of this broad family of vascular pathologies are problematic: developmental malformations (some called hamartomas) and benign neoplasms. For these two, difficulty in their distinction is acknowledged and definitive hallmarks remain to be identified; proffered diagnoses can be confusing and are sometimes contentious. When occurring in the central nervous system (CNS) of domestic animals, disagreement in diagnostic terms is compounded by the sparsity of cases and thus a paucity of literature to consult.
Pathologists draw on historical, clinical, and imaging data in concert with their evaluation of patients’ tissues, utilizing conventional histologic morphology, aided by histochemical and immunohistochemical (IHC) staining, nucleic acid hybridization protocols, and sometimes ultrastructure. The pathologists’ role is also to anticipate clinical outcome but for the CNS parenchyma there are complications intrinsic to any expansile mass lesion arising there, vascular or otherwise. Such sequelae are dependent on neurolocalization, as when essential structures are effaced, cerebrospinal fluid flow is obstructed, or any induced vasogenic edema is sufficient to cause brain herniation. Consequently, the prognosis for a low-grade lesion may be intrinsically favorable but grim, if in an area prone to the complications described.
The goal of this article is to review the literature pertaining to this diverse group of proliferative vascular lesions arising within or otherwise involving the CNS in domestic animals, propose a rationale for selection of the most appropriate diagnostic terms, and discuss the associated controversies. This review does not include the perivascular (vascular wall) neoplasms nor those of lymphatic vascular origin. In terms of the neoplastic cohort that we will discuss, preferred names vary around the globe, and for the sake of this discussion, we equate angioma with hemangioma, angiomatosis with hemangiomatosis, and so on. Different rules apply for vascular endothelial malignancies; the veterinary profession avails itself of the terms hemangiosarcoma or lymphangiosarcoma to convey blood and lymphatic vessel endothelial malignancies respectively, whereas the medical profession prefers the single term angiosarcoma, which includes both possibilities, to be further defined by additional procedures. For ready comparison, the entities to be discussed are summarized in Table 1.
Synopsis: Proliferative Vascular Lesions of the Central Nervous System in Domestic Animals.
Abbreviations: CNS, central nervous system; FVIII-RAg/vWF, factor VIII-related antigen (von Willebrand factor).
a Indicates published reports of lesions with similar features to those described herein, but not given that diagnosis in the published report.
Reactive Vascular Proliferations: Systemic Reactive Angioendotheliomatosis
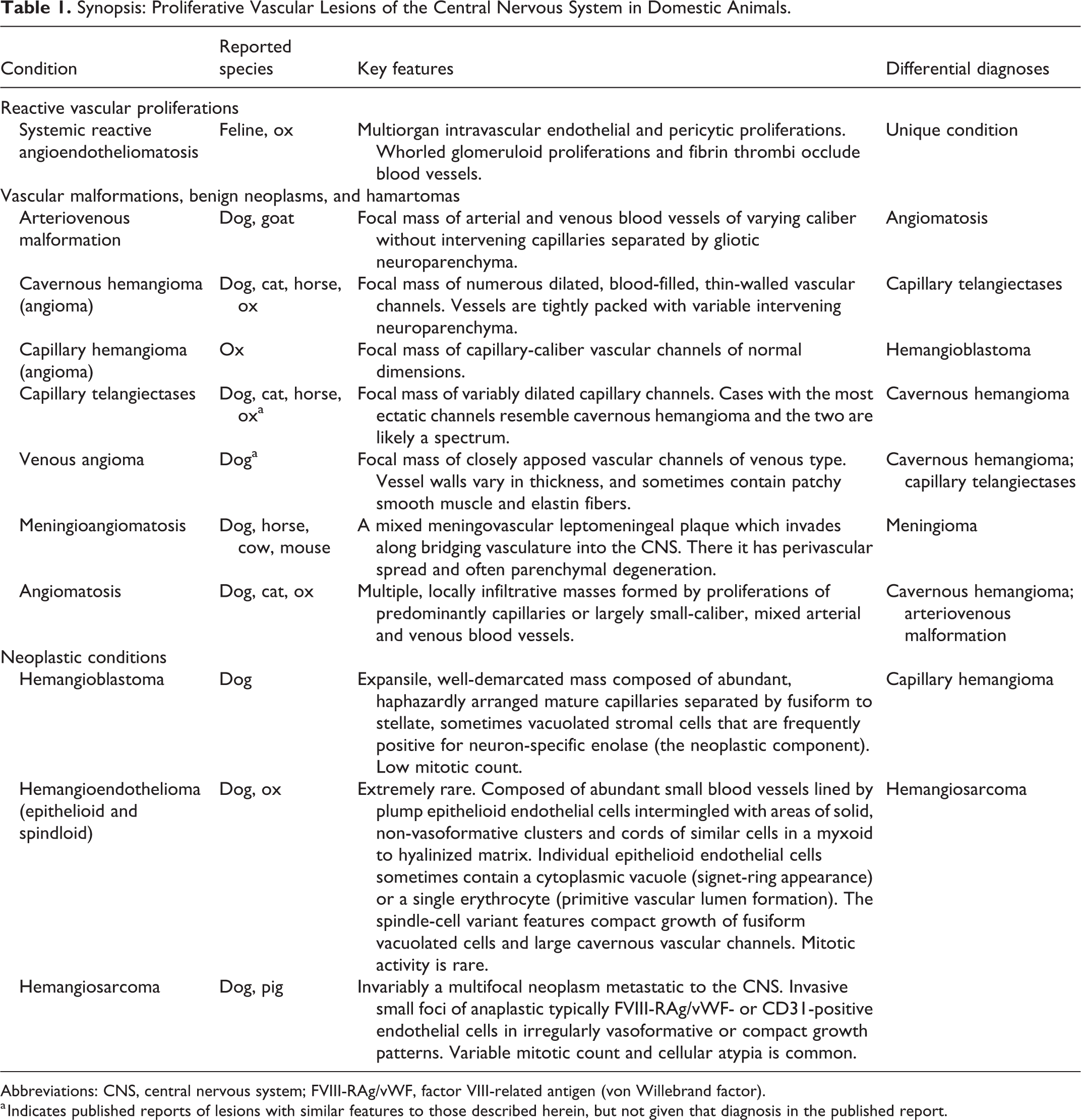
Systemic reactive angioendotheliomatosis is a nonneoplastic proliferative disorder of unknown etiology characterized by multifocal intravascular endothelial and lesser pericytic proliferation in blood vessels throughout the body. Affected vessels are partially to completely occluded by whorled glomeruloid proliferations of spindle cells (Fig. 1), which, using IHC markers and ultrastructural studies, consist of an admixture of endothelial cells and pericytes. 29 The abnormal vessels are sometimes further occluded by fibrin thrombi. Historically, the term angioendotheliomatosis was utilized in humans and domestic animals to identify novel, widespread, purely intravascular neoplastic proliferations which proved in many cases not to be of vascular origin but examples of intravascular or angiotropic lymphoma. 25
Systemic reactive angioendotheliomatosis, cerebellum, cat. Within the cerebellar granular layer are multiple whorled glomeruloid vascular proliferations with fibrin partially to completely occluding lumina. Hematoxylin and eosin (HE).
In veterinary medicine, systemic reactive angioendotheliomatosis is best described in cats, a syndrome termed feline systemic reactive angioendotheliomatosis. To date, fewer than 15 cases have been reported, all with similar vascular lesions to those described above. 20,29,84,90,102 In these reports, the organs most commonly affected included the heart, kidneys, spleen, lymph nodes, gastrointestinal tract, brain including meninges, eyes, and pancreas. More rarely affected were liver, spinal cord, adrenal glands, and thyroid glands, and in only one case each were lesions in the sciatic nerve, subcutis, lung, bone marrow, and urinary bladder. Involvement of the myocardial vasculature is most common and some cats may present with cardiac insufficiency and pulmonary edema. While CNS involvement is recorded, neurological signs do not feature in the clinical records. Affected cats sometimes have concurrent coagulopathies, manifesting as petechiae and ecchymoses; however, it is unknown if they are contributory to the lesion or simply a consequence of them. An unpublished report proposed that 2 cats with FRSA qualified as thrombotic thrombocytopenic purpura (American College of Veterinary Pathologists [ACVP] Annual Meeting, 2004, Abstract #121).
