Abstract
In a retrospective survey of caprine neoplastic disease, eight masses were diagnosed as cutaneous vascular tumors. The typical clinical presentation was a solitary raised, bleeding mass. No predilection with regard to age, breed, sex, or anatomic location was found. Reevaluation of the microscopic features of the masses resulted in diagnoses of hamartoma (2), hemangioma (4), and hemangiosarcoma (2). An endothelial cell origin was confirmed in all seven tumors tested immunohistochemically for factor VIII-related antigen. Although rarely reported, goats display a range of cutaneous vascular growth abnormalities similar to those observed in other domestic animals.
Neoplastic disease is less frequently diagnosed in the goat than in most other domestic animals.1–3,9,10 In a 5-year survey of the occurrence of tumors in domestic animals reported to the Veterinary Medical Data Program by 12 North American veterinary colleges, only four caprine cases were found, the lowest total for any of the common domestic animals.8 Cutaneous vascular neoplasia is most common in the dog, cat, and horse9 but has rarely been documented in the goat.1,12 Here, we describe eight examples of caprine cutaneous vasoproliferative lesions diagnosed at the Oregon State University Veterinary Diagnostic Laboratory (OSU VDL) and at the New York State Diagnostic Laboratory of Cornell University (CU).
A computerized search of caprine pathology submissions to each laboratory identified potential cases for this study. At CU a list of all cases of caprine neoplastic disease was generated, whereas at the OSU VDL the list consisted of all diagnoses obtained for caprine pathology submissions. Cases qualifying for this study were then obtained by manually checking these lists for diagnoses pertaining to vascular disorders (e.g., hemangioma, hemangiosarcoma, and angiomatosis). The time frame searched for the CU and OSU VDL databases was 1977–1997 and 1991–2000, respectively. Qualifying case records were retrieved, and follow-up information was solicited from owners and attending veterinarians by either a mailed questionnaire or a telephone interview. Histologic features of each mass were assessed in 7-μm-thick hematoxylin and eosin (HE)-stained sections from archived formalin-fixed, paraffin-embedded tissue.
Immunohistochemical (IHC) detection of factor VIII–related antigen (FVIIIR-Ag), an endothelial cell marker, was attempted on paraffin-embedded tissue from goat Nos. 1–7; the paraffin block from goat No. 8 was not available. A strepavidin–biotin–peroxidase method was used (Zymed LAB-SA System, Zymed Laboratories, South San Francisco, CA). The technique included a trypsin digestion step and incubation with 10% normal goat serum prior to exposure to a 1:1,000 dilution of primary antisera (Factor VIIIR-Ag, Dako, Carpinteria, CA). The biotinylated secondary antibody (goat anti-rabbit; Zymed) was then incubated with the streptavidin–peroxidase conjugate, and bound antigen was detected using 3,3-diaminobenzidine. Endothelial cells lining normal vasculature served as internal positive controls. Nonimmune rabbit serum (Dako) was substituted for the primary antibody on negative control slides. All sections were counterstained with Gill's No. 1 hematoxylin.
Neoplastic disease was diagnosed in 94 of 1,344 caprine submissions (7%) during the 20-year study period at CU. At the OSU VDL, 31 neoplasms were diagnosed from 456 caprine pathology submissions (7%) during the 9 years studied. Cutaneous vascular tumors were diagnosed in three CU cases and in five of the OSU VDL cases (about 3% and 16% of caprine neoplastic disease diagnoses, respectively).
The case signalment, size of the mass, and the length of time the growth was noted to be present prior to surgical removal are summarized in Table 1. Goats originated from eight different premises. A solitary cutaneous growth was the presenting clinical sign in seven animals. Goat No. 3 presented with three cutaneous masses, only one of which was included in this study. Bleeding at the site of the lesion was noted in seven goats and in some instances was the impetus for requesting surgical removal of the mass. Concurrent systemic disease was not identified in any of the goats. Therapy consisted of surgical excision of the mass, with the exception of goat No. 4, which also received cryotherapy. Follow-up information was obtained for five goats (Table 1).
Characteristics of eight cases of vasoproliferative lesions in the skin of goats.
All animals remained disease free at date of follow-up.
Euthanatized 1 week after the tumor was removed.
In goat No. 1, enlargement and bleeding of a previously quiescent raised mass prompted the owners to request surgical removal of the growth. Microscopically, this unencapsulated mass was consistent with a hamartoma. It was composed of lobular groupings of tightly packed, well-organized vascular structures, ranging from capillaries to small arterioles. A grenz zone was evident, and the abnormal vessels did not extend beyond the deep dermis. Endothelial cell nuclei featured one or two small nucleoli and mild anisokaryosis. No mitotic figures were observed in 10 random 40× high-power fields (HPFs). Most capillaries were invested with a small quantity of smooth muscle, and pericytes were often present. The superficial portion of the mass was infiltrated by many eosinophils, and a few clusters of lymphocytes were evident in the deep dermis.
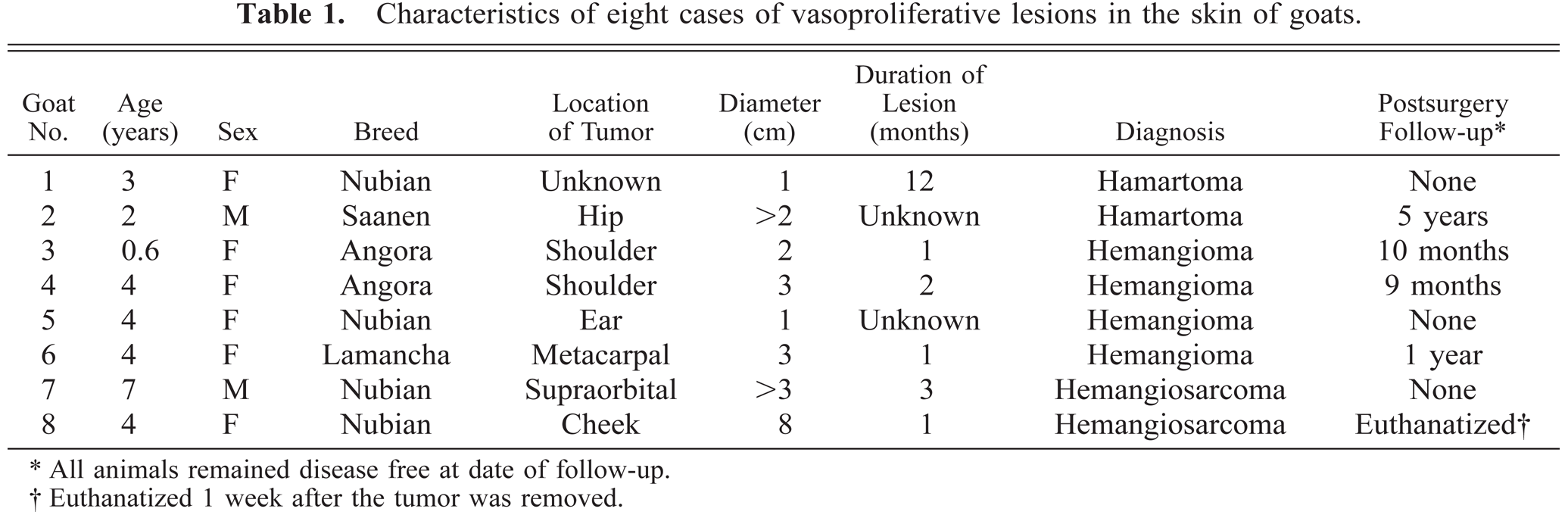
The unencapsulated and poorly defined mass removed from goat No. 2 was located in the deep dermis and was also consistent with a hamartoma. The mass contained lobular aggregates of enlarged arterioles and veins accompanied by clusters of less distinct capillary-like structures (Fig. 1). The lobules were separated by normal dermal collagen. Nuclei were unremarkable, and no mitotic figures were seen in the endothelial cells lining these vascular structures.
Cutaneous vascular hamartoma; goat No. 2. One of the lobular aggregates of blood vessels in the subcutis. Immunostained with anti–factor-VIII-related antigen and Gill's hematoxylin counterstain. Bar = 100 μm.