There is one case report of a comparable systemic vasoproliferative disorder in a 2-year-old steer. The vascular changes were similar to those in the feline cases, and again, these abnormal vessels were most prominent in the heart, though also involved the liver, lung, lymph nodes, kidney, adrenal gland, and brain. 12 IHC stains as employed in feline studies similarly highlighted intraluminal proliferations of endothelial cells and pericytes. 12 This steer was persistently infected with bovine viral diarrhea virus (BVDV), and IHC labeling for BVDV antigen was occasionally associated with the glomeruloid intravascular proliferations. 12 Whether BVDV infection played a direct role in the development of the systemic vasoproliferative lesions is not known. 12
A similar widely disseminated entity has not been reported in humans. The histologic appearance most resembles human reactive angioendotheliomatosis in which the lesions are limited to the skin. 53 Vasoproliferative conditions have been linked to infections with Bartonella spp. in humans, termed bacillary angiomatosis. In one study, Bartonella spp. DNA was isolated from formalin-fixed paraffin-embedded tissue samples of the lesions from 4 cats with feline systemic reactive angioendotheliomatosis and the previously described steer, but the significance of this finding in regards to the pathogenesis of vasoproliferation remains to be established. 5
Vascular Malformations, Benign Neoplasms, and Hamartomas
There would be little resistance from either medical or veterinary pathologists to the proposal that differentiating between some vascular malformations and some benign vascular neoplasms is next to impossible or only arbitrary. 22,25 Given this state of affairs, the medical profession has for many years pragmatically chosen to employ these diagnostic terms seemingly as synonyms, such as equating cavernous angioma (aka hemangioma) with cavernous malformation. In veterinary medicine, the distinction between the 2 groups is maintained, although, in practice, there is considerable overlap in the choice of terms. Only rare veterinary reports have employed the medical approach, such as Zaki’s 1979 paper entitled “Vascular Malformation (Cavernous Angioma) of the Spinal Cord in a Dog.” 118 This fundamental difference in the practice of pathology highlights the difficulty in directly relating the medical and veterinary literature for cases in which the discussion of some vascular malformations and some benign neoplasms is inseparably woven together and often used interchangeably. A further point of divergence is that unlike in the medical profession, in veterinary pathology, the term vascular hamartoma is used as a further diagnostic category. Consider a focal hemorrhagic mass within the CNS composed of abundant, variably sized, thin-walled vessels, which is examined by 2 pathologists. The MD pathologist might favor the diagnosis of “cavernous angioma (also known as cavernous malformation)” while the DVM pathologist might advocate any of 3 diagnoses: “cavernous malformation,” “cavernous hamartoma,” or “cavernous angioma/hemangioma.” The hamartoma option presents its own controversies and further muddies the waters, as, for example, one veterinary pathologist’s hamartoma may be another’s hemangioma.
As routine histopathologic examination alone of these cases is problematic, other informative alterations such as genetic changes have been sought. In humans, the expression of the glucose transporter protein GLUT1 has been used to label neoplastic endothelial cells in capillary hemangioma in skin and soft tissues, contrasted with vascular malformations, which do not express this protein. 25 However, in humans, the endothelium in cerebral congenital vascular malformations appear to also express GLUT1, limiting its diagnostic potential in the CNS. 62 In veterinary medicine, no such differentiating marker has been determined, so one is left with the unfortunate uncertainty of attempting to tease out an accurate diagnosis using histomorphological pattern, a limited array of histochemical and IHC stains, and clinical presentation. Some aspects of the histogenesis of vasoproliferative disorders are novel. Cutaneous infantile hemangioma is common in childhood; it has long been hypothesized that the transformed endothelia are of placental or maternal (rather than host) origin, thus accounting for the congenital nature of the mass. This proposal remains under debate with evidence provided for and against. 4,81 In the capillary type, over 70% show spontaneous regression, 112 such that surgical intervention may not be required. This has been recorded in a calf with congenital cutaneous hemangiomas, which within 8 months had disappeared. 78
CNS vascular malformations and related benign neoplasms are considered together. CNS vascular malformations in human patients occur in the brain and spinal cord parenchyma, the leptomeninges, and dura.
15
Those of the neuroparenchyma are generally identified in humans as one of the following: Arteriovenous malformations Cavernous angiomas Venous angiomas Capillary telangiectases
Together, these vascular malformations within the CNS come to clinical attention over a wide age range in infants and adults. As discussed above, in some quarters, the choice to name some vascular malformations as angiomas is an understandable cause of confusion.
Arteriovenous Malformations
Arteriovenous malformations are the most serious of this quartet in humans, representing a potential cause of fatal hemorrhage or ischemia. A tangled mass of arterial and venous blood vessels without intervening capillaries occurs in the territory of one of the major cerebral arteries, separated by gliotic tissue (Fig. 2). Blood at arterial pressure directly enters and distorts the venous channels that become dilated and thickened. In contrast to the other types, vessels of arteriovenous malformations (mainly the arterial component) have prominently thickened walls and are lined by a single layer of endothelial cells.
While common in humans, remarkably few arteriovenous malformations have been identified in the veterinary literature. Of three in dogs, two were young, aged 6 months and 10 months, while one was 8 years. The variation in age may indicate that these lesions can occur as congenital malformations which may be clinically silent, possibly for years. In the few canine cases recorded, they show a preference for the meningeal vasculature of the cervical, caudal thoracic, and thoracolumbar spinal cord segments. 18,39,67 These lesions in the leptomeninges or the extradural space of the vertebral canal can be visualized grossly and via imaging modalities such as computed tomography (CT), magnetic resonance imaging (MRI), and angiography. 18,39,65,67 In one canine case, anomalous vessels also penetrated and effaced the spinal cord parenchyma. 39 The abnormal vessels typically have thickened walls, reported to be due to a mixture of collagen and smooth muscle. 18 The arterial vessels may also have a demonstrable elastic tunic, highlighted by Verhoeff-Van Gieson staining. 39,65
A somewhat similar vascular mass lesion in a 10-month-old goat was composed of artery-like vascular channels. It was closely associated with the dorsolateral aspects of the T1–T3 vertebrae and extended into the adjacent subcutis and skeletal muscle, the vertebral canal, and left hemithorax. 65 Compression of the cranial thoracic spinal cord resulted in progressive paraparesis and thoracic limb hypermetria. The vessels composing the mass were thick-walled and were immunolabeled for desmin and actin, indicative of a smooth muscle component. This case was not deemed to be an arteriovenous malformation but a vascular hamartoma, a vague term that is further discussed in a later section of this article. Other cases of compressive myelopathy resulting from intradural-extraparenchymal vascular anomalies have been described in the dog. 11,114 These also were not viewed as examples of arteriovenous malformation and described as vascular ectasias or anomalies, respectively.
Cavernous Angiomas
Cavernous angiomas (or hemangiomas) in human patients are also accepted as cavernous malformations. These vasoproliferations occur widely in the CNS parenchyma and the meninges with a male predominance and come to attention in the first 4 decades of life with roughly 50% of patients developing seizures. 15 Cavernous angiomas can coexist with capillary telangiectases and they may be a spectrum.