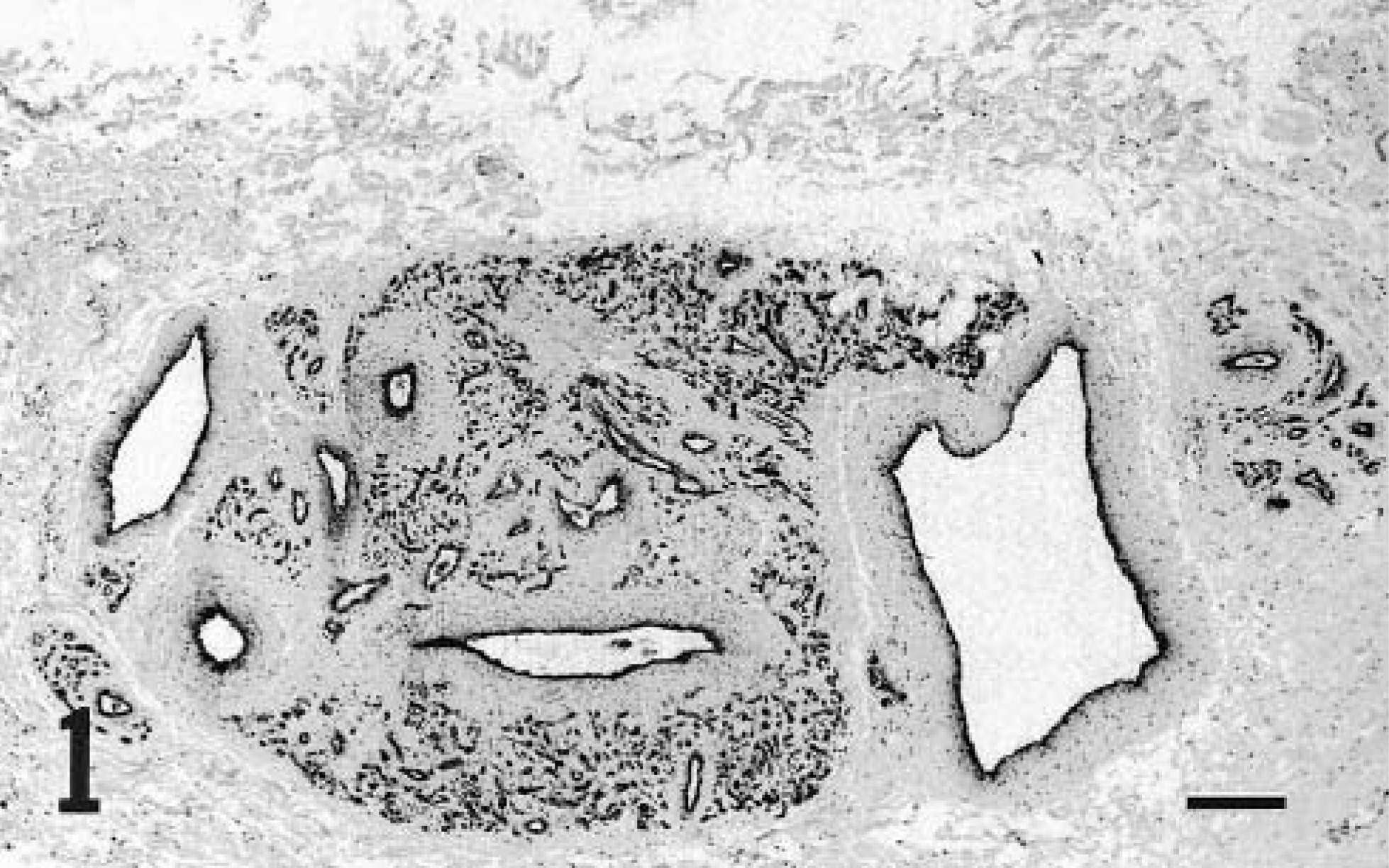
A pedunculated hemorrhagic mass from the shoulder of goat No. 3 was surgically excised, and two wartlike growths on the contralateral shoulder were left untreated. These small growths spontaneously regressed within a few weeks; none had recurred at follow-up 6 months later. Microscopically, the excised mass was consistent with a hemangioma composed of many randomly oriented blood-filled capillaries loosely packed in a collagen-poor stroma (Fig. 2). The abnormal vascular channels extended from the dermoepidermal junction to the deep dermis. The epidermis was largely ulcerated, with consequent infiltration of neutrophils. Four mitotic figures were observed in 10 random HPFs.