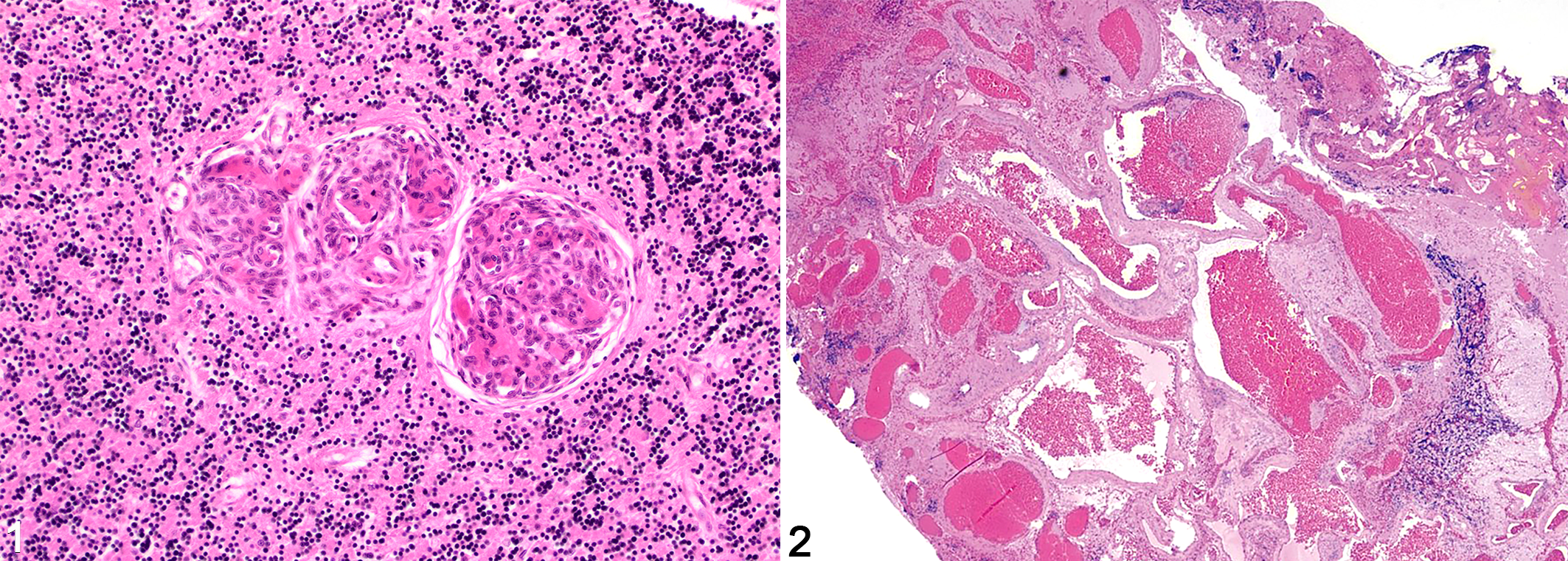
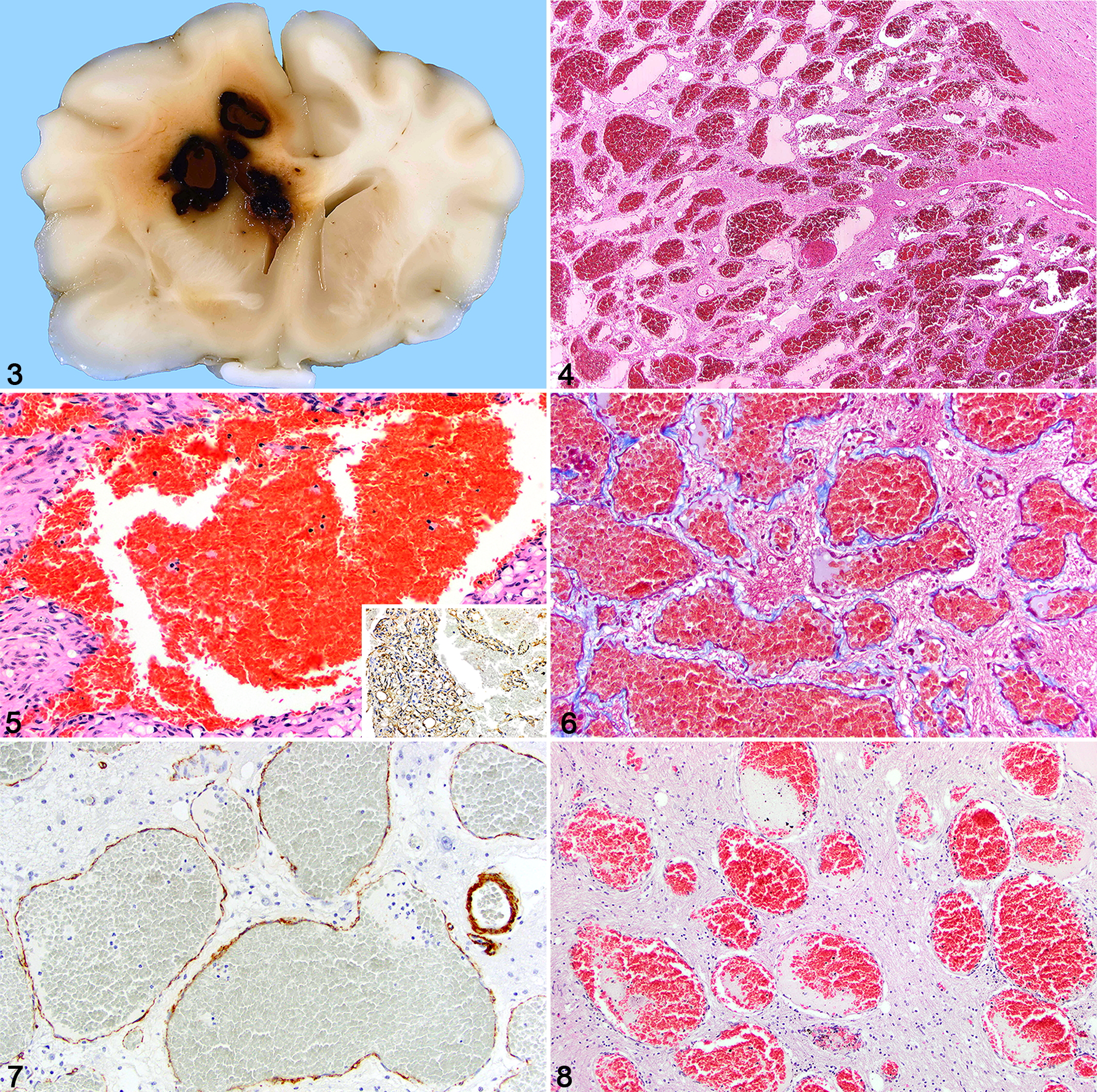
In veterinary medicine, cavernous and capillary hemangiomas (angioma is less frequently used) are also encountered in the CNS. In domestic animals, there are more reports than of arteriovenous malformations, and it is probable that some cavernous types have been published as vascular hamartomas. Most CNS hemangiomas are recognized in dogs, and the clinical signs are related to the location, size, and degree of hemorrhage associated with the space-occupying mass. 18,21,46,87,91,94,118 Grossly, these masses are typically nodular to multilobular, expansile to mildly invasive, cavitated with dark red gelatinous blood contents, and compress or efface the surrounding neuroparenchyma, sometimes with areas of surrounding hemorrhage (Fig. 3). 18,21,94,118 Microscopically, numerous small to large thin-walled and variably hyalinized vascular channels are tightly packed, sometimes thrombosed, and separated by a variable amount of intervening neuroparenchyma and sometimes hemosiderin and mineralization (Figs. 4–7). They occur in the brain (3 affecting the rostral cerebrum and olfactory bulbs 21,91 and 1 the temporal lobe 94 ) and the spinal cord (2 cervical 18,118 and 3 thoracic 18,46,87 ). Cases with similar masses are reported in a cat, 60 a foal, 31 and a calf. 41
Cavernous hemangioma, brain, cat. A demarcated hemorrhagic mass expands from the centrum semiovale into the corpus callosum and caudate nucleus. The adjacent edematous white matter is blood-tinged while widespread brain swelling has caused a midline shift.
As the names imply, capillary hemangioma is distinguished from cavernous hemangioma by the smaller size of vascular channels that form the mass. One case diagnosed as an angiomatous vascular malformation in the L5–L6 spinal cord of a Hereford calf could be considered consistent with a capillary hemangioma. 16
In humans, cavernous angioma has an excellent prognosis with surgical excision. 51,58,89 In the documented canine cases, the 2 reports of complete surgical removal resulted in improvement or resolution of neurologic clinical signs and no evidence of recurrence of the mass at 13 and 14 months, respectively, by postoperative MRI. 87,94
Venous Angiomas
Venous angiomas are typically detected as an incidental finding on imaging the human brain and only occasionally are symptomatic; they can be compared to varicose veins. They are dilated veins and their branches, with thickened collagenous walls. A common location is the subarachnoid space of the thoracic spinal cord, and these qualify as a varix. To our knowledge, there are no reports in domestic animals, although one case described by MacKillop et al in 2007, diagnosed as a cavernous malformation, could be consistent with a venous angioma. 59 Occurring in the L4 spinal cord parenchyma, the mass was histologically composed of closely apposed vascular channels lined by a single layer of uniform endothelium with variably thin to slightly thickened walls. IHC labeling for smooth muscle actin (SMA) highlighted a patchy layer of smooth muscle cells in the thickened vascular walls, and Verhoeff-Van Gieson staining revealed accumulation of elastic fibers in these areas, suggestive of venous morphology. 59
Capillary Telangiectases
Capillary telangiectases of the CNS in humans are also an incidental finding in most cases. Telangiectasis describes a single dilated vessel, whereas telangiectases refers to a cluster of dilated capillaries characterized by a small intraparenchymal expansile mass formed by mostly thin-walled, variably sized blood vessels with intervening neuroparenchyma separating the vascular spaces. The vessels are lined by a single layer of endothelium and supported by a thin collagenous stroma, and may be variably dilated, occluded by thrombi, and/or associated with adjacent parenchymal hemorrhage. The endothelial lining is identified by IHC for factor VIII–related antigen (von Willebrand factor), and adjacent ablumenal SMA-positive cells are interpreted as pericytes, while a collagen matrix is highlighted by Masson’s trichome stain. 42,59,86,97 Intervening neuroparenchyma can be identified by appropriate IHC markers such as synaptophysin, glial fibrillary acidic protein (GFAP), myelin basic protein, and neurofilament. 42,86,97
The precise nature of such lesions in humans has been questioned: Are these true capillaries or simply dilated venules? 15 Capillary telangiectasis would appear as a group of dilated, very thin walled vessels, but there appear to be no reported cases in domestic animals (Fig. 8). Furthermore, some of the cases reported as canine vascular hamartomas may qualify; lesions with this histologic pattern have been described in various breeds with no sex predisposition and ages ranging from 4 to 17 years. 23,42,59,86,97 Canine vascular hamartomas typically occur as a solitary mass and are reported most commonly in the cerebrum, with one case each in the cerebellum, and lumbar and thoracic spinal cord. Clinical signs are related to the location and size of the mass, due to their space-occupying nature, and associated hemorrhage and ischemia. In the reported cases, intracranial masses often resulted in seizures, although in one case, the mass was an incidental finding 86,97 as is common in humans. Spinal cord lesions resulted in stereotypic signs of an ectatic compressive mass correlated to the location of the lesion. 59
In other species, reported cases with described histologic features of capillary telangiectases are more rare, with reports in a cat, 100 a horse, 10 and an ox. 107 The prognosis and potential for lesion progression in domestic animals is unknown, as all animals in the reported cases either died or were euthanized secondary to neurologic clinical signs.