Dermal hemangioma; goat No. 3. Randomly oriented capillaries are lined by plump endothelial cells. HE. Bar = 50 μm.
A papillated, multilobular mass was submitted from goat No. 4. A similar mass at this site had been clipped off 2 months earlier during shearing. Microscopic features of this focally ulcerated structure were consistent with a hemangioma. The mass included a locally extensive area of loose granulation tissue with a subjacent zone of tightly packed capillaries lined by plump endothelial cells. Nuclear characteristics of these cells included mild anisokaryosis, lightly clumped chromatin, an occasional small nucleolus, and less than one mitotic figure per HPF. The neoplastic cells extended to the dermoepidermal junction in some locations and were accompanied by locally extensive lymphocytic infiltrates and large numbers of more diffusely distributed neutrophils. Macrophages containing hemosiderin were common. Epidermal hyperplasia was present at the periphery of the mass.
Microscopic features of the smooth-surfaced unencapsulated mass removed from goat No. 5 included locally extensive edema with a central area of hemorrhage rimmed by randomly oriented vascular elements typical of a hemangioma. The mass was focally ulcerated with superficial bacterial colonization and a strong neutrophilic response. The proliferated vascular elements were similar to those of goat Nos. 3 and 4 and consisted of blood-filled capillaries of variable caliber, loosely packed within a collagen-poor stroma. The endothelial cell nuclei were large and hyperchromatic. Two mitotic figures were observed in 10 random HPFs. Small numbers of hemosiderin-laden macrophages were scattered through the stroma.
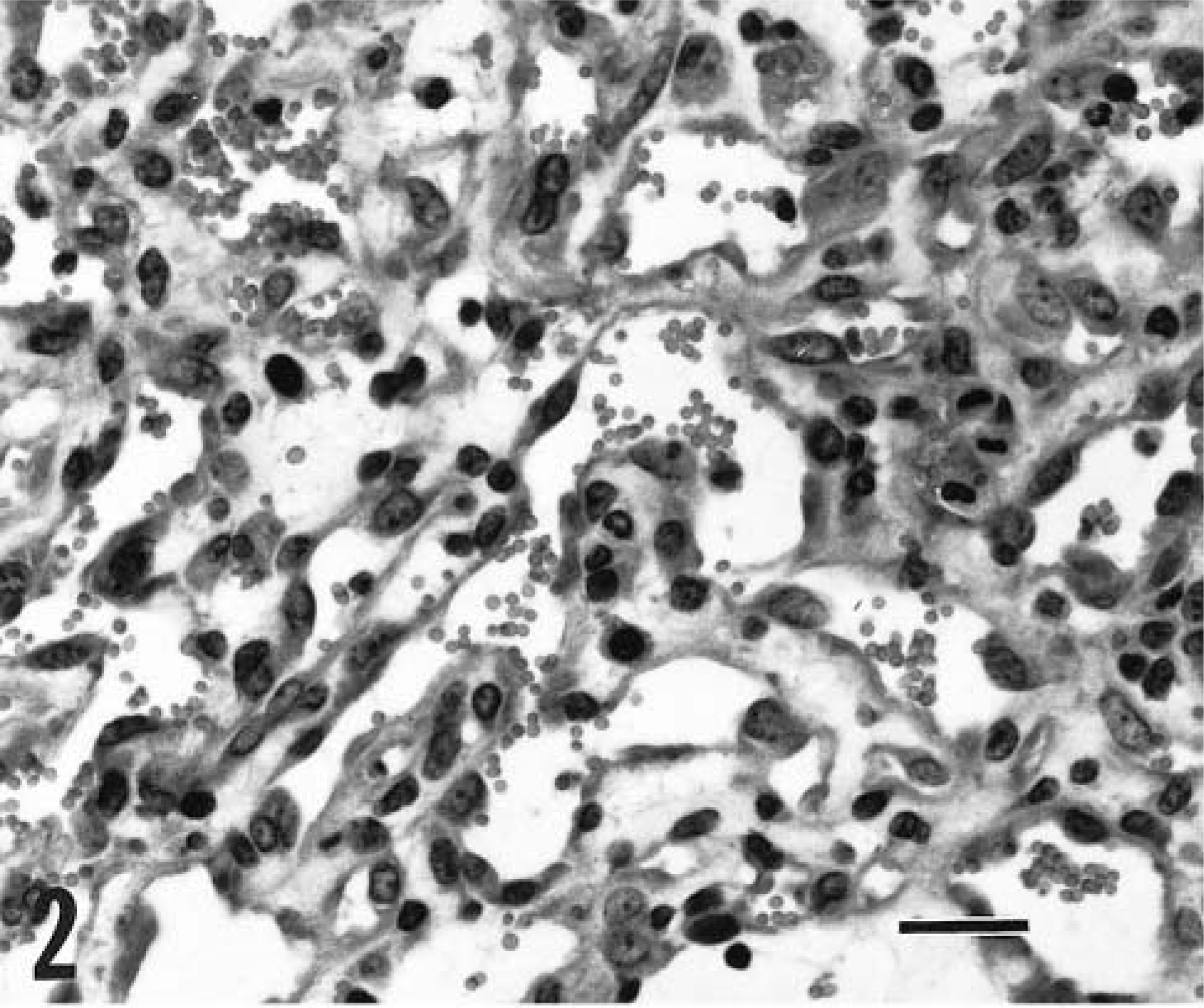
The ulcerated, unencapsulated mass removed from goat No. 6 was composed of large interconnecting cavernous sinusoids (Fig. 3). These vascular spaces contained various combinations of erythrocytes, fibrin, and neutrophils and were supported by broad collagenous septa that were sometimes expanded by a fibrinocellular infiltrate plus a few hemosiderin-laden macrophages. The endothelial cells were oval, plump, and occasionally clumped together but lacked nucleoli. No mitotic figures were observed in 10 random HPFs, and the mass was diagnosed as a hemangioma.
Dermal hemangioma; goat No. 6. Interconnecting blood-filled sinuses formed by thick septa are lined by plump endothelial cells. HE. Bar = 60 μm.
The tumor removed from goat No. 7 was consistent with a hemangiosarcoma. This mass was composed of interlacing sheets of tightly packed pleomorphic cells that occasionally included well-defined vascular channels. The tumor was ulcerated and partially encapsulated and contained extensive areas of