Some of these 4 syndromes may be related and a classification which recognizes 3 rather than 4 variants of CNS vascular malformation may be more sustainable. Cavernous hemangioma has similar histologic and immunohistologic features to capillary telangiectases. Depending on the chronicity of the lesions, the histologic patterns of capillary telangiectases and cavernous hemangioma may reflect a spectrum of the same entity, as compression from congested blood-filled vascular spaces is noted to cause pressure atrophy of the intervening neuroparenchyma. 46,87,91,94,118 While the suffix “-oma” typically reflects a benign neoplastic entity, it is unclear if all of these lesions are truly neoplastic, a long acknowledged issue. Masses with the phenotype of cavernous hemangioma have been reported in dogs as young as 4 months; therefore, a malformation may also be considered. 118 In the absence of objective and readily applied means of differentiation, the practice in human neuropathology of equating some vascular malformations and benign tumors is understandable.
The diagnosis of a hamartoma is generally made in the setting of a focal mass lesion, in some cases congenital, which is composed of malformed or overabundant tissue elements appropriate to that site. In humans, hamartomas are recognized in many organs and tissues such as liver, heart, spleen, and lung, to name a few. 25 Some are well characterized clinically and pathologically such as fibrous hamartoma of infancy, for which the body sites of predilection and a male preference are recognized. 25 A considerable number are also recorded in domestic animals arising in the skin, liver, gut, and more, 63 and a few, such as collagenous hamartoma, are common enough to have gained diagnostic utility.
Within the human CNS, the hypothalamic hamartoma is a disorderly overgrowth of neurons and glial cells, and this is also recorded in dogs. 103 In contrast, the designation of a malformed vascular mass as a hamartoma (neural or other) has largely disappeared from the human lexicon, while in the veterinary literature, vascular hamartomas (arising at various sites and at all ages) continue to be reported, a source of diagnostic confusion and inconsistency. Published cases of vascular hamartomas appear morphologically similar if not identical to others reported as some form of vascular malformation or benign angiomatous neoplasm. 59,67,86,87,118 The assorted names that have been used to describe these include CNS vascular hamartoma, angiomatous hamartoma, and telangiectatic hamartoma, while other comparable cases are deemed to be examples of cerebral angiomatosis, cavernous vascular malformation, cavernous angioma, hemangioma, and arteriovenous fistula. Cases diagnosed as vascular hamartomas of the CNS have occurred in dogs ranging in age from 4 months to 15 years, 18,39,59,87,97,118 likely reflective of the heterogeneous nature of this group, each with their own pathogenesis. Similar lesions termed vascular hamartomas have been reported in other domestic species as well, including the cat, 60,74,100 horse, 10,31 ox, 107 and goat. 65
From critical review of the cases that our literature search identified, CNS vascular hamartoma emerges as a diagnostic term which has been assigned to sporadic, focal, vasoproliferative mass lesions in the brain and spinal cord of young to aged animals. It is striking how frequently the microscopic features differ markedly from one case to another, 65,74,86,87 while in contrast, cases with common morphologic features suggest only one or two alternative diagnoses such as cavernous hemangioma or capillary telangiectases. 10,60,86,97,100
In the absence of any distinguishing or defining features of the CNS vascular hamartomas, however, it must be asked what purpose the continued use of this term serves. If abandoned, diagnosis would fall between some form of recognized vascular malformation or, if more fitting, a low-grade vascular neoplasm. Veterinary pathologists could also consider the option of equating vascular malformations and benign vascular neoplasms of the CNS as an acknowledgement that their distinction is currently subjective and lacking in firm criteria.
Meningioangiomatosis
Meningioangiomatosis (MA) is a rare, complex, and heterogeneous proliferative disorder with dual meningothelial/fibroblastic and vascular (“meningovascular”) components found primarily in the leptomeninges and cerebral cortex in humans, 105 and with a suggested predilection for the brainstem in dogs. 9 The nature of MA remains elusive and it may not be a single pathogenetic entity. Proposed pathogeneses include developmental (often described as hamartomatous), dysplastic, or reactive, rather than neoplastic. 9,105
In humans, MA is typified by a leptomeningeal and neocortical plaque occurring in children and young adults sporadically or in association with neurofibromatosis type 2 (NF2). The most common location is the frontal lobe, followed by the temporal, parietal, and occipital lobes in sporadic MA cases, and can involve subcortical white matter and other brain regions such as the cerebellum and deep gray matter. 105 The clinical presentation is a highly variable spectrum from asymptomatic to focal deficits or seizures, depending on the location of the lesion and whether the case is sporadic or a manifestation of NF2. Radiographically, it can present as multifocal tumor-like or cystic lesions, or the brain may appear normal. 105 Human MA has often been reported in association with other lesions of the CNS, most often with an overlying meningioma, 76 as was reported in one dog. 32
This disorder is only described in 12 dogs, 9,32,35,57,79,82,93,101 one horse, 26 one cow, 43 and one mouse, 3 while 3 additional canine cases reside in our archive (unpublished data). Previous potentially misidentified cases may include reports of murine meningeal tumors diagnosed as “meningeal sarcoma” 66 and “malignant meningioma,” 68 which were so classified based on the pattern of infiltration into brain along the adventitia of small blood vessels. 48
In the dozen canine cases published, MA has no specific breed or sex predilection, but, as in humans, it is most commonly found in young animals with a progressive course. One small case series reported the age range of affected dogs as 4 to 14 months, 9 although in a solitary case report, the dog was 9 years old at the time of diagnosis. 79 In addition to the brainstem, canine MA was also reported in the cervical 9 and thoracolumbar spinal cord, 35 while spinal lesions of MA have not been reported in the human literature. The clinical presentation of MA in animals again depends on the involved area, but common clinical signs include those characteristic of local brain invasion and compression. The MRI findings of MA are highly variable, and the degree of contrast enhancement is likely to vary according to the degree of vascularity of the lesion. 35,57 Cerebrospinal fluid abnormalities have not been described in canine MA, in contrast to most cases of meningiomas, where cerebrospinal fluid alterations are commonly recorded. 32 The prognosis in dogs is generally poor due to degenerative changes caused by the infiltrative nature of the lesions, which makes surgical resection challenging if not impossible. 9
Gross changes may not be apparent externally. MA forms circumscribed cellular leptomeningeal plaques on the surface of the brain or spinal cord that extends from the subarachnoid space along the perivascular spaces into the adjacent parenchyma. 103 The parenchymal lesions may be nodular, variably well-demarcated, firm, and white to gray on cut surface. 57 The histopathologic hallmark of MA is a leptomeningeal plaque that extends along the bridging vasculature and invades the adjacent neural parenchyma, with perivascular spread of spindle cells (presumed meningeal fibroblasts) with a variable vasoformative component (Figs. 9–11). The connection with the overlying meninges may not be evident depending on the level of the tissue section. 9,103,105 Infiltrative meningioangiomatous tissue forms collars around multiple parenchymal blood vessels (Figs. 12, 13). This incursion may extend deeply in some areas such as the brainstem and characteristically elicits considerable parenchymal degeneration with myelin ballooning, axonal swellings, some loss of tissue (Fig. 14), gitter cells, and variable gliosis. This putative malformation can be as destructive as the most neuroinvasive of neoplasms such as malignant nerve sheath neoplasms.
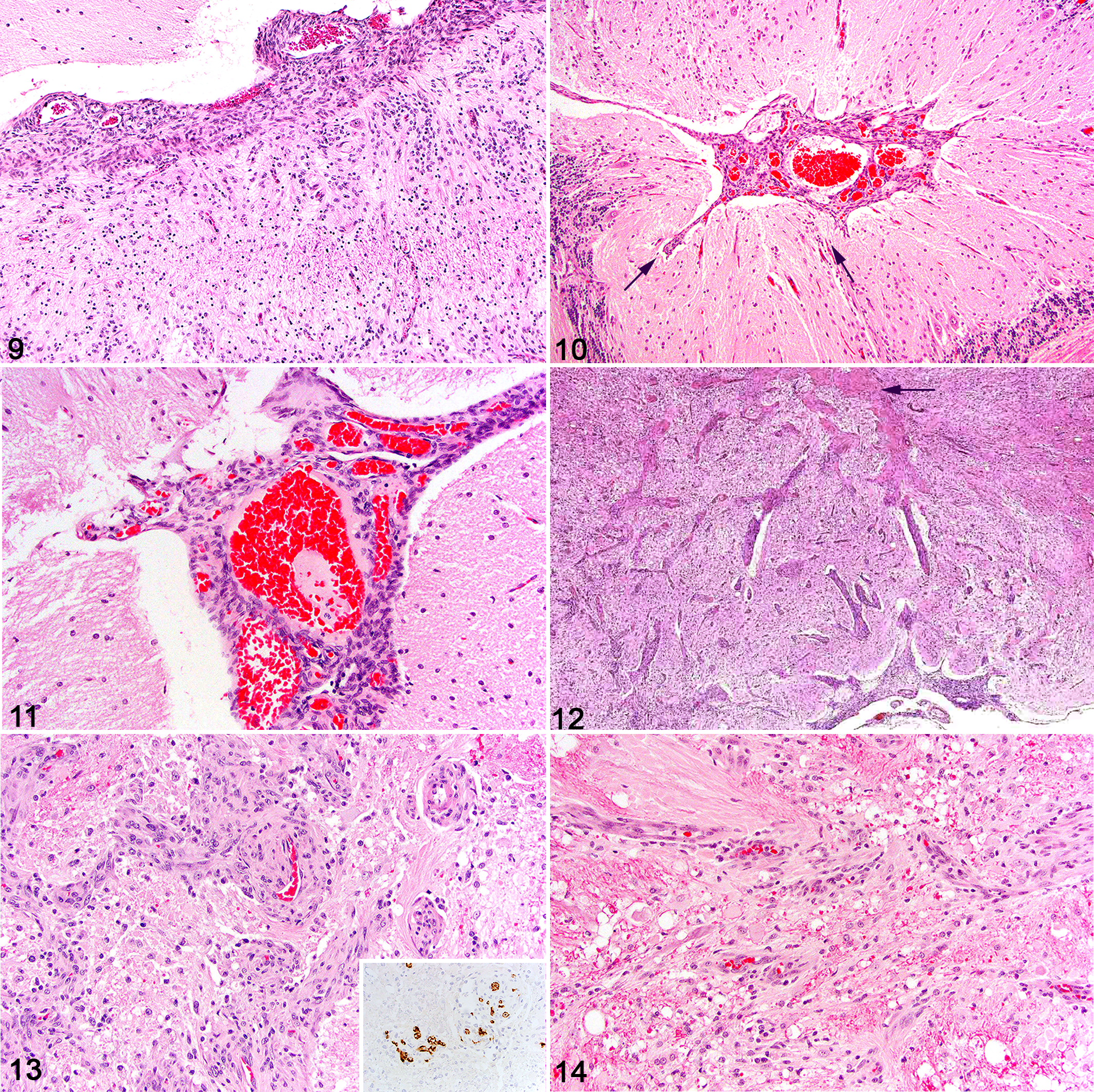
Meningioangiomatosis, brain, dog.
The relative proportion of spindle cell and vascular components can vary based on area and case examined. While focally this proliferative tissue may be highly vascularized (Figs. 10, 11); overall, the meningofibroblastic element predominates. Canine MA typically lacks the usual calcification, psammoma bodies, and neurofibrillary tangles that are seen with MA in humans. 9,105
Immunohistochemistry yields variable results among MA cases in humans and domestic animals, and only vimentin has been reported to be consistently immunolabeled in the proliferating spindle cells, supporting their mesenchymal origin. 3,9,111 These cells also have variable immunolabeling for α-SMA, suggesting some degree of myofibroblastic differentiation. 3,9 The Ki-67 proliferation index is typically very low 111 but is irrelevant if the lesion invades deeply. As would be expected, the endothelial cells lining the proliferative vasculature express factor VIII–related antigen, 3 and the admixed spindled cells have been suggested to have a fibroblastic origin. 79 It has been debated in human literature whether all of the spindle cell component is derived from leptomeningeal tissues or perhaps some of the intraparenchymal proliferation is locally derived from blood vessels. However, IHC and ultrastructural findings do not support suggestions of perivascular neural plexi, muscular, or pericytic derivation and show contradictory results for a meningothelial derivation. 34,79 GFAP-positive astrogliosis frequently surrounds the lesions, with immunolabeled glial processes extending into the Virchow-Robin spaces. 3,9,34 In one of our cases (unpublished data), the vascular proliferation was highlighted by strong immunolabeling for CD31 expressed in seemingly well-differentiated endothelial cells lining the disorganized blood vessels. Immunohistochemistry for ionized calcium binding adaptor molecule 1 (Iba1) labeled marked infiltration by macrophages with different reactive morphologies. Immunohistochemistry (GFAP, oligodendrocyte transcription factor [Olig2], and myelin protein zero) did not support a glial or Schwann cell origin as a component of, or accounting for, the spindle cell proliferation.
The classification of MA is a thorny issue with a developmental (hamartomatous) hypothesis most commonly accepted. This is supported by the association of MA with NF2 in humans, in which MA would be included among the “neurocristopathies” as a developmental abnormality of neural crest cell migration. 88 The hypothesis of a developmental malformation is also sustained in dogs; many come to attention before 15 months of age. One 3-month-old dog had a unique combination of MA and astrocytic thalamic hamartoma. 93 Another dog at 9 months of age (Fig. 10) had coexistent MA and cerebellar cortical dysplasia adjacent to the meningeal plaques, thus indicating a spatial relationship between the 2 abnormalities, and suggesting that the MA lesion was most likely present in utero or in the early neonatal period. 9 In this novel case, the cerebellar dysplasia likely was a consequence of cerebellar meningeal (pial) disruption by the MA rather than a de novo second hamartomatous component. 36 A second theory to explain its histogenesis focuses on the vascular aspect of MA, suggesting that the lesion begins as a vascular malformation with subsequent proliferation of meningothelial cells 45,82,103,115 or fibroblastic cells from vessel walls, 79 or from pluripotent arachnoid cap cells in Virchow-Robin spaces that could differentiate into the various cell types. 34,111 Finally, a third theory postulates invasion by a meningioma into the brain with subsequent evolution of the MA, this supported by the frequent association of MA with meningiomas. 76 However, Perry et al studied 24 cases of human MA or MA with concurrent meningioma and found evidence for a clonal neoplastic process in 9/10 cases in the latter group. 76 This was interpreted as perivascular extension of the meningioma mimicking the growth pattern of MA. 76 Other associated conditions linked to human MA, including post-radiation MA, suggest that the pathogenesis is not nearly a simple developmental abnormality but may also manifest as a reactive change secondary to a number of different processes.
The differential diagnosis for MA is limited, but may include malignant meningioma, malignant nerve sheath tumors, and other vascular malformations. Indicators for the diagnosis of MA in dogs are the typical juvenile presentation; the characteristic ribbon-like arrangement of MA in the subarachnoid space with vascular, meningothelial, and/or fibroblastic components; the lack of atypia, pleomorphism or high mitotic activity; and the remarkable brainstem infiltration.
Angiomatosis/Hemangiomatosis
Human angiomatosis (or hemangiomatosis as some prefer) is an uncommon vasoproliferative disorder encountered in children and young adults. It affects the superficial and deeper soft tissues of the lower extremities (even reaching down to the skeleton) or can have a multiorgan presentation. Angiomatosis has 2 microscopic patterns: either a mixture of blood vessel types, some cavernous, or a predominance of capillaries. Lobules of mature adipose tissue characteristically accompany the predominant vascular element.
In domestic animals, angiomatosis has been identified as a mass lesion within the skin and subcutis of cats and dogs, often in early life. Bovine cutaneous angiomatosis was identified in 1963. 19 Cases are recorded in the skeleton (often vertebrae), and we have seen a canine vertebral case with angiomatosis also in the spinal cord parenchyma.
Feline vertebral angiomatosis is a fairly well-documented condition, typically affecting younger cats and often resulting in spinal cord compression. 27,37,83,92 Reported in the thoracic and lumbar spine, the lesions consist of multifocal proliferations of well-differentiated, small-caliber arterioles and veins accompanied by fibrous stroma which form expansile masses throughout the vertebral medullary cavities. 37,92
A generalized pattern is chronicled in calves, with involvement of the heart, lung, kidney, and more. Angiomatosis is generally viewed as nonneoplastic but some bovine cases have been published as “disseminated hemangioma,” 69 one with brain involvement. 2 The lesions of cutaneous angiomatosis in some domestic animals have been described as progressive and invasive as might be seen with a locally extensive hemangioma.