hemorrhage. The neoplastic cells featured marked nuclear hyperchromasia, pleomorphism, and anisokaryosis (Fig. 4). About three mitotic figures were present per HPF, and a few multinucleate cells were visible.
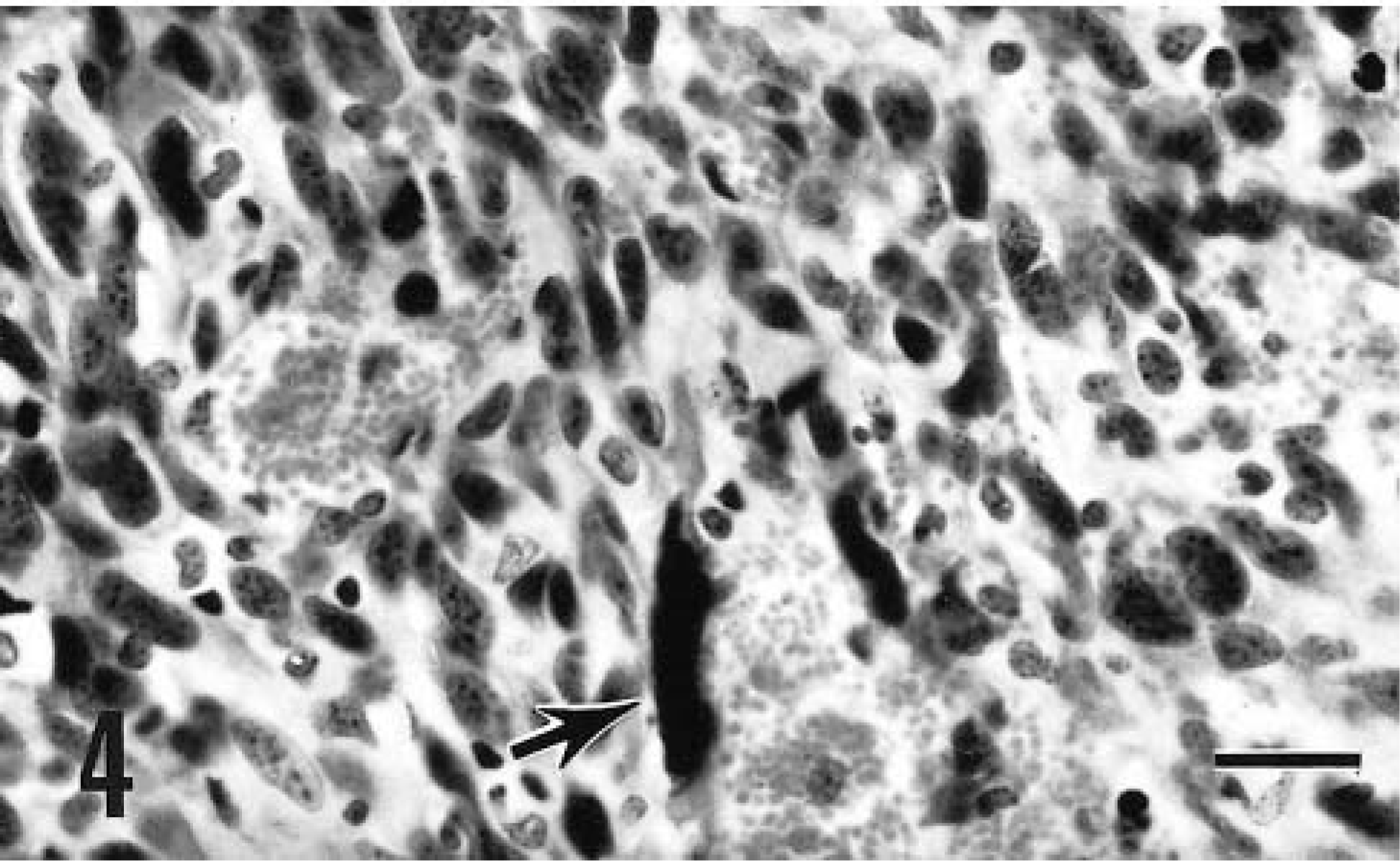
Cutaneous hemangiosarcoma; goat No. 7. Sheets of tightly packed fusiform mesenchymal cells with a few blood-filled spaces make up the tumor. Note enlarged and distorted nucleus (arrow). HE. Bar = 50 μm.
The tissue received from goat No. 8 was a portion of an ulcerated, roughly spherical mass that extended from the cutaneous aspect of the cheek into both the nasal and oral cavities. Despite this extensive tissue involvement, the goat remained bright and alert and had an excellent appetite. The microscopic features of this neoplasm were consistent with a hemangiosarcoma and included extensive hemorrhage with poorly defined sinusoidal to capillary-like channels separated by cells with ill-defined boundaries (Fig. 5). The oval nuclei had a vesicular chromatin pattern and there was marked anisokaryosis with one or two large nucleoli. About two mitotic figures were present per HPF, and multinucleate cells were occasionally seen. Large numbers of neutrophils infiltrated this ulcerated lesion. The goat was euthanatized 1 week after the biopsy procedure because the neoplasm had extended into the surrounding soft tissues.