A case was reported as cerebral angiomatosis in a 2-year-old domestic shorthair cat that presented with generalized seizures. 50 Microscopic vascular lesions, which occurred bilaterally in the cerebral grey matter, comprised multiple small compact nests of fusiform cells which contained differentiated capillaries of varying caliber. Vascular subendothelial layers were thickened and sometimes vessels were encircled with a homogeneous eosinophilic material with dystrophic mineralization found to include laminin and von Willebrand factor proteins.
A more complex syndrome is human encephalofacial or meningofacial angiomatosis (Sturge-Weber syndrome). 17 This is the third most common neurocutaneous disorder or phakomatosis after neurofibromatosis and tuberous sclerosis, but is not inherited. In this condition, a unilateral leptomeningeal and cerebral vascular malformation is accompanied by a similar facial hemangiomatosis which produces a so-called port wine birthmark. 17
A case resembling Sturge-Weber syndrome has been reported in a horse for which the term meningocerebral hemangiomatosis was employed. 61 This 3-year-old horse presented with intermittent generalized seizures. Grossly, the leptomeninges covering most of the left cerebral hemisphere were opaque, and there was thinning and collapse of the ipsilateral piriform lobe, with moderate dilation of the lateral ventricles. Leptomeningeal opacity was due to vascular proliferation and meningothelial hyperplasia. Redundant tortuous arterial and venous vessels were prominent within the subarachnoid space over the gyri and in sulci (Fig. 15) and extended into some regions of the subjacent cortex, where there was neuronal loss, ectopia, and disorganization (dysplasia). Further vascular proliferation affected choroid plexi of the lateral and fourth ventricles, while milder vascular lesions were present in the leptomeninges of the ventral brainstem, right cerebrum, spinal cord, and in the eye. The left trigeminal nerve was distorted by swollen fascicles containing onion bulb-like structures, shown to be of Schwann cell origin. 61
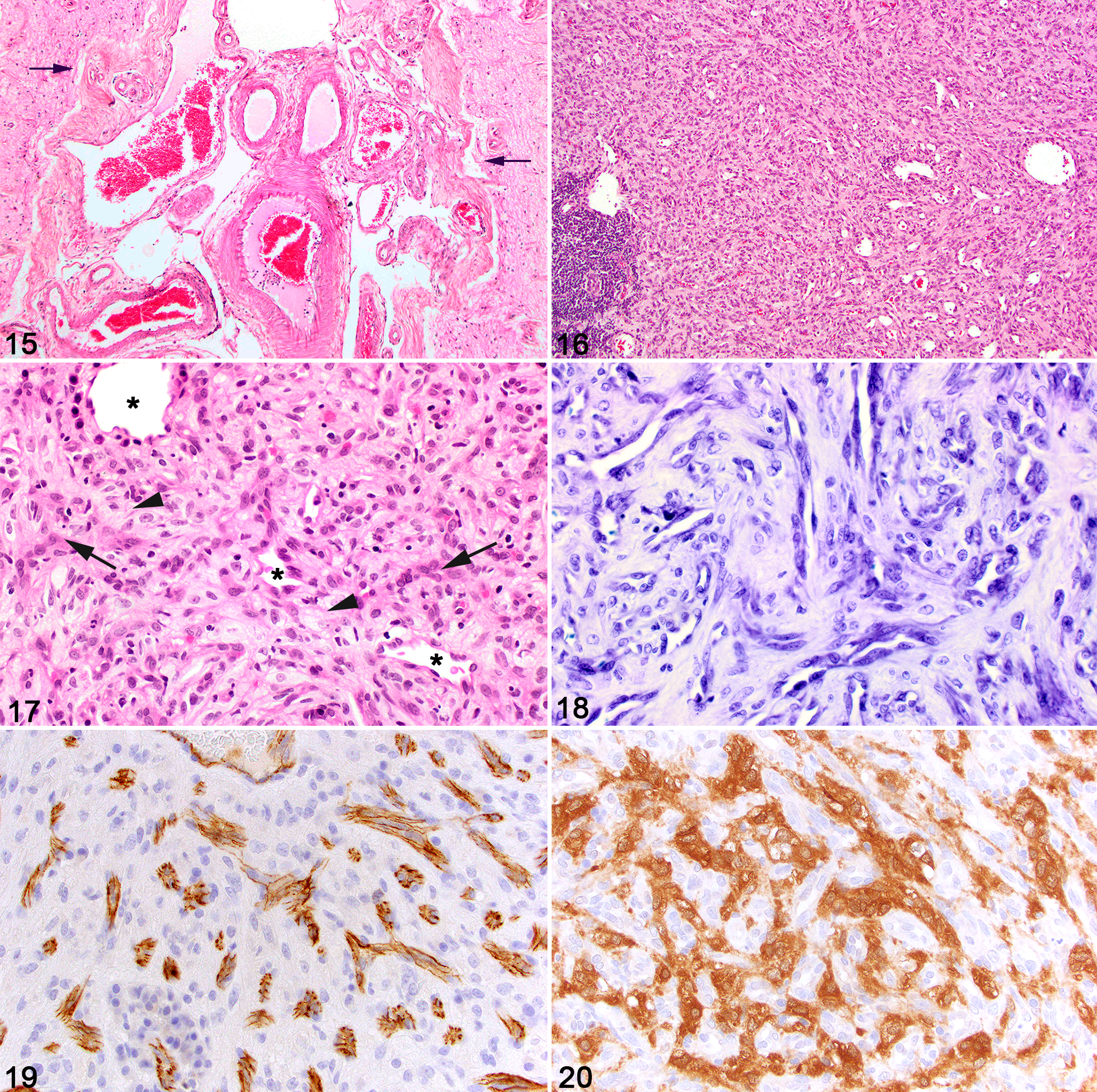
Meningocerebral angiomatosis, brain, horse. From the affected cerebral hemisphere, a sulcus (delineated by arrows) is expanded by a collection of arteries and veins. At left, note the thin-walled cavernous vessel. Diffusely, mature collagenous tissue thickens the leptomeninges. Hematoxylin and eosin (HE).
The primary abnormality was the meningocerebral hemangiomatosis, which resulted in dysplastic changes in the developing cerebral cortex and it was suggested to have initiated the episodic seizure activity. The relationship of the trigeminal nerve changes to the meningocerebral hemangiomatosis is unclear, but a compound anomaly in embryological development resulting in dysplasia and a proliferative onion-bulb neuropathy may be involved. In about 10% of human cases, the vascular abnormalities are limited to the brain, 17 and so in the absence of facial hemangiomatosis, this equine case is similar (but not identical) to an “incomplete” Sturge-Weber syndrome.
Neoplastic Disorders
A review of cavernous and capillary hemangiomas of the CNS would be anticipated here but they have been considered with the vascular malformations. This choice is in acknowledgement of the great difficulty, if not impossibility, in differentiating on morphological grounds between some vascular malformations and benign vascular tumors of the CNS. Recognizing this problem, the human medical pathology literature equates these 2 entities.
Hemangioblastoma
Hemangioblastoma is a slow-growing vascular neoplasm in humans that is considered benign (World Health Organization grade I); the neoplastic component is unique. 58 Some cases arise spontaneously anywhere in the CNS, though most commonly, in the cerebellum where it is a manifestation of von Hippel-Lindau disease caused by a mutation in the tumor suppressor gene VHL. 56,110 Characterized by a well-demarcated mass, microscopically, hemangioblastoma is composed of an abundance of haphazardly arranged capillaries that are separated by neoplastic stromal cells which are of unknown histogenesis (Figs. 16–18). The prominence of the capillaries, which is emphasized by endothelial or pericytic IHC stains, can overshadow the stromal cellular component in some canine cases. Careful inspection for a potential stromal component will help differentiate this tumor from a capillary hemangioma. Indeed, 2 cases of hemangioblastoma were recovered from the archives of one author (BAS) masquerading as capillary hemangioma years before the diagnosis of canine hemangioblastoma was recognized. The stromal cells are fusiform to stellate with lightly eosinophilic cytoplasm which often contains small lipid vacuoles. Their nuclei are typically large and oval with an open chromatin pattern and one to two distinct nucleoli. Anisocytosis and anisokaryosis can vary, with occasional bi- and multinucleated cells; mitotic activity is low. Plasma cells may be scattered throughout the mass, with perivascular or peripheral aggregates of plasma cells and lymphocytes.
In humans, 2 histologic subtypes of hemangioblastoma are recognized: (1) reticular, with an even mixture of capillaries and (in between them) stromal cells, and (2) cellular, with a predominance of the stromal cell component. 38 In humans, the more prominent stromal cells are often highly vacuolated, resulting in a characteristic clear-cell or lipoblast-like morphology, 58 which can mimic metastatic clear cell renal carcinoma. Patients with von Hippel-Lindau disease develop hemangioblastoma of the brain and retina and, in other organs, a variety of cysts and further tumors.
The fascination with human hemangioblastoma lies in the histogenesis of the puzzling stromal cells that, unlike the capillary vessels, do not express common endothelial antigens such as factor VIII–related antigen and CD31. Rather, interstitial cell markers include those associated with neurons (neuron-specific enolase, neural cell adhesion molecule), a cytoskeleton-binding protein (ezrin), and a gonadal hormone (inhibin-α), as well as the VHL protein. Inhibin-α is an important marker to differentiate hemangioblastoma from the microscopically similar renal cell carcinoma, which is negative. 58 Several studies analyzing von Hippel-Lindau disease tumorigenesis and protein expression in the stromal cells suggest the cell of origin may be the hemangioblast (multipotent with both hematopoietic and endothelial potential) or hemangioblast progenitor cell. 33,95,109
While well-documented in humans, hemangioblastoma is a rare neoplasm of the canine CNS first described in 2003 14 with a predilection for the spinal cord based on cases reported to date. Cases are of the spontaneous category, as von Hippel-Lindau disease is unknown in domestic animals. Four examples are published in dogs, 2 to 9 years old, 8,14,54,64 while an additional 2 cases, reported in 1979 as hemangioma, share the characteristic histologic pattern of hemangioblastoma and are more consistent with this entity than hemangioma. 18 Of these 6 cases, 5 cases occurred in the spinal cord, with 3 cases in the cervical spinal cord and 1 case each in the thoracic and lumbar regions. Clinical signs were related to spinal cord degeneration and necrosis due to compression and effacement caused by the space-occupying mass. The sixth case occurred in the right olfactory and frontal lobe, resulting in a rapid onset of cluster seizures, abnormal mentation, and proprioceptive deficits.
In the 4 cases published as canine hemangioblastoma, capillary endothelia but not stromal cells expressed factor VIII–related antigen and CD31 antigens (Fig. 19). 8,14,54,64 Neuron-specific enolase (NSE) was consistently expressed by stromal cells and a similar pattern has been seen with cases in our archive (Fig. 20). The NSE immunolabeling is an important diagnostic criterion, as for the unwary, the histology of hemangioblastoma can overlap with capillary hemangioma. One report also noted patchy stromal cell labeling with vimentin and GFAP, 8 while S100 was patchy to negative. 8,14 IHC labeling for inhibin-α was used in only one of the reports of canine cases, and the stromal cells were negative for this marker, as are a subset of human cases. 8 Prognosis after complete surgical resection of spontaneous hemangioblastoma in humans is excellent. 58 In 2 of the 6 reported cases in dogs, their spinal cord masses were successfully removed with improvement or resolution of neurologic signs and no evidence of postoperative regrowth at 9 and 12 months, respectively. 8,64 In one case, postsurgical neurologic improvement was followed by rapid deterioration resulting in euthanasia. A necropsy was not authorized and the authors speculated that surgical complications were the most likely cause of death. 54
Hemangioendothelioma
While histologically dissimilar to other endothelial neoplasms, in behavioral terms human hemangioendothelioma sits as an intermediate entity between hemangioma and hemangiosarcoma. 58 It is considered a low-grade malignancy. First recognized in 1982, 113 epithelioid, spindle cell, and kaposiform hemangioendotheliomas have been described, although the second has been “downgraded” from spindle cell hemangioendothelioma to spindle cell hemangioma. 58 They occur in many organs and soft tissue but in humans, only epithelioid hemangioendothelioma is recognized in the CNS. 58
Human epithelioid hemangioendothelioma, recently reclassified as a World Health Organization grade 3 tumor, occurs only rarely in the brain. 58 Largely lacking the formation of conventional vascular channels with lumina, instead human epithelioid hemangioendothelioma is characterized by cords and clusters of epithelioid cells amid an abundant myxoid stroma with frequent individual cell cytoplasmic vacuole (microlumen) formation. Due to the paucity of cases, behavior can often be difficult to predict, but local invasion and occasional metastasis have been reported. 71
In the veterinary literature, there are but 2 reported cases of mass lesions occurring in the brain for which the term hemangioendothelioma was used: a 9-year-old dog and a 5-month-old calf. 24,117 Remarkably, these 2 cases both showed epithelioid and spindle cell hemangioendothelioma differentiation.
Histologically, both cases were composed of plump epithelioid endothelia lining abundant small, proliferating blood vessels or were organized as interspersed solid clusters and cords, supported by a myxoid to hyalinized stroma. 24,117 In some areas, the endothelial cells exhibited a more spindled morphology. The epithelioid endothelial cells were typically large and polygonal with eosinophilic cytoplasm and round to oval nuclei with vesicular chromatin, and the spindle cells had similar nuclear features but lacked cytoplasmic vacuoles. Cellular and nuclear pleomorphism in all areas was mild and mitotic activity was low. Individual cells occasionally contained a large cytoplasmic vacuole, lending a signet ring appearance, which sometimes contained a single erythrocyte, consistent with primitive single-cell lumen formation. Vascular channels were often blood-filled and there were areas of stromal hemorrhage. IHC markers for endothelium (factor VIII–related antigen, vimentin, CD31, and CD34) readily labeled the neoplastic population.
Another, and less common, variant is retiform hemangioendothelioma, so named because of its elongated arborizing networks of blood vessels lined by plump, hobnailed endothelium said to be reminiscent of the rete testis. 13 In some areas, neoplastic cells may form intraluminal papillae supported by collagenous cores. While a primary retiform hemangioendothelioma has not been reported in the CNS of a domestic animals, one report describes 2 dogs with malignant vascular tumors, which in areas had developed the retiform hemangioendothelioma pattern (Fig. 21). 55 One of the 2 affected dogs had disseminated disease, including metastatic lesions in the brain and spinal cord.
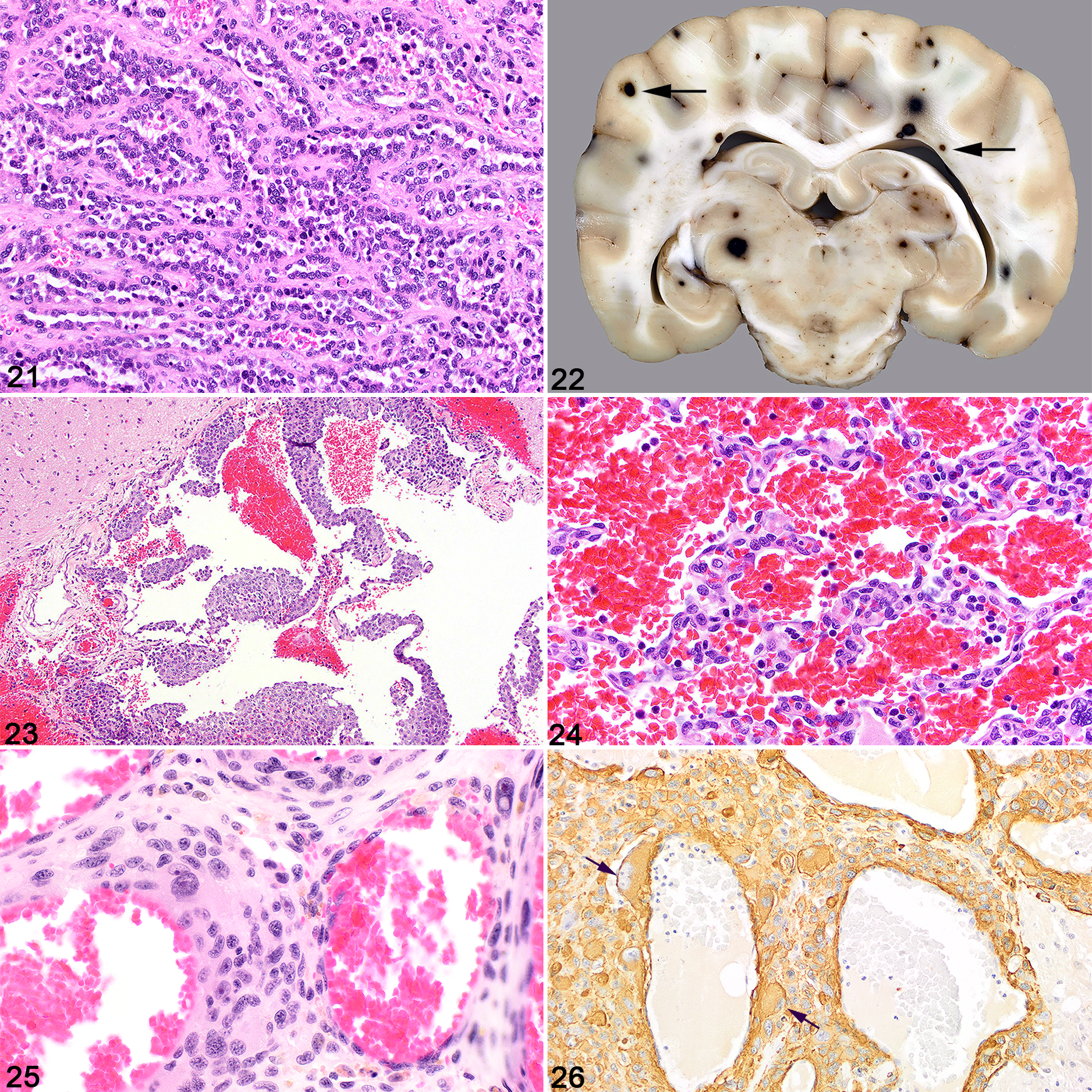
Metastatic hemangiosarcoma with a pattern resembling human retiform hemangioendothelioma, brain, dog. In retiform areas which resemble delicate tubules, this neoplasm is composed of arborizing network of blood vessels lined by rounded, crowded, and sometimes layered endothelium. Hematoxylin and eosin (HE).