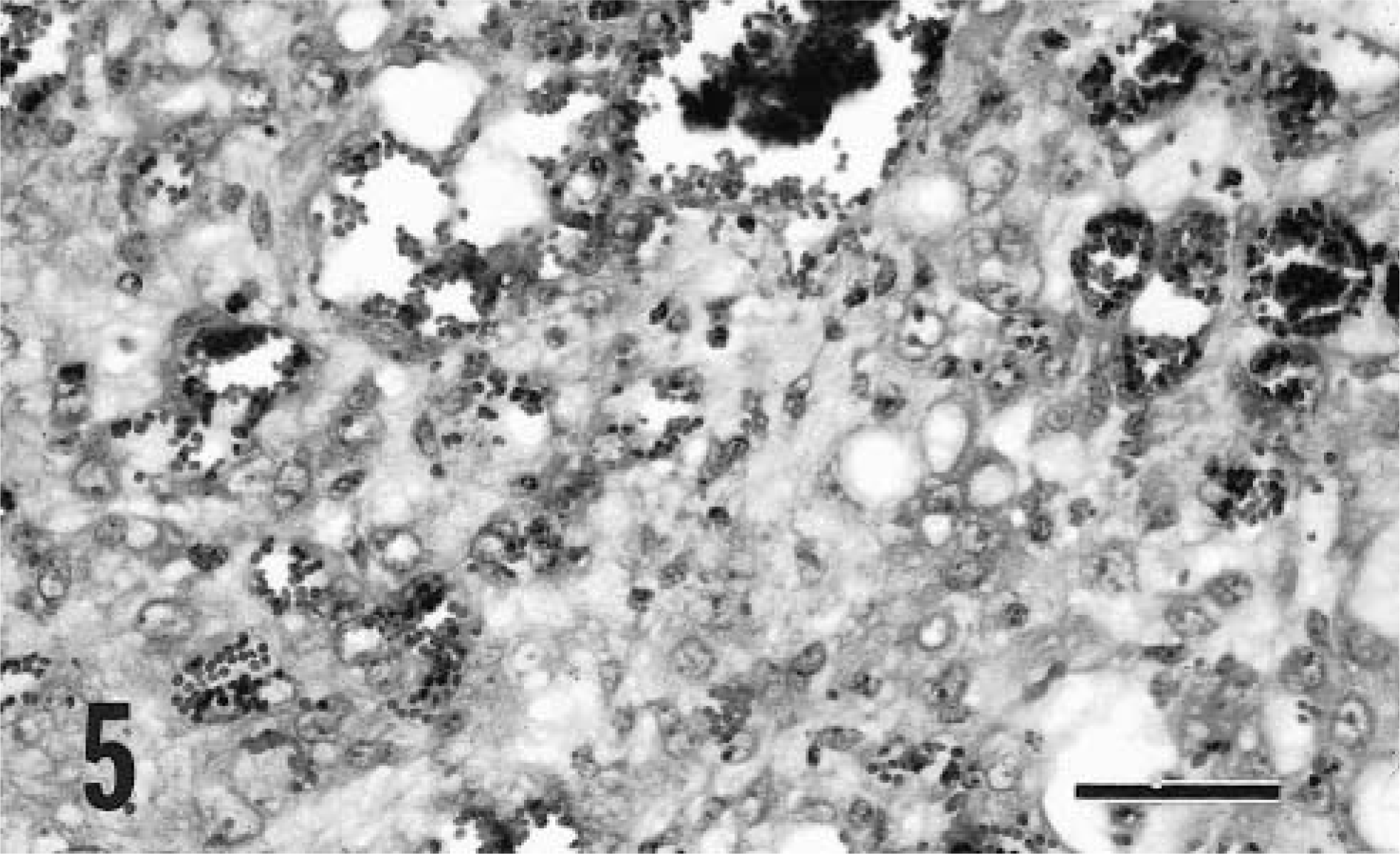
Cutaneous hemangiosarcoma; goat No. 8. The tumor contains cells that have poorly defined boundaries and round to oval nuclei with vesicular chromatin. Many blood-filled spaces are present. HE. Bar = 60 μm.
FVIIIR-Ag was detected within neoplastic cells of all seven goats tested. Diffuse and intense staining was visible in the neoplastic cells of goat Nos. 1–6 (Fig. 1). Staining was very weak and restricted to a few scattered neoplastic cells in the tissue from goat No. 7. Antigen was readily identifiable within adjacent normal endothelial cells.
The final diagnosis assigned to each mass is included in Table 1. The mass from goat No. 1 was originally diagnosed as a cutaneous hemangioma, but we reclassified it as a vascular hamartoma, based on the involvement of different types of blood vessels and the presence of smooth muscle and pericytes around the rudimentary capillaries. Although situated in the subcutis and deep dermis, the mass from goat No. 2 featured the same general organization and cellular composition and was also diagnosed as a vascular hamartoma. The difficulty of distinguishing vascular hamartomas from hemangiomas has been discussed,5,14 and we do not profess to have found the solution. The lobular architecture and proliferation of arteriolar and capillary components within these masses was similar to that described for cutaneous hemangiomas of young horses and dogs and human infants and to the condition known as bovine cutaneous angiomatosis.5,14 Both of these caprine hamartomas were first noted when the goats were 2 years of age.