Hemangiosarcoma
In contrast to the previously discussed vascular neoplasms, involvement of the human CNS with hemangiosarcoma (angiosarcoma) is rare. In domestic animals, visceral hemangiosarcoma is a highly malignant neoplasm of vascular endothelium with a high rate of metastasis. It is most commonly diagnosed in dogs, less commonly in cats, and rarely in other domestic species. 63 Typical primary sites in the dog include the spleen, liver, the right auricle of the heart, and the skin and subcutis. When the CNS is affected by metastasis, gross findings are of many scattered pinpoint to larger and variably shaped dark red hemorrhages evident both on the surface of the brain and spinal cord and following sectioning. Small rounded foci are sometimes located at the cerebrocortical-white matter junction, supposedly reflecting the point at which the branching arterial vessels have diminished to a size in which emboli become trapped (Fig. 22). Imaging the brain can identify the multifocal intracranial lesions and suggest the likely diagnosis. Microscopically, the embolic neoplasm is characterized by irregular fingers and ribbons of anaplastic, hyperchromatic endothelial cells in which anisocytosis and anisokaryosis and a variable mitotic activity are encountered. As hemangiosarcomatous foci expand, irregular blood-filled vascular channels form between expanding slender fingers and larger trabeculae of compact neoplastic tissue lacking vasoformation (Figs. 23, 24). The neoplastic cells may be labeled by IHC markers for endothelium such as factor VIII–related antigen and CD31, although negative results are encountered with some malignancies. 7 Often, the histologic pattern alone is adequate for diagnosis, although in some CNS micrometastases, hemorrhage is abundant while the endothelial population is inconspicuous. Metastatic epithelioid hemangiosarcoma, which can be mistaken for carcinoma, is also documented in the CNS of one dog. 96 Multifocal disease involving metastatic lesions in the CNS is particularly common in the dog, though is documented in other species. 40,47,52,104,106,112 Hemangiosarcoma is the most frequent cause of metastatic CNS neoplasia in the dog, in one review accounting for 51/177 secondary canine brain tumors, most commonly affecting the cerebrum and resulting in disturbances of mentation. 98 There are two purported cases of primary CNS hemangiosarcoma in the veterinary literature: in the cerebrum of a 6-week-old dog 30 and in the meninges overlying the left olfactory bulb in a juvenile pig. 99
There are several reports of primary hemangiosarcoma within the epidural/extradural space, resulting in compressive myelopathy in dogs 28,44,72 and in one horse. 6 Primary vertebral hemangiosarcoma is more common, though still rare, and can similarly cause compressive myelopathy if the neoplasm invades the vertebral canal. 49,70,73,75,80 Other variants of metastatic vascular neoplasms in the CNS are almost unknown, but one such was a canine angioleiomyosarcoma of undefined source which embolized to the lung and the brain (Figs. 25, 26).
Discussion and Conclusion
We have reviewed and summarized the variety of proliferative vascular lesions recorded in the CNS of domestic animals (Table 1). They are not common and are mostly recognized in the dog, with a few recorded in cats, horses, ruminants, and mice. Sporadic examples occur at all ages with somewhat more coming to attention either in early life or in mature-to-aged animals. Cases in juvenile animals raise the potential of a malformation, but some in the aged are microscopically similar and perhaps have been clinically dormant throughout life.
The goal of revisiting this published literature was to critically review the pathologic data with the aim of clarifying the terminology used to identify and delineate each syndrome. One fundamental question can be stated this way: Why did authors opt for any one particular diagnosis over another and what feature(s) distinguished and so differentiated each diagnostic entity? We found that even for well-described cases, the identification of discriminating attributes which could be considered syndrome-specific was lacking. In addition, human textbooks and journals are commonly used resources, and in some of the cases under discussion here, their use of terminology can cause confusion and even appear contradictory.
Disorders that are rarely encountered can be a cause of diagnostic difficulty for the pathologist, certainly in veterinary medicine. This appears to hold also for CNS vascular malformations in domestic animals, as many of the terms used in publications to name them are nonspecific, even noncommittal, indicating these authors’ discomfort in naming the syndrome, including vascular malformation, 18 vascular anomaly, 114 venous malformation, 31 angiomatous vascular malformation, 16 and vascular ectasia. 11 The human CNS vascular malformations that come to clinical attention are arteriovenous malformations (common, and sometimes very serious) and cavernous malformations (also called cavernous angiomas in humans), though these are much less common; in comparison, examples of these subgroups in domestic animals are rare. 18,59 While most vascular malformations of the human brain fall into one of the groups enumerated above, one large review of 280 cases identified 14 mixed cases with pathologic features of more than one type. 1 This may be of relevance to veterinary medicine as further cases are encountered. Following patients with these vascular disorders and repeating scans over a number of years has shown their capacity for growth, such as of cavernous angiomas. 77 Ultrastructural studies of human cases are limited in number; one small comparison of arteriovenous malformations with cavernous malformations found thinner walls and a rarity of tight junctions between endothelial cells in the latter group and so proposed an explanation for their tendency to recurrent microhemorrhages. 116
Hamartomas are also acknowledged as malformations and a number of CNS vascular hamartomas are published, in dogs and other domestic animals. Often, they are morphologically heterogeneous and do not form a unified group which can readily be compared with each other or a single human counterpart. In the past, these have been studied microscopically almost exclusively at postmortem examination. In contrast, a recently published report describes the outcome following resection of an intracranial vascular hamartoma in a 13-year-old dog. 108 At 27 months follow-up, the dog was clinically normal. As intracranial intervention becomes ever more the norm in dogs and cats, the pathologist needs to employ diagnostic terms of some specificity so that with time, vascular lesion type and clinical outcome can be correlated. While some vascular hamartomas of the CNS were observed in young animals, 10,74,97 other case reports include a 13-year-old cat and two 15-year-old dogs. 60,97 A few vascular hamartomas seem highly compatible with published cavernous hemangiomas 21 and perhaps indeed are benign neoplasms and not malformations as claimed. On the other hand, is it possible that such neonatal disturbances in growth can remain dormant in the CNS throughout life until the later years?
In conclusion, the challenge lies with appropriate assignment of term and differential diagnosis. This review of the literature did not reveal any unique or objective clinical or pathologic features of so-called CNS vascular hamartomas in domestic animals that authors have cited in support of this diagnosis. Some authors point to the presence or absence of intervening parenchyma between cavernous vessels as a guide to the differentiation of hemangiomas from other diagnoses. The vessel walls of human cavernous hemangiomas in close apposition are said to leave little intervening parenchyma, while in contrast it has been proposed that brain parenchyma is present between those thought not to be neoplastic. 86,97 However, our experience is that finding some vessels abutting and others separated by neuropil or white matter in the same field in these cases is common (see Fig. 9). Vascular hamartoma appears a term of convenience for an ill-defined group of lesions. 85 It seems highly probable that the literature of published CNS vascular hamartoma is contaminated with other entities and for these reasons we recommend that for the CNS the term “hamartoma” be abandoned. Importantly, it should be noted that pericytic cell components are an element that can be seen in benign and sometimes malignant vascular endothelial neoplasms and cannot be used as a differentiating feature. Last, although rare, vascular neoplasms distinct from hemangiosarcoma such as hemangioblastoma, hemangioendothelioma, and angioleiomyosarcoma must also be considered as part of the diagnostic algorithm based on a combination of histology, histochemical, and immunohistochemical features.
Footnotes
Acknowledgement
We thank Dr. Craig Horbinski of the Feinberg School of Medicine, Northwestern Medicine, Chicago, IL for providing Figure 2. We also thank Jen Patterson for her assistance with the images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