The mass from goat No. 6 was classified as a hemangioma based on the well-defined nature of the cavernous vascular channels and a lack of mitotic activity. However, the endothelial cells in this tumor were generally larger and lacked the flattened outline characteristic of hemangiomas in most species.
Classification of the tumors from goat Nos. 3, 4, and 5 was difficult. These ulcerated masses lacked the organization of granulation tissue but featured mitotic activity within endothelial cells without any evidence of tissue invasion by these cells. We chose to classify these growths as hemangiomas, but based on the presence of the occasional mitotic figures others might identify these masses as malignant tumors. One or all of these masses may actually have been cutaneous papillomas, in which necrosis of the epidermal component had resulted in a mass dominated by reactive rather than neoplastic vascular elements. A highly vascular dermal core has been described for some caprine papillomas.12 Experimental inoculation of the teat of a goat with a papilloma extract resulted in the formation of a cutaneous hemangioma, unaccompanied by epidermal hyperplasia.12 Only one of the three masses in question was verrucose (goat No. 4), but all were raised and somewhat pedunculated. Although not confirmed histologically, the two masses on the contralateral shoulder of goat No. 3 were grossly compatible with cutaneous papillomas, and both regressed spontaneously. Despite these reservations, the highly vascular nature and random orientation of the blood vessels in the specimens from these three goats justifies diagnoses of vascular neoplasia.
Two of the masses examined had histopathologic features consistent with malignancy (goat Nos. 7 and 8). This malignant character was also observed at the gross level; invasive growth into soft tissues was described for both masses at the time of removal. Unfortunately, necropsies were not performed to check for sites of internal metastasis. The IHC stain for FVIIIR-Ag in the mass from goat No. 7 gave only a weak positive result, consistent with a poorly differentiated hemangiosarcoma. Although similar data are not available for caprine vascular tumors, in the dog 89% of hemangiosarcomas and 100% of hemangiomas were reported to be positive for FVIIIR-Ag.13 An inconsistent FVIIIR-Ag reactivity pattern similar to that seen in the neoplastic cells from goat No. 7 was also noted in some canine hemangiosarcomas.13 Although IHC confirmation could not be attempted, the mass from goat No. 8 featured distinct vascular channels lined by pleomorphic cells, consistent with hemangiosarcoma.
Review of the case records did not reveal any predisposing factors to connect the cases in this study. Nubian goats were most commonly affected, but this breed also accounts for most caprine submissions seen by our diagnostic laboratories. No site predilection was identified, although both highly invasive tumors were found on the head. In four of five goats for which coat color was known the tumor was located on a white area. A correlation between lightly haired, poorly pigmented skin has been noted for some forms of vascular neoplasia in the dog.4 Solar elastosis of the superficial dermis was noted in several of the affected dogs. Elastosis was not visible in any of the HE-stained sections in the present study, but minimal intact superficial dermis was available for examination in most cases.
Cutaneous neoplasms are uncommon in the goat, with papillomas, squamous cell carcinomas, and melanomas accounting for most documented cases.6 At least one cutaneous hemangiosarcoma has been reported in this species, although the anatomic location and microscopic features were not described.1 Reports of vascular tumors in any anatomic site in the goat are rare, but hemangiosarcoma has been found in the bulbourethral gland and lung of this species.2,11 A vascular hamartoma has also been identified in the spinal column of a goat.7 The two laboratory databases searched during the present study did not include any diagnoses of vascular tumors in any noncutaneous location in goats.
The small number of cases identified in this study confirms that vascular tumors are rare in the goat. Nevertheless, the masses identified here provide evidence that this species does develop the range of vascular growth abnormalities observed in other domestic animals. Any conclusions on the biologic behavior of vasoproliferative lesions in goats will require the study of a larger number of cases.